Abstract
Nothapodytes nimmoniana (J. Graham) Mabb., commonly known as Narkya, holds immense potential as a medicinal plant, exhibiting a wide array of pharmacological activities. This review provides a comprehensive overview of the methods used for extracting bioactive compounds from different plant parts, along with an exploration of their diverse pharmacological activities. The plant’s significance stems from its high content of camptothecin (CPT), a potent anti-cancer alkaloid. We delve into the plant’s anti-microbial, antioxidant, anti-tumor, anti-helminthic, anti-malarial, and anti-inflammatory properties. Additionally, we highlight the challenges faced by N. nimmoniana (J. Graham) Mabb., due to overexploitation and its endangered status. To address these concerns, the review underscores the importance of sustainable conservation strategies and the implementation of advanced techniques like plant tissue culture and endophyte production for increased CPT production. This holistic analysis not only sheds light on the plant’s multifaceted benefits but also underscores the need for responsible utilization and conservation efforts.
Keywords
Introduction
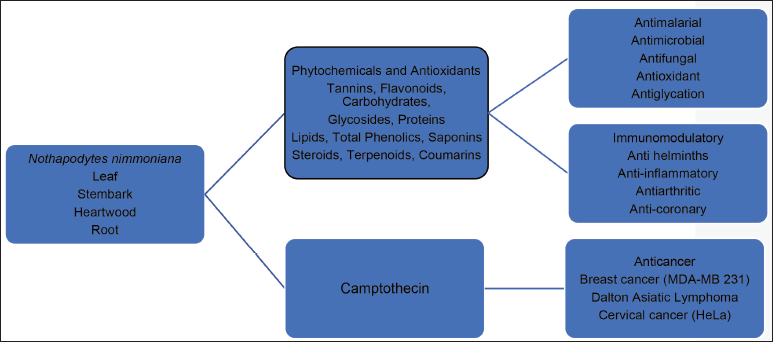
Plant Information
Nothapodytes nimmoniana (J. Graham) Mabb., is a compact tree that reaches a height of 4–10 m and features a spreading crown. This small shrubby tree boasts broad, dark green leaves and flowers, as detailed in the study by Khan et al. (2012). Indigenous to several Asian countries including Sri Lanka, China, Southeast Asia, Taiwan, Philippines, and India, this plant holds a notable presence. Within India, it is exclusively found in the Western Ghats regions of Maharashtra, Tamil Nadu, Goa, Kerala, Assam, Jammu and Kashmir. In these areas, it goes by various local names such as Narkya, Kalgur, Ghanera, and Amruta (referred to as Marathi names), as well as Foetid Mappia in English, as documented by Namdeo et al. (2010) and Khan et al. (2012). The reproductive nature of species is quite diverse, exhibiting a range of breeding types. These include male, female, hermaphrodite, monoecious, andromonoecious, gynomonoecious, and trimonoecious trees, as expounded upon in the study by Khan et al. (2012).
According to the prioritized plant’s brochure by the additional principal chief conservator of forests, the scientific classification of N. nimmoniana (J. Graham) Mabb. places it in the Kingdom Plantae, and its family is identified as Icacinaceae. This shrub, characterized by its dark green leaves and flowers, is predominantly discovered within the Western Ghats and Himalayan regions of India. The plant is particularly notable for containing the anti-cancer compound camptothecin (CPT), which serves as its active component (Figure 1). While the synthesis of CPT has been successfully achieved in lab settings, its commercial viability remains limited (Isah & Mujib, 2015). As a result, the plant’s natural reservoir is tapped into to meet the demand, with India emerging as the primary supplier to markets including the USA, Japan, and Spain. It is noted that while around 1,000 tons of this material are officially reported and exported, the actual quantity of unreported material is estimated to be twice as much (Patwardhan, 2006). This unregulated harvesting has significantly contributed to the alarming decline in the population of N. nimmoniana (J. Graham) Mabb. species. To comprehend its geographic variations, population studies on N. nimmoniana (J. Graham) Mabb. plants have been conducted across a wide expanse of the Western Ghats (Ankad et al., 2015; Suhas et al., 2007).
