Abstract
Background
Parkinson’s disease (PD), the second most prevalent neurological illness, is treated with pharmacologic and nonpharmacologic techniques, including medicinal plants and their extracts.
Purpose
We assessed the anti-neurodegenerative properties of aridanin against 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride (MPTP)-induced PD in mice.
Materials and Methods
In the current investigation, the neuroprotective potential of aridanin in the MPTP-induced PD animal model via behavioral analysis, oxidative stress, pro-inflammatory cytokines, and histopathology was determined.
Results
The results showed that mice treated with MPTP had mobility, motor issues, and behavioral dysfunctions. When mice were challenged with MPTP, they had low antioxidant levels. The levels of malondialdehyde (MDA) and pro-inflammatory cytokines were high in MPTP-treated mice. There are signs of PD based on these results. Behavioral effects were improved, antioxidant levels increased, and MDA and proinflammatory cytokines levels decreased in animals treated with aridanin.
Conclusion
Thus, in combination, aridanin therapy guards against MPTP-induced neuronal loss, neuroinflammation, oxidative stress, and motor dysfunction in PD mice.
Keywords
Introduction
Parkinson’s disease (PD) is a well-known clinical phenomenon with several causes and clinical symptoms. Except for infectious etiology, the rapidly increasing frequency of PD throughout the globe is similar to several traits commonly seen during a pandemic. The effects of PD in an individual are profound. A degenerative disease’s distinctive characteristic is that it can last for several years. For those impacted, the normal prognosis is a gradual decline with more impairments (Bloem et al., 2021). An early and noticeable loss of dopaminergic neurons in the substantia nigra pars compacta (SNpc) characterizes PD, a neurodegenerative condition. As a result, the basal ganglia are depleted of dopamine, causing Parkinsonian-like motor symptoms. Many other nonmotor symptoms of PD are also linked to the disorder, some of which appear more than 10 years before the motor impairment (Kalia & Lang, 2015).
In the United States, around one million people are affected by PD. By 2030, this number is predicted to upsurge to 1.2 million. Followed by Alzheimer’s disease, PD is the second most common neurological illness. About 90,000 US people receive a PD diagnosis annually. Over 10 million individuals around the world are affected by PD. Although the rises with age, four percent of PD patients are thought to receive a diagnosis before turning 50. By 2030, 1.2 million Americans will have PD, predicts the Parkinson’s Foundation Parkinson’s Prevalence Project. The Prevalence Project was formed in 2014 to evaluate the accurate prevalence of PD in North America. This study incorporates data from a wide range of demographic groups (Ben-Shlomo et al., 2024).
PD is 1.5 times more prevalent in males than in females. It was previously believed that environmental variables were the primary trigger of PD; however, research is showing that genetics and environment interact intricately to generate the condition. PD is brought on by nongenetic variables, including dietary factors, inflammation, estrogens, heavy metals, pesticides, herbicides, and smoking (De Lau & Breteler, 2006). Premotor PD symptoms include nonmotor clinical signs like neuropsychiatric disorders, rapid eye movement (REM) behavior sleep problems, olfactory defects, and sadness. Biological indicators for PD are used to identify high-risk patients, track the illness’s course, and investigate the impact of therapy in preventing dopaminergic cell loss. Early diagnostic techniques for PD include markers including nonmotor clinical symptoms, cerebrospinal fluid testing, and several imaging modalities (Bhat et al., 2018).
Genetic models and toxin models are the two primary experimental animal models used to evaluate the pathogenesis of PD. Acute animal models of PD are induced by the environmental toxin 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine hydrochloride (MPTP). When MPTP is administered, neurons in the striatum and SNpc terminate. Researchers have an exceptional potential to examine the neuroprotective qualities of new medicines using the MPTP-triggered PD models. Through the specific inhibition of complex-I of the mitochondrial electron transport chain, MPTP in the brain is transformed into the hazardous metabolite MPP+. Dysfunction of the mitochondria leads to disruption of adenosine triphosphate (ATP) and aberrant metabolism of lipids and amino acids (Atiq et al., 2023).
MPTP, a drug used in PD treatment, is administered intraperitoneally or intravenously to mice, which metabolizes into toxic MPP+, damaging dopaminergic neurons and causing behavioral assessments. These tests assess motor function and cognitive function, as well as neurochemical analysis, such as dopamine measurement and immunohistochemistry. Post-mortem analysis can also be conducted to examine neuronal loss and Lewy body formation. The success of these models is often confirmed by confirming the presence of characteristic PD-like pathologies, such as selective loss of dopaminergic neurons, motor defects, and alpha-synuclein aggregates formation (Duty & Jenner, 2011; Mustapha & Mat Taib, 2021).
