Abstract
Background
Panax japonicus (P. japonicus) belongs to the Panax genus of Araliaceae and is used as medicine mainly with the bamboo whiplike rhizome, which has the functions of dispelling blood stasis and hemostasis, attenuating swelling and pain, eradicating phlegm and relieving cough, tonifying, and strength.
Objectives
This review intends to summarize the chemical constituents and pharmacological activities of P. japonicus to provide a scientific and systematic basis for better utilization of its rational applications.
Methodology
The literature was searched using PubMed, Baidu Scholar and Science Hub search engines.
Results
The chemistry components isolated from P. japonicus are mainly saponins, besides also including polysaccharides, amino acids, volatile oils, inorganic elements, etc. P. japonicus exhibits extensive pharmacological activities including anti-inflammatory, antioxidant, lipid lowering activities, and therefore show extensive protective effects on the central nervous system, cardio-cerebrovascular system, digestive system.
Conclusion
This article included a thorough summary of the botany, phytochemistry, and pharmacology of P. japonicus and provided evidence for its further research and clinical applications.
Introduction
Panax japonicus C. A. Meyer belongs to the Araliaceae Panax family in the plant kingdom (China Pharmacopoeia Commission, 2020). At an elevation of 1,800–2,600 m, it thrives in valley broadleaf forests and is distributed in the south of the Yangtze River basin, east to Japan, and west to the Hengduan Mountains (Jia & Zhang, 2016; Lin et al., 2007). The typical characteristics of P. japonicus are the transverse rhizomes in the shape of bamboo whips which can be used as medicine. They have the functions of dispelling blood stasis and hemostasis, attenuating swelling and pain, eradicating phlegm and relieving cough, tonifying, and strength (Song, 1999).
The advancement of technology in separation and identification has allowed for the successful isolation and identification of over 100 compounds (College of Traditional Chinese Medicine of Nanjing Pharmaceutical University, 1976; He et al., 2019; Ohtani et al., 1989; Wang, 2020; Wu & Yi, 1992; Xiang, 2009; Yang et al., 2014). Modern pharmacological research on this plant has demonstrated that its primary ingredients, which include volatile oils, polysaccharides, amino acids, and saponins, have extensive pharmacological effects on the central nervous system, cardio-cerebrovascular system, digestive system, immune system, and so on.
This review intends to summarize the chemical constituents and pharmacological activities of P. japonicus to provide a scientific and systematic basis for better utilization of its rational applications.
Methodology
The literature search was conducted using PubMed, Baidu Scholar, and Science Hub search engines. The search was performed using keywords such as Panax japonicus and phytochemistry, Panax japonicus and pharmacological activities, Panax japonicus and cardioprotection, Panax japonicus and hepatoprotective, Panax japonicus and metabolic system, Panax japonicus and obesity, etc. In total, approximately 75 articles on 118 compounds from 1976 to 2023 were compiled. This article aimed to compile literature focused solely on P. japonicus.
Botany
P. japonicus is a perennial herbaceous plant that has transverse rhizomes and grows to a height of 30–60 cm. The petiole is slender and 4–9 cm long. The central leaf is obovate or elliptic obovate with 5–15 cm long, 2–5.5 cm wide, base cuneate, rounder or subcordate, margin serrate, double serrate or incised-serrate, glabrous above and sometimes pilose below. Umbel: single terminal, rare few branches; the peduncle is about 12 cm. The flowers are small and in clusters of 20–50. The calyx is cup-shaped with five parts. There are five pale yellow-green petals and an oval triangle. The drupe is berry-shaped, and red when ripe and often black at the apex. May through June is blooming time, while July through August is fruiting time (Institute of the Academy of Medical Sciences of China, 1979), as shown in Figure 1.

Chemical Constituents
This review identifies over 120 chemical components isolated from P. japonicus based on the literature, including saponins, saccharides, amino acids, volatile oils, and inorganic elements.