Applications of Nothapodytes nimmoniana.
Plant Tissue Culture of N. nimmoniana (J. Graham) Mabb.
Sundravelan et al. (2004) successfully cultivated CPT from callus cultures of N. nimmoniana (J. Graham) Mabb. leaves. This cultivation took place on a Murashige and Skoog medium supplemented with picloram, N6-benzyladenine, and gibberellic acid. The analysis of the cultured tissue unveiled the presence of the cytotoxic quinoline alkaloid CPT and its derivative (Sundravelan et al., 2004).
Cryopreservation of embryonic axes has emerged as an approach for the short-term conservation of this species. Scaling up the production of CPT from N. nimmoniana (J. Graham) Mabb. using bioreactors holds the potential for meeting the demand of the pharmaceutical market and reducing the strain on natural populations. In their study, Radha et al. (2010) assessed the current challenges and progress in CPT production from N. nimmoniana (J. Graham) Mabb. using in vitro techniques.
Puri et al. (2005) documented the immunomodulatory activity of an extract from the novel fungal endophyte Entrophospora infrequens, which was isolated from N. nimmoniana (J. Graham) Mabb. Their investigation focused on assessing the immunomodulatory potential of chloroform and methanolic extracts of E. infrequens through in vitro and in vivo (in BALB/c mice) experiments. Remarkably, the endophyte E. infrequens was found to produce CPT, particularly in the chloroform extract. This study marked the first identification of immunomodulatory potential in this novel CPT-producing endophyte originating from N. nimmoniana (J. Graham) Mabb.
Mohinudeen et al. (2021) explored the use of somatic embryogenesis to facilitate clonal propagation and alkaloid production, revealing proteome expression changes that indicate higher metabolic and physiological processes in embryogenic callus cultures, offering insights into optimizing culture conditions for sustainable CPT production and conservation.
N. nimmoniana (J. Graham) Mabb. is considered a valuable plant source for large-scale extraction of the anti-cancer alkaloid CPT, contributing to a billion-dollar annual trade. Isah et al. (2022) studied the somatic embryogenesis showing promise in clonal propagation and alkaloid production, with a study revealing proteome expression changes indicating higher metabolic and physiological processes in embryogenic callus cultures, providing insights for optimizing culture conditions to address the endangered status of N. nimmoniana (J. Graham) Mabb. while meeting industrial demand for CPT.
Plant Indications
This plant displays a multitude of pharmacological properties encompassing anti-cancer, anti-HIV, anti-malarial, antibacterial, antioxidant, anti-inflammatory, and antifungal activities. Furthermore, it has demonstrated efficacy in treating conditions such as anemia and various sarcomas, including lung, breast, and uterine cervical cancers. The plant’s potential extends to both in vitro and in vivo settings, exhibiting considerable effectiveness against an array of cancer types. Notably, its profound impact on breast cancer cell lines has been documented by Dharmalingam et al. (2014). Ongoing research is focused on exploring the untapped metabolites within N. nimmoniana (J. Graham) Mabb., adding to its therapeutic potential (Table 1).
