Abstract
Background
Aluminum chloride (AlCl3) is an environmental neurotoxin that affects cerebral functions and causes health complications. However, the role of AlCl3 in arbitrating glia homeostasis and pathophysiology remains obscure.
Objectives
In this study, we analyzed the role of AlCl3 in causing reactive gliosis in the brain of rats and the ability of Shikonin to attenuate reactive gliosis and neuronal inflammation.
Materials and Methods
In our study, animals were divided into five different groups. The status of AlCl3-induced alterations in neurobehavioral status, estimation of acetylcholinesterase (AChE) and Aβ1 peptide in rat brain, oxidative stress (lipid peroxidation [LPO] and nitric oxide [NO]), antioxidant levels (superoxide dismutase [SOD], Catalase [CAT], and glutathione [GSH]), inflammatory marker levels (interleukin-1β [IL-1β], interleukin-6 [IL-6], and tumor necrosis factor-alpha [TNF-α]), and histopathological changes were analyzed and the neuroprotective effect of Shikonin investigated.
Results
Treatment with Shikonin significantly restored the changes in the open field test, Morris water maze test, and Y-maze spontaneous alteration test in Alzheimer’s disease (AD) rats induced by AlCl3. Shikonin treatment significantly reduced levels of AChE and Aβ1 peptide content in AD rat brain tissues induced with AlCl3. Furthermore, Shikonin treatment significantly reduced the levels of oxidative stress markers and restored the levels of endogenous antioxidants in AlCl3-induced AD rats. Levels of inflammatory cytokines were significantly reduced upon Shikonin treatment in AD rats induced with AlCl3. In accordance with the aforementioned benefits, treatment with Shikonin significantly restored the histological aberrations and infiltration of granular cells white blood cells (WBCs) in brain tissues of AD rats induced with AlCl3.
Conclusion
Collectively, our results showed that Shikonin significantly restored altered neurobehavioral changes and antioxidant levels, and reduced oxidative stress and the inflammatory milieu. Additionally, histological abnormalities support its neuroprotective effectiveness under experimentally induced Alzheimer’s disease conditions caused by AlCl3 in rats.
Introduction
Numerous age-related neurodegenerative conditions contribute to brain dysfunction and associated deaths worldwide and possess an array of etiological factors (Hamdan et al., 2022). Alzheimer’s disease (AD) is a major neurodegenerative disease condition worldwide with a significant socioeconomic burden on global society. Similar to other neurodegenerative conditions, AD is also age-associated and shows patterns similar to memory loss due to hippocampus neuronal loss or neuronal dysfunction, cognitive dysfunction, and abnormal motor functions due to deposition of extracellular protein aggregate called amyloid beta fibrils (Aβ fibers in brain tissues; Heilman & Nadeau, 2022; Ju et al., 2022). Aβ fibrils are insoluble, extracellular protein deposits found in the brain and other tissues that induce a variety of disease conditions (Fandrich et al., 2007). Various proteins in human tissues can induce Aβ formation, which include neuronal Aβ fibrils-AD, atrial natriuretic factor-cardiac arrhythmias, serum amyloid A protein—rheumatoid arthritis, islet amyloid polypeptide (insulin)—diabetes mellitus type II, etc., (Ow et al., 2014). However, the effect of Aβ fibrils in brain tissue has been highly investigated in recent decades as it induces progressive neurodegenerative conditions as aging progresses (Bowler et al., 1998). Unlike rapidly progressive dementias (RPDs) that are fatal within a few months to years after its onset (Geschwind et al., 2007), AD is a slowly progressive dementia that leads to inevitable death between 4 and 20 years after diagnosis in patients above 60 years of age (Vermunt et al., 2019). Hence, studies in recent decades are highly focused on understanding the mechanisms of neurodegenerative etiological agents such as AlCl3, and also on finding avenues that can exert significant neuroprotection during fatal neurodegenerative conditions such as AD (Long & Holtzman et al., 2019).
