Abstract
Background
Skin moisture management is important for maintaining healthy skin and treating inflammatory skin diseases, and skin barrier function can be assessed by measuring trans-epidermal water loss (TEWL) and skin moisture content.
Aim
This study was designed to investigate the effects of a topical cream containing 17% herbal extract (Cream17) on skin barrier function.
Materials and Methods
In this study, the ability of investigational product (IP) to improve skin barrier function was investigated in 33 subjects using a randomized and assessor-blinded four-week clinical trial. In our study, the effects of IP on TEWL, skin water content, and the degree of itching were investigated.
Results
After four weeks of application, TEWL was significantly reduced by IP (p < 0.001), and skin water content was significantly increased (p < 0.001) compared to non-treated control. In addition, topical application of IP significantly lowered the visual analog scale at the second and fourth weeks after treatment commencement as compared with those in the untreated control group (visit 3 at the second week, p < 0.05; visit 4 at the fourth week, p < 0.001).
Conclusion
These results indicate that Cream17 can improve the skin barrier function, which is important for managing dry skin conditions such as atopic dermatitis.
Introduction
The skin barrier, commonly known as the stratum corneum, plays an important role in maintaining skin moisture and protecting the body from exogenous chemical, physical, and biological stimuli. Therefore, a normal skin barrier function is the most basic and essential requirement for maintaining healthy, beautiful skin. On the other hand, damage to the stratum corneum causes water loss, flare, irritation, and hypersensitivity, and these phenomena are associated with skin barrier function impairment, which is a causative factor and major symptom of atopic dermatitis (AD) and other chronic skin diseases (David et al., 2017).
Trans-epidermal water loss (TEWL) and skin moisture content are important physiological indicators of skin barrier damage and recovery. TEWL provides an important auxiliary, objective means of evaluating skin barrier function and the healing of atopic and dry skin because when the function of the stratum corneum deteriorates, TEWL increases, and skin moisture content reduces (Verdier-Sévrain & Bonté, 2007).
Dry skin is both a cause and a symptom of AD. Dry skin refers to a skin condition that causes discomfort due to dryness and clinically describes a skin condition with slight erythema, fissures, scales, and a rough surface. Furthermore, it is important to use an appropriate moisturizer and avoid excessive washing when the skin is dry because washing removes natural moisturizing factors that protect the skin from harmful environmental stimuli (Abels & Angelova-Fischer, 2018).
Itching is a distress factor for those suffering from diseases such as AD and psoriasis and a major exacerbating factor in various skin diseases, including inflammatory skin diseases. In addition, dry skin conditions can cause itching, and patients with dermatological problems accompanied by itching often complain of dry skin, and thus, tend to use moisturizers (Moniaga et al., 2020).
Herbal extracts have been used for centuries in traditional medicine and skincare. Many of these extracts contain bioactive compounds that can offer various benefits for the skin, including improving skin inflammation status and enhancing the function of the skin barrier. The herbal extract used in this study (CeBiNES) is composed of natural ingredients that moisturize and provide nutrition to problematic skin areas (Table S1, Supplementary Data 1). The active ingredients of herbal extract are those extracted from various natural products. Among the active ingredients of herbal extract, extracts of Quercus acutissima Carr. bark (a major component of herbal extract) (Lee et al., 2011), Ulmus davidiana var. japonica Nakai. root bark (Lyu et al., 2013), and Houttuynia cordata Thunb. Doi et al. (2014) were demonstrated to have anti-dermatitis effects in mice with contact dermatitis or AD. In addition, Ecklonia cava Kjell. extract inhibited the release of inflammatory factors from HaCaT keratinocytes induced by exposure to airborne particulate matter (Ha et al., 2019). Centella asiatica (L.) Urb. Extract is known for its wound healing, skin inflammation suppressing, and moisturizing effects, as a known therapeutic agent (Bylka et al., 2014), and sesame oil can restore damaged skin barrier functions (Lin et al., 2017).
