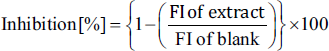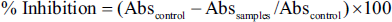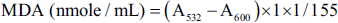Abstract
Background
Metabolic syndrome (MS) is considered a challenging health problem worldwide. However, the search for alternative treatments to alleviate MS is lacking.
Purpose
This study defines Silybum marianum (milk thistle) ethanol seed extract (SMESE) in terms of polyphenol and flavonoid content, antiglycation effect, α-amylase activity, and hypolipidemic and hypoglycemic effects in streptozotocin (STZ)-induced diabetic rats.
Materials and Methods
S. marianum seeds were collected, ground, and extracted with 80% ethanol. The α-amylase inhibitory activities of 10, 30, and 100 µg/mL SMESE were evaluated using spectrophotometric methods. The inhibitory activity of SMESE against advanced glycation end-product formation was evaluated using fluorescence spectrophotometry. In vivo, 48 Wistar male albino rats were used. Eight animals served as the normal control (group 1), and 40 rats were injected with 60 mg/kg STZ to induce diabetes. Diabetic rats were divided into five groups: diabetic control (group 2) and positive control that received the hypoglycemic reference drug metformin at 100 mg/kg (group 3). Groups 4, 5, and 6 were administered the seed extract orally at 30, 100, and 300 mg/kg, respectively. Body weight, serum glucose, lipid profile, and malondialdehyde (MDA) levels were measured weekly for five weeks after SMESE administration.
Results
SMESE administration inhibited BSA glycation and α-amylase activity. In vivo, SMESE administration increased body weight (BW), decreased serum glucose levels, and reduced triglyceride, total cholesterol, low-density lipoprotein, very low-density lipoprotein, and MDA levels but increased high-density lipoprotein levels.
Conclusion
Data indicate the beneficial hypoglycemic and antihyperlipidemic effects of SMESE, suggesting its potential use in controlling lipid metabolism.
Introduction
Metabolic syndrome (MS) refers to a group of metabolic problems, including resistance to insulin, dyslipidemia, central obesity, and high blood pressure. The pathogenesis of MS includes genetic and acquired entities that belong to insulin resistance and chronic mild inflammation (Fahed et al., 2022).
In humans, MS risk factors include increased belly circumference, high plasma triglycerides, hypertension, hyperglycemia, and low high-density lipoprotein (HDL) in the plasma. The presence of three out of five major risk factors is sufficient to diagnose a patient with MS (Masenga et al., 2023). In the early diagnosis of MS, it is important to change the lifestyle of the patient and therefore modify the risk factors. When MS is advanced, however, drug therapy is indicated, aiming to treat individual components of MS. Some nutraceuticals have been shown to have benefits in treatment (Ambroselli et al., 2023).
Medicinal plants are utilized worldwide, and there are regulations to define their proper use, including the identification of the correct species name and the verification of the presence, purity, and concentration of the active ingredients (Palhares et al., 2015). Plant products are generally safe, and in most cases, few side effects have been reported (Mondal et al., 2014).
Because of their therapeutic capacities, medicinal plants are considered a source of relief for MS, including diabetes mellitus (Joy et al., 2021). These capacities are attributed to the content of nutraceuticals and active compounds, such as phenols, flavones, lignans, tannins, alkaloids, volatile oils, steroids, resins, and terpenoids (Ríos & Recio, 2005), which can be found in different parts of medicinal plants, such as seeds, leaves, fruits, roots, and flowers (Jamshidi-Kia et al., 2018).
The plant Silybum marianum, which has the common name milk thistle (
The main objective of this study was to examine the potential glucose- and lipid-lowering effects of ethanol seed extracts of the wild medicinal plant S. marianum (milk thistle) on streptozotocin-induced diabetes in rats.
Materials and Methods
Silybum Marianum Seed Collection and Extraction
S. marianum cultivars were taxonomically identified by a professor of plant taxonomy. A voucher specimen was deposited at the herbarium of the Department of Biological Sciences under number 1210. S. marianum was collected between May and July 2020. The seeds were air-dried in the shade at room temperature to give 1 kg dry weight, which was crushed into a fine powder using a grinder (Philips, India). S. marianum seed powder (300 g for a batch) was extracted with 80% ethanol in a 1: 10 ratio of powder to solvent(w/v). Mixing was performed using an Ultra-Turrax mixer and sonicated for 15 min at 40°C using an ultrasonic water bath. The S. marianum ethanol seed extract (SMESE) was separated from the residual material by filtration through the Whatman No. 1 filter paper. The solvent was removed by evaporating to dryness in vacuo at 40°C for 3–4 hours using a Heidolph rotary evaporator (Laborota 4001, Heidolph, Schwabach, Germany) at 40°C. The yield of SMESE was 13% of the seed dry weight, and the extract was stored in sterile dark bottles in a refrigerator at 4°C.