The primary medications used as first-line treatment for PD are levodopa and dopamine agonists, and the patients are also treated with anticholinergic medicines as well as other prescriptions. Owing to the progressive nature of PD, researchers are looking for neuroprotective drugs that can halt the root cause of the diseased state and halt additional neuronal loss. Numerous medicinal plants, as well as their chemical constituents and extracts, may have neuroprotective benefits against the degeneration of neural cells, according to the literature (Abushouk et al., 2017).
Aridanin, a triterpenoid glycoside, is obtained from the root of Tetrapleura tetraptera, which belongs to the family Fabaceae. T. tetraptera, also known as Aidan fruit or Prekese, is a West African plant used in folk medicine for its anti-inflammatory, antioxidant, digestive, and respiratory benefits, but research is ongoing. The compound’s biological effects as an antibiotic and in preventing the spread of schistosomiasis have proven to be a significant lead in the creation of novel medications of therapeutic significance. The substance showed a wide range of pharmacological potential, particularly in terms of anti-bacterial, anti-viral, anti-cancer, molluscicidal, and anti-schistosomal properties (Sikam et al., 2022). Aridanin’s ability to modulate cholinergic, serotonergic, and opioid receptors results in its anti-convulsive properties (Saliu et al., 2021).
In this work, the aim was to determine the neuroprotective potential of aridanin in the MPTP-induced PD animal model via behavioral analysis, oxidative stress, pro-inflammatory cytokines, and histopathology.
Materials and Methods
Materials
A phytocompound, aridanin (C38H61NO8; Mw 659.89), and MPTP hydrochloride (MPTP-HCl) were procured from Sigma–Aldrich (St. Louis, USA) and acquired. We bought tumor necrosis factor-alpha (TNF-α), interleukin (IL)-1β, and IL-6 assay kits from R&D systems (Minneapolis, MN). The investigations employed only analytical-grade chemicals and reagents, and all other chemicals, unless otherwise stated, were from Sigma (St. Louis, USA).
Animals and Experimental Design
In this investigation, male C57BL/6 mice weighing 25–30 g were employed for the experiment. For 7 days, the mice were acclimated. The mice were kept in a controlled environment (22 ± 2°C) with 12 hours of light and 12 hours of dark cycle, and they were also given unrestricted access to meals and water. The institutional ethics committee authorized every experimental protocol that was used. Ethical approval no. (2023-K168) was granted.
The mice were assigned to four groups, each consisting of six mice: Group I (control) received 0.5 ml of normal saline. To simulate PD, animals in group II (disease group) were given intraperitoneal injections of MPTP (30 mg/kg b.w. in saline) once a day for consecutive 5 days. Group III (aridanin + MPTP) was treated with aridanin (30 mg/kg b.w.) + MPTP (30 mg/kg b.w.) for 14 days. MPTP was given from the 10th to the 14th day of therapy. The animals in group IV were given a single oral dosage of aridanin (30 mg/kg body weight) for 14 days. Behavioral tests were performed after the prescribed course of therapy. Animals were euthanized and sacrificed after behavior pattern analysis, and brain tissues were taken right away and stored at –80°C for additional examination.
Behavioral Analysis—Pole Test
The pole test machine consisted of a 40 cm-tall hardwood pole with a 10 mm diameter. Mice, with their heads positioned close to the top of a rough-surfaced wooden pole, were used to measure how long it took them to reach the level. Three trials of the test were conducted, each lasting 10 minutes. We used the mean of three trials to assess the behavioral changes (Matsuura et al., 1997).
Open Field Assessment
Utilizing an open field configuration with dimensions of 40 × 40 cm in diameter and 40 cm in height, the spontaneous mobility behavior was determined. The mice were tracked using video tracking software 3.0 as they moved across the 16 uniformly sized squares that collectively make up an open field box. The mice were allowed several hours to acclimate to their new environment before the experiment. In the same corner, every mouse is placed separately. To get rid of odors in between tests, the open area was washed with 10% ethanol and allowed to dry. We measured how far the mice traveled overall and how long they stayed in the center. The distance is measured in centimeters, while the duration is expressed in seconds (Seibenhener & Wooten, 2015).