Various etiological agents were reported to induce the development of age-related AD. Among such, AlCl3 has been widely studied in recent years as it was found to be deposited in the cerebral tissues of AD patients (Exley & Clarkson, 2020). Anthropogenic sources of AlCl3 are antacids, medicines, drinking water, cosmetics toothpaste, etc., which increase the risk of human AD development (Alasfar & Isaifan, 2021). Furthermore, AlCl3 was reported to induce neurotoxic niche, brain oxidative stress and neuronal cell death, cholinergic degradation, Aβ deposition, and hippocampal dysfunction in human brain tissues (Miu et al., 2006; Singh et al., 2018). Similarly, Aβ deposition was reported to induce neuronal, synaptic loss, brain atrophy via oxidative stress, neuroinflammation, and also cholinergic dysfunction (Abdel-Salam et al., 2021). Mitigating the above pathological events is widely believed to be a useful avenue in the clinical management of neurodegenerative disease conditions such as AD (Markovic et al., 2022; Yan et al., 2022). In this context, natural plant-based phytochemicals and their derivatives were widely used in the avenues for finding neuroprotective agents against neurodegenerative diseases like AD. In this study, we have used Shikonin (SHK), a natural naphthoquinone pigment found in the root of the plant species Lithospermum erythrorhizon. Reports denote SHK, a naphthoquinone compound derived from the root of the traditional Chinese medicine SHK, has favorable biological activities such as anti-inflammatory, antibacterial, immunomodulatory, anticancer, and wound-healing-promoting pharmacological activities (Song et al., 2023); however, the neuroprotective effect of SHK remains largely unexplored.
Here, we investigated if treatment with SHK had beneficial effects against AlCl3-induced neurodegeneration in experimental rat brain tissues. Our preliminary findings revealed SHK-exerted significant neuroprotection via its ability to restore the changes in behavioral studies such as open field test, Morris water maze test, Y-maze spontaneous alternation test, and antioxidant marker, while reducing levels of AChE and Aβ1 peptide in rat brain, including levels of oxidative stress and inflammatory cytokine levels during histological analysis.
Materials and Methods
Chemicals and their Sources
Aluminum chloride (AlCl3: Cat. No. 237051), SHK (Cat. No. S7576), bovine serum albumin (BSA; Cat. No. A4161), hematoxylin and eosin stain (H&E stain), acetylcholine, donepezil hydrochloride (DON) acetylthiocholine iodide, reduced glutathione, thiobarbituric acid, and 5,5’-dithiobis-(2-nitrobenzoic acid) (DTNB) were obtained from Sigma Chemical Company, St Louis, USA. Various other chemicals and substances used in this study were of analytical grade unless otherwise stated. Coverslips, dibutyl phthalate xylene (DPX) mountant, and microscopic glass slides were purchased from Thermo Fisher Scientific, USA.
Experimental Animals
Male albino Wistar rats (150–180 g) were housed for about one week for acclimatization and then assorted into five various groups with (n = 6/group). Animals were housed under standard laboratory conditions with 25 ± 2ºC, a humidity of 50% ± 5%, and a constant 12:12 h light–dark cycle. Animals were housed in proper cages made of sterile polypropylene cages with food and water ad libitum. The study was approved by the Ethics Committee of Central Hospital Affiliated to Shandong First Medical University (protocol number 2023-096).
Experimental Design
Animals were divided into five different groups with (n = 6) rats per group.
Orally for 28 days (Olowoyeye et al., 2018);
Following the induction of the neurotoxicity schedule, the rats were administered orally with SHK 1% dimethyl sulfoxide (DMSO) in phosphate-buffered saline (PBS) (vehicle of SHK) for 8 weeks. Following, treatments with SHK, rats were euthanized and the brain tissues were immediately excised and the fresh brain tissues were immediately used for biochemical analysis and histological studies. Simultaneously, other sets of rats after treatment with SHK, and also DON were trained for behavioral studies like open field test, Morris water maze test, and Y-maze spontaneous alteration test, for up to two weeks during the SHK treatment schedule and then the behavioral analyses were carried out.
Behavioral Studies
Open Field Test
The open field test was conducted using an automated machine purchased from Commat Ltd., Turkey. The apparatus consists of a plexiglass chamber with size, measuring 40 cm × 40 cm × 35 cm, on which the locomotor activities of experimental animals were monitored and analyzed using the software. Parameters like ambulation frequency, grooming latency, defecation, and the exploratory rearing numbers were analyzed in this open field test after treatment with SHK to AlCl3-induced rats. The experiment was conducted with a standard protocol with slight modifications as reported previously (Tasdemir & Colak, 2021).
Morris Water Maze Test
This test is useful in the evaluation of the learning and memory abilities of control and experimental groups of rats. Briefly, the test was conducted on a stainless-steel platform with a diameter of 150 cm and a height of 60 cm, where water is filled within 25 cm level. Rats were trained in water by allowing them to swim for 60 s before one week before experimenting. During the test, the time taken to find the platform within 120 seconds is recorded and analyzed. The test was performed using standard procedures as reported previously with necessary modifications as per our study (Morris, 1984; Yagoubi et al., 2015).
