Abstract
Aim
The present study aims to investigate the toxicity profile and neuroprotective effect of ethanol crude extract of Aristolochia bracteolata (EEAB) leaves in mice.
Materials and Methods
EEAB was subjected to preliminary phytochemical screening and gas chromatography–mass spectrometry (GC–MS) analysis. To determine the toxicity profile in Swiss albino mice, acute and subchronic toxicity experiments were performed according to Organisation for Economic Co-operation and Development criteria. A behavioral test in mice with drug-induced learning and memory impairment was performed to determine neuroprotective effects.
Results
In the study GC–MS, 19 compounds were detected. Compared to the control group, no clinical signs of toxicity were detected in the plant-treated group in both the acute and subchronic toxicity experiments at the highest dose of 2,000 mg/kg. It was calculated that the mean oral lethal dose (LD50) > was 2,000 mg/kg. Scopolamine-induced cognitive deficits in mice were greatly reduced after pretreatment with the EEAB at a dose of 100 mg/kg p.o. This was demonstrated by the reversal of the improvement in spontaneous alternation in the Y-maze task and by the significant improvement in latency in the passive avoidance task.
Conclusion
Based on the results, EEAB might contain potent secondary metabolites that would primarily enhance the neuroprotective effects and cognitive deficits induced by cholinergic dysfunction.
Introduction
Alzheimer’s disease (AD), the most common form of dementia, is a neurological condition that is more common elderly population. AD has turned into a global health issue due to populations getting older worldwide. 1 The pathological manifestation of AD is the accumulation of extracellular-amyloid peptide (Aβ) in the brain tissue, as well as neurofibrillary tangles and neuronal loss formed by the accumulation of intracellular hyperphosphorylation of tau protein. These pathological manifestations result in the loss of cognitive function, cholinergic dysfunction, oxidative stress, and neurodegeneration, leaving affected individuals bedridden and in need of round-the-clock care.2, 3
Seven medications—donepezil, rivastigmine, galantamine, memantine, and memantine coupled with donepezil, aducanumab, and lecanemab—have received FDA approval for the treatment of AD. They do not cure AD, are not suitable for everyone with AD, and also have side effects. 4 Finding a possible medication to treat AD is therefore necessary.
The discovery of new medications has benefited greatly from the utilization of medicinal plants’ phytochemicals, which serve as structural models for semi-synthetic pharmaceuticals or can be utilized directly as medical remedies. 5 Since Aristolochia bracteolata Lam. has been used as ethnomedicine for centuries and has been shown to have antioxidant and anti-inflammatory properties, we have chosen this plant to investigate its neuroprotective effects against AD. 6
In this study, we have focused on the preliminary phytochemical analysis, in vivo neuroprotective study, and toxicity profiles of the ethanol extracts of A. bracteolata (EEAB) for the possibility of use as a neuroprotective agent.
Materials and Methods
Chemicals
Eserine, scopolamine hydrobromide, and all other materials were bought from Sigma-Aldrich in India.
Plant Collection and Extraction
The leaves of A. bracteolata were gathered from several locations in Tamil Nadu and verified by the Botanical Survey of India, South region, Coimbatore. The leaves were powdered after being shade-dried. Using a soxhlet system, the plant materials were continuously heated and extracted in 80% ethanol. Under low pressure, the solvents were evaporated, and after that, they were lyophilized. The dried extracts were kept in a −20°C freezer for further use.
Experimental Animals
Swiss albino adult male mice weighing 25–30 g were housed in a regular habitat (25°C ± 100°C, 55% ± 5% humidity, and a 12 h/12 h light/dark cycle). The animals had unrestricted access to regular laboratory mice chow and tap water. The protocol was approved by our institutional animal ethics committee, which is a subcommittee of the Committee for Control and Supervision of Experiments on Animals (India) (CPSCEA), and the care and handling of the mice were done following the generally recognized standards for using animals.