Rotarod Assessment
To evaluate motor coordination, a rotarod was utilized. The evaluation is contingent upon the amount of time that mice can stay on to a revolving rod. Before the experiment, every animal had a 1-minute trial with the moving rod. For 300 seconds, their motor coordination was measured while they were positioned on a revolving rod with speed ranges of 5–15 rpm. There was a noticeable lag in the rolling rod fall. Regular mice were able to stay on the revolving rod for an endless amount of time. Three times a day, at 30-minute periods, the motor performance was assessed, and the average retention time was computed (Campos et al., 2013; Rozas et al., 1997).
Grip Assessment
The grip test was conducted using the earlier methodology (Leiter et al., 2011). In summary, the test setup includes two 40-cm elevated supports on a level surface and 50 cm of copper wire linked between them. The mice were carefully flipped upside down and set on the steel wire. The animals were permitted to remain on the metal wire for a maximum of 90 seconds. Every animal received 10 tries at intervals of 60 seconds. The value with the longest delay to collapse or release both hind legs was noted (Leiter et al., 2011).
Footprint Assessment
The foot-printing test was carried out a bit differently than in the prior study (Yadav et al., 2014). In brief, nontoxic paints were applied to the forepaws and hind paws of the mice, and they were then placed to walk continuously on a white piece of paper. The analysis of the stride length included calculating the distance taken on the same-side limbs with each step (Yadav et al., 2014).
Preparation of Brain Tissue Extract for Enzyme Assay
The assays of superoxide dismutase (SOD), catalase (CAT), glutathione (GSH), and thiobarbituric acid reactive substances (TBARS) were performed on this brain tissue extract. 1.5 ml of phosphate buffer (pH 7.2) containing K2HPO4 (50 mM) was used to sonicate the brain tissues on ice. The brain homogenates were centrifuged at centrifuge rates tailored for each test (10,000g for TBARS for 10 minutes at 4°C, 10,000g for CAT and GSH for 15 minutes at 4°C, and 1,500g for SOD for 5 minutes at 4°C). Supernatants were collected and kept at 80°C until biochemical analysis.
Estimation of TBARS, SOD, CAT, and GSH
1,1,3,3-Tetramethoxypropane was used as a reference in fluorometric analysis to determine the amounts of TBARS. In a nutshell, 50 µl of tissue samples were loaded in a tube with 1 ml of demineralized (DM) water. The samples were in a water bath at 95–100°C for an hour, when they were exposed to 1 ml of a solution that included 29 µmol/l of 2-thiobarbituric acid (TBA) mixed in acetic acid (8.75 mol/l). The samples were treated with 3.5 ml of n-butanol to extract the reaction product, which was then vortexed and centrifuged at 3,000g for 10 minutes. Following the separation of the butanol phase comprising the extracted reaction product, the fluorescence was recorded at 525 nm for excitation and 547 nm for emission wavelengths using spectrophotometry (Botsoglou et al., 1994). The CAT (Johansson & Borg, 1988), SOD (Malstrom et al., 1975), and GSH (Baker et al., 1990; Vandeputte et al., 1994) in the brain tissues were determined using assay kits and spectrophotometric studies. The results were represented in units per milligram of protein.
Assay of IL-1β, TNF-α, and IL-6 Cytokines
Enzyme-linked immunosorbent assay (ELISA) kit was employed to quantify the IL-1β, TNF-α, and IL-6 levels. The 96-well plates were filled with brain samples, and the appropriate antibodies (IL-1β, IL-6, or TNF-α) were added. The plates were then incubated at 4°C for an entire night. The block was then exposed to 1% BSA for 1 hour at 37°C. Following the addition of 50 µl of horseradish peroxidase (HRP), all of the wells underwent incubation at room temperature for 45 minutes. Lastly, the plate was read at 450 nm, and the protein concentration was represented in parts per milligram (pg/mg) using kits from R&D systems (Minneapolis, MN).
Histoarchitecture Analysis
The brain tissues of both control and PD mice treated with ariadanin were processed using a saline buffer. Formalin was used to dry the brain tissue, and the same was then embedded in paraffin. The tissues that had been preserved in wax were cut into 6 µm pieces and then stained using hematoxylin and eosin (H&E). Lastly, an optical microscope was used to examine the stained sections to look for changes in the histology.