Based on the above, this study was designed to investigate the effect of topical cream containing 17% of the herbal extract (Cream17) on skin barrier function and itching. For this purpose, we investigated the effect of Cream17 on TEWL, skin moisture, itching, and visual findings of skin symptoms through a randomized four-week trial. Before this trial, the herbal extract was confirmed to be suitable for skin use (non-comedogenic) based on the results of clinical trials conducted before the present study (Table S2, Supplementary Data 2). In the four-week trial conducted, no specific adverse skin reactions were observed in 20 subjects administered topical herbal extract 12 times on alternate days.
Materials and Methods
This study was designed following the principles of the Declaration of Helsinki and the guidelines published by the Ministry of Food and Drug Safety, Republic of Korea (Guideline-1123-01) (Ministry of Food and Drug Safety, 2021). The methods and results of the study were described according to CONSORT recommendations (Gagnier et al., 2020).
Study Design
The randomized 4 trial was conducted from September to October 2021 at Cutis Biomedical Research Center (Seoul, Republic of Korea) on September 27, 2021 (CTS-IRB-21901-15, date of final protocol approval September 27, 2021).
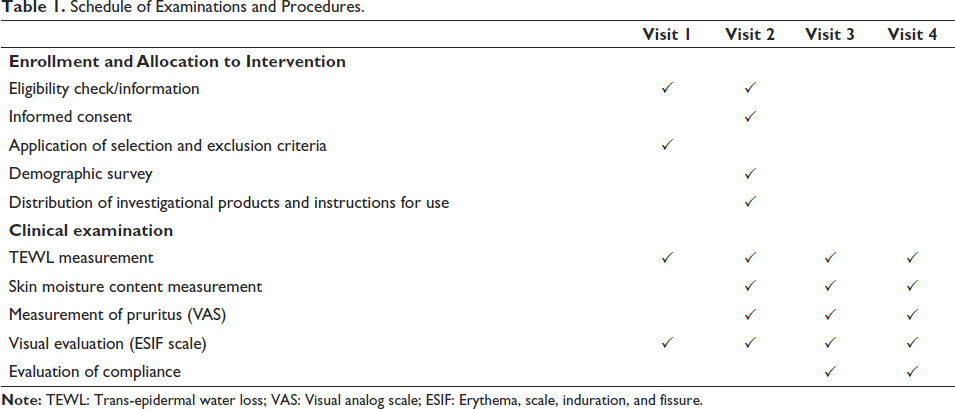
Baseline measurements were taken on the second visit day for subjects selected at the first visit, and measurements were obtained after two weeks of application and again at the end of the study (four weeks later). To evaluate the efficacy of the investigational product (IP), non-treated sites were used as controls. An overview of the study is presented in Table 1.
Schedule of Examinations and Procedures.
Subjects
The number of test subjects was determined based on the effectiveness evaluation guidelines for functional cosmetics and the guidelines for cosmetic labeling and advertisement verification test methods (more than 30 people) (Ministry of Food and Drug Safety, 2021).
Inclusion criteria for this clinical study encompassed individuals with a history of pruritus or xerosis and a TEWL value equal to or greater than 12 g/m2/h at designated test sites. Exclusion criteria included individuals with skin conditions necessitating medical treatment, a score of 6 or higher on the erythema, scale, induration, and fissure (ESIF) scale, or recent use of antibiotics, steroids, immune-suppressants, anti-histamines, retinoids, or phototherapy for a skin condition within the four weeks preceding the commencement of the study. Additionally, participants with physical or chemical damage to the skin barrier (e.g., skin peeling) were also ineligible.
Intervention and Compliance Evaluation
The Research Institute of Natural Extracts for Healthy Skin suggested a topical cream (Cream17). The IP was applied topically twice daily for four weeks. After cleaning test sites, residual water was removed, and an appropriate amount (at least 2 fingertip units, 1 g of cream per site) was applied. The two test sites were within the cubital fossa (below the epicondylar line) or the popliteal fossa. Medication compliance was assessed by measuring the amount of cream remaining for each subject from the first to the last day of the study, and this was further verified by the study monitor upon completion of the treatment.
Efficacy and Safety Outcomes
The primary endpoint concerning efficacy in improving skin barrier function was significant changes in TEWL, skin moisture, and the degree of itching using a 10-point visual analog scale (VAS) compared to controls.
