Abstract
Background
The rationale for the pathogenesis of Alzheimer’s disease (AD) is the production of amyloid-beta (Aβ) and its resultant toxic effects.
Purpose
In this work, rats with AD were used as models to assess the modulatory effects of tanshinone IIA (tan IIA) in alleviating cognitive impairment, oxidative damage, inflammation and apoptosis.
Materials and Methods
Aβ1–42 was injected bilaterally in the hippocampus to establish an AD rat model. The effects of tan II A on the learning and memory capacities of AD rats were detected by ethological experiments. Tan IIA’s anti-inflammatory and antioxidant capacities were examined in this study using biochemical and immunohistochemical methods on rat hippocampus tissues.
Results
The experimental data showed that the cognitive ability of rats could be significantly improved by tan IIA. Furthermore, fewer injured neuron apoptosis was demonstrated in the tan IIA rats than in the AD group. More importantly, on the one hand, we discovered that the tan IIA group had considerably lower levels of interleukin 1 beta, tumour necrosis factor-alpha and inducible nitric oxide synthase. On the other hand, tan IIA was discovered to prevent oxidative stress by raising glutathione and superoxide dismutase activity and lowering malondialdehyde, protein carbonyl and 8-hydroxy-2′-deoxyguanosine levels.
Conclusion
Tan IIA can alleviate cognitive impairment and neuronal cell damage by inhibiting oxidative stress and inflammatory responses during AD.
Introduction
As an insidious neurodegenerative disease, Alzheimer’s disease (AD) is clinically characterised by full-blown dementia manifestations such as memory impairment, aphasia, dysarthria, executive dysfunction as well as personality and behavioural changes, which can affect the quality of life as well as even death (Ferrari & Sorbi, 2021). With a prevalence rate of up to 50% among people 85 and older, Alzheimer’s is a disease that afflicts seniors worldwide. It was estimated that more than 46.8 million people around the world are AD patients, and with the extension of the life of this figure will be doubled in 2030 (Mushtaq et al., 2015). AD is the fourth greatest cause of mortality globally, behind cardiopathy, cancer and stroke. Because its pathophysiology is complicated and unclear, there is no clinical drug that can treat it completely.
Aggregation of insoluble amyloid-beta (Aβ) and degenerative neuritis are the main causes of the neuropathological feature senile plaques (SP) in AD (Durazzo et al., 2014). Aβ is a peptide with 39–42 amino acids that is created when the amyloid precursor protein is cleaved enzymatically. Additionally, the Aβ peptide comes in two varieties: one has 40 amino acids, while the other has 42 (Cheignon et al., 2018). Soluble oligomers are thought to be the most harmful type of Aβ1–42 (Kayed et al., 2003). The generation, metabolism and toxicity of Aβ are the central link of AD pathological mechanisms.
Furthermore, AD may be triggered by increased levels of oxidative stress and pronounced inflammation associated with Aβ induced (Uruno et al., 2020). On the one hand, Aβ may have the assistance of metal ions in causing reactive oxygen species (ROS) production. Conversely, Aβ peptides weaken the cellular antioxidant mechanisms, which leads to oxidative stress (Ionescu-Tucker & Cotman, 2021). At present, multiple mechanisms of oxidative stress as an early factor of AD have been identified (Bai et al., 2022). Aβ deposition and associated neuroinflammation are considered to be the important factors leading to cognitive deficits in AD. In addition, it has also been found that inflammation is often associated with the brain in AD patients. Activated microglia, astrocytes and neuronal loss are characteristics of local inflammation. The neuroinflammation induced by Aβ forms the pathological features of AD, and the neuroinflammation further exacerbates cognitive decline in aged mice (Sy et al., 2011). Oxidative stress and neuroinflammation have been recognised in existing studies as key factors that induce and exacerbate pathological processes in AD.
With the prevalence of AD increasing globally every year, the need for treatments that can accurately target the disease with fewer side effects has become more urgent. The Chinese herb Salvia miltiorrhiza contains a lipid-soluble compound called tanshinone IIA (tan IIA) (Figure 1). It is well known to possess biological properties such as promoting blood flow and eliminating blood stasis, as well as reducing pain and promoting mental clarity. Tan IIA has been utilized extensively recently because of its anti-inflammatory, anti-bacterial, anti-tumour and nerve cell-protecting characteristics (Subedi & Gaire, 2021; Zhang et al., 2022). Inhibition of Aβ deposition and protection of neuronal cells are the possible targets of tan IIA in the pathology of AD in existing studies, but the detailed mechanism of action is not clear (Li et al., 2016; Zhang et al., 2018).
The Chemical Structure of the Tanshinone ⅡA.