Statistical Analysis
The findings are shown as the mean ± standard deviation (SD) and were assessed using the SPSS software (version 12.0) for one-way analysis of variance (ANOVA). Duncan’s multiple range test (DMRT) was utilized to measure substantial variations between the groups at p < 0.05.
Results
The Potential Effect of Aridanin Against Behavioral Dysfunction in MPTP-Intoxicated Mice
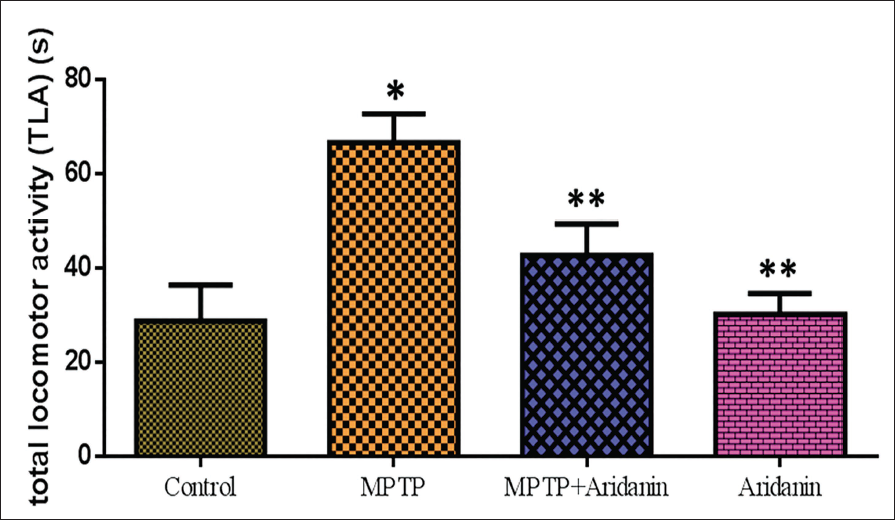
The protective effect of aridanin against MPTP-induced behavioral impairment in mice intoxicated with MPTP is shown in Figure 1. In the pole test, the MPTP mice revealed a markedly longer time to change positions and approach downhill; however, this impact was greatly mitigated by the aridanin injection.
Protective Effect of Aridanin Against 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-Induced Behavioral Dysfunction in MPTP-Intoxicated Mice. Vehicle or Doses (30 mg/kg) Aridanin was Administered Orally Once per Day for 14 Days, and MPTP was Administered 30 mg/kg on the Last Day Of Aridanin Treatment. Arrival at the Bottom [Total Locomotor Activity (TLA)] of the Pole was Recorded with a Cut-Off Time of 70 s for Pole Test Performance. Values are Given as Mean ± Standard Deviation (SD) for Six Mice in Each Group. *p < 0.05, Compared with the Control Group; **p < 0.05, Compared with the MPTP Group.
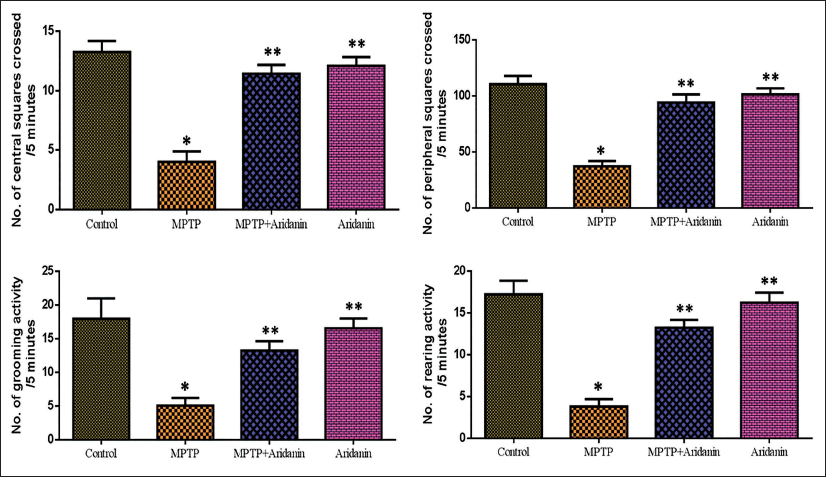
The Potential Effect of Aridanin on Behavioral Assessment in MPTP-Intoxicated Mice by Open Field Test
The behavioral study contains an open-field test that illustrates how aridanin helps the animals receiving MPTP move more freely. An open-field exam covers a wide range of traits, including rearing, grooming, line central, and peripheral. There was a significant reduction in grooming, rearing, and central and peripheral locomotor activity when the MPTP mice were compared to the typical control. Aridanin remarkably elevated the number of rearing as well as the activity of the muscles. Similarly, aridanin-administered mice displayed a significant increase in grooming behavior (Figure 2).