Safety and tolerability were assessed by monitoring the incidence and duration of adverse events. In case of any adverse reactions occurring during the period of product usage after receiving the IP, participants were instructed to promptly report them to the responsible study personnel. In addition, the severity of skin lesions by visual evaluation was also investigated using the ESIF scale.
Measuring Conditions
After wiping test sites with detergent, all subjects stayed for 30 minutes in a constant temperature and humidity environment (23°C ± 1°C and 50% ± 5% relative humidity). During this period, water intake and leaving the room were prohibited. One researcher measured TEWL and skin moisture content in the same area in the same way to ensure consistency of results.
Measurements of TEWL and Skin Moisture Content
TEWL was measured using a Tewameter TM300 (Courage+Khazaka electronic GmbH, Germany), and skin moisture contents were measured using a corneometer CM825 (Courage+Khazaka electronic GmbH, Germany). Average measurements taken at three different sites within each skin area were subjected to analysis.
Measurement of Pruritus
The itching degree over the previous 24 hours was self-assessed using a VAS. Degrees were represented by points on a 10 cm scale, on which a rating of 10 represented the most severe itch imaginable.
Visual Evaluation by Experts
A dermatologist evaluated degrees of ESIF scale at each test site using a 4-point scale (0, none; 1, mild; 2, moderate; 3, severe). Lesion severity was evaluated using a 0–12-point scale by summing scores using a modified version of the method described by Kumar et al. (2004). Evaluation criteria are shown in Supplementary Data (Table S3, Supplementary Data 3).
Statistical Analysis
Statistical analysis was performed using IBM SPSS for Windows (Version 27; Armonk, NY, USA). Data are presented as means ± standard deviations. The D’Agostino and Pearson omnibus normality test was used to determine the normality of the data distribution. Experimental and control group values were compared using Friedman’s multiple comparisons test. The independent t-test and Mann–Whitney U test were used to assess the homogeneity of normally distributed and non-normally distributed groups (baseline values). p values < 0.05 were considered significant.
Results
Demographic Information
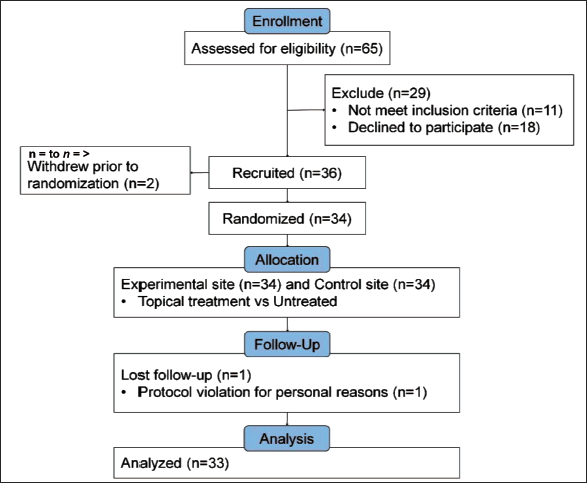
After applying the inclusion and exclusion criteria, 36 women aged 19–69 years were enrolled in this study. Among them, the participants who completed the trial totaled 33 individuals, with an average age of 49.5 ± 5.9 years. The age distribution of participants was as follows: 16 individuals aged 40–49 (48.485%), 16 individuals aged 50–59 (48.485%), and 1 individual aged 60–69 (3.030%) (Supplementary Data 6).
Dropouts
A total of three volunteers were dropped out during the entire trial period, two before randomization and one at the fourth-week visit. The reasons for these losses were withdrawal of consent (two cases) and non-compliance with the schedule (one case). The overall progress of the clinical study is summarized in Figure 1.
Flow Diagram of the Study.