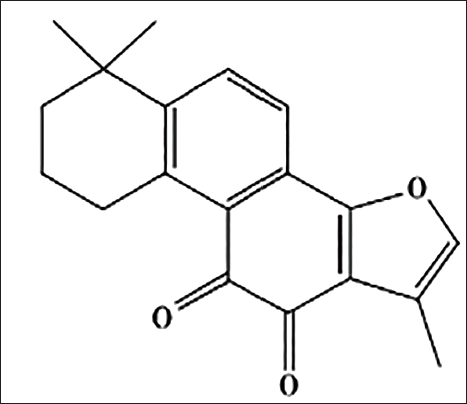
In our study, we explored the effects of tan IIA on learning memory, oxidative stress and inflammatory responses in an AD model.
Materials and Methods
Reagents
Tan IIA (purity level: >98%) from Hongsen Biotechnology Co., Ltd. (Xi’an, China). GL Biochemistry Ltd. (Shanghai, China) provided Aβ1–42. Protein carbonyl, superoxide dismutase (SOD), glutathione (GSH) and malondialdehyde (MDA) assay kits were provided by Nanjing Jiancheng Bioengineering Research Institute (Nanjing, China). DAB chromogenic and terminal deoxynucleotidyl transferase dUTP nick end labelling (TUNEL) staining kits were acquired from Zhongshan Biotechnology Co., (Beijing China) and Roche Molecular Biochemicals (Germany). The secondary antibodies, anti-tumour necrosis factor-alpha (TNF-α), anti-interleukin 1 beta (IL-1β), anti-inducible nitric oxide synthase (iNOS) and anti-8-hydroxy-2′-deoxyguanosine (8-OHdG) utilized in this work were acquired from Beijing Bioss Co., Ltd. (Beijing, China).
Preparation of Aggregated Aβ1–42
In short (Stine et al., 2003), 1,1,1,3,3,3-hexafluoro-2-propanol (HFLP) that had been pre-cooled was mixed with Aβ1–42 at a final concentration of 1 mM. The solution is then allowed to stand and evaporate at room temperature in a fume hood. At −80°C, the peptide films were kept in storage. Peptides were resuspended in dry dimethyl sulfoxide to 5 mM for oligomer formation. The concentration was diluted to 2.5 µg/µL (pH 7.4) in phosphate-buffered saline (PBS) and incubated at 4°C for 24 hours. Lastly, for future usage, the soluble Aβ1–42 oligomers were kept at −80°C.
Animal Treatment
Seventy-two male Sprague-Dawley rats, weighing between 200 and 220 g, were provided by Vital River Laboratory Animal Technology Co., Ltd. (Licence No. SCXK (Jing) 2021-0006). Freedom to eat and drink. All rats were randomly divided into sham (control) group, model group, huperzine A group (0.2 mg/kg) and L, M, H (25, 50, 100 mg/kg) dose groups of tan IIA (n = 12).
AD Model Establishment by Injection of Aβ1–42
Bilateral intraperitoneal doses of 2% sodium pentobarbital (0.35 mL/100 g) were used to anaesthetize the rats, and the rats’ heads were fixated using stereotaxic equipment. After the skin was sterilized, the exposed skulls of cut rats were treated with 5% H2O2. Creating a 26-gauge needle attached to a microinjector in the rat stereotaxic coordinate system in the hippocampal CA1 region of the skull, according to the atlas of the rat brain (Kjonigsen et al., 2015). To construct the model, 5 µL of Aβ1–42 (2.5 µg/µL) was injected in the rat hippocampal CA1 area, and the sham group was replaced with 0.01M PBS. After injection, the needle was kept in that position for more than 15 minutes and then slowly withdrawn. After that, an intramuscular injection of penicillin (80,000 U) was taken to prevent infection. The rats in each group were given intragastric administration (1 mL/100 g) on the second day after operation and were administered for 15 days, once a day.
Morris Water Maze
Positional navigation tests were performed on days 11–14 and spatial probe tests on days 15 after the Aβ1–42 injection (Estévez-Silva et al., 2022; Samman et al., 2023). During training, rats were placed with their backs to the centre point in one of four quadrants, which changed from day to day. To test the rats to find from the entry point and climb up the table the time required for the escape latency (EL). The training was stopped after the platform was found by a rat within 90 seconds, and the time was recorded as EL. Instead, if the platform was not found, an EL of 90 seconds was recorded, but it was necessary to guide it to reach the platform and hold it for 20 seconds. Each rat was subjected to three consecutive trials from one to four days. The probe test was conducted approximately 24 hours after the position navigation test. After removing the platform, each rat was given 60 seconds to swim through the maze in search of it, using the same entry point to enter the water. The probe latency and search frequency were recorded and evaluated.