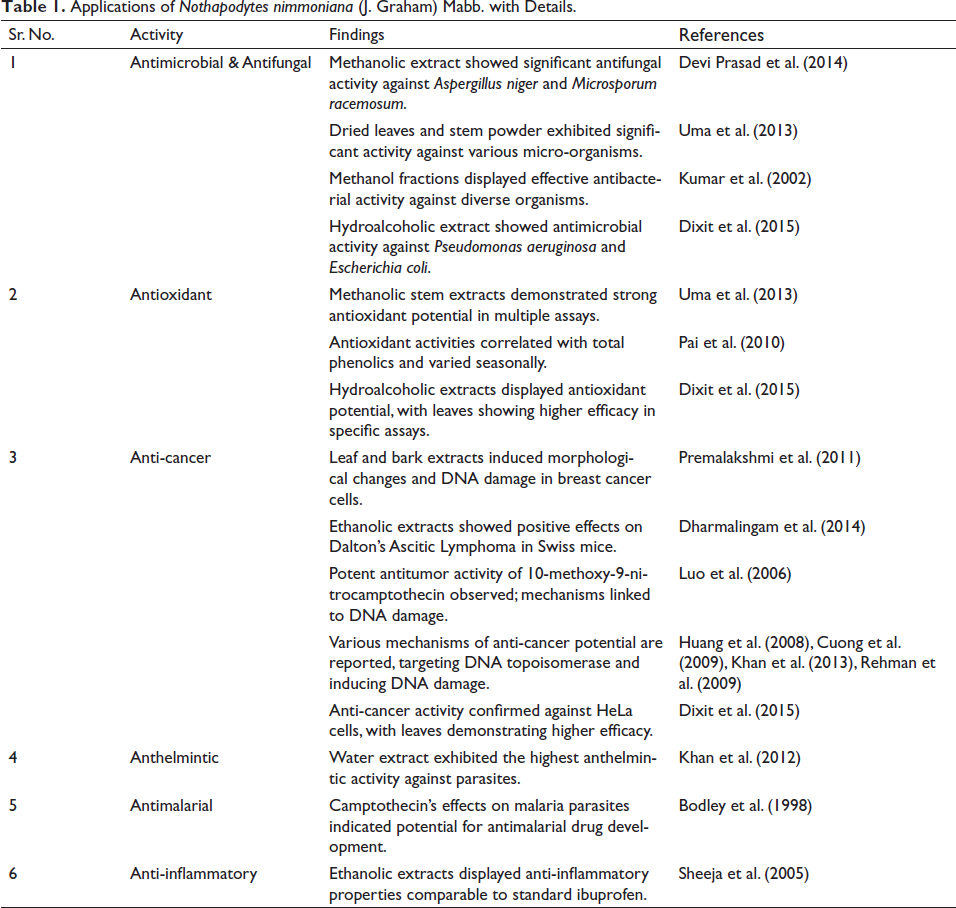
Applications of Nothapodytes nimmoniana (J. Graham) Mabb. with Details.
Active Component
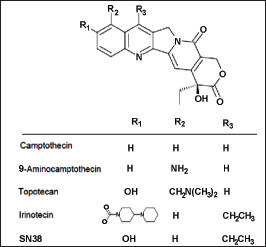
The plant contains an alkaloid known as CPT, renowned for its significant role as a commercial product with potent anti-cancer properties. This compound was initially extracted from Camptotheca acuminata, a tree native to China, which has been traditionally employed in Chinese Medicine for treating cancer (Isah & Mujib, 2015). While CPT exhibited promising anti-cancer effects in early clinical trials, its practical use was hindered by challenges such as limited solubility and a high incidence of adverse drug reactions. To overcome these drawbacks, several derivative compounds were developed to enhance the therapeutic advantages of CPT, yielding favorable outcomes (Figure 2). Notably, two analogs of CPT, namely topotecan and irinotecan, have gained approval and are now utilized in cancer chemotherapy (Isah & Umar, 2019).
CPT belongs to the category of cytotoxic quinoline alkaloids. Its primary mode of action involves the inhibition of the DNA-modifying enzyme known as topoisomerase I (topo I). This enzyme plays a pivotal role in relieving torsional stress within double-stranded DNA molecules (Svejstrup et al., 1991). CPT effectively enhances the binding of topo I to DNA. When combined with ongoing DNA synthesis, this interaction can lead to the creation of potentially lethal damage within the DNA structure.
Extraction Methods
The researchers conducted extraction experiments utilizing various methods, including stirring extraction, Soxhlet extraction, ultrasonic extraction, and microwave-assisted extraction (MAE). The quantification of CPT and 9-methoxycamptothecin (9-Me-CPT) extraction percentages from N. nimmoniana (J. Graham) Mabb. was performed using high-performance liquid chromatography (HPLC). Significantly higher extraction percentages of CPT (2.67%, w/v) and 9-Me-CPT were achieved through the application of the MAE technique using Methanol (90%, v/v) in comparison to ethanol (90%, v/v) (Fulzele & Satdive, 2005). The outcomes indicated that the MAE technique exhibited greater efficiency in extraction and notable time savings compared to other methods.