Baseline Characteristics
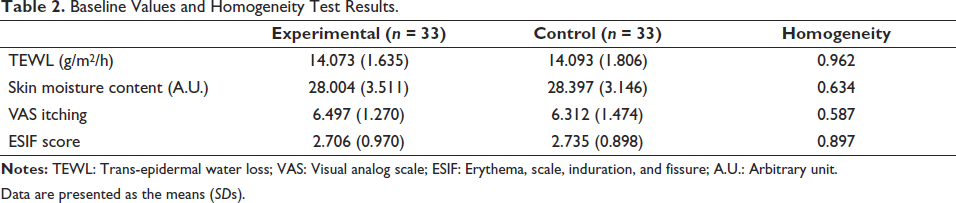
Baseline values were measured at second visits (zero week), and the homogeneity values for TEWL, Skin moisture content, VAS itching, and ESIF score were 0.837, 0.634, 0.587, and 0.897, respectively (Table 2).
Baseline Values and Homogeneity Test Results.
Efficacy
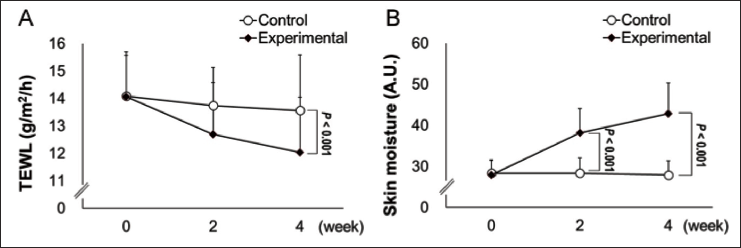
TEWL values after four weeks of IP administration in the sample group (12.04 ± 2.00 g/m2/h) were significantly lower than those in the control (untreated) group (13.57 ± 2.48 g/m2/h). Two weeks of topical application did not significantly affect TEWL values (Figure 2A). In addition, after two to four weeks of topical treatment, the skin moisture content in the experimental group was 38.19 ± 5.89 and 42.91 ± 7.48, respectively, significantly higher than those in the control group (p < 0.001) (Figure 2B). In addition, the VAS scores of the experimental group were 4.30 ± 1.82 and 2.26 ± 1.65 at weeks 2 and 4, which was significantly lower than those of the control group (5.80 ± 1.83 at week 2 and 4.88 ± 2.03 at week 4) (Table 3).
The Results of TEWL Measurement (A) and Skin Moisture Content Measurement (B) Data are Presented as Means + SDs of 33 Participants.
The Results of the VAS Itching Assessment.
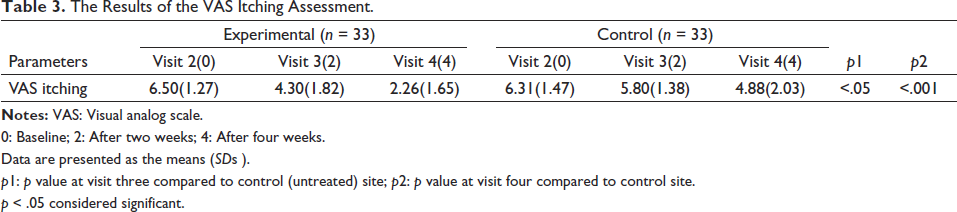
0: Baseline; 2: After two weeks; 4: After four weeks.
Data are presented as the means (SDs).
p1: p value at visit three compared to control (untreated) site; p2: p value at visit four compared to control site.
p < 0.05 considered significant.
Safety
Over four weeks, the IP was applied twice daily to the predetermined site, with approximately 1 g of IP used per application, resulting in a total application of 56 g. Regardless of causality, we monitored adverse reactions in all participants from the outset of the study. Throughout the study period, no adverse reactions, such as stinging, burning, tightness, or prickling, were observed in any of the participants. As shown in Table 4, the ESIF score of the experimental group was significantly lowered by two or four weeks of treatment (p < 0.001) (Table 4).
The Results of the ESIF Score Evaluation.

0: Baseline; 2: After two weeks; 4: After four weeks.
Data are presented as the means (SDs).
p1: p value at visit three compared to control (untreated) site; p2: p value at visit four compared to control site.
p < 0.05 considered significant.
Discussion
The results of this study showed that applying Cream17 for four weeks significantly reduced TEWL, increased skin water content, and significantly reduced VAS compared to the non-treated control. Additionally, no side effects were reported, and ESIF results decreased to a significant level. These results indicate that Cream17 can improve the skin barrier function.