Passive Avoidance Test
The passive avoidance apparatus (shuttle box) consists of a light box and a dark box (RWD Lifescience Co.). On postoperative day 13, rats were subjected to a passive avoidance test. On the first day, the rat was positioned in the illuminated enclosure with its back to the guillotine door, so that the rats are free to explore the device and have free access to the darkroom. The rats were put inside the bright compartment during the 14 days of training when the door was opened. When the rat enters the darkroom, the door is closed and a mild electric shock (0.8 mA for 3 seconds) is administered. The only way for the rats to escape from electric shock is to return to the safe illuminated compartment. After 24 hours of training, the latency of their response to entering the dark room within 5 minutes was recorded when the rats were placed in a luminous area. In contrast, the recording latency was 300 seconds.
Brain Sample Preparation
After the behavioural experiment, eight rats (each group) were decapitated. The brain tissue was quickly dissected and removed from the skull and the hippocampal tissue was stripped from the brain tissue. After rinsing each tissue in pre-cooled saline to eliminate blood, then refrigerate at −80°C. To achieve deep anaesthesia, sodium pentobarbital was administered to the remaining animals in each group. Intracardiac perfusion was then performed with 0.1M PBS and 4% paraformaldehyde (pH 7.4). Brain tissues were removed from the 4% paraformaldehyde fresh configuration. After that, they were equilibrated in a gradient concentration of sucrose in 0.1M PBS, respectively, at 4°C. Then it is cut the needle before and after coronary surgery in the paraffin-embedded area of the wax block. The prepared paraffin blocks were placed on the slice of coronal slice of 5 µm, which were cut and used to investigate the morphological changes of the neurons in the brain and immunohistochemistry.
Measurement of SOD, GSH, MDA and Protein Carbonyl Activities
In accordance with the assay kit requirements, the activities of SOD, GSH, MDA and protein carbonyls in hippocampus tissue were determined.
Nissl Staining for Cell Morphology in the Rat Hippocampus
Cut paraffin sections are dewaxed and rehydrated in a series of decreasing levels of ethanol (100%, 95% and 70%). They were then combined with 1% toluidine blue at 55°C for 30 minutes and then rinsed. They were treated with ethanol (70%, 95% and 100%) and xylene, covered with coverslips and observed (Du et al., 2020; Zhao et al., 2019).
TUNEL Assay
The TUNEL assay (Roche Molecular Biochemicals, Germany) was used to measure the quantity of DNA breaks (Sharma et al., 2016; Takeda et al., 2015). In brief, the paraffin sections of hippocampal tissues were fully dewaxed with xylene, placed in anhydrous ethanol, 90% ethanol and 70% ethanol, respectively, and reacted with prepared Tris/HCl solution (pH 7.4–8.0) for 15–30 minutes at room temperature, then washed well with PBS to remove the proteinase K, and then treated with methanol containing 3% H2O2 for 10 minutes. Finally, the samples were incubated in a buffer containing fluorescein-dUTP and TdT at 37°C for 60 minutes, and then washed with PBS for observation.
Immunohistochemical Staining for 8-OHdG, IL-1β, TNF-α and iNOS Expression
After removing paraffin by rapidly submerging the tissue sections in xylene, the sections were hydrated using ethanol solutions of different concentrations. Tissues are incubated in boiling Tris-EDTA buffer solution (pH 9.0) for 5 minutes followed by 3% hydrogen peroxide for 15 minutes. Incubate with primary and secondary antibodies for 20 minutes at 37°C with serum prior to binding and then incubate with the markers for 20 minutes. Following three PBS rinses, the sample was reacted with DAB solution for 10 minutes. In the end, the sections were dehydrated, coverslips were blocked and observed under the microscope. The staining intensity of the proteins was analysed using Image ProPlus 6.0 software.
Statistical Analysis
Statistical analyses were performed using the software SPSS 19.0. The mean ± standard deviation was used to express the data. The results between groups were checked using one-factor ANOVA together with Bonferroni’s post hoc test, and two-factor ANOVA with repeated measures and Bonferroni’s multiple comparison test. Statistical significance was indicated when p < 0.05.
Results
Effects of Tan ⅡA on Learning and Memory on the Morris Water Maze (MWM) Test
Rats in each group were examined using the MWM experiment to assess their learning memory capacity, taking three times a day of training as the average value of the day’s training results. All groups were trained for four consecutive days, we found that the EL of rats in each group showed a decreasing trend with time (Figure 2a). The EL of rats in the model group was significantly prolonged from day 2 of training compared to the sham group (p < 0.01), which was significantly improved by treatment with tan IIA or huperzine A (p < 0.05).