Y-maze Spontaneous Alternation Test
Briefly, the apparatus consists of a platform with three arms measuring, 50 cm long, 11 cm wide, and 10 cm high (Plexiglas apparatus with maze platforms 120º to each other), in the shape of a letter Y. The surface of the apparatus was spread using sawdust to prevent the mice from getting olfactory-based sensation cues that could cause bias in the experimental data. During the trials, the rats were allowed to explore the pseudo arm with one of the arms closed and allowed to explore the setup for up to 5 min. During the test session, the rats were allowed to explore all three arms in the maze for up to 5 min and the time spent on the novel arm was recorded and analyzed calculated as a percentage of time spent on all three arms during the first 2 min of analysis. The test was conducted on standard procedures as previously reported with slight modifications (Yau et al., 2007).
Estimation of AChE and Aβ1 Peptide in Rat Brain
The enzymatic activity of AchE (Cat. No. MBS8243242) was analyzed by a standard protocol based on Ellman et al. (1961). The reaction mixture consists of 100 mmol/l of Potassium K+-phosphate buffer, pH 7.5, and 1 mmol/l of 5, 5-dithiobisnitrobenzoic acid. The enzyme (50 mg of protein obtained from fresh brain tissue homogenate) was pre-incubated for 2 min and the reaction was initiated by adding 0.8 mmol/l of acetylthiocholineiodide and enzyme activity expressed in µmol of acetylcholine iodide hydrolyzed/min/mg protein.
Aβ1 peptide levels in control and experimental rat brain tissues were determined using enzyme-linked immunosorbent assay (ELISA). Standard buffer containing eight volumes of guanidine–Tris buffer (5.0 M Guanidine HCl/50 mM Tris–HCl, pH 8.0) was used for tissue lysis of freshly harvested brain tissues. Reaction mixture contents were mixed at room temperature for 3-4 h and then subjected to sandwich ELISA to measure the specific rat Aβ1 as per manufacturer’s instructions (My Biosource Company, San Diego, USA, Cat. No. MBS726579).
Oxidative Stress and Antioxidant Markers Level
The status of LPO was determined by quantifying the formation of thiobarbituric acid reactive substances (Ohkawa et al., 1979). Levels of nitric oxide (NO) in rat brain tissues were determined using commercially available kits obtained from Abcam (Cat. No: ab65328). NO levels were spectrophotometrically quantified at 550 nm based on the manufacturer’s instructions. Activity levels of Superoxide dismutase (SOD) were determined based on the method of SOD by Marklund and Marklund (1974). The SOD-specific activity was calculated per mg of protein and expressed in U/mg. The activity of Catalase (CAT) was determined based on the procedure described by Sinha (1972) via hydrogen peroxide (H2O2) as substrate. Data were expressed as µ moles H2O2 consumed/min. Glutathione [GSH] levels were quantified using the standard procedures as reported earlier by Moron et al. (1979) The absorbance of tissue supernatants was performed at 412 nm and compared using GSH standards. Data were expressed as nmol/mg of protein.
Analysis of Inflammatory Cytokine Levels
Levels of inflammatory cytokines interleukin-1β (IL-1β) (Abcam, Cat. No: ab100769), interleukin-6 (IL-6) (Sigma Chemical Company, Cat. No: ab100769), tumor necrosis factor-alpha (TNF-α) (Abcam, Cat. No: RAB0312) were quantified from the brain tissues of rats using commercially available ELISA kits purchased from Abcam. The measure of inflammatory cytokines was performed based on the manufacturer’s instructions.
Histological Analysis
Rat brain tissues were freshly harvested and washed in ice-cold isotonic saline (sterile grade), dried on filter paper, and post-fixed in 4% paraformaldehyde (PFA) for 4 h. Formalin-fixed brain tissues were dehydrated in grading alcohol series (0%–100% alcohol series). Tissues were cleared in xylene and embedded in paraffin at 56º C in a hot air oven for 24 h. Paraffin tissue blocks were mounted on m microtome and sectioned as thin slices with 4µM thickness. Sliced brain tissues (coronal tissue sections) were fixed on microscopic slides, and then processed using standard procedures and stained by H&E for evaluating the tissue morphology and its homeostasis/dyshomeostasis.
Statistical Analysis
Data were expressed as mean ± SD of three discrete experiments. One-way analysis of variance (ANOVA) followed by Tukey’s post hoc test assay was used in our study for statistical analysis. A statistical value of p < 0.05 was used for evaluating statistical significance.