Phytochemical Screening
Standard techniques described in Trease & Evans and Harborne were used to conduct preliminary phytochemical analysis on the EEAB.7–9
Gas Chromatography–Mass Spectrometry (GC–MS) Analysis
A Perkin-Elmer GC Clarus 680 system gas chromatograph interfaced to a Mass Spectrometer Clarus 6080 outfitted with an Elite-5MS fused a capillary column (30 × 0.25 m ID × 0.25 m DF) was utilized to conduct the GC–MS analysis for EEAB. For GC–MS detection, an electron ionization system operated in electron impact mode with an ionization energy of 70 eV. Helium gas (99.9%) is used as a carrier gas, at a constant flow rate of 1 mL/min and an injection volume of 1 µL (10:1 as the split ratio). The ion-source temperature was 240°C, and the injector temperature was also 240°C. At 70 eV, a scan interval of 0.5 second, and fragments ranging in size from 50 to 600 Da, mass spectra were recorded. The system’s solvent delay ranged from 0 to 2 minutes. The running time for the GC/MS was fixed at 36 minutes. By comparing the component’s average peak area to the total areas, the relative percentage quantity of the component was calculated. The mass detector employed for the analysis was Turbo-Mass ver-5.4.2. Based on a comparison between the constituent’s mass spectra and those in the NIST library, the compounds were described and identified.
Acute Toxicity Studies
Three animals in each group were used in the acute toxicity research, which was carried out by Organisation for Economic Co-operation and Development (OECD) guideline 423.10, 11 The test animals were all weighed before and after being fasted for about 12 hours. According to the fasted mice’s body weight, an oral dose of the EEAB reconstituted in normal saline was given. The dosage of the EEAB was 5, 50, 300, and 2,000 mg/kg, and it was given orally by gavage. After treatment, the animals were routinely and individually monitored for mortality and clinical symptoms at 0, 1, 2, 4, 5, and 6 hours as well as on day 1, with particular focus placed on the first 4 hours. The observation was then kept up every day for a total of 14 days. 12 On day 15, all the research mice were euthanized by a chloroform overdose, and the internal organs were inspected for any indications of abnormalities.
Sub-chronic Toxicity Studies
The repeated dose oral toxicity study in rodents (OECD: 408) was carried out by the recommendations of the OECD. Four groups of six male and six female mice in each group were used. The ethanol extracts of EEAB were diluted in saline and given orally in single doses of 500, 1,000, and 2,000 mg/kg (satellite dose) of body weight for 28 days at a dose of 5 mL/kg each day. An identical volume of ordinary saline was given orally to mice in the control groups. These doses were chosen by the acute toxicity studies’ LD50 value. All groups’ body weights were gauged during the trial period. Additionally, the mice used in the study were checked for mortality, behavioral abnormalities, and morphological alterations. After a 12- to 16-hour fast after the study, all of the mice were anesthetized using chloroform. To assess hematological and biochemical parameters, blood samples were drawn into dry and EDTA-coated tubes. The mice were then euthanized, and their organs were taken out for necropsy, weight measurements of the organs, and histological analysis.
Y-maze Task
The Y-maze test was conducted by the instructions provided by Kim et al. in 2006. 13 Mice were given EEAB (12.5, 25, 50, and 100 mg/kg, p.o.) or eserine (10 mg/kg, p.o.) as a positive control an hour before the test. Scopolamine (1 mg/kg, i.p.) was administered 30 minutes after the oral dose of EEAB, eserine, or 0.9% saline solution to cause cognitive impairment. Animals used as controls only received 0.9% saline solution. Locomotor activity was measured by the number of arm entries. The following formula was used to calculate percentage alternation.
% Alternation = [(Number of alternations)/(Total arm entries − 2)] × 100.
Passive Avoidance Task
Training and testing for the passive avoidance task were conducted by prior research.13, 14 One hour before the acquisition trial, different doses of EEAB (12.5, 25, 50, and 100 mg/kg, p.o.) or eserine (10 mg/kg, p.o.) were given as a positive control. Scopolamine (1 mg/kg, i.p.) was administered half an hour after EEAB, eserine, or 0.9% saline solution to cause cognitive impairment. A 0.9% solution of standard saline was given to the control groups. The same mice underwent a retention study 24 hours after the acquisition trial. Latency time was measured as the length of time it took the mice to enter the dark compartment.
Statistical Analysis
The mean and standard error of the mean are used to express the results of the behavioral investigations. To assess significant mean differences between treatment groups, one-way analysis of variance with the Dunnett test was used in the statistical study. P values lower than 0.05 were regarded as significant. GraphPad Prism software was used for all statistical analysis.