Skin barrier function and immunological abnormalities are the major causative factors of AD, which are commonly encountered in clinical practice. Furthermore, the skin of patients with AD is usually dry and rough due to reductions in epidermal moisture and lipid levels (Rahman et al., 2011). Characteristically, atopic skin also has a relatively high pH compared to normal skin (Seidenari & Giusti, 1995). and contains lower levels of free amino acids, which moisturize the stratum corneum, than normal skin. Furthermore, the ability to biosynthesize ceramides is reduced (Rahman et al., 2011). For these reasons, the management of patients with AD requires that focus be placed on the restoration of skin barrier function and the suppression of skin inflammation.
Our TEWL and skin moisture content measurements showed that topical application of IP (Cream17) for four weeks decreased TEWL values and increased skin moisture contents significantly based on intergroup comparisons (Figure 2). We believe the components of IP primarily responsible for its moisturizing effect are H. cordata and C. asiatica extracts and sesame oil. H. cordata extract can upregulate filaggrin expression on human keratinocytes (Doi et al., 2014), and sesame oil restores skin barrier function (Lin et al., 2017). C. asiatica has been reported to improve skin barrier function among batik workers (Ratz-Łyko et al., 2016) and to have a moisturizing effect in cosmetic formulations (Anggraeni et al., 2021). The moisturizing effects of the ingredients in Cream17 are believed to work individually or in combination to ultimately enhance skin barrier function.
In our results, Unlike the effect on skin moisture, IP application for two weeks did not improve TEWL to a significant level. This appears to be partly because the variation in TEWL values is greater than the skin moisture content. Another possibility is that TEWL recovery may take more time. A simple moisturizing effect can increase skin moisture content, but reducing TEWL levels requires time for the damaged skin barrier function to recover.
Itching is a common symptom of dry skin and AD and an exacerbating factor for various inflammatory skin conditions. Doctors managing these conditions place great importance on controlling itching and often use adjuvant treatments such as bandaging or gloves. In the present study, mean VAS scores differed significantly at two and four weeks after commencing administration in the experimental and control groups (Table 3). On the other hand, VAS scores decreased by ~34% from 6.497 at baseline to 4.303 after two weeks of IP treatment and to 2.255 after four weeks, a decrease of 65%, whereas no significant difference was observed between VAS scores (baseline, two-week, and four-week) in the control group. Q. acutissima bark has also been reported to inhibit mast cell degranulation and the release of allergic mediators from basophils (Lorenz et al., 2016), extracts of H. cordata and E. cava, and dieckol isolated from E. cava inhibited anaphylaxis and allergic responses (Ahn et al., 2015; Han et al., 2009, 2020; Sun et al., 2022). In addition, C. asiatica has been reported to have anti-pruritic effects (George et al., 2009; Klövekorn et al., 2007). In addition, CeBiNES was also found to suppress phorbol 12-myristate 13-acetate and A23187-induced degranulation in RBL-2H3 cells and inhibit the productions of interferon-γ and interleukin-8 in tumor necrosis factor-α stimulated HaCaT cells (Figures S1–S3, Supplementary Data 4). The anti-itching effect of IP observed in this study appears to be due to the anti-inflammatory or anti-allergic effect and moisturizing effect of CeBiNES or its ingredients.
Unlike healthy skin that remains slightly acidic (pH 5.4–5.9), the skin of individuals with AD or dry skin tends to have a relatively high pH (Seidenari & Giusti, 1995). The pH of the skin is influenced by numerous intrinsic factors, including skin moisture, sweat, sebum, anatomical location, genetic factors, and age. Additionally, extrinsic factors such as detergents, cosmetics usage, occlusive dressings, and topical antibiotics can also impact the pH of the skin (Schmid-Wendtner & Korting, 2006). In addition, the low pH value in the extracellular space plays a crucial role in enzyme activity regulation, particularly in epidermal keratinization and barrier regeneration (Schmid-Wendtner & Korting, 2006). Cream17 has a pH of 4.0 and can temporarily adjust the skin pH of patients suffering from inflammatory skin diseases or dry skin.