Results
Shikonin Improves Behavioral Defects in AlCl3-induced Alzheimer’s Rats
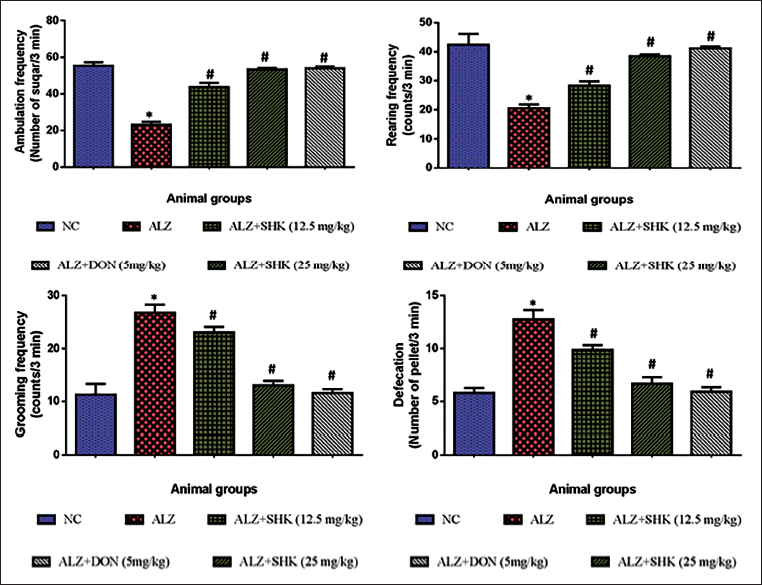
In Figure 1, AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in grooming frequency and defecation and degreased ambulation frequency and rearing frequency as compared to control rats. SHK-treated Alzheimer’s rats (12.5 and 25 mg/kg b.wt) showed a significant (p < 0.05) reduction in the total number of grooming frequency and defecation conditions and an increase in ambulation frequency and rearing frequency as compared to AlCl3-induced Alzheimer’s rats. Positive control rats treated with DON showed results similar to control rats.
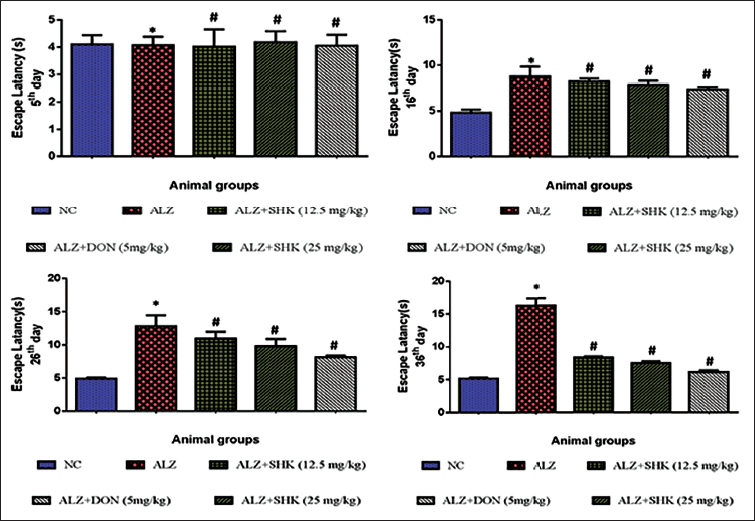
Similar to the above, from the results of the Morris water maze test (Figure 2), AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in ability to reach the platform, that is, an increase in escape latency when compared to control rats from 16th, 26th, and 36th days after induction of Alzheimer’s disease. SHK treatment significantly (p < 0.05) reduced the escape latency phenomenon from the 26th and 36th days in group II rats. Group I rats and group V rats did not show any significant (p < 0.05) alterations between themselves in this test analysis.