Effect of Aridanin on 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-Induced Impairment of Spontaneous Movement and Activities in an Open Field Test for 5 Minutes. If the Animal Crosses the Central Nine Squares Sixteen Outer Squares Drawn on the Bottom of the Arena were Measured by Manually Counting. Rears and Grooms were Manually Counted. Values are given as Mean ± Standard Deviation (SD) for Six Mice in Each Group. * p < 0.05, Compared with the Control Group; ** p < 0.05, Compared with the MPTP Group.
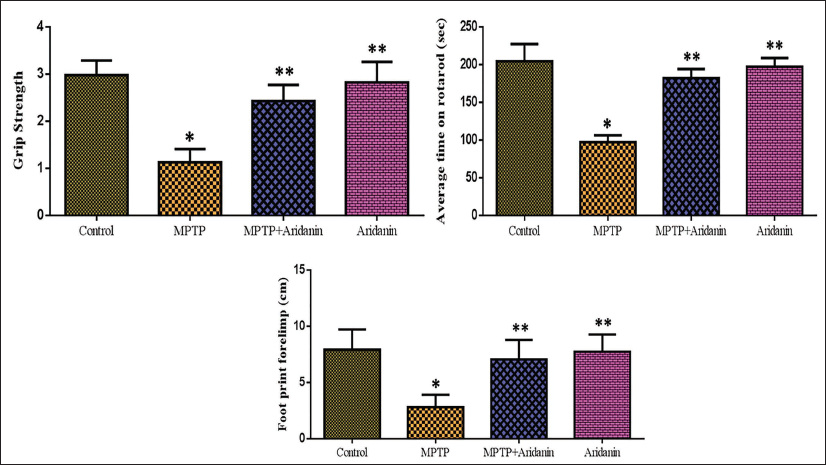
The Potential Effect of Aridanin on Behavioral Assessment in MPTP-Intoxicated Mice by Grip Test, Footprint, and Rotarod Test
The grip test findings for the MPTP-intoxicated mice receiving aridanin treatment are shown in Figure 3. The hanging time in MPTP mice was much lower than in control mice. In contrast, mice who were induced with MPTP and given aridanin beforehand hung for noticeably longer periods than those who were induced with PD. Figure 3 shows the motor coordination test results for the experimental animals using a rotarod. Compared to control groups, mice injected with MPTP showed a shorter retention period as well as poorer neuromuscular coordination. Aridanin pretreatment markedly increased balancing capacity and retention time and prevented disorientation in mice treated with MPTP. Abridged stride length is one of the primary features of the abnormal gait associated with PD. The results of our investigation demonstrated that the forelimb and hindlimb strides of the MPTP-induced mice were notably shorter. Conversely, compared to the MPTP-treated group, the mice treated with aridanin + MPTP had longer strides. Moreover, no variations in stride lengths were seen between aridanin alone (Figure 3).
1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP)-Induced Group Significantly Decreased Grip Strength, Motor Coordination, and Forepaw Stride Length Compared to the Control, and Aridanin Treatment Recovered in the Aridanin + MPTP-Induced Group Compared to MPTP-Alone Induced Animals. Values are Given as Mean ± Standard Deviation (SD) for Six Mice in Each Group. * p < 0.05, Compared with the Control Group; ** p < 0.05, Compared with the MPTP Group.