Effects of Tan ⅡA on Escape Latency of Morris Water Maze in Aβ1–42-induced Rats. (a) Average Escape Latency of Each Group onto a Hidden Platform. Three Training Trials were Administered Each Day for Four Continuous Days. (b) The Probe Trial Test. The Escape Latency was Defined as the First Time When a Rat Crosses the Former Location of the Removed Platform. (c) Search Frequency is Defined as the Number of Times the Rat Crosses the Target Area. Data Are Expressed as Mean ± SD (*p < 0.05, **p < 0.01 Versus the AD Model Group; #p < 0.05, ##p < 0.01 Versus Sham Group).
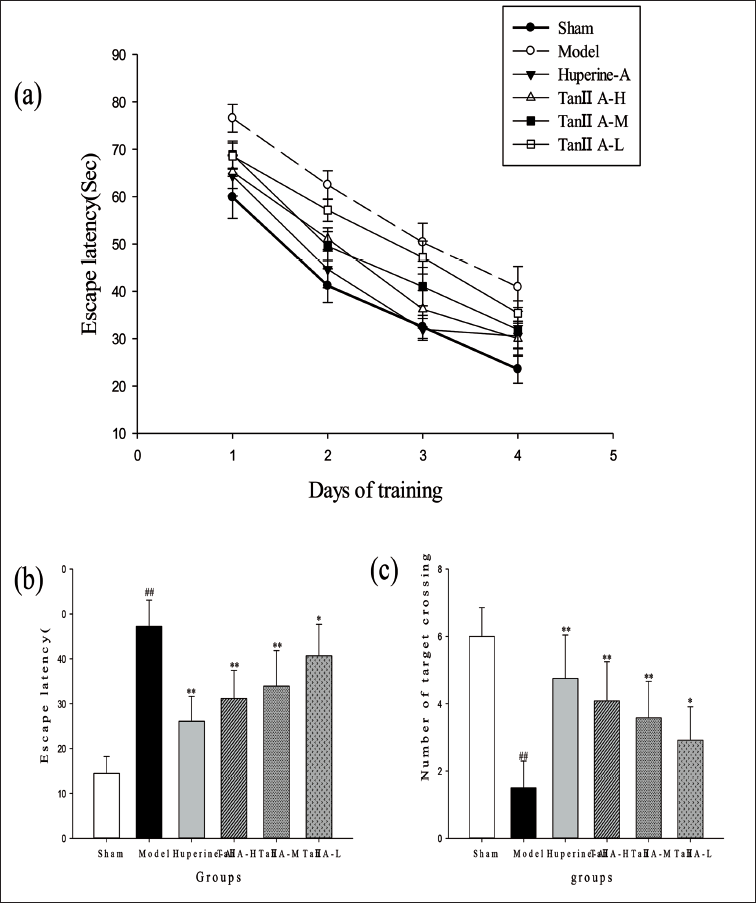
In the spatial probe test, Aβ1–42-injected rats were less able to recognise the platform and had a significantly longer EL than PBS-injected rats, as seen in Figure 2b. Treatment with tan ⅡA-M, tan ⅡA-H and huperzine A group (0.2 mg/kg) resulted in considerably shorter escape latencies (p < 0.01, respectively) compared to the model group. The AD model rats’ number of targets crossing the platform was considerably less than the sham group (p < 0.01) (Figure 2c). In comparison, tan ⅡA-M, tan ⅡA-H and huperzine A treatment significantly alleviated the deficit (p < 0.01), and tan ⅡA-L group is the same (p < 0.05). These findings imply that tan IIA can alleviate learning disabilities in AD rats.
Effects of Tan ⅡA on the Passive Avoidance Test
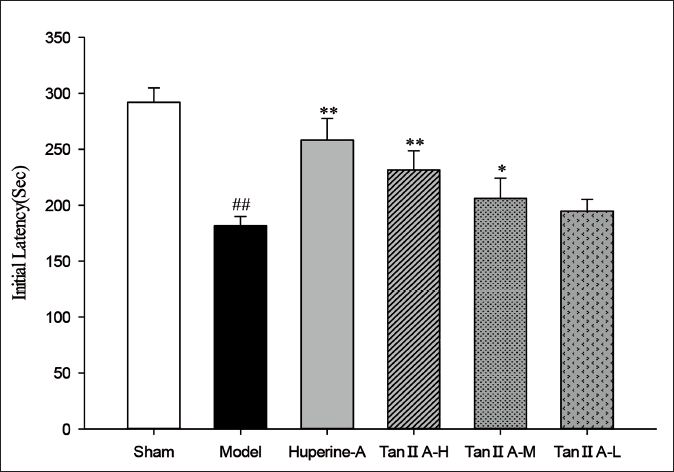
The results showed that the model group had the shortest response latency in the darkroom and was statistically different (p < 0.01) compared to the sham group (Figure 3). The tan ⅡA-H treated rats and huperzine A-treated rats showed significantly increased step-through latency (p < 0.01), tan ⅡA-M group is the same (p < 0.05).