Estimation of the Total Phenolic Content of SMESE
SMESE (100 µL), distilled water (500 µL), and Folin-Ciocalteu reagent (FCR) (100 µL) were mixed well in a test tube and left to stand for 6 min. Seven percent sodium carbonate (1,000 µL) and another 500 µL of distilled water were added to the tube, and the mixture was then shaken at room temperature and incubated at 37°C for 90 min. Finally, absorbance was measured at 760 nm. A gallic acid standard curve was established. The total phenolic content in the samples was calculated from the standard gallic acid calibration curve and expressed as milligram gallic acid equivalent per gram (mg GAE/g) of dry extract (Shirazi et al., 2014).
Estimation of the Total Flavonoid Content of SMESE
The total flavonoid content was determined using the aluminum chloride colorimetric technique (Kim et al., 2003). Briefly, 1 mL of the extract was added to a test tube containing 4 mL of distilled water, followed by the addition of 0.3 mL of 5% sodium nitrite. Five minutes later, 0.3 mL of 10% aluminum chloride was added to the mixture. In the sixth minute, 1 mL of 1 M NaOH was added. Next, the mixture was adjusted to 10 mL with distilled water and thoroughly mixed using a vortex mixer. The absorbance of the reaction mixture was measured at 510 nm. The total flavonoid content of the extract was expressed as milligrams of quercetin equivalents per gram (mg QE/g) of dry extract.
Determination of the Anti-glycation Activity of SMESE
This was performed following the procedure described by Szawara-Nowak et al. (2014). BSA (10 mg/mL) and glucose (90 mg/mL) were separately dissolved in phosphate buffer (pH 7.4). Then, 1 mL of previously prepared concentrations of SMESE was mixed with 1 mL of BSA and 1 mL of glucose solution in a 5 mL polypropylene test tube. A blank was prepared using 1 mL of phosphate buffer, 1 mL of BSA solution, and 1 mL of glucose solution. A positive control was prepared by mixing 1 mL of aminoguanidine (AG) solution (1 mol/L) with 1 mL of BSA and 1 mL of glucose (Alhadid et al., 2022). The test solution also contained 0.01% NaN3 to protect against microbial growth. The tubes were capped and incubated for 72 hours at 37°C in the dark in a temperature-controlled incubator. The following equation was used (FI: fluorescence intensity).
Determination of SMESE Effect on α-Amylase Activity
In vitro α-amylase inhibitory activity of the extracts was assayed as described by McCue et al. (2005). Briefly, the reaction mixture was made of 0.1 mL of different dilutions (3, 10, and 100 µg/mL) of the SMESE, 0.5 mL buffer, and 0.5 mL α-amylase enzyme. The mixture was then incubated at 25ºC for 10 min. After that, 0.5 mL of starch was added, and the mixture was incubated at 25ºC for 10 min. One milliliter of 3,5-dinitro salicylic acid was added, and the mixture was incubated in a water bath for 5 min. Finally, 7.8 mL of distilled water was added to the reaction mixture. The control sample was prepared similarly, but without the extract. The maltose concentration of each sample was determined by adding 0.1 mL of each sample to 1 mL of the calorimetric reagent 3,5-dinitro salicylic acid and 8.9 mL of distilled water; the test tubes were then covered and boiled in a water bath for 10 min at 95°C. The absorbance was measured at a wavelength of 540 nm.
The percentage α-amylase inhibitory activity of SMESE was calculated as follows:
SMESE Effect on Lipid Peroxidation
Lipid peroxidation was evaluated by the determination of malondialdehyde (MDA) concentration in plasma samples, according to Lima et al. (2012). A plasma sample of 250 µL was mixed with 500 µL of 0.6% 2-thiobarbituric acid (TBA) (T5500, Sigma Aldrich, USA). The mixture was heated in a water bath at 94°C for 1 hour, then left to cool to room temperature.
One milliliter of n-butanol was added, vortexed, and centrifuged at 3,500 rpm for 10 min to divide the mixture into two phases. One milliliter of the chromogen-colored supernatant was pipetted into a cuvette and read on a spectrophotometer (Elico SL150 UV-visible spectrophotometer, India) at 532 and 600 nm wavelengths using distilled water as a blank solution. MDA was calculated using the following formula:
where (A532–A600) is the absorbance due to the MDA-TBA2 adduct, the extinction coefficient of the MDA-TBA2 adduct at 532 nm is 155 m/M/cm, the light path is 1 cm, and the volume of the reaction is 1 mL.