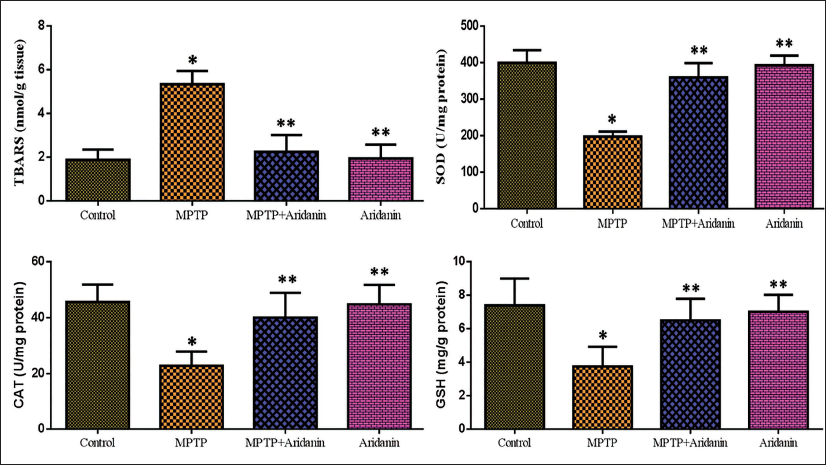
Potential Effects of Aridanin on Antioxidant Levels in MPTP-Intoxicated Mice
Levels of TBARS and antioxidants in the experimental mice’s brains reveal the potential effect of aridanin on MPTP-intoxicated mice. Tests for CAT, SOD, and GSH—the main components of the natural antioxidant-defined system—were used to evaluate oxidative stress. When compared to normal mice, the MPTP group’s CAT, SOD, and GSH levels showed a statistically significant decline. The pretreatment of aridanin significantly lessened the MPTP-induced decrease of CAT, SOD, and GSH in the midbrain area. We measured oxidative stress using the lipid peroxide (LPO). TBARS is one LPO biomarker that demonstrates malondialdehyde (MDA) generation and lipid breakdown. In this work, the MDA of the MPTP mice was considerably elevated than the control. The MPTP-induced parkinsonism-causing mice had significantly lower MDA levels than the MPTP group after receiving aridanin (Figure 4).
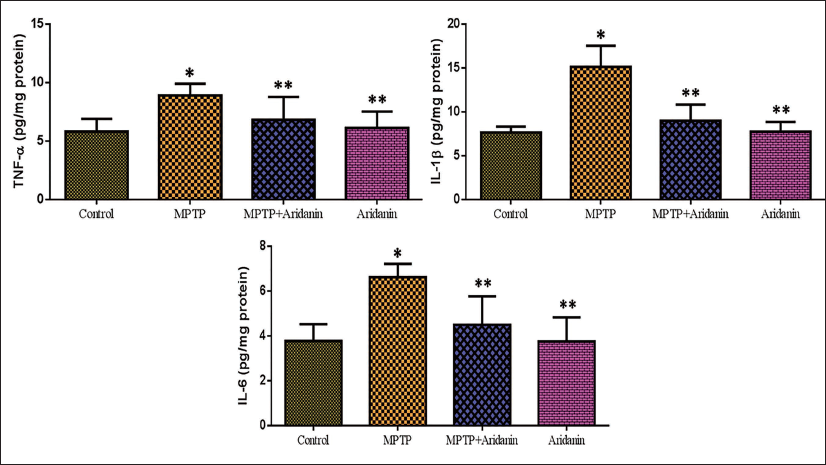
Effects of Aridanin on Pro-Inflammatory Cytokine in MPTP-Triggered Mice
The impact of aridanin on pro-inflammatory cytokines in rats intoxicated with MPTP is shown in Figure 5. The brain tissue of MPTP-triggered rats revealed a remarkable upsurge in IL-1β, TNF-α, and IL-6 levels. However, in comparison to the MPTP alone group of mice, aridanin pretreatment considerably reduced the IL-1β, TNF-α, and IL-6 levels in MPTP-induced mice. There were no appreciable differences observed in either the aridanin-only treatment group or the untreated control group.
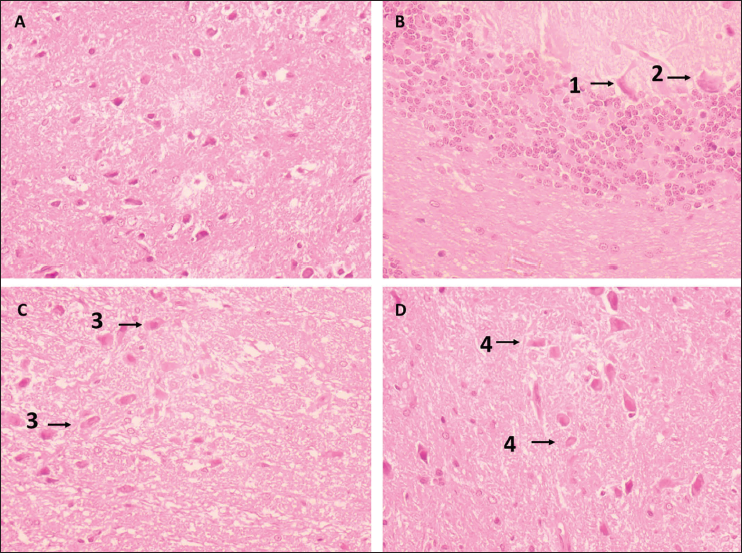
Effects of Aridanin on Brain Histoarchitecture of MPTP-Intoxicated Mice
The brain tissue of control and PD-affected mice given aridanin is depicted in Figure 6. The MPTP mice demonstrated substantial inflammation, glial cell loss, and neuronal ganglion loss compared to the control. Conversely, the aridanin-treated PD animals demonstrated robust neuroprotection by decreasing inflammation, which shielded the glial cells from harm along with normal tissue structures and ganglion layers.