Effects of Tan ⅡA on Initial Latency in Passive Avoidance Test. The Initial Latency of the Rats Entering the Dark Compartment was Recorded 24 Hours After the Training Session (*p < 0.05, **p < 0.01 Versus the AD Model Group; # p < 0.05, ## p < 0.01 Versus Sham Group).
Tans IIA Suppresses Oxidative Stress in AD Model Rats
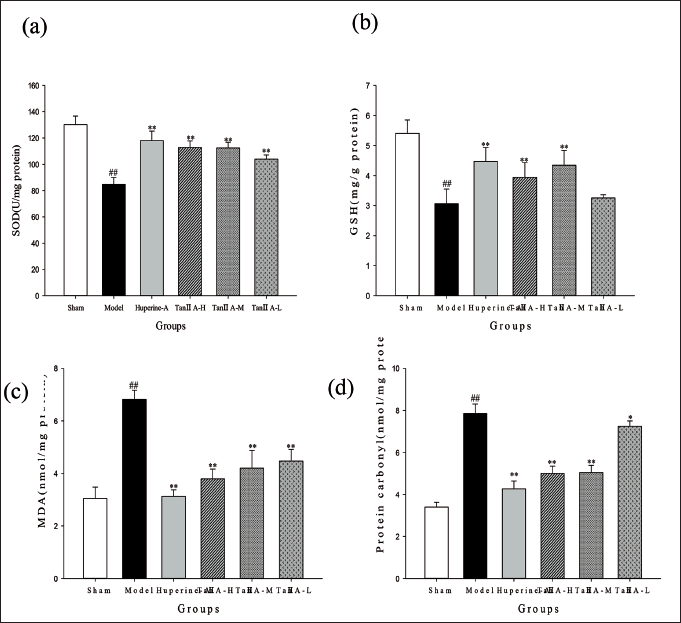
The oxidative stress indications in each group were displayed in Figure 4, together with the changes in SOD, MDA, GSH and protein carbonyl activity in the hippocampal tissue. The rats in the model group showed a decrease in SOD and GSH activities and an increase in the levels of MDA and protein carbonyls, both of which were significantly different from those in the sham group (p < 0.01). In contrast, the tan IIA and huperzine A groups significantly ameliorated these effects (p < 0.05 and p < 0.01).
Effect of Tan ⅡA on the Activities of (a) SOD, (b) GSH and (c) MDA, (d) Protein Carbonyl Leaves in the Hippocampus (*p < 0.05, **p < 0.01 Versus the AD Model Group; #p < 0.05, ##p < 0.01 Versus Sham Group).
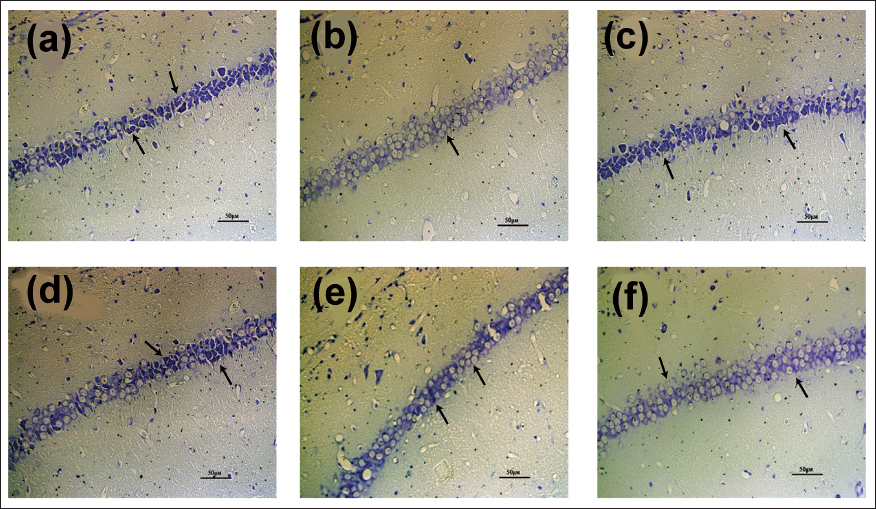
Changes in Nissl Bodies in the Hippocampus Tissues
The quantity of Nissl bodies, a distinctive feature of neurons, indicates the morphological change and neuronal status (Figure 5). Almost undetectable damaged neurons in the sham group. Light microscopy showed that clear staining of the nidus and structure of the hippocampus with regularly arranged nerve cells. Conversely, a sizable fraction of the neurons in the AD model group were injured, displaying a wide range of degenerative alterations, such as diminished or absent cytoplasmic Nissl bodies, poorly aligned cells, shrunken cytoplasma, oval or triangular nuclei, and disordered hippocampal structures. However, tan ⅡA treatment can reverse the damage to a certain extent.