In a study by Upadhya et al. (2014), the impact of varying temperature ranges while maintaining a consistent solvent pressure of ethanol and methanol (0.5 mg mL−1) on the extraction efficiency of the alkaloid CPT from the stem bark of N. nimmoniana (J. Graham) Mabb. was explored (Upadhya et al., 2014). The results demonstrated that the highest CPT yield was achieved at 80°C, highlighting the correlation between increased temperature and enhanced CPT extraction.
Hsiao et al. (2008) employed direct injection capillary electrophoresis to determine CPT in dimethyl sulfoxide (DMSO) extracts of N. nimmoniana (J. Graham) Mabb. The hydrophobic compound was extracted from plant tissue using DMSO, a water-miscible organic solvent, at an elevated temperature (60°C). The resulting extract was directly injected into the separation capillary, and CPTs were successfully separated and quantified within a short migration time. This approach was deemed suitable for monitoring CPT levels during the cultivation of the medicinal plant (Svejstrup et al., 1991).
Additionally, Puri et al. (2005) reported the separation of 9-Me-CPT and CPT from N. nimmoniana (J. Graham) Mabb. using semi-preparative HPLC. The purity of the isolates was confirmed through physicochemical assessments and liquid chromatography-mass spectrometry (LC-MS).
Characterization of N. nimmoniana (J. Graham) Mabb.
A group of researchers employed the GC-MS technique to identify the phytochemicals present in the methanolic leaf extract of N. nimmoniana (J. Graham) Mabb. A total of 26 compounds were detected through GC-MS analysis. The methanolic leaf extract was found to be rich in phenolics, fatty acids, flavonoids, and antioxidants. The primary compound identified was 9,12,15-octadecatrienoic acid, [Z, Z, Z], commonly known as linolenic acid, which possesses properties such as anti-inflammatory, anti-arthritic, anti-coronary, and anti-cancer effects (Uma et al., 2013).
In a study conducted by Gayakwad et al. (2020), experiments were conducted to showcase the potential of N. nimmoniana (J. Graham) Mabb. By observing changes in the protein and metabolite levels. Cancer progression stages were monitored before and after treatment with stem and leaf extracts of N. nimmoniana (J. Graham) Mabb. On HeLa cells. The study revealed that cancer-specific markers significantly decreased following treatment with the leaf methanolic extract compared to the stem bark extract, even in comparison to commercial CPT. Proteins such as Zinc finger protein 788, transcription factor RFX3, and specific metabolites like 3-hydroxysuberic acid were characterized as cancer-specific markers. The study also indicated that the crude extract, in combination with phytochemicals, yielded more favorable results than pure CPT.
Kavitha et al. (2010) conducted chemical profiling of 48 individual plants of N. nimmoniana (J. Graham) Mabb. From different regions in the Western Ghats of Karnataka. Ethanol (80%) was used for extraction, and CPT and 9-Me-CPT were analyzed using reverse-phase HPLC. The root bark yielded the highest amount of CPT and 9-Me-CPT (0.29% ± 0.21%), followed by stem bark (0.09% ± 0.06%). The stem exhibited a relatively lower content (0.12% ± 0.08%), followed by leaves (0.04% ± 0.02%). A positive correlation between CPT and 9-Me-CPT was observed in all plant parts. The relationship between CPT and 9-Me-CPT accumulation was linear in the stem, stem bark, and root bark, but not in leaves.
Sharma et al. (2012) conducted a preliminary pharmacognostical screening of N. nimmoniana (J. Graham) Mabb. Stem to authenticate the plant and distinguish CPT from other species. Various physicochemical parameters, percentage yield, extractive value, and chemical evaluation were analyzed as per WHO recommendations. Authentic phytochemical procedures were examined using HPLC. Qualitative tests indicated the presence of alkaloids, carbohydrates, saponins, steroids, terpenoids, phenolics, coumarins, and fixed oil.