Saponins
Saponins are considered to be the main bioactive ingredients of P. japonicus, and their contents vary with different producing areas. It was reported that the content of saponins in the rhizome of P. japonicus was about 5% in China and that in Japan was 23.6%. Up to now, 118 triterpenoid saponins have been isolated from P. japonicus (Table 1), which were mainly oleanane type and dammarane type. They could be classified into oleanolic acid (OA), ursolic acid (UA), dammarenediol II (DD-II), protopanaxadiol (PPD), protopanaxatriol (PPT), ocotillol (OCT), and miscellaneous subtypes, as shown in Figures 2 and 3.
The Constituents from P. japonicus.

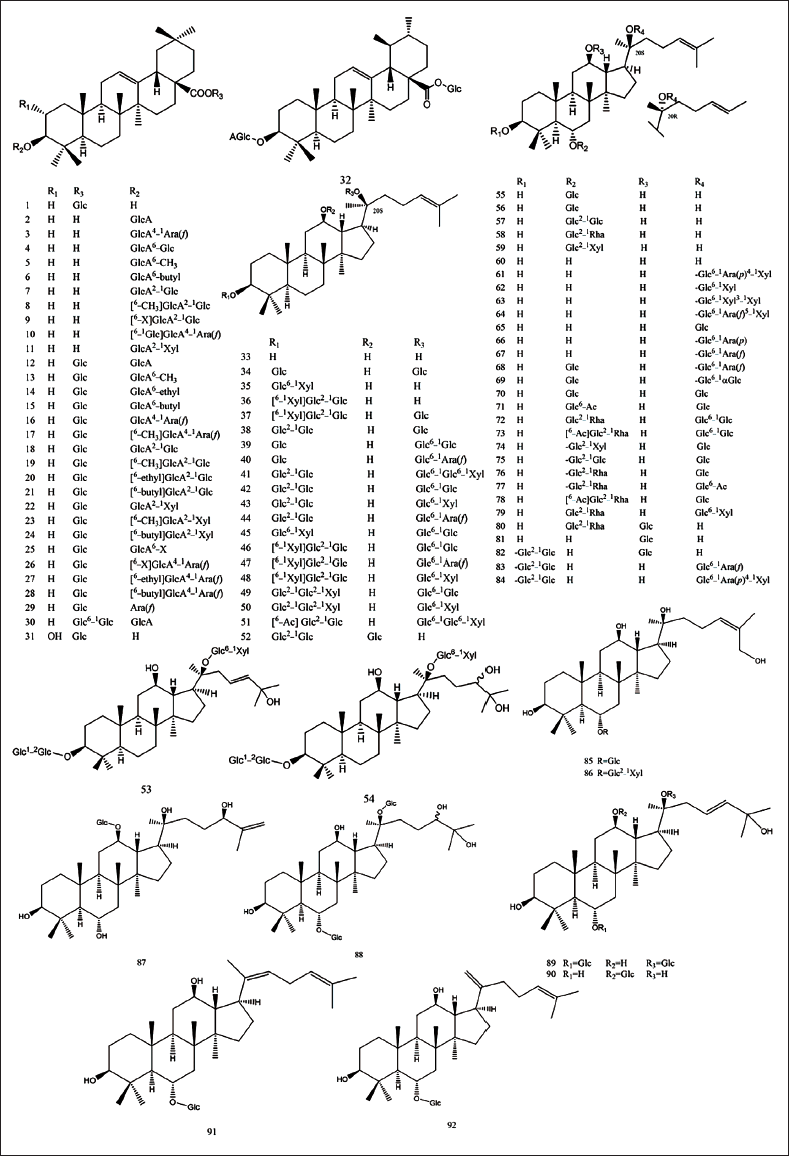
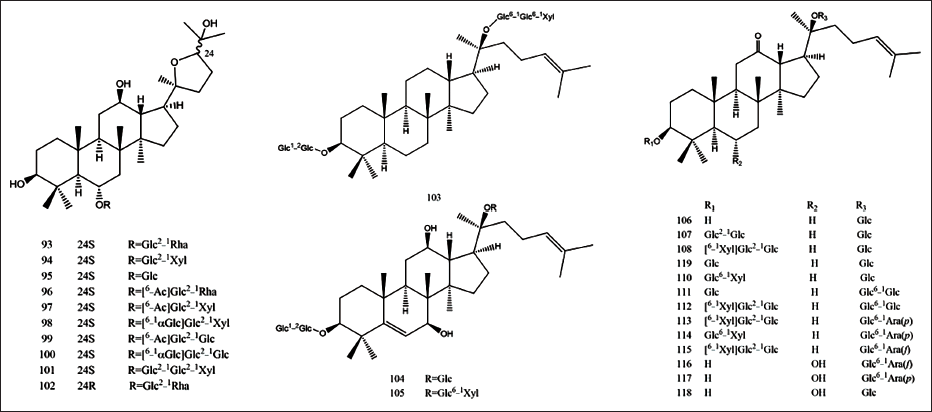
Saccharides
Zhao et al. demonstrated that the total saccharide content, reducing sugar content, and total polysaccharide content in P. japonicus were 70.011%, 5.819%, and 64.192%, respectively, through 3,5-dinitrosalicylic acid colorimetric method (Zhao et al., 2010). Ohtani et al. isolated two reticuloendothelial system-activating polysaccharides, which were named tochibanan-A and tochibanan-B, from P. japonicus. One is β-1,4-galactosan with a molecular weight of about 23,000, and the other is a complex polysaccharide with a molecular weight of about 40,000 containing 7.9% L-arabinose, 87.1% D-galactose, 1.6% D-glucose, and 3.5% galacturonic acid (Ohtani et al., 1989). Yang et al. isolated five kinds of polysaccharides with different molecular weights from P. japonicus, and PP1–PP5, and all of them have activities of scavenging free radicals and protecting DNA damage (Yang et al., 2014).
Amino Acids
A large number of amino acids had been obtained from P. japonicus, including aspartic acid, glutamic acid, arginine, lysine, valine, leucine, isoleucine, proline, phenylalanine, tyrosine, and histidine. Among them, seven compounds such as methionine, threonine, and phenylalanine are essential for human health, which indicates that P. japonicus has high nutritive value (Wu & Yi, 1992; Xiang, 2009).
Volatile Oils
The volatile oils were isolated from P. japonicus using steam distillation and identified by gas chromatography–mass spectrometry (GC–MS) as reported in the literature. The volatile oil content of P. japonicus was 0.016% (Dun & Yuan, 2006), and the main components were β-sandalene and β-farnesene (Zuo & Yuan, 2005).