Results
Phytochemical Analysis
The phytochemical analysis of the EEAB leaves revealed the presence of phenol, glycosides, saponins, carbohydrates, tannins, and flavonoids.
GC–MS Analysis
Phytoconstituents present in the EEAB were identified by GC–MS analysis. Compounds were identified based on the retention time and NIST library search. GC–MS spectrum for EEAB is shown in Figure 1. A list of compounds identified, and their name, molecular formula, molecular weight, retention time, peak area, and percentage peak area are given in Table 1. The GC–MS analysis of EEAB yielded 19 compounds. Among them, n-hexadecanoic acid and hexadecanoic acid ethyl ester contributed 31.82% and 12.76%, respectively. The other compounds including 9,12,15-octadecatrien-1-ol (9.30%), 3,7,11,15-tetramethyl-2-hexadecen-1-ol (7.84%), benzaldehyde, 2, 4-dimethyl (5.01%) and the remaining chemical constituents are comprised from 0.93% to 4.41%.
GC–MS Spectrum of EEAB.
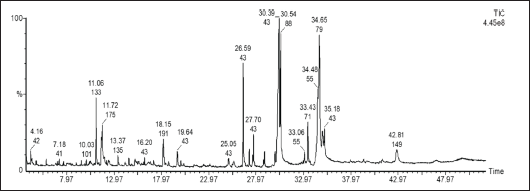
GC–MS Spectral Profile of EEAB.
Acute Toxicity Studies
The toxic effect of EEAB on the appearance and general behavioral pattern of mice was observed for up to 14 days. No toxic symptoms or mortalities were observed. Behavioral patterns of the animals were observed for the first 4 hours and then 24 hours after administration. Animals in both vehicle and extract treated groups were normal and showed no significant changes in behavior, skin effects, respiration, impaired food and water intake, postural abnormalities, and hair loss. Based on the observations, the EEAB is classified as Category −5 (Globally Harmonized System of Classification and Labeling of Chemicals), and the LD50 value was reported to be greater than 2,000 mg/kg.
Subchronic Toxicity Studies
All animals in the tested group treated with plant extract at doses of 500, 1,000, and 2,000 mg/kg body weight daily survived the 28 days. No clinical signs of toxicity were observed in the plant-treated group compared to the control group.
Hematological and serum biochemical results are shown in Table 2. After chronic administration, no significant differences in body weight were observed between the vehicle-treated group and the treated group. The hematological parameters including WBC, RBC, and hemoglobin levels were studied in both male and female mice treated with plant extracts. The other parameters, including ALT, AST, creatinine, and blood urea levels, were also measured, and the results showed no significant changes compared to the control mice.
Hematological and Biochemical Properties in Swiss Albino Male and Female Mice Treated with 2,000 mg/kg of the EEAB.
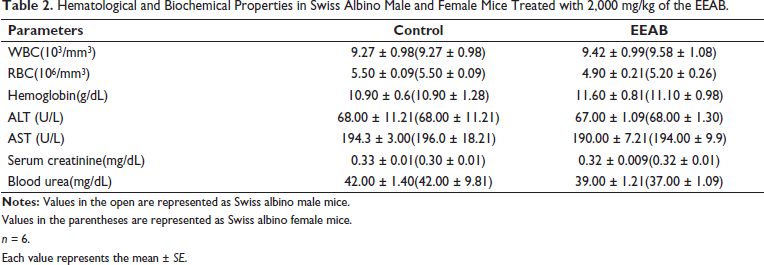
Values in the parentheses are represented as Swiss albino female mice.
n = 6.
Each value represents the mean ± SE.
Macroscopic examination of vital organs was performed after treatment. Major vital organs, such as the heart, liver, kidneys, lungs, and brain, were surgically removed, cleaned with normal saline, and then weighed and examined for inflammation. No significant changes were found compared to the control.