Another study investigated the alkaloid content of CPT from different parts of N. nimmoniana (J. Graham) Mabb. Collected from October to February, utilizing HPLC analysis. The study emphasized that CPT synthesis varies in different parts of N. nimmoniana (J. Graham) Mabb., and the content fluctuates periodically.
Namdeo et al. (2010) concluded that methanolic extracts of different parts of N. nimmoniana (J. Graham) Mabb. Exhibited maximum phenolic content, antioxidant activity, and anti-lipid peroxidation effects in fruits.
Patil et al. (2014) carried out the phytochemical evaluation, identification, isolation, purification, and extraction procedure of CPT. Soxhlet extraction was performed, and thin-layer chromatography (TLC) fingerprinting was compared with standard CPT. The compound’s unique functional group was confirmed through Fourier-transform infrared spectroscopy spectrophotometric data. TLC bio-autography techniques were used to correlate the anti-cancer ability with the antioxidant and antibacterial potential of the plant, indicating its multifaceted chemo-preventive properties.
Dixit et al. (2015) quantified various phytochemical components of the hydroalcoholic extract of N. nimmoniana (J. Graham) Mabb., including tannins, flavonoids, glycosides, carbohydrates, proteins, lipids, and total phenolics. The stem bark extract exhibited higher quantities of these components compared to the leaf extract.
The same researchers conducted a glycation assay to assess advanced glycation end-product (AGEs) production, which was reduced in the presence of stem bark and leaf extracts. The quenching assay suggested that the extracts bound to proteins induced a conformational change, impacting the production of AGEs, which are implicated in various diseases.
Quantitation of CPT in Different Parts of N. nimmoniana (J. Graham) Mabb.
Observations have indicated that the plant N. nimmoniana (J. Graham) Mabb. From Mahabaleshwar in Maharashtra exhibits the highest concentration of CPT in its roots, while in general, the stem from Patan in Maharashtra displayed the highest CPT content. The study provided a rough estimate that the concentration of CPT in both stem and roots is significantly higher than that found in fruits and leaves. The researchers have underscored the notable influence of geographical and climatic conditions on the CPT content in N. nimmoniana (J. Graham) Mabb (Namdeo, 2010).
Yamazaki et al. (2003) reported the isolation of CPT-related alkaloids from methanolic extracts of Ophiorrhiza pumila, C. acuminata, and N. nimmoniana (J. Graham) Mabb. The plants were characterized and identified using reverse-phase HPLC combined with online photodiode array detection and electrospray-ionization ion-trap mass spectrometry.
According to Padmanabha et al. (2006), the root barks of N. nimmoniana (J. Graham) Mabb. Were found to contain a higher amount of CPT compared to the content in the root wood and stem bark. Leaves, on the other hand, exhibited the lowest CPT content. The variation in CPT content between male and female plants was not significant. The research indicated that the observed variations might be attributed to the genetic makeup and environmental factors of the plants.
Since N. nimmoniana (J. Graham) Mabb. is the sole source of the alkaloid CPT, the increasing demand is placing stress on the natural plant population. The researchers have proposed high-yielding clonal multiplication and in vitro development as alternative methods to alleviate this pressure on the plant. It was also noted that the variance in CPT content across plant parts is not dependent on the plant’s age or geographical distribution (Suhas et al., 2007).
Dighe et al. (2007) introduced a sensitive and dependable high-performance thin-layer chromatographic method for quantifying CPT in the dry stem powder of N. nimmoniana (J. Graham) Mabb. The method utilized a methanolic extract of the dry powder chromatographed on silica gel 60 F254 plates with a mobile phase of toluene—acetonitrile—glacial acetic acid (6.5 + 3.5 + 0.1 v/v). Densitometry scanning in fluorescence mode at λ = 366 nm was used for detection and quantification, employing a mercury lamp. The accuracy of the method was validated through recovery determination using the standard addition approach, yielding a recovery rate of 99.49%. The average CPT content in the powder was found to be 0.059%. This method proved to be rapid, straightforward, and precise.