Pharmacological Activities
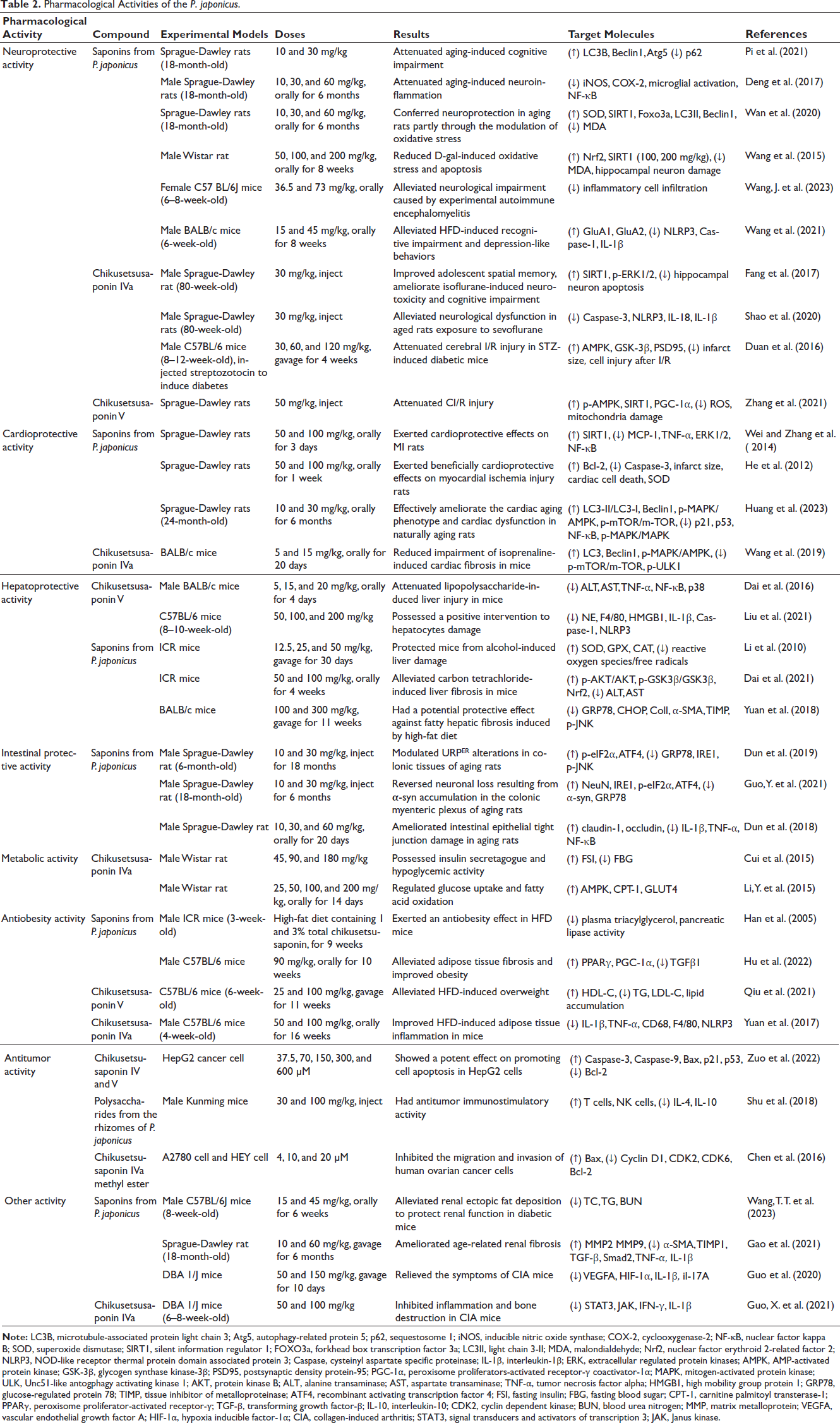
Mounting studies have demonstrated that P. japonicus exhibits extensive pharmacological effects such as antioxidant, lowering lipid, and anti-inflammation, and therefore show extensive protective effects on the central nervous system, cardio-cerebrovascular system, digestive system, and so on. We summarized the pharmacological activities in Table 2.
Pharmacological Activities of the P. japonicus.
Effects on the Central Nervous System
With aging, the structure and function of the human central nervous system gradually undergo degenerative changes. Several studies have demonstrated that P. japonicus confers neuroprotection in naturally aging rats. Saponins from P. japonicus (SPJ) can reduce cognitive dysfunction in aged rats by upregulating microglial M1 to M2 polarization, resulting in reduced microglial inflammation and increased microglial autophagy capacity (Pi et al., 2021). Deng et al. indicated that SPJ has antineuroinflammatory effects in aging rats via regulation of the mitogen-activated protein kinase (MAPK) signaling pathway (Deng et al., 2017). And Wan et al. reported that SPJ can reduce age-related apoptosis and promote antioxidant enzyme activities and mitochondrial function (Wan et al., 2020).
