Abstract
Background
Scutellaria barbata D. Don, also known as “Banzhilian” in China, the diterpenes of Banzhilian (Bzl-D) is a commonly used herbal medicine for treating various tumors including colorectal cancer (CRC).
Objectives
This study aimed to examine the function of Bzl-D in inhibiting CRC development and explore its potential targets and mechanisms involving the Hedgehog (HH) pathway.
Materials and Methods
To determine the major components in Bzl-D, we performed ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-qTOF-MS) analysis. The proliferation, migration, invasion, and apoptosis abilities of the CRC HT-29 cell line were examined, and the related protein expression was tested. Tumorigenesis after treatment was detected by in vivo experiments.
Results
Nineteen diterpenoids were detected, Bzl-D significantly inhibited the cell proliferation and metastasis and induced the apoptosis of HT-29 cells through binding to Sonic HH (SHH) and down-regulating the key proteins (SHH, patched, smoothened, and glioma-associated oncogene) of the HH pathway, thus. Of note, Bzl-D remarkably suppressed the growth of HT-29 xenografts in nude mice.
Conclusion
This research demonstrates that Bzl-D is a promising anti-rectal cancer drug, providing a new entry point for targeted therapy of CRC.
Introduction
Due to increasing morbidity and mortality, colorectal cancer (CRC) has gradually become one of the hotspots in global research (Bray et al., 2018). For the treatment of CRC, the commonly used strategies nowadays include local excision, radiotherapy, chemotherapy, immunotherapy, and so on. Patients in the early stage of CRC can be cured with excision surgery, while the prognosis of patients in the late stage is poor and often accompanied by serious side effects, such as violent weight loss and hair loss. Currently, targeted therapy alone or in combination with other strategies has further reduced the risk of side effects and enhanced the efficacy of CRC treatment (Chang et al., 2007).
The Hedgehog (HH) signaling pathway has a crucial role in tumor proliferation and migration and is linked to embryonic development (Haycraft et al., 2005; Jiang, 2022). The HH signaling pathways can be divided into two categories, classical and non-classical signaling pathways (Skoda et al., 2018). In the classical pathway, binding Sonic HH (SHH) ligand leads to the patched (PTCH) receptor activating the transmembrane protein smoothened (SMO), which induces translocation of the glioma-associated oncogene (GLI) nuclear transcription factor to the nucleus, ultimately leading to the revitalization of genes associated with cell proliferation and differentiation (Gulino et al., 2012; Niewiadomski et al., 2014). The non-classical HH signaling pathway represents an alternative mode of HH signaling, involved with the activation of many signaling cascades (Briscoe & Therond, 2013; Liu et al., 2018).
There are many investigations on the molecularly targeted inhibitors of the HH pathway, most of which bind to downstream genes as targets, such as the SMO inhibitor vismodegib, the GLI inhibitor GANT-61, etc., but further experiments have been halted by their safety and side effects. Numerous reports have shown that the HH pathway is overactivated in CRC predominantly through a paracrine ligand-dependent mechanism (Douard et al., 2006; Wang et al., 2012). Therefore, opening up upstream SHH target inhibitors may be a new hope for opening up new anticancer drugs.
Traditional Chinese Medicines are the favored resource in the exploration of anticancer drugs due to low toxicity, low cost, low side effects, multi-components, and multi-targets. It has been reported that the diterpenoids isolated from Scutellaria barbata D. Don showed more potential cytotoxic activities against several tumor cell lines, due to the specific chemical structures. For example, scutebarbatine B displayed obvious cytotoxic effects against HONE-1, KB, and HT-29 cells, with IC50 values of 4.4, 6.1, and 3.5 µM (Dai, Tao, et al., 2006). However, there are fewer reports on antitumor effects in vivo and further investigations of the mechanism action of these effects because of low content (Zhang et al., 2014; Zhang et al., 2017). This study will purify and investigate the diterpenoid-enriched extract (diterpenes of Banzhilian [Bzl-D]) from this plant, associated with inhibiting CRC activity and the potential mechanisms involved with the HH signaling pathway. This research will clarify the mechanism action of the diterpenoids as active ingredients of S. barbata D. Don against CRC, as well as bring new thoughts for the exploitation of targeted inhibitors for CRC.