Ramesha et al. (2008) disclosed that the highest levels of CPT (approximately 0.3%, w/w) were identified in N. nimmoniana (J. Graham) Mabb. Using both HPLC and LC-MS techniques. They also unveiled the presence of minor CPTs, including 10-hydroxy CPT, in the stem and root extracts of the tree.
The hydroalcoholic extracts of N. nimmoniana (J. Graham) Mabb. Collected from Mulshi, Pune demonstrated the greatest concentration of CPT in the stem bark (303.81 µg/mL), as determined by Dixit et al. (2015) through a colorimetric assay (A252). This result was corroborated by LC-MS conducted in +ESI mode. The LC-MS analysis revealed a mass of 349.1743 m/z for CPT with major product ions at 231.0965 and 160.0946 m/z, which precisely matched the standard CPT (molecular weight 348.352 g/mol).
Pharmacological Studies
Antimicrobial and Antifungal Activity
Recently, researchers conducted a study to assess the antibacterial and antifungal properties of aqueous, diethyl ether, and methanol extracts of N. nimmoniana (J. Graham) Mabb. Leaves. This investigation targeted Staphylococcus aureus, Escherichia coli, Aspergillus niger, and Microsporum racemosum, using the well diffusion method. Notably, the methanolic extract of N. nimmoniana (J. Graham) Mabb. Exhibited significant antifungal effects against A. niger and M. racemosum (Devi Prasad, 2014).
Furthermore, another group of researchers explored the antimicrobial potential of dried leaves and stem powder of N. nimmoniana (J. Graham) Mabb. They tested these extracts against a range of micro-organisms, including Bacillus thuringiensis, S. aureus, E. coli, Serratia marcescens, Proteus vulgaris, Salmonella paratyphi as well as fungal species like Paecilomyces lilacinus, Mucur spp., Trichoderma viride, Azospirillum lipofererum, Verticillum lecanii, and Penicillium spp. The extracts demonstrated notable activity against the tested organisms (Uma et al., 2013).
Kumar et al. (2002) successfully investigated the antibacterial potential of petroleum ether, chloroform, and methanol extracts from N. nimmoniana (J. Graham) Mabb. leaves and stems. Among these fractions, the methanol extracts exhibited the highest efficacy against all tested organisms.
In a separate study, Dixit et al. (2015) assessed the antimicrobial properties of the hydroalcoholic extract of N. nimmoniana (J. Graham) Mabb. using a tetrazolium microplate antimicrobial assay. The leaf extract displayed antimicrobial activity against Pseudomonas aeruginosa (with a minimum inhibitory concentration of 50 mg/mL), while the stem bark extract exhibited antimicrobial effects against E. coli (MIC at 25 mg/mL) and P. aeruginosa (MIC at 25 mg/mL).
Antioxidant Activity
Maximum antioxidant activity was observed in the methanolic extracts derived from the stem of N. nimmoniana (J. Graham) Mabb. This activity was assessed through various in vitro models including DPPH (1,1-diphenyl-2-picryl hydrazine), ABTS+ (2,2′-azinobis-(3-ethylbenzothiazoline-6-sulfonic acid)) assays, ferrous iron chelating ability, and reducing power (Uma et al., 2013).
Pai et al. (2010) established a strong correlation between the antioxidant activities of N. nimmoniana (J. Graham) Mabb. and its total phenolic content using assays such as di(phenyl)-(2,4,6-trinitrophenyl) iminoazanium (DPPH) and Ferric reducing ability of plasma (FRAP). This correlation held true across all seasons, with the highest antioxidant activity observed during summer and the lowest during the monsoon. During the winter month, total phenolic content exhibited a particularly strong correlation with DPPH and FRAP antioxidant assays.
In the work conducted by Dixit et al. (2015), antioxidant assays were performed to assess the free radical scavenging potential of hydroalcoholic extracts from N. nimmoniana (J. Graham) Mabb. The IC50 values (in µg/mL) for different assays were determined for stem bark and leaf samples. Specifically, the values were 17.01 and 21.93 µg/mL for the ABTS assay, 18.82 and 16.74 µg/mL for the DPPH assay, 336.04 and 409.03 µg/mL for the FRAP assay, and 18.56 and 34.72 µg/mL for the thiobarbituric acid reactive substances (TBARS) assay. Notably, the results indicated that the leaf extract exhibited comparatively higher antioxidant potential than the stem bark extracts in the DPPH and TBARS assays.