Furthermore, substantial studies have found that P. japonicus shows therapeutic potential for drug- or disease-induced models by regulating multiple signaling pathways. D-gal-induced animal models are commonly used in brain aging studies. It has been reported that SPJ can ameliorate D-gal-induced neuronal injury and cognitive impairment, which may be related to nuclear factor erythroid 2-related factor 2 (Nrf2) and silent information regulator 1 (SIRT1)-mediated antioxidant pathways (Wang et al., 2015). Chikusetsusaponin IVa, the main monomers of SPJ, was reported to improve sevoflurane or isoflurane-induced cognitive impairment and neuroinflammation through activation of the SIRT1/extracellular regulated protein kinases (ERK)1/2 pathway and inhibition of the NOD-like receptor thermal protein domain associated protein 3 (NLRP3)/cysteinyl aspartate specific proteinase (caspase)-1 pathway (Fang et al., 2017; Shao et al., 2020). In STZ-induced diabetic rats, Chikusetsusaponin IVa can also prevent cerebral ischemia-reperfusion (I/R) injury via phosphorylating glycogen synthase kinase-3β (GSK-3β) through the APN-LKB1 pathway and AMP-activated protein kinase (AMPK) (Duan et al., 2016). Another active component of SPJ is Chikusetsusaponin V, which has been found to alleviate cerebral I/R injury through attenuating oxidative stress and mitochondrial damage, enhancing the deacetylation of peroxisome proliferators-activated receptor-γ coactivator-1α (PGC-1α), and activating AMPK and SIRT-1 (Zhang et al., 2021). SPJ could alleviate the neurological impairment caused by experimental autoimmune encephalomyelitis. The mechanism could be associated with modulation of the gut microbiota and nuclear factor kappa B (NF-κB) pathway (Wang, J. et al., 2023). In addition, SPJ can prevent cognitive deficits and depressive-like behaviors induced by a high-fat diet (HFD) by inhibiting the NLRP3 inflammasome, which increases AMPA receptor expression and influences the CaMKII-CREB pathway (Wang et al., 2021).
Effects on Myocardial Injury
Several studies have found that P. japonicus has protective effects on myocardial injury. SPJ can improve myocardial ischemia injury and attenuate myocardial apoptosis through multiple signaling pathways, including NF-κB, SIRT1, and MAPK. SPJ decreased the serum TNF-α and MCP-1 levels, increased the Bcl-2/Bax expression ratio, and inhibited NF-κB p65 expression, ERK1/2, and p38 MAPK, and enhanced the SIRT1 protein expression (Wei and Zhang et al., 2014). SPJ can also maintain oxidative and antioxidative balance through Nrf2 pathway (Wei and Yuan et al., 2014), eventually alleviating cardiac cell death and myocardial ischemia injury (He et al., 2012). In naturally aging rats, SPJ can improve cardiac injury through enhancing autophagy and activating AMPK pathway (Huang et al., 2023). Additionally, Chikusetsusaponin IVa alleviated isoprenaline resulted in myocardial fibrosis mediated by up-regulation of p-AMPK and inhibition of p-mTOR, thereby decreasing the phosphorylated ULK1 757 and enhancing autophagy (Wang et al., 2019).
Effects on the Liver Injury
SPJ has been proven to exert protective effects on liver injury of different models through some important pathways such as anti-inflammation, antioxidation, and regulation of endoplasmic reticulum stress (ERS). It was found that SPJ prevented alcohol-induced liver damage by preventing Kupffer cell activation and decreasing toll-like receptor 4 (TLR4) expression, subsequently alleviating ROS production and inflammatory insult (Yuan et al., 2013). Chikusetsusaponin V can decrease the lipopolysaccharide (LPS)-induced production of inflammatory cytokines by inhibiting the MAPK/NF-κB pathway, ultimately improving liver injury (Dai et al., 2016). In addition to regulating the neutrophil extracellular traps, Chikusetsusaponin V also suppresses the high mobility group protein 1 (HMGB1) release and Caspase-1 activation, thereby reducing the neutrophils recruitment and the inflammation as well as the mitochondrial oxidative stress, which contributes to improving the liver damage caused by acetaminophen (Liu et al., 2021).