Materials and Methods
Reagents and Materials
The dried whole plant was obtained from Hangzhou Huadong Traditional Chinese Medicine Co., Ltd (Lot:20210926; Hangzhou, China). CCK-8 working solution was supplied from APE x BIO (Houston, TX, USA). Annexin V-FITC Apoptosis Detection kit, Cell Cycle and Apoptosis analysis kit, and phosphate-buffered saline (PBS) were obtained from Beyotime (Shanghai, China). Primary antibodies were purchased from Santa Cruz (CA, USA). Secondary antibodies were obtained from Beyotime (Shanghai, China), and other reagents were available commercially.
Preparation of the Bzl-D from S. barbata D. Don
The dried S. barbata D. Don was initially crushed into coarse powder and then percolated with 85% aqueous ethanol. The percolation liquid was subsequently dried by a rotary evaporation instrument, and then extracted with ethyl acetate. The dichloromethane extract was purified by polyamide column from the extract of ethyl acetate eluting with dichloromethane. Bzl-D purifying from dichloromethane extract by passing the C18 silica open column twice.
Chemical Composition Analysis
Bzl-D was dissolved in methanol and assayed on an ACQUITY UPLC CSH C18 column (2.1 × 100 mm. 1.7 µm; Waters USA) with gradient elution using 0.1% formic acid in water and acetonitrile as mobile phases. The gradient program was as follows: acetonitrile concentration from 10% to 25% for the first 5 minutes; from 25% to 30% for 5–15 minutes; from 30% to 60% for 15–40 minutes; and from 60% to 100% for 40–60 minutes. Data were processed using MassLynx V4.1 software (Waters Corporation) and mass spectra were plotted using Original 2019 software.
Cell Proliferation Assay
Cells were cultured into 96-well plates and then exposed to different concentrations (ranging from 10 to 40 µg/mL) of the tested compounds after 24 hours. At the end of the treatment, cell proliferation was detected using the CCK-8 assay. Optical density was recorded at 450 nm using a Bole 680 automated microplate reader (Bio-Rad, USA) and cell viability (%) was calculated.
Apoptosis Assay
After inoculation of cells into 24-well plates, cells were treated with Bzl-D (5–20 µg/mL) for 24 hours. Rinsed adherent and floating cells with cold PBS, then centrifuged and discarded the supernatant, and resuspended with Annexin-binding buffer. The cells were incubated with fluorescent dye under light for 20 minutes. Fluorescence values were measured by flow cytometry (BD, FACS Cellesta), FlowJo 10.8 software was used to analyze the results.
Cell Cycle Analysis
The cell culture and drug administration were the same as in the previous section. Cells were resuspended in pre-cooled 70% ethanol and allowed to stand at 4°C for 30 minutes. The cells were rinsed of residual ethanol with cold PBS. Add The staining buffer, pyridinium iodide staining solution, and RNase A after mixing. Incubated in a dark environment for 30 minutes, and then the fluorescence values of the treated cells were measured by flow cytometry.
Cell Scratch Assay
The influence of Bzl-D on cell migration capability was evaluated by a monolayer cell scratch assay. Cells were inoculated into six-well plates and reached 80% confluency. A horizontal line was drawn on the monolayer with a 10 µL pipette tip, and residual cells were rinsed and incubated for 24 hours with different concentrations of Bzl-D (5–20 µg/mL). Images were taken by an inverted microscope (OLYMPUS, Japan) and processed with ImageJ.
Transwell Assay
The migratory and invasive ability of Bzl-D on HT 29 cells was used in the Transwell cell invasion assay. Matrigel was diluted by the basal medium and evenly coated in the upper chamber for 4 hours of incubation before inoculating HT-29 cells. Bzl-D (5–20 µg/mL) was administered for 24 hours. Excess cells were gently swabbed with a cotton swab. The invaded cells were fixed with 4% paraformaldehyde, stained with 0.1% violet crystal, and then air-dried appropriately. Images were acquired by inverted microscope and analyzed by ImageJ software.
Western Blotting Analysis
Cells were incubated with Bzl-D (5–20 µg/mL) for 24 hours and then protein was extracted. Proteins of the same concentration and volume were separated by constant pressure 110 V on SDS-PAGE for 2 hours and transferred 2 hours to PVDF membrane (0.45 µm) by cross-flow 280 mA. The PVDF membrane was blocked for 2 hours with 5% skim milk then incubated with the primary antibody (1:1,000) overnight at 4°C. After washing, the secondary antibodies (1:1,000) were added and incubated at room temperature for 2 hours. Protein bands were imaged and analyzed by Evolution-Fusion software (Vilber, France).