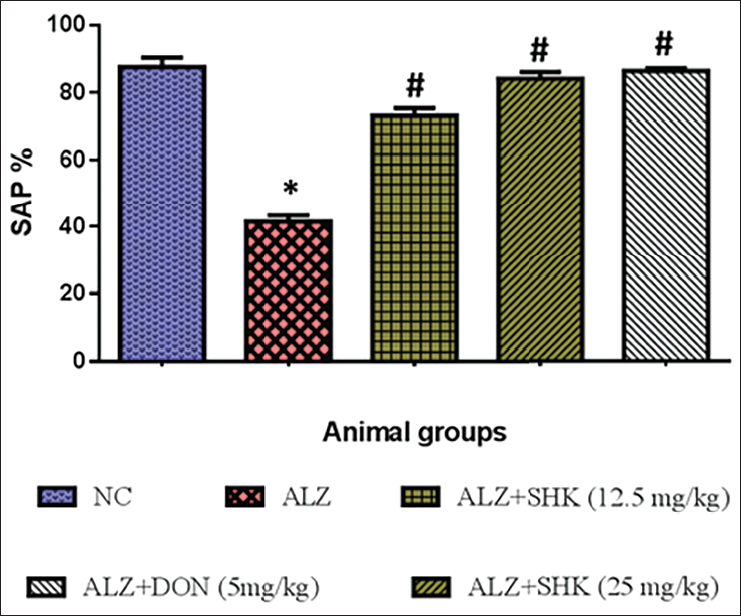
Shikonin Improves Y-maze Spontaneous Alternation Test in AlCl3-induced Alzheimer’s Rats
In Figure 3, AlCl3 administered to Alzheimer’s rats showed a significant (p < 0.05) decrease in novel arm exploratory time (expressed in percentage) as compared to control rats. SHK-treated Alzheimer’s rats (12.5 and 25 mg/kg b.wt) showed a significant (p < 0.05) increase in the total number of novel arm exploratory time when compared to AlCl3-induced Alzheimer’s rats. Positive control rats treated with DON showed results similar to control rats.
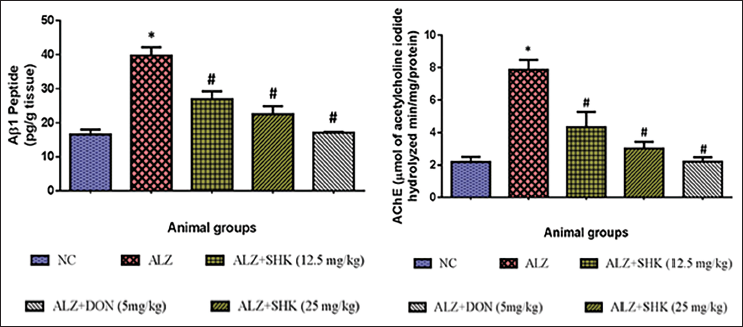
Effect of Shikonin on AChE and Aβ1 Peptide Levels in the Brain Tissues of AlCl3-induced Alzheimer’s Rats
In Figure 4, AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in the levels of AChE activity and Aβ1 peptide contents as compared to control rats. SHK-treated Alzheimer’s rats (12.5 and 25 mg/kg b.wt) showed a significant (p < 0.05) reduction in the levels of AChE activity and Aβ1 peptide contents when compared to AlCl3-induced Alzheimer’s rats. Positive control rats treated with DON showed results similar to control rats.
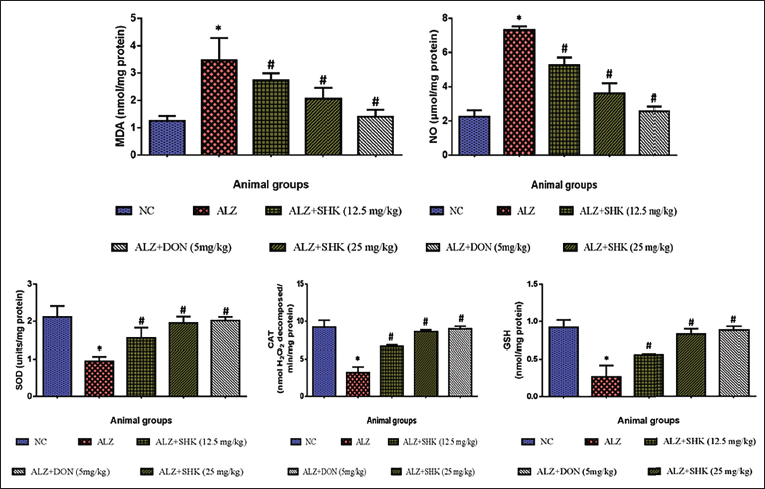
Effect of Shikonin on the AlCl3-Activated Oxidative Stress and Antioxidant Markers Level in Alzheimer’s Rats
In Figure 5, AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in malondialdehyde (MDA), and NO, while the levels of antioxidant enzymes SOD, CAT, and intracellular antioxidant GSH were significantly reduced (p < 0.05) as compared to control rats. SHK-treated Alzheimer’s rats (12.5 and 25 mg/kg b.wt) showed significant (p < 0.05) reduction in the levels of MDA, NO while the antioxidant enzyme SOD, CAT, intracellular antioxidant GSH were significantly increased when compared to AlCl3 induced rats. Positive control rats treated with DON showed results similar to control rats.