SPJ also protected liver injury through antioxidant effects. SPJ can directly scavenge oxygen free radicals and upregulate the expression of antioxidation enzymes superoxide dismutase (SOD), Cat, GPX, etc., thereby attenuating alcohol-induced liver injury (Li et al., 2010). SPJ enhanced intracellular antioxidant capacity through regulating protein kinase B (AKT)/GSK-3β/Nrf2 signaling pathway, thus ameliorating CCL4-provoked hepatic fibrosis in mice (Dai et al., 2021). Moreover, P. japonicus modulates ERS to attenuate hepatic fibrosis by decreasing glucose-regulated protein 78 (GRP78) and CHOP expression (Yuan et al., 2018).
Effects on the Intestine Injury
Recent studies indicate that P. japonicus may be an effective treatment for intestinal diseases. SPJ can improve intestinal pathology by regulating the intestinal tight junction barrier and ERS in aged rats. SPJ can downregulate GRP78 and URPER alterations, contributing to the myenteric plexus neurons’ recovery in aged rats (Dun et al., 2019; Guo, Y. et al., 2021). Additionally, SPJ can remarkably modulate the damage of intestinal epithelial tight junction by upregulating the expression of claudin-1 and occludin, and its mechanism might include the reduction of inflammation, downregulation of NF-κB, and inactivation of MAPK (Dun et al., 2018).
Effects on the Metabolic System
Research has shown that the ethanolic extract from the roots of P. japonicus mainly inhibited α-glucosidase activity (Chan et al., 2010). Six SPJ including Chikusetsusaponin IV, Chikusetsusaponin IVa, Chikusetsusaponin V, Notoginsenoside R1, Ginsenoside Rb1, and Ginsenoside Rd could exhibit α-glucosidase inhibitory effect using a novel assay (Li, S. et al., 2015). Recently, more attention has been paid to Chikusetsusaponin IVa among the above six saponins. It was reported that Chikusetsusaponin IVa can decrease blood glucose levels and increase serum insulin levels in type 2 diabetes mellitus (T2DM) rats (Cui et al., 2015). Chikusetsusaponin IVa has also been proven to decrease the levels of low-density lipoprotein-cholesterol (LDL-C), free fatty acid (FFA), and total cholesterol (TC) in T2DM rats (Li, Y. et al., 2015).
Effects on the Adipose Tissue
Numerous studies have confirmed the effects of P. japonicus on adipose tissue. For example, SPJ exerts an antiobesity effect in mice treated with HFD, which is possibly mediated by preventing uptake of dietary fat from the intestines by inhibiting the activity of pancreatic lipase (Han et al., 2005). Hu et al. reported that SPJ could ameliorate obesity and reduce adipose tissue fibrosis via activating peroxisome proliferator-activated receptor-γ (PPARγ)/PGC-1α and suppressing transforming growth factor-β1 (TGFβ1)/Smad2 pathway (Hu et al., 2022). Furthermore, Chikusetsusaponin V significantly alleviated HFD-induced overweight (Qiu et al., 2021). Meanwhile, Chikusetsusaponin IVa inhibited NLRP3 inflammasome and NF-κB signaling pathway, thereby improving HFD-induced adipose tissue inflammation in mice (Yuan et al., 2017).