Anti-cancer Activity
Extracts obtained from various parts of the N. nimmoniana (J. Graham) Mabb. plants have exhibited the ability to induce cell death in breast cancer cell lines MDA-MB 231. At IC50 concentrations of 450 µg/mL for leaves and 500 µg/mL for bark, these extracts led to morphological changes, mitochondrial potential loss, altered protein expression levels, and significant DNA damage (Premalakshmi et al., 2012).
A recent study by Dharmalingam et al. (2014) explored the impact of ethanolic extracts derived from the heartwood and bark of N. nimmoniana (J. Graham) Mabb. on Dalton’s Ascitic Lymphoma (DAL) in Swiss mice. Their findings revealed enhanced life span, reduced cancer cell count, decreased tumor weight, and stabilized hematological parameters post-extract treatment. The ethanolic extracts of heartwood and bark proved to be effective against DAL.
Luo et al. (2006) highlighted the potent antitumor activity of 10-methoxy-9-nitrocamptothecin. This compound’s ability to elevate cellular DNA damage accumulation aligned with its high cytotoxic potency. This observation suggested the involvement of cell cycle regulation in the anti-cancer effects of 10-methoxy-9-nitrocamptothecin, warranting further development as an anti-cancer agent.
Huang et al. (2008) noted that CPT induced arrest at the S phase or G2-M phase and triggered the homologous recombination repair pathway within tumor cells.
Cuong et al. (2009) demonstrated the capacity of the plant alkaloid CPT to induce DNA damage by specifically targeting DNA topoisomerase, effectively obliterating a wide range of tumors.
Khan et al. (2013) reported the discovery of a novel naturally occurring alkaloid, acetylcamptothecin, along with 17 known compounds. Among these, scopoletin, CPT, 9-O-methoxycamptothecin, and O-acetylcamptothecin exhibited substantial cytotoxic activity.
Rehman et al. (2008) explored the in vitro cytotoxicity of an endophytic fungus isolated from N. nimmoniana (J. Graham) Mabb. The ethyl acetate fraction from the endophyte displayed significant growth inhibition against all cell lines tested.
In a recent investigation by Dixit et al. (2015), the anti-cancer potential of N. nimmoniana (J. Graham) Mabb. leaf and stem bark extracts were assessed using HeLa cell lines cultivated in Minimal Essential Medium. The MTT cell cytotoxicity assay yielded IC50 values of 178 µg/µL for leaves, 1540 µg/µL for stem bark, and 29 µg/µL for standard CPT. The study indicated that leaves exhibited a lower IC50 compared to stem bark extracts, emphasizing their efficacy as an anti-cancer drug against cancerous cells. Morphological changes in HeLa cell lines were also observed through Inverted Microscopy following the MTT assay.
Anthelmintic Activity
Conducting phytochemical screening and assessing the anthelmintic potential of N. nimmoniana (J. Graham) Mabb., Khan et al. (2012) determined that extracts derived from chloroform, methanol, and water exhibited anthelmintic activity. Notably, the water extract demonstrated the highest level of activity.
Anti-malarial Activity
In their study, Bodley et al. (1998) investigated the impact of CPT, a potent and selective inhibitor of topo I, on erythrocytic malaria parasites in vitro. Their research on Plasmodium falciparum revealed that CPT led to the formation of protein-DNA complexes, which in turn hindered nucleic acid biosynthesis and exerted cytotoxic effects. These findings substantiated the notion that topo I could be considered a susceptible target for the development of novel antimalarial drugs.