Effect of Tan ⅡA on Nissl Bodies in the Hippocampus Tissues. Nissl Staining in the Hippocampus for Each Group. In Physiological Conditions, the Nissl Bodies were Big and Abundant, Showing that the Function of Neuronal Protein Synthesis was Strong; on the Other Hand, when Nerve Cells were Damaged, the Number of Nissl Bodies Would be Reduced or Even Disappear. Representative Photomicrographs of Nissl Bodies in the Hippocampus (400× magnification; n = 4 in each group). (a) Sham (Control) Group, (b) AD Model Group, (c) Huperine A Group, (d) Tan ⅡA-H Group, (e) Tan ⅡA-M Group, (f) Tan ⅡA-L Group.
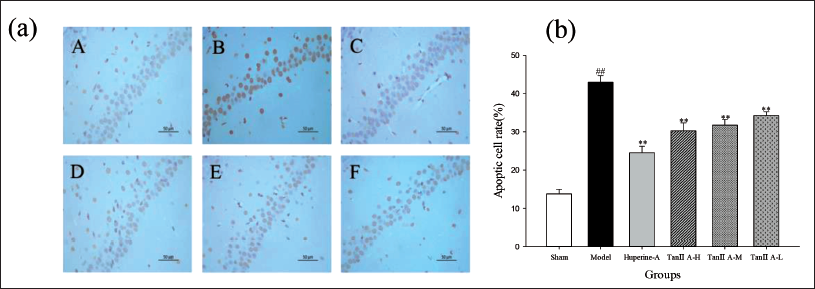
Tan IIA Ameliorated Neuronal Apoptosis
In the hippocampal tissues, the TUNEL assay demonstrated neuronal death (Figure 6). These results showed that apoptosis in the hippocampal tissue, which was originally higher in the model group, was improved and significantly reduced after treatment with tan IIA and huperzine A (p < 0.01), suggesting that apoptosis can be inhibited by tan IIA.
Effect of Tan ⅡA on the TUNEL Staining in the Hippocampus of Aβ1–42-treated Rats. (a) Representative Photomicrographs of TUNEL-positive Cells (magnified ×400). (A) Sham (Control) Group, (B) AD Model Group, (C) Huperine A Group, (D) Tan ⅡA-H Group, (E) Tan ⅡA-M Group, (F) Tan ⅡA-L Group. (b) Quantification of TUNEL-positive Cells in Each Group. Data Are the Mean ± SD of Each Individual (*p < 0.05, **p < 0.01 Versus the AD Model Group; #p < 0.05, ##p < 0.01 Versus Sham Group).
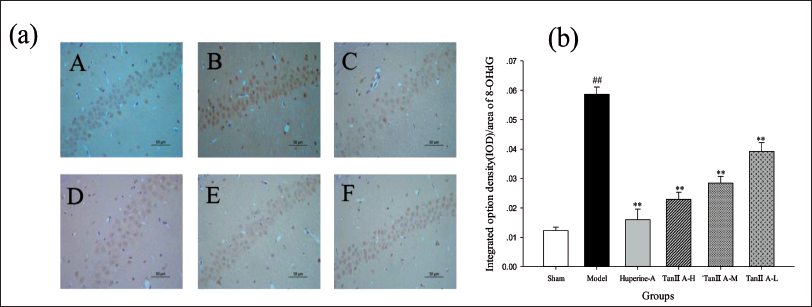
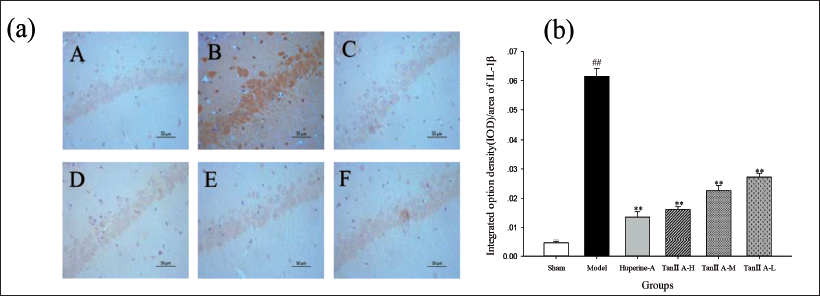
Effects of Tan IIA on the Expression of Related Proteins
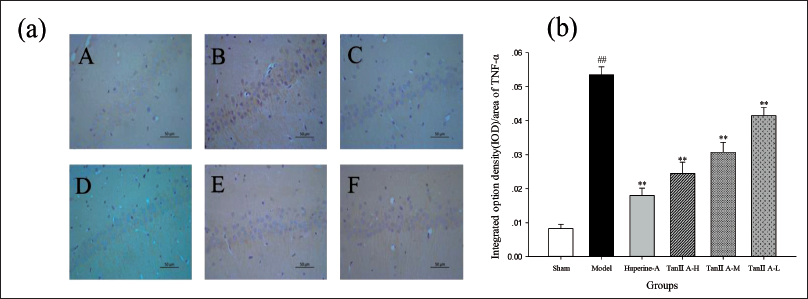
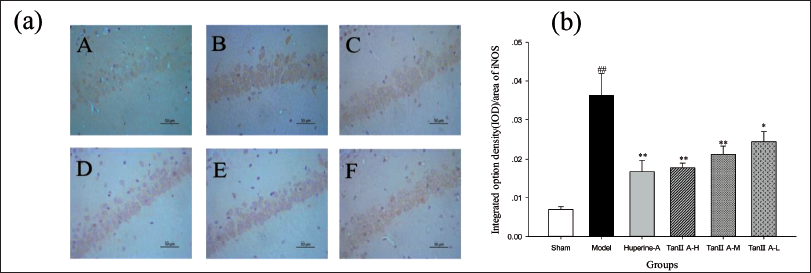
Figures 7–10 displayed the protein expression of 8-OHdG, IL-1β, TNF-α and iNOS. After tan IIA treatment, the high level of expression of all four proteins in the original model group was significantly suppressed (p < 0.01), and the data also indicated a dose-dependent manner.