Effects on Tumor Cachexia
There is growing evidence that P. japonicus can show antitumor activity and improve tumor cachexia in various types of tumor models. Zuo reported that Chikusetsusaponin IV and V promoted HepG2 apoptosis through the p53-mediated apoptosis pathway (Zuo et al., 2022). Also, Shu reported that polysaccharides isolated from the rhizomes of P. japonicus (PSPJ) have antitumor immunostimulatory activity, specifically as follows: PSPJ increased spleen/thymus indexes and splenocyte proliferation induced by ConA/LPS while reduced CD4+T cells of mice harboring H22 hepatoma cells. PSPJ decreased the production of immunosuppressive factors in tumor-associated macrophages (Shu et al., 2018). Furthermore, Chikusetsusaponin IVa methyl ester inhibits ovarian cancer cell proliferation, causes cell cycle arrest, promotes apoptosis, as well as inhibits invasion and migration (Chen et al., 2016).
Other Effects
Besides the biological activities described above, P. japonicus was reported to have some other pharmacological activities. Wang et al. reported that P. japonicus prevented renal cell apoptosis through the Bcl-2/caspase 3 pathway and attenuated renal injury in diabetic mice (Wang, T. T. et al., 2023). SPJ also ameliorated age-related renal fibrosis through inhibiting NF-κB and TGF-β1/Smad pathways and enhancing Nrf2 signaling pathway (Gao et al., 2021). Moreover, P. japonicus has effects on rheumatoid arthritis via anti-inflammatory and antiangiogenic biological properties. SPJ was reported to modulate the hypoxia inducible factor-1α (HIF-1) or vascular endothelial growth factor (VEGF) pathways via inhibiting signal transducers and activators of transcription 3 (STAT3) and non-receptor tyrosine kinase, which inhibited angiogenesis and inflammation, and significantly reduced the arthritis indices in collagen-induced arthritis (CIA) mice (Guo et al., 2020). Guo et al. also reported that Chikusetsusaponin IVa attenuated bone destruction and inflammation mediated by Janus kinase (JAK)/STAT pathway in CIA mice, contributing to the clinical treatment of rheumatoid arthritis (Guo, X. et al., 2021).
Conclusion
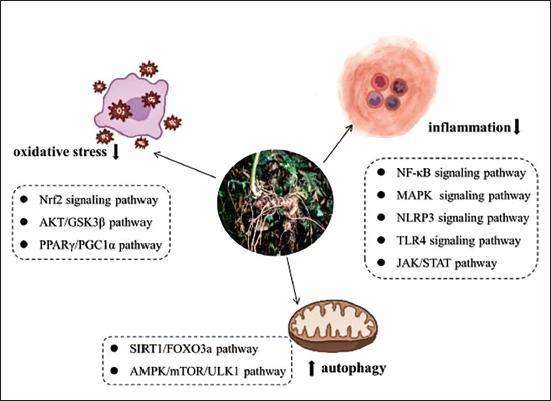
In this article, we have summarized the botany, phytochemistry, and pharmacological effects of P. japonicus. Substantial evidence has shown that the total saponins are the major active ingredient of P. japonicus, which have a long history in traditional Chinese medicine because of possessing the functions of dispelling blood stasis and hemostasis, relieving swelling and pain, eliminating phlegm and relieving cough, tonifying, and strength. Mounting studies have demonstrated that P. japonicus exhibits extensive pharmacological activities through multiple signal pathways (Figure 4), and therefore shows extensive protective effects on the central nervous system, cardio-cerebrovascular system, and digestive system. But more clinical trial supports are needed to further verify these pharmacological actions of P. japonicus. Meanwhile, the available data on the chemical composition of the various parts of P. japonicus and their wide range of pharmacological properties are scarce, which makes it more attractive to study it in depth.
Footnotes
Acknowledgments
None.
Author Contributions
Li Wang, Ao-jia Zhou, Yi-dan Zhang, Cheng Fan, and Shan-shan Hu prepared the manuscript which was guided by Zhi-yong Zhou, Ding Yuan, and Ting Wang.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
Not applicable.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (NSFC81673778); the Research Foundation of Wuhan Municipal Health Commission (WX21D02); and the Research and Innovation Fund of Wuhan Asia General Hospital (2022KYCX1-B14).