Cellular Thermal Shift Assay (CETSA)
Bzl-D affects the HH pathway with SHH as a binding target validated by CETSA as described before (Zhang et al., 2022). Cells were inoculated in six-well plates and treated with basal medium containing Bzl-D (10 µg/mL) for 4 hours. Trypsin was added to digest and collect the cells. The cells were equally divided into five, and each sample was heated at different temperatures over 3 minutes (40°C–48°C), centrifuged and PBS discarded. The proteins were extracted by adding Radio Immunoprecipitation Assay buffer and analyzed by Western blotting.
In vivo Tumor Xenograft Studies
A mouse CRC model was established using axillary inoculation of tumor cells as our previous method (Zhang et al., 2022). The control (Ctrl) group (n = 6) was provided with saline and the treatment group (n = 6) was provided with 50 mg/kg Bzl-D, intraperitoneally once a day for 16 days at the end of treatment. The average volume of the tumors was calculated according to this formula: volume = (length × width × width)/2, and the data were recorded every two days. At the end of treatment, the mice were euthanized, and the tumors were aseptically excised and weighed. Tumor tissues were stained by hematoxylin–eosin (H&E) for pharmacodynamic analysis.
Data Analysis
All experiments were conducted three times independently. Significance analyses and bar graphs were performed using GraphPad Prism 8.0 software. p < 0.05 was considered statistically significant.
Results
Chemical Components of Bzl-D
The dichloromethane extract (30.0 g) was isolated from 85% aqueous ethanol of S. barbata D. Don (dry weight 4.0 kg), and a total of 14.9 g of purified Bzl-D was obtained from the dichloromethane extract. As shown in Figure 1 and Table 1, 19 detected peaks were identified from Bzl-D by comparing the spectral data with published data, and the exact mass values of the protonated molecular ions matched the theoretical values.
The Chemical Composition Identified from Bzl-D.
Bzl-D Inhibits the Proliferation of HT-29 Cells
After 24 hours of Bzl-D treatment, the cell viability of the in vitro CRC cell line showed a regular decrease with increasing drug concentrations (Figure 2A). Moreover, different cell lines of CRC also displayed different degrees of sensitivity to Bzl-D. HT-29 cells showed higher sensitivity to Bzl-D compared to HCT116 and SW480 cells. Approximately, 40 µg/mL of Bzl-D resulted in 50% cell death on HCT116 and SW480 cells, whereas 20 µg/mL of Bzl-D was able to induce about 50% growth inhibition on HT-29 cells, with the value of IC50 22.75 µg/mL. Consequently, HT29 cells were chosen for further investigation in subsequent experiments.
Bzl-D Induces Apoptosis in CRC Cells
The ratio of apoptotic cells in Bzl-D-treated HT29 cells increased from the initial 22.9% to 33.4%, 55.8%, and 80.4%, respectively, with increasing concentration of the administered drug after 24 hours of dosing. From the changes in the data, we can infer that the proportion of early apoptotic cells in HT29 cells is increasing with a dose within a certain range (Figure 2B).
Bzl-D Induces G2/M Arrest in CRC Cells
As shown in Figure 2C, compared with the Ctrl group, the proportion of G2/M phase was augmented dose-dependently on HT-29 cells after drug treatment. It suggested that Bzl-D has the potential to inhibit the proliferation of tumor cells by inducing G2/M phase arrest.
Bzl-D Inhibits Migration and Invasion of CRC
Cell scratch assay showed that Bzl-D dramatically reduced the migratory capacity of HT-29 cells. Compared with the Ctrl, the percentage was approximately 53.2%, 35.1%, 25.8%, and 23.1% fewer migratory cells in the different concentrations of Bzl-D-treated groups (Figure 3A and B). Similarly, in vitro, Transwell assays showed that the amount of migrating and invading cells was markedly and regularly decreased in Bzl-D-treated cells (Figure 3C–E). The number of migrating cells decreased from 525 in the Ctrl to 242, 141, and 71 with the increase of administered dose, the number of invading cells decreased from 460 in the Ctrl group to 202, 110, and 58. Analysis of the data using Ordinary one-way ANOVA showed a significant decrease in the migration and invasion ability of the cell groups treated with Bzl-D using Ctrl as the baseline for comparison (p < 0.001).