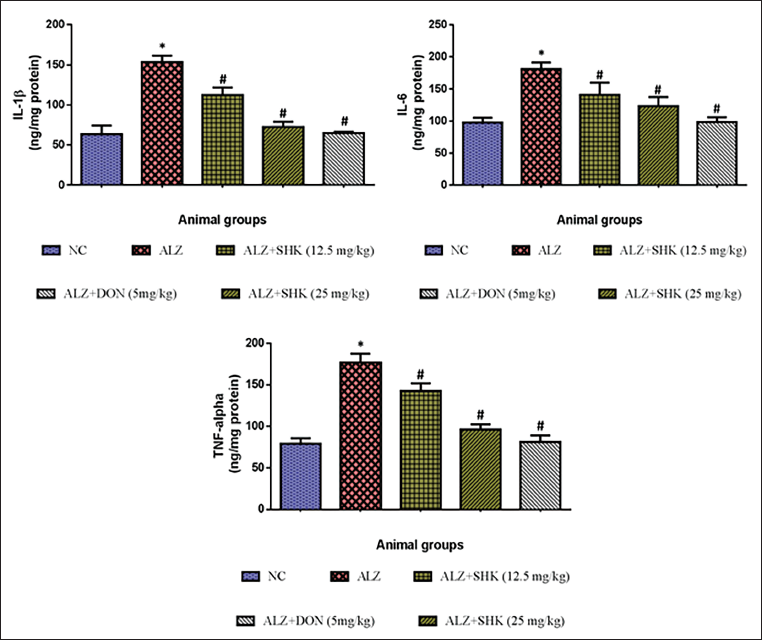
Effect of Shikonin on the Levels of Brain Inflammatory Cytokines Alzheimer’s Rats
In Figure 6, AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in the levels of IL-1β, IL-6, and TNF-α when compared to control rats. SHK-treated Alzheimer’s rats (12.5 and 25 mg/kg b.wt) showed significant (p < 0.05) reduction in the levels of IL-1β, IL-6, and TNF-α when compared to AlCl3-induced rats. Positive control rats treated with DON showed results similar to group I rats.
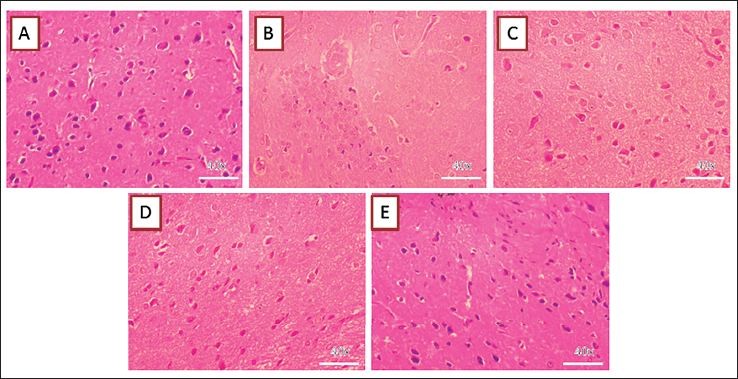
Effect of Shikonin on the Levels of Brain Histological Changes in Alzheimer’s Rats
Figure 7 shows the status of brain tissue histology of rats used in our study. AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in the atrophic cells, condensed cells, shrunken nuclei-containing cells, and infiltration of granular cells compared to control rats. SHK-treated Alzheimer’s rats (12.5 and 25 mg/kg b.wt) showed significant (p < 0.05) reduction in the atrophic cells, condensed cells, shrunken nuclei-containing cells, infiltration of granular cells when compared to AlCl3-induced rats. Positive control rats treated with DON showed results similar to control rats.
Effect of Shikonin on Histopathological Changes in AlCl3-induced Alzheimer’s Rats. Effect of Shikonin on the brain histopathology of the AlCl3-activated AD rats. Control animals exhibited the typical histological structures of the hippocampus (A). The AlCl3-triggered AD animals demonstrated diverse degenerating cells, inflammatory regions, and reduced cell density (B). The hippocampus of the 12.5 and 50 mg/kg of Shikonin and 5 mg/kg of standard drug DON-administered animals demonstrated near normal hippocampus structures with reduced histological alterations (C–E).
Discussion
In recent years, the global role of phytochemicals has gained more importance, as it exerts long-term health-promoting efforts against various human disease conditions. Notably, many medicinal plants were found to exert a beneficial role against diseases without having any nutritional value (Alhazmi et al., 2021). In this context, the necessity to investigate the biological functions of ancient and novel plant components is greatly warranted to utilize their medicinal value. In our study, we have investigated the neuroprotective ability of SHK, a plant naphthoquinone present in the roots of plants like L. erythrorhizon Siebold & Zucc. It has been used for centuries in Chinese herbal medicine and also in the Himalayan Mountain ranges (Song, 2023). We have used SHK in our study to treat the rats for experimentally induced AD using AlCl3. As stated earlier, it is well known that AlCl3 is a neurotoxicant to brain cells like neurons, astrocytes, glial cells, and vascular cells in both in vivo and in vitro experimental conditions (Bittencourt et al., 2022; Yang et al., 2019) and was also reported to be present in brain tissues of age-related dementia and Alzheimer’s patients (Exley & Clarkson, 2020).