Histopathology
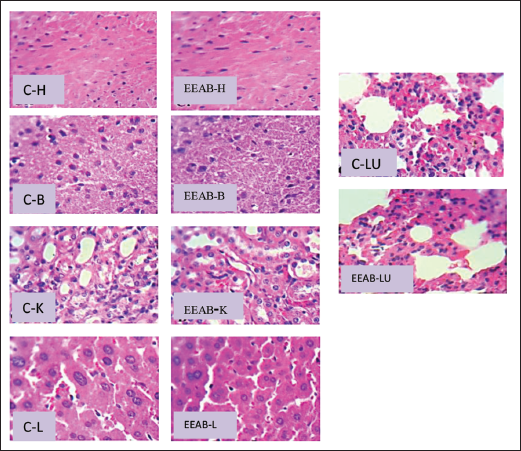
The results of the histopathological examinations are shown in Figures 2 and 3. No obvious changes were observed in the major organs of the animals treated with the tested plant extract. The brain section of the animals treated with EEAB showed the normal configuration and architecture of the neurons in the cerebral cortex, indicating no neurotoxicity. Liver cells showed normal hepatocytes, and no inflammatory changes, necrosis, fibrosis, or local fatty degeneration were observed in the liver, indicating no obvious changes. Microscopic observation of the myocardium showed no changes in the cell nuclei. Lung section of animals treated with plant extracts showed no changes in alveolar macrophages, and the kidney showed a normal configuration of the renal cortex with proximal tubules.
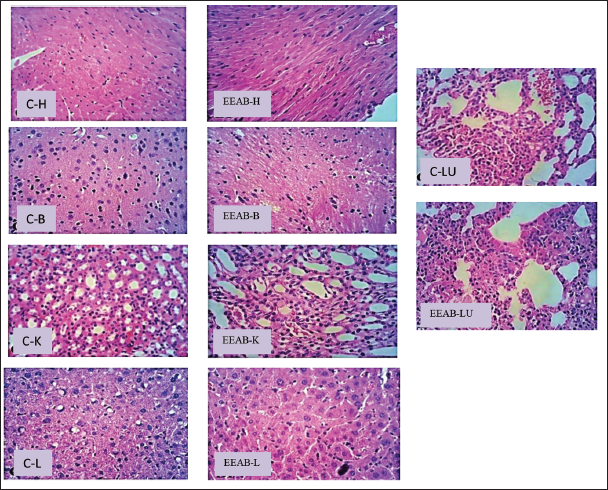
Effect of Tested Plant Extract on Histopathology of the Treated Female Mice: Heart, Brain, Kidney, Liver, and Lungs.
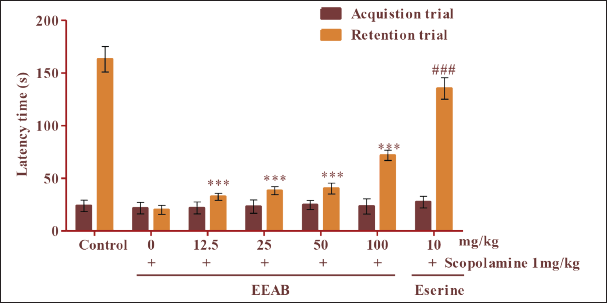
Passive Avoidance Task
The observation of the passive avoidance task is shown in Figure 4. Latency did not show much difference between groups during the acquisition trial. In the retention trial, the latency of the scopolamine-treated group to enter the dark compartment was significantly shorter than that of the control group, indicating memory impairment. The reduced latency on the retention test caused by scopolamine treatment was improved in a dose-dependent manner by treatment with EEAB (12.5, 25, 50, and 100 mg/kg). EEAB treatment significantly reversed the scopolamine-induced reduction in latency.
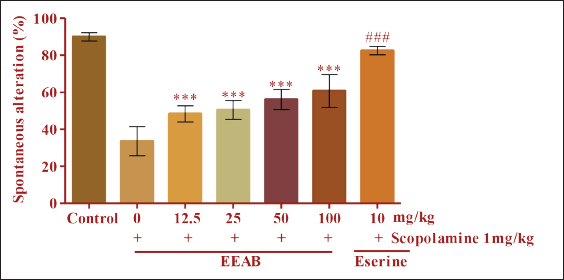
Y-maze Task
The effects of EEAB on short-term memory function examined using the Y-maze task. As shown in Figure 5, scopolamine (1 mg/kg, i.p.) significantly decreased the percentage of spontaneous alternation. The scopolamine-induced reduction in spontaneous alternation was significantly restored by EEAB (12.5, 25, 50, and 100 mg/kg) treatment in a dose-dependent manner, suggesting an improvement in memory. The total number of arm movements between groups did not differ, suggesting that locomotor activity was not affected by treatment with scopolamine, esterol, or EEAB. Average spontaneous alternation was significantly higher at 100 mg/kg than at the lower EEAB doses.