To sum up, Bzl-D was capable of remarkably inhibiting the migration and invasion of HT-29 cells, and the change was more intense as the concentration increased within the administration range we set.
Bzl-D Down-regulate Key Proteins of the HH Signaling Pathway
To investigate whether Bzl-D exerts anti-CRC effects by inhibiting the classical HH pathway, we examined the protein expression in Bzl-D-treated HT-29 cells after 24 hours. The protein expression levels of SHH, PTCH, SMO, and GLI were down-regulated in HT-29 cells treated with Bzl-D (Figure 4A–E). These results suggest that Bzl-D inhibits the occurrence of cell development-related events such as cell proliferation involved in the classical HH signaling pathway.
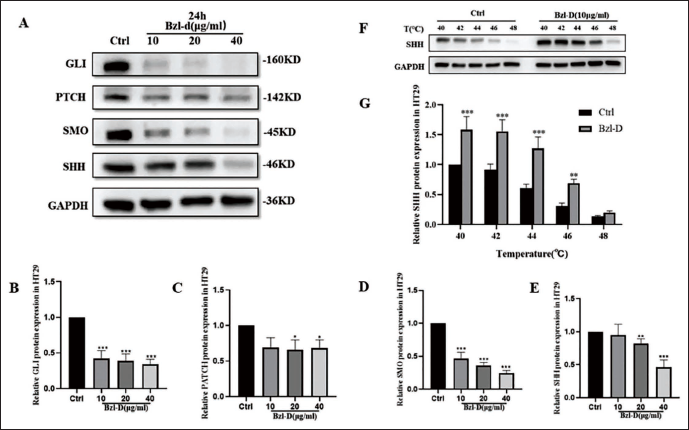
Bzl-D Inhibited HH Pathway Signaling by Targeting SHH on HT-29 Cells
CETSA is a method based on thermal stability to detect binding targets. We next assess whether SHH was the binding target of Bzl-D acting on the classical HH pathway after 4 hours of treatment. The results showed that with the gradient temperature changed, SHH was much more stabilized to detect in HT-29 cells exposed to Bzl-D, compared with Ctrl (Figure 4F and G). The increased stability suggested that Bzl-D may interact with SHH protein to modulate its function in the HH signaling pathway. Collectively, this result indicated that Bzl-D binds specifically to SHH in HT-29 cells.
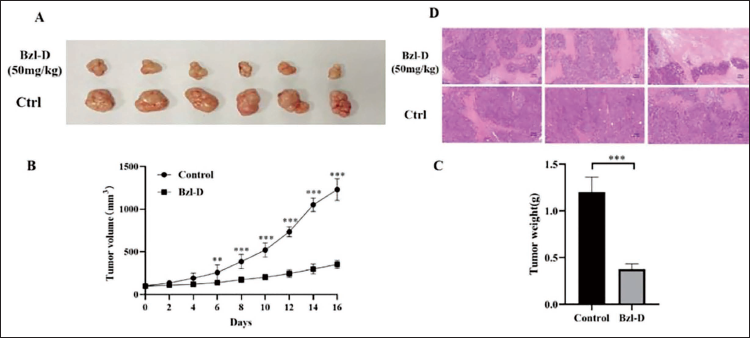
Effects of Bzl-D Administration on Xenograft Tumor Growth
From the results in Figure 5A to C, the tumor weight and volume in the Bzl-D treatment group were markedly lower than those in the Ctrl group. Furthermore, H&E analysis of the tumor tissues showed a significant reduction in pathological mitosis and a more orderly cellular arrangement in the tumor tissues of mice in the Bzl-D treatment group (Figure 5D). Overall, animal experiments proved that Bzl-D could effectively inhibit tumor growth in the xenograft model of CRC.