Effect of a 14-Day Aridanin Treatment on the Histoarchitecture of Mice’s Striatum After 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP) Pretreatment. A: Normal Architectural Design. B: Neuronal Cells (1) That Showed Inflammation, Glial Cell Destruction, Neuronal Ganglia Loss, and Degeneration, as well as Atrophy, Necrosis, and Occasionally Apoptosis (2). C: Neurons with a Moderate Degree of Degeneration have Hollow Spaces Surrounding (3) Them and Shrinking Neuronal Cells. D: Disintegration, a Modest form of Neuronal Degeneration Accompanied by Neuronal Hypertrophy (4).
Discussion
With over 6 million cases worldwide, PD is the second-highest prevalent neurological illness. One of the leading causes of neurological damage today is PD, with a prevalence that has grown by 2.5 times over the previous generation. PD pathology includes Lewy bodies and Lewy neurites, as well as diminished cells in the substantia nigra and other brain areas (Tolosa et al., 2006). PD can be described by the mechanism of Lewy body accumulation, which is abnormal intracellular builds made up of proteins like alpha-synuclein (aSyn) and ubiquitin, as well as the exhaustion or deterioration of dopaminergic neurons in the SNpc. Approximately 60–70% of SNpc neurons are damaged before symptoms emerge. Research has demonstrated that other peripheral and central nervous system components are also implicated in the degenerative process of PD, in addition to the dopaminergic neurons of the SNpc. As the disease progresses, Lewy body pathology spreads to limbic and neocortical brain areas from its origins in cholinergic and monoaminergic brainstem neurons and olfactory system neurons. Once the end-stage illness has developed, the dopaminergic neuron loss that was once limited to SNpc becomes more prevalent (Radhakrishnan & Goyal, 2018).
MPTP is metabolized in the blood–brain barrier (BBB), is converted to MPP+, and is released in the extracellular space. Aridanin has been found to have neuroprotective potential against MPTP intoxication-mediated Parkinsonism in a mice model (Rehman et al., 2022). Motor assessments revealed motor-related markers in MPTP mice, including akinesia, muscle strength, bradykinesia, gait patterns, and synchronized motor functions (Mustapha & Mat Taib, 2021). The study found that MPTP-mediated bilateral partial striatal dopamine terminal lesions increased reaction time and immobility, leading to a decline in stepping action (Meredith & Rademacher, 2011). This neuronal loss in the SN is a significant characteristic of PD, with motor function significantly altered by MPTP intoxication.
Oxidative stress, synucleinopathy, neuroinflammation, dopaminergic loss, and motor impairment are among the pathological characteristics of PD. Thus, we concentrated on these variables in the present investigation and employed aridanin to specifically target these problematic components. The current work offers the first concrete proof that aridanin inhibited the MPTP-triggered PD mice model. The current study’s behavioral assessment results, which included grip, open field, rotarod, pole, and footprint analysis tests, indicate that receiving aridanin over an extended period enhanced muscle activity and movement. It is commonly recognized that MPTP induction causes cognitive impairments in mice and nonhuman primates (Mustapha & Mat Taib, 2021). As a result, measuring behavioral function offers a sensitive assessment of aridanin’s capacity to offer neuroprotection. All of the mice that were given MPTP showed a substantial change in their cognitive abilities, with poor results in tasks involving passive avoidance and wide fields.