Skin infection is a major exacerbator factor of inflammatory skin diseases. In patients with AD, bacterial, viral, and fungal infections occur at higher frequencies than in normal individuals due to immune response imbalance caused by, for example, T-cell dysfunction (David et al., 2017). For example, Staphylococcus aureus colonies are found on the skins of only around 5% of healthy individuals but on the skins of more than 90% of AD patients (Geoghegan et al., 2018). For this reason, agents with antibacterial and antifungal effects are useful management tools for treating inflammatory skin diseases.
Our research team confirmed that CeBiNES inhibits the growth of Cutibacterium acnes, a dominant flora in sebaceous follicles, using an agar-well diffusion assay (Table S4, Supplementary Data 5). In addition, CeBiNES contains ingredients with anti-fungal properties such as H. cordata (Liu et al., 2021) and propolis extracts (Forma & Bryś, 2021). Taking these factors into account, Cream17 is expected to be beneficial for individuals with inflammatory skin conditions due to its antimicrobial and antifungal effects.
During the trial period, adverse reactions were monitored in the form of self-reports, questionnaires, and measurements. No subjects reported adverse effects such as itching, pain, burning, or stiffness and did not use escape medication. In addition, no aggravating factors were observed in the visual evaluation by a dermatologist (ESIF score). These results mean that no special side effects were observed within the observable range of this study.
In this study, applicants with an ESIF score of 6 or higher were excluded to eliminate individuals with moderate to severe skin diseases that require medical treatment. In addition, during the four weeks of this trial, the dermatologist evaluated safety using the ESIF score. In our results, the ESIF scores of IP topical applications were significantly lower than those of the non-treated control (Table 4). This means more than just the fact that no special adverse reactions occurred on the skin surface. The improvement in skin surface symptoms, as evidenced by a significant decrease in the ESIF score, appears to be attributed to the moisturizing, skin barrier function recovery, and itching-suppressing effects of Cream17.
In this study, it was not possible to establish an appropriate placebo group due to the nature of IP, a complex of various natural extracts. Due to the unique odor of natural extracts, it is difficult to produce samples with similar odors that subjects cannot identify. For this reason, this study could not use a double-blind technique. Not using a placebo group may affect the results of the questionnaire. Therefore, in this study, the VAS score is less reliable than the measurement result, TEWL value, or skin moisture content. In addition, this study has limitations in terms of national and genetic diversity because it targeted women aged 19–69 years in Korea.
Another limitation of this study is the difficulty in presenting a clear therapeutic mechanism. To overcome this, additional research using cells and animals will need to be conducted. Now we are researching the anti-inflammatory and anti-allergic effect of CeBiNES using RAW 264.7 cells and HaCaT cells. These studies will improve confidence in the therapeutic effect of Cream17, which contains natural products as its main ingredient.
Conclusion
Topical application of IP (Cream17) decreased TEWL, increased skin moisture contents, and suppressed itching. These results suggest IP can be used to manage dry skin and inflammatory skin diseases by improving skin barrier function.
Abbreviations
A.U.: Arbitrary unit; AD: Atopic dermatitis; Cream17: Cream containing 17% herbal extract; ESIF: Erythema, scale, induration, and fissure; IP: Investigational product; SD: Standard deviation; TEWL: Trans-epidermal water loss; VAS: Visual analog scale.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest concerning the research, authorship, and/or publication of this article.
Ethical Approval
The study was conducted in accordance with the Declaration of Helsinki, and approved by the Institutional Ethics Committee of Cutis Biomedical Research Center (CTS-IRB-21901-15, date of final protocol approval 27 September 2021).
Funding
This research was supported by a grant of the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health & Welfare, Republic of Korea (grant number: HF21C0089) and the National Research Foundation of Korea grant funded by the Korean government (MSIP; Grant no. 2022R1A2C2091960).
Informed Consent Statement
No protocol changes were made after the trial was initiated. Written information about the nature of the trial was provided to all subjects, and all signed a consent form.
Supplementary Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