Tan ⅡA Attenuated 8-OHdG Expression in the Hippocampus of Aβ1–42-treated Rats. (a) Representative Photomicrographs of 8-OHdG Expression (400× magnification). (A) Sham (Control) Group, (B) AD Model Group, (C) Huperine A Group, (D) Tan ⅡA-H Group, (E) Tan ⅡA-M Group, (F) Tan ⅡA-L Group. (b) Quantification of 8-OHdG Labelling in Each Group Employed Integral Optical Density (IOD)/Area. Data Are Expressed as Mean ± SD of Each Individual (*p < 0.05, **p < 0.01 Versus the AD Model Group; #p < 0.05, ##p < 0.01 Versus Sham Group).
Tan ⅡA Attenuated IL-1β Expression in the Hippocampus of Aβ1–42-treated Rats. (a) Representative Photomicrographs of IL-1β Expression (400× Magnification). (A) Sham (Control) Group, (B) AD Model Group, (C) Huperine A Group, (D) Tan ⅡA-H Group, (E) Tan ⅡA-M Group, (F) Tan ⅡA-L Group. (b) Quantification of IL-1β Labelling in Each Group Employed Integral Optical Density (IOD)/Area. Data Are Expressed as Mean ± SD of Each Individual (*p < 0.05, **p < 0.01 Versus the AD Model Group; #p < 0.05, ##p < 0.01 Versus Sham Group).
Tan ⅡA Attenuated TNF-α Expression in the Hippocampus of Aβ1–42-treated Rats. (a) Representative Photomicrographs of TNF-α Expression (400× Magnification). (A) Sham (Control) Group, (B) AD Model Group, (C) Huperine A Group, (D) Tan ⅡA-H Group, (E) Tan ⅡA-M Group, (F) Tan ⅡA-L Group. (b) Quantification of TNF-α Labelling in Each Group Employed Integral Optical Density (IOD)/Area. Data are Expressed as Mean ± SD of Each Individual (*p < 0.05, **p < 0.01 versus the AD Model Group; #p < 0.05, ##p < 0.01 versus Sham Group).
Tan ⅡA Attenuated iNOS Expression in the Hippocampus of Aβ1–42-treated Rats. (a) Representative Photomicrographs of iNOS Expression (400× Magnification). (A) Sham (Control) Group, (B) AD Model Group, (C) Huperine A Group, (D) Tan ⅡA-H Group, (E) Tan ⅡA-M Group, (F) Tan ⅡA-L Group. (b) Quantification of iNOS Labelling in Each Group Employed Integral Optical Density (IOD)/Area. Data are Expressed as Mean ± SD of Each Individual (*p < 0.05, **p < 0.01 versus the AD Model Group; #p < 0.05, ##p < 0.01 versus Sham Group).