The Nigro-striatal pathway is crucial for the production of dopamine neuronal cells and TH, an essential neurotransmitter for movement and balance. In mice, MPTP-administered mice show a decline in dopamine neuronal cell number and TH expression levels. However, aridanin treatment reduces the loss of dopamine neurons and rescues the decrease in TH expression mediated by MPTP. Aridanin prevents MPTP-mediated decreased dopamine levels and their metabolites, defending dopamine neurons and restoring declined TH expression in MPTP-mediated PD mice. Neuroinflammation is primarily mediated by microglial cell activation, and MPTP can damage dopamine neurons (Huang et al., 2017). MPTP induction blocks mitochondrial complex I, leading to excessive reactive oxygen species (ROS) production (Bourdy et al., 2014). Inhibiting ROS overproduction might be essential for protecting cascade-dependent neuroinflammation and neurodegeneration. Activated microglial-induced neuroinflammation plays a significant role in PD pathogenesis, with NF-kB nuclear translocation increasing proinflammatory cytokine marker release (Villadiego et al., 2023).
Oxidative stress is caused by an inconsistency in the body’s ability to employ antioxidants to counteract the detrimental impacts of free radicals and their generation. SOD, GSH, CAT, and uric acid are the main antioxidants in the human body. On the other hand, free radicals like hydroxyl, H2O2, NO, and superoxide radicals are the ones that seriously damage cells. MDA is a noteworthy consequence of lipid peroxidation. The onset of PD has been linked to the antioxidant defense system’s inability to prevent the production of free radicals, which destroy all of the cell’s constituents, including lipids, deoxyribonucleic acid (DNA), and proteins. This ultimately results in cell death (Wei et al., 2018). In this work, the level of MDA was high, and the level of antioxidant enzymes was low in MPTP-treated animals. On treatment with aridanin, the level of antioxidant enzymes increased, and the MDA level decreased, which indicates the anti-oxidative property of aridanin (Sikam et al., 2022).
Aridanin’s anti-inflammatory action suppresses NF-kB expression, leading to the release of iNOS, Cox-2, TNF-α, IL-6, and IL-1β (Zhang et al., 2023). Effective drugs with antioxidative and anti-inflammatory properties are recommended as therapeutic molecules for neurodegenerative disorders. Triterpenoid derivative compounds are excellent scavengers, anti-inflammatory, and neuroprotective in demyelinating and neurodegenerative disorders (Kwon et al., 2013; Won et al., 2006).
One factor that is crucial to PD is the neuroinflammatory response. Following microglial cell activation (IFN-γ), pro-inflammatory markers like TNF-α, IL-1β, and IL-6 are secreted, which enhances the neurotoxicity and activates the NF-κB signaling (Rodríguez-Cruz et al., 2020). In this study, the MPTP mice had elevated IL-1β, IL-6, and TNF-α levels in the midbrain. The amounts of every pro-inflammatory cytokine that was assessed dropped considerably when aridanin was administered as therapy compared to the MPTP group. The result suggests that inflammation is controlled in PD, most likely due to suppression of the NF-κB pathway.
In line with the biochemical data and those from previous studies, our histological findings revealed neuronal degenerations in the brain tissue of MPTP-intoxicated mice, including inflammation, glial cell destruction, and neuronal loss. On treatment with aridanin, there was a considerable reduction in the histopathological changes.
Conclusion
In the end, our results provided evidence that aridanin may be able to shield the brain against MPTP-triggered PD in mice. Treatment with aridanin improves motor function and facilitates movement. In MPTP-treated mice, further aridanin therapy decreased MDA levels and raised antioxidant levels, which in turn decreased oxidative stress. Treatment with aridanin also decreased levels of other inflammatory cytokines. Hence, aridanin treatment demonstrates neuroprotective activity against MPTP-induced PD in mice by inhibiting oxidative stress, neuronal loss, inflammation, and motor defects.
Abbreviations
SNpc: Substantia nigra pars compacta; PD: Parkinson’s disease; MPTP: 1-Methyl-4-phenyl-1,2,3,6-tetrahydropyridine; CAT: Catalase; SOD: Superoxide dismutase; GSH: Glutathione; IL-6: Interleukin-6; IL-1β: Interleukin-1beta; TNF-α: Tumor necrosis factor-alpha; PFA: Paraformaldehyde; H&E: Hematoxylin–eosin; MDA: Malondialdehyde; NO: Nitric oxide.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was approved by the Ethics Committee of the Second Affiliated Hospital of Heilongjiang University of Traditional Chinese Medicine [Ethical approval no. (2023-K168)].
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