Preliminary tests using SHK to find its ability to reduce behavioral defects caused by AlCl3-induced AD conditions revealed significant (p < 0.05) beneficial outcomes. SHK alters abnormal defects like increased grooming frequency and defecation as evaluated from open field tests in rats after AlCl3 neurotoxicity. Similarly, the results of the Morriz water maze test revealed SHK-treated Alzheimer’s rats demonstrated a significant (p < 0.05) increase in the ability to find the platform placed in a water well. In support of the above, the exploratory behavior of the Alzheimer rats treated with SHK was also increased significantly (p < 0.05) in our results. All these data denote the ability of SHK to modulate brain functions after AlCl3 intoxication. This denotes SHK can improve motor neuron functions (open field test) in rat brains (Tatem et al., 2014), hippocampus neuron functions (Morris water maze test-spatial memory) (D’Hooge & De Deyn, 2001), and functions of regions like hippocampus, septum, basal forebrain, and prefrontal cortex as observed from Y-maze test (Kraeuter et al., 2019) (Figures 1–3). Furthermore, we analyzed the ability of SHK to alter biochemical parameters like oxidative stress and antioxidant levels based on our preliminary findings of behavioral analyses. Moreover, the ability of SHK to improve behavioral defects is in turn supported by a recent study. Furthermore, AlCl3-induced Alzheimer’s rats showed a significant (p < 0.05) increase in AChE activity and increased deposition of Aβ1 peptides in rat brain tissues. This denotes that AlCl3-induced acetylcholine metabolism might trigger the formation of amyloid 1 beta fibrils in brain tissues and thereby promote Alzheimer’s disease conditions (Alvarez et al., 1997). The reduction in levels of AChE and Aβ-1 in aluminum-induced rats denotes the neuroprotective ability of SHK; however, its underlying mechanisms have not been studied in detail in our study. The results of our study are also supported by another report denoting that SHK can protect neuronal cells from amyloid-induced toxicity in vitro in PC12 cells (Tong et al., 2018).
Next, SHK treatment to AlCl3 induced Alzheimer’s rats showed a significant (p < 0.05) decrease in the levels of oxidative stress markers MDA, NO and simultaneous elevation in levels of antioxidant enzymes SOD, CAT, and intracellular antioxidant GSH, which were lowered after AlCl3 induction to experimental rats. AlCl3-induced increase in levels of MDA, NO, and reduction in SOD, CAT, and GSH denotes, AlCl3 due to its lipophilic characteristics compromised blood-brain barrier tight junctions and enters the brain tissues and caused significant (p < 0.05) damage to cellular membranes of neurons, astrocytes, and glial cells and also to brain vasculature cells in vivo thereby causing lipid peroxidation, leading to increase in MDA and NO generation via its ability to induce generation of reactive oxygen species (ROS) (Auti & Kulkarni, 2019; Lu et al., 2020). Though AlCl3 does not have redox ability like other transition metals due to its ability to bind biological membranes and its ability to facilitate ferrous iron-mediated radical chain reactions, can cause peroxidation of phospholipid bilayers and eventual generation of oxidative stress was reported in vivo and in vitro conditions, as observed in this study (Yoko Yamamoto et al., 2002). Notably, SHK treatment reduced the levels of MDA and NO denotes the ability of SHK to inhibit AlCl3-induced ROS generation and oxidative stress. Furthermore, the increased levels of SOD, CAT, and GSH can be due to the ability of SHK to inhibit oxidative stress and as well to enhance the induction of antioxidant genes via other in-cell regulatory mechanisms, probably like the induction of Nrf-2 keap 1 gene, which deserves further in-depth analysis. However, such speculative abilities were reported in other experimental studies (Du et al., 2022). Together results of our study are supported by other studies that highlight the ability of SHK to regulate oxidative stress and enhance antioxidant systems (Du et al., 2022; Yoshida et al., 2016).