Discussion
AD is one of the major causes of dementia, and overproduction of Aβ42, aberrant post-translational modification of tau proteins, and neuronal loss are its main pathologic features. The main clinical manifestations of AD were progressive memory impairment, cognitive dysfunction and personality disorder. Many studies have confirmed that amyloid toxicity and endogenous build-up, as well as disease behaviours associated with memory deficits, can be observed in models constructed by artificial injection of Aβ. Therefore, the injection of soluble Aβ became an effective method for establishing the AD model. In our work, the metrics of the MWM experiment were somewhat improved by tan IIA treatment, with a significant reduction in EL and a significant increase in the number of crossings of the target platform and passive avoidance latency. Our results also suggest that fewer injured nerve cells and loss of neurons than in the AD rats’ hippocampal tissue.
In recent years, oxidative injury has become a hot issue. Oxidative stress occurs when the toxicity produced by ROS exceeds the antioxidant capacity of the cell. Reynolds’ study shows that induction of Aβ causes oxidative stress in cultured human cerebrovascular smooth muscle cells (Reynolds et al., 2016). According to a prior study, the structure of biological biomolecules, such as proteins, may heel change with oxidative stress and lead to cell death (Thapa & Carroll, 2017). Tan IIA can reduce oxidative stress in both in vivo and in vitro experiments by modulating target proteins of different signalling pathways (Shu et al., 2016). In this work, treatment with tan IIA increased the levels of antioxidant enzymes SOD and GSH and decreased the peroxidation levels of MDA and protein carbonyls. GSH is able to increase antioxidant levels by scavenging free radicals from the body, which in turn reduces oxidative stress. 8-OHdG, a marker of DNA oxidative damage, is regulated by SOD, suggesting a close relationship between oxidative stress and DNA damage. The ability of tan IIA to protect neuronal cells and reduce apoptosis has been shown in the current study to work by downregulating the expression levels of p53 and pp53 (Li et al., 2015). The ability of tan IIA to reduce neuronal cell damage was also demonstrated in this study.
In addition to ROS, excessive production of NO may contribute to the genesis of numerous illnesses, such as AD. Macrophages and microglia induce iNOS in response to inflammatory mediators, producing large amounts of NO, which directly or indirectly leads to neuronal necrosis and apoptosis. High expression of iNOS is one of the key factors of AD. In addition, microglia activation will greatly strengthen the inflammatory response to SP, inducing local cell factors of acute phase reaction, activation of the complement cascade and inflammation-related enzymes, and then the expression of iNOS was increased (Gong et al., 2017). Consistent with previous studies, our work illustrates that tan IIA reduces iNOS expression in hippocampal tissue (He et al., 2015; Jiang et al., 2014).
Inflammation cannot be ignored in the pathogenesis of AD. According to recent research, inflammation might be a third crucial element that, once malfunction arises, could accelerate the course of the illness (Onyango et al., 2021). The inflammatory response could be an early factor in AD development and a cause of mild cognitive impairment. The major cytokines involved in the regulation of AD inflammatory response are IL-1β, TNF-α and others. According to Zhang et al. (2013), atorvastatin reduces the amounts of TNF-α, IL-6 and IL-1β produced in the hippocampal regions of rats treated with Aβ1–42 to simulate AD. Consistent with existing studies, the reduction in the levels of the inflammatory factors IL-1β and TNF-α after tan IIA treatment in the present study also showed a dose-dependent pattern.
Conclusion
In conclusion, our work suggests that tan IIA is neuroprotective against oligomeric Aβ1–42-induced neurotoxicity and also contributes to the improvement of learning and memory capacity. In the present study, the modulation of antioxidant indices and inflammatory cytokine levels by tan IIA suggests that its neuroprotective effects are closely related to its inhibitory effects on oxidative stress. Our findings strongly imply that tan ⅡA can enhance learning and memory while alleviating nerve cell damage. Based on the current general context of medicinal plants for the treatment of chronic diseases, tan IIA needs to be further explored for the treatment of AD.
Abbreviations
Footnotes
Acknowledgments
The authors thank all participants and Beijing Union University.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval
All experimental protocols were approved by the Experimental Animal Ethics Committee of the Health Food Function Testing Center, College of Applied Arts and Sciences, Beijing Union University.
Funding
This work was supported by grants from the National Natural Science Foundation of China (11975048); Beijing Natural Science Foundation (6164030); Beijing Key Laboratory of Bioactive Substances and Functional Foods Research Project, the Academic Research Projects of Beijing Union University (ZK80202102); and Education Reform Project of Beijing Union University (JY2023Y008).