Discussion
The EEAB was subjected to GC–MS analysis, which revealed the presence of 19 components, of which n-hexadecanoic acid was the most abundant. Hexadecanoic acid has been attributed biological properties, including antioxidants, hypocholesterolemic properties, nematicides, and pesticides. 15 The presence of these bioactive compounds in certain plants raises hopes for their potential usefulness in drug discovery.
According to the results of an acute toxicity study, EEAB can be safe up to 2,000 mg/kg body weight in treated mice, and the LD50 was determined to be more than 2,000 mg/kg. Any pharmaceutical substance or drug with an oral LD50 greater than 1,000 mg/kg can be considered safe and of low toxicity. 16 Accordingly, a single dose of EEAB up to 2,000 mg/kg body weight is practically non-toxic.
The hematological and biochemical studies showed that EEAB did not lead to significant changes compared to the control. Blood cells are formed in the bone marrow, and several phytochemicals extracted from plants have an effect on the number of red blood cells. 17 The hematopoietic system, one of the most sensitive targets in the biological system, is the most important target for toxic substances. 18 The red blood cells do not show any detectable changes in our studies. Consequently, the investigated plant extract may have no negative effects on bone marrow function and may help to clarify why EEAB is safe, as it does not cause anemia at any dose.
The biomarker enzymes ALT and AST were used to evaluate normal liver function. 19 In our study, ALT and AST did not change significantly during the evaluation of liver function by EEAB, indicating that the selected plant is not toxic to liver function. The most sensitive biomarkers of kidney damage are blood urea and serum creatinine. 20 The blood urea and serum creatinine levels of the mice were not significantly different from those of the control group in the current study.
Histological analysis of the chronic intake of EEAB in the current study revealed that EEAB had no adverse effects on the morphology of the heart, liver, kidneys, lungs, or brains of the mice. This assertion is supported by the results of the biochemical and hematological analyses and by the acute toxicity data.
The primary organ affected by toxicity is the liver, which is exposed to xenobiotics that are absorbed in the intestine and converted into other compounds that may or may not be hazardous to the liver of mice. 21 In our study, no necrosis, fibrosis, or localized fatty degeneration were detected in the liver cells of the treated mice, indicating that the plant extract did not induce inflammatory changes. Based on the results of the current study, EEAB appears to be generally safe in mice at doses up to 2,000 mg/kg for a duration of 28 days and may not cause serious treatment-related toxicity.
The prolonged latency in the passive avoidance task and the improvement in spontaneous change in the Y-maze test compared with control indicate that EEAB may have some neuroprotective effect against the memory deficit caused by scopolamine in mice.
The central cholinergic system essentially controls cognitive functions, and the loss of cholinergic neurons in the hippocampus is the hallmark of AD. Memory impairment and reversible symptoms of senile dementia have been associated with scopolamine, a cholinergic muscarinic receptor antagonist, in young volunteers. 22 In our work, EEAB reduces scopolamine-induced memory impairment, suggesting that it may be a source for the development of neuroprotective drugs.
Conclusion
The ethanol extracts from A. bracteolata leaves showed remarkable improvement in memory of mice with cholinergic impairment. In addition, the acute and subchronic toxicity studies did not result in any adverse events or mortality in the treated mice. A. bracteolata, which may act as an enhancer of cholinergic activity, could be considered a novel therapeutic option in the treatment of a variety of neurodegenerative diseases, including AD.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study was approved by our institutional animal ethics committee, Ref no. AUROT/IAEC/DEC2014-011.
Funding
This work was funded by Department of Science and Technology, Government of India to P. Selvamani, Assistant Professor, Department of Pharmaceutical Technology, Centre for Excellence in Nanobio Translational Research (CENTRE), Anna University, Bharathidasan Institute of Technology Campus, Tiruchirappalli, Tamil Nadu, India (DST: Ref. No. NO.SR/FT/LS-41/2010 Dated 04.05.2012.).