Discussion
Various extracts of S. barbata D. Don (e.g., ethanolic extracts, chloroform-based extracts, and flavonoid extracts) have exhibited significant antitumor activity against CRC (Chen et al., 2017; Gao et al., 2014; Lin et al., 2014). It was reported the ethanolic extracts of S. barbata D. Don were able to induce apoptosis of CRC cells and inhibit tumor angiogenesis by modulation HH pathway (Wei et al., 2012). Moreover, S. barbata D. Don extracts were co-administered with 5-FU, a commonly used agent in the treatment of a diversity of cancers, and showed much superior efficacy on CRC than used it alone (Xu et al., 2013). Based on the outstanding performance in inhibiting the growth of CRC cells, we believe that S. barbata D. Don has a high potential for the treatment of CRC.
We isolated and purified the 85% aqueous ethanol of S. barbata D. Don, and obtained a diterpene mixture enriched with Bzl-D. By analysis of UPLC-qTOF-MS, some diterpenes have been identified, which of them have been confirmed to exert favored anti-cancer efficacy. For example, scutebarbatine A can induce apoptosis in cancer cells through the intrinsic apoptotic pathway to achieve anticancer effects (Feng et al., 2021; Yang et al., 2014), and scutebarbatine B at a low concentration inhibits cancer cell proliferation by regulating the MAPK signaling pathway (Hao et al., 2022). In our in vitro assay, Bzl-D dose-dependently inhibited the growth of different CRC cell lines, with HT-29 cells being the most sensitive cell line. In addition, Bzl-D inhibited HT-29 cell metastasis and induced apoptosis. Based on these external manifestations, we explored the mechanisms responsible for their generation.
The critical role of the HH pathway in the developmental changes of CRC has been previously reported. Numerous studies have recognized that the activation of the HH pathway in CRC can occur through various mechanisms, including genetic alterations, signaling cross-talk, and changes in the tumor microenvironment (Hung et al., 2014; Šuštić et al., 2018). SHH, one of the members of the HH signaling pathway, binds with PTCH to activate the classical HH pathway and blocks PTCH-induced apoptosis, and its overexpression has been positively correlated with CRC tumorigenesis (Bian et al., 2007; Tiwari et al., 2018), making it a promising target for HH pathway-associated cancers (Bissey et al., 2020).
After Bzl-D treatment, HH pathway protein expression showed a significant down-regulation trend, we speculated that Bzl-D may cause many external manifestations of cancer cells by inhibiting the activation of the HH pathway. It is worth noting that in targeted therapies for CRC, many inhibitors of downstream and midstream targets of the HH pathway have already been marketed, but there is still plenty of room for exploration of upstream target inhibitors. The results of CETSA demonstrated that Bzl-D may target SHH to inhibit the HH signaling pathway, which provides additional evidence to support our hypothesis.
The results of this study provide an important reference for the development of new anticancer drugs and open up new ideas for the development of new targeted therapeutic drugs. Nevertheless, there are still some flaws in the overall experimental design, and more trials are required to accurately validate the relationship between Bzl-D and SHH targets, and more experiments and data are required to confirm that the action of Bzl-D in mice is indeed related to the HH pathway, and these aspects of our work will be the focus of our subsequent work.
Conclusion
This research suggested that Bzl-D could specifically target SHH and inhibit the expression of proteins connected with the HH pathway in vitro, thus affecting the progress of cancer cells. In vivo, experiments further verified the pronounced inhibitory effect of Bzl-D on tumor growth. These findings provide a scientific basis for the rational development and utilization of S. barbata D. Don, as well as the discovery of novel antitumor drugs with potent effects and well-defined components for the treatment of CRC.
Abbreviations
Bzl-D: Diterpenes of Scutellaria barbata D. Don; CETSA: Cellular thermal shift assay; CRC: Colorectal cancer; Ctrl: Control H&E: hematoxylin–eosin; GLI: Glioma-associated oncogene; HH: Hedgehog; PBS: Phosphate-buffered saline; PTCH: Patched receptor; SHH: Sonic Hedgehog; SMO: Smoothened; UPLC-qTOF-MS: Ultra-performance liquid chromatography-quadrupole time-of-flight mass spectrometry.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Animal experiments have passed the examination of the Animal Ethics and Research Committee of Zhejiang (Approval No. ZJCLA-IACUC-20020035).
Funding
This research was funded by the Medical Health Science and Technology Project of Zhejiang Provincial Health Commission (2021KY638 and 2021KY644).
Informed Consent
Animal experimental procedures and animal care were carried out by the ethical standards of the Ethics Committee of Zhejiang Province to maximize animal welfare and minimize suffering.