Furthermore, levels of inflammatory cytokines IL-1β, IL-6, and TNF-α were markedly (p < 0.05) elevated in AlCl3-induced Alzheimer rats. This denotes the involvement of oxidative stress and subsequent induction of cellular damage which might trigger the induction of inflammatory cytokine generation, which results in subsequent tissue, and organ damage via its ability to induce immune activation, T-cell and B-cell hyperactivity, and autoantibodies generation (Umare et al., 2014). Treatment with SHK reduced the levels of IL-1β, IL-6, and TNF-α in our study denotes the ability of SHK to protect neuronal cell and tissue damage from the toxic niche of inflammation caused by AlCl3 induction. This can be attributed to the antioxidant ability to reduce oxidative stress during AlCl3 induction in our experimental study. From this, we conclude SHK can regulate inflammation caused by aluminum-induced neurotoxicity. Results of our study are in turn supported by reports highlighting the anti-inflammatory activity of SHK in various experimental models (Liao et al., 2017).
In support of the above protective abilities of SHK, our histological analysis revealed supportive evidence for SHK’s neuroprotective ability. AlCl3 induction caused shrunken nuclei-containing cells, cell swelling, and necrotic and apoptotic cell bodies together with infiltration of granule-containing cells and also showed damage to vasculature cells in histological sections. Our results were in turn supported by other reports showing similar damage to brain tissues during experimental aluminum-induced neurotoxicity (Pan et al., 2015). SHK treatment in AlCl3-induced rats showed a significant reduction in histological aberrations and this can also be attributed to the ability of SHK to scavenge ROS, inhibit oxidative stress, and enhance antioxidant defenses and also can be via other indirect regulatory mechanisms like inhibiting aluminum-mediated ferrous iron-mediated LPO in rat brain tissues. However, this has to be studied in detail using in vitro experimental systems. Results of this study are in turn supported by another report highlighting the protective abilities of SHK in brain toxicity studies (Wang et al., 2010).
Conclusion
In conclusion, our results highlighted the neuroprotective ability of SHK against experimentally AlCl3-induced Alzheimer’s disease. The neuroprotective ability was correlated to the ability of SHK to inhibit oxidative stress, inflammation, and enhance behavioral defects and antioxidant systems in brain cells. In support of the above protective abilities of SHK, our histological analysis revealed supportive evidence for SHK’s neuroprotective ability. However, the cellular mechanism of SHKs’ neuroprotective functions has to be studied in detail using suitable experimental systems. Further research and optimization of this formulation are necessary to enhance its efficacy and potential application in Neurotherapy therapy.
Summary
AD is a major neurodegenerative disease condition worldwide, with significant socioeconomic burden on global society. Similar to other neurodegenerative conditions AD is also age-associated and shows patterns like due to hippocampus neuronal loss or neuronal dysfunction, cognitive dysfunction, and abnormal motor functions due to the deposition of extracellular protein aggregate called amyloid beta fibrils. Various etiological agents were reported to induce the development of age-related AD. Among such, AlCl3 has been widely studied in recent years as it was found to be deposited in the cerebral tissues of AD patients. In this context, natural plant-based phytochemicals and their derivatives were widely used in the avenues for finding neuroprotective agents against neurodegenerative diseases like AD. In this study, we have used SHK, a natural naphthoquinone pigment found in the root of plant species L. erythrorhizon. Reports denote SHK possesses various biological effects like anti-inflammation and anticancer, effects in experimental models. Our preliminary findings revealed SHK exerted significant neuroprotection via its ability to mitigate behavioral defects, oxidative stress, and neuroinflammation and protect neuronal cell morphology against AlCl3-induced experimental neurotoxicity. In conclusion, our results highlighted the neuroprotective ability of SHK against experimentally induced Alzheimer’s disease. The neuroprotective ability was correlated to the ability of SHK to inhibit oxidative stress, and inflammation and enhance behavioral defects and antioxidant systems in brain cells. However, the cellular mechanism of SHKs’ neuroprotective functions has to be studied in detail using suitable experimental systems.
Abbreviations
AlCl3: Aluminum chloride; AD: Alzheimer’s disease; CAT: Catalase; SOD: Superoxide dismutase; IL-6: Intereukin-6; H&E: Hematoxylin-eosin; RPD: Rapidly progressive dementias; BSA: Bovine serum albumin; SHK: Shikonin; LPO: Lipid peroxidation; IL-1β: Interleukin-1β; TNF-α: Tumor necrosis factor-alpha; PFA: Paraformaldehyde.
Footnotes
Acknowledgments
The authors thank the Department of Neurology, Central Hospital Affiliated to Shandong First Medical University, Shandong, China, for supporting and providing facilities for this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
Ethical approval for the study was obtained from the institutional ethics committee.
Funding
The authors received no financial support for the research, authorship and/or publication of this article