Anti-inflammatory Activity
In their research, Sheeja et al. (2005) documented the anti-inflammatory potential of N. nimmoniana (J. Graham) Mabb. using the carrageenan-induced hind paw oedema method in rats. The effects of the extracts were evaluated against both a control group and the standard drug ibuprofen. Administration of all substances was done orally. Comparatively, the anti-inflammatory efficacy of the ethanolic extract was more pronounced than that of the petroleum ether extract. Specifically, the ethanolic extract at a dosage of 200 mg/kg exhibited a significant reduction in inflammation (p < 0.01), which was comparable to the effect of the standard ibuprofen.
Discussion
The preceding review of literature encompasses a range of extraction methods and biochemical assays employed in the assessment of N. nimmoniana (J. Graham) Mabb. The primary emphasis lies on the collective activities demonstrated by the entirety of the plant extract, rather than being solely centered on the alkaloid component, CPT. It can be deduced that the diverse activities exhibited are an amalgamation of various phytochemicals, secondary metabolites, and notably, the extensively discussed CPT and its derivatives. In terms of extraction efficiency, the microwave-assisted method emerged as the most effective among the investigated techniques.
Throughout the research, the compound CPT has taken the spotlight, which is unsurprising considering its stature as a commercial chemotherapeutic product against cancer. From this comprehensive analysis, one can conclude that N. nimmoniana (J. Graham) Mabb. boasts a multifaceted nature, exhibiting a spectrum of activities ranging from antifungal to anti-cancer properties. Notably, the whole extract demonstrated anti-cancer efficacy against specific types of cancer such as Breast cancer (MDA-MB 231), Dalton Asiatic Lymphoma, and Cervical cancer (HeLa), underscoring its potential across these malignancies.
Unfortunately, the extensive utilization of this versatile plant for commercial purposes, coupled with rampant smuggling, has led to a decline in its population. This drastic exploitation has caused N. nimmoniana (J. Graham) Mabb. to become listed as an endemic species. To counteract this decline, intensive research is being conducted to explore novel techniques for enhancing production. Areas of active investigation include plant tissue culture, cryopreservation, and endophyte production, all aimed at bolstering the propagation of this valuable plant.
Conclusion
N. nimmoniana (J. Graham) Mabb.’s multifunctional properties, particularly its high CPT content, make it a plant of significant interest for pharmaceutical and medical research. This review has provided a comprehensive understanding of its extraction methods, various pharmacological activities, and the potential threats to its survival due to overexploitation. Addressing these challenges requires a concerted effort toward sustainable conservation methods and advanced production techniques. By embracing responsible utilization and exploring innovative cultivation strategies, N. nimmoniana (J. Graham) Mabb. can continue to contribute to the field of medicine while ensuring its long-term survival.
Future Prospects
As the demand for CPT grows, developing efficient and sustainable production methods will be crucial. Plant tissue culture and endophyte-based approaches offer promising avenues to meet this demand while minimizing the pressure on natural populations. Furthermore, exploring the plant’s interactions with its environment and genetic factors affecting CPT production can guide targeted cultivation efforts. By integrating these approaches and promoting awareness about the plant’s ecological importance, we can pave the way for a brighter future for N. nimmoniana (J. Graham) Mabb. and its potential contributions to medicine and research.
Abbreviations
9-Me-CPT: 9-methoxycamptothecin; AGEs: Advanced glycation end products; CPT: Camptothecin; DAL: Dalton’s Ascitic Lymphoma; DMSO: Dimethyl sulfoxide; DPPH: Di(phenyl)-(2,4,6-xstrinitrophenyl) iminoazanium; FRAP: Ferric reducing ability of plasma; HPLC: High-performance liquid chromatography; LC-MS: Liquid Chromatography-Mass Spectrometry; MAE: Microwave-assisted extraction; MTT: (3-(4,5-dimethylthiazol-2-yl)-2,5-Diphenyltetrazolium Bromide); N. nimmoniana (J. Graham) Mabb.: Nothapodytes nimmoniana (J. Graham) Mabb.; TBARS: Thiobarbituric acid reactive substances; TLC: Thin-layer chromatography; topo I: topoisomerase I
Footnotes
Acknowledgments
The authors would like to acknowledge the DG, CCRAS, New Delhi for the moral support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