Experimental Design of Streptozotocin-induced Diabetes in Rats
Adult Wistar albino male rats weighing 190–230 g (7–8 weeks old) were used in the experiments. Rats were kept for two weeks in a laboratory environment for acclimation in accordance with the guidelines of the Committee for the Purpose of Control and Supervision of Experiments on Animals and with the guidelines and regulations of the university. An Internal Review Board approval (#105, dated October 7, 2021) was obtained. The animals were housed in plastic cages in a room with a temperature of 24 ± 2°C and a humidity of 45–64%. The animals were fed a balanced commercial diet that was made of 58.1% carbohydrates, 16.51% protein, 4.23% crude fat, 6.31% crude fiber, 8.63% moisture, and 6.22% total ash (Hammouda Feed Company, Amman, Jordan) (Harb et al., 2019) during the course of the experiment. Three days before the start of the experiment, the rats were divided into weight-matched control and experimental groups. On the day of injection, all rats were fasted, except for water, for eight hours prior to STZ injection.
Induction of Diabetes by STZ in Rats
STZ was dissolved in cold fresh 0.05 M sodium citrate buffer (pH 4.5) immediately before intraperitoneal injection at a dose of 60 mg/kg. After STZ injection, the rats were administered 5% D-glucose for 3 days as well as were provided a normal rat diet and drinking water routinely. Fasting blood sugar levels were then measured. The control animals had free access to citrate buffer (pH 4.5) and to a normal rat diet. A rat was considered diabetic if the blood sugar level was greater than 200 mg/dL (Akbarzadeh et al., 2007).
Experimental Design
A total of 48 rats were used in this study. Rats were distributed into six groups of eight each after the induction of diabetes, as follows:
Group 1: Normal control rats (negative control group) were orally administered 2 mL of 0.9% NaCl.
Group 2: Diabetic control rats (positive control group) were orally administered 2 mL of 0.9% NaCl.
Group 3: Diabetic rats administered metformin orally at a dose of 100 mg/kg (Cheng et al., 2006).
Group 4: Diabetic rats orally administered 30 mg/kg S. marianum seed extract.
Group 5: Diabetic rats orally administered 100 mg/kg S. marianum seed extract.
Group 6: Diabetic rats orally administered 300 mg/kg S. marianum seed extract. Doses of SMESE were designed based on extrapolation from the in vitro experiments followed by pilot experiments.
Blood Sample Collection
Blood samples were collected from the retro-orbital plexus of the eye. The animals were fasted overnight for 14 hours on the last day of the experiment and lightly anesthetized with ketamine. Standard heparinized micro-hematocrit capillary tubes were used to collect 2 mL of blood using sterile vacutainers. The blood was centrifuged for 10 min at 3,000 rpm (Zentrifuge Heraeus Labofuge, model I HC122, Germany). The serum was separated and stored in 1 mL Eppendorf tube at −20°C for analysis of biochemical parameters (Panda, 2021).
Estimation of Lipid Profile and Serum Glucose
A lipid profile is a test that measures the amount of total cholesterol (TC), triglyceride (TG), and high-density lipoprotein (HDL) in the serum. The serum TC, TG, HDL, and glucose levels were measured enzymatically using commercial assay kits (HUMAN Diagnostics Worldwide, Germany), according to the manufacturer’s instructions. All biochemical tests were performed using an Elico SL150 UV-visible spectrophotometer (India).
Statistical Analysis
Statistical analyses were performed using GraphPad Prism software version 9.1.2 (GraphPad Software, LLC, San Diego, USA). Data are presented as mean ± SEM. The IC50 values were calculated using the software package, and the data were analysed for statistical significance using a one-way analysis of variance (ANOVA) followed by Fisher’s LSD test. Differences were considered statistically significant at p < 0.05.
Results
Quantification of Phytochemicals in SMESE
The quantitative analysis of SMESE showed the presence of phenols (191.87 ± 0.61 mg GAE/g extract) and flavonoids (19.46 ± 0.72 mg QE/g extract). The extract also contained alkaloids, tannins, and saponins.
In Vitro α-Amylase Inhibitory Activities of SMESE
Supplementary Figure 1 shows that S. marianum seed extract significantly inhibited the activity of α-amylase in vitro. SMESE had an IC50 of 31.46 µg/mL against α-amylase.
SMESE Had an Antiglycation Activity
The antiglycation activities of SMESE compared with aminoguanidine (positive control) are shown in Supplementary Figure 2. The highest antiglycation activity detected was (72.42 ± 3.02%). SMESE showed an IC50 value of 40.82 µg/mL.
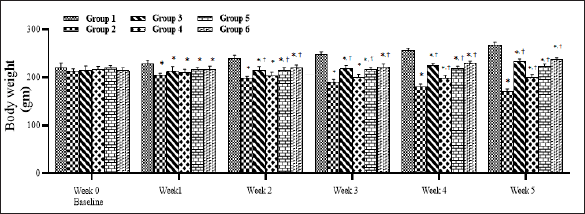
STZ Caused Rats to Lose Weight, and SMESE Reversed This Effect
The body weight of the rats was measured weekly. At the beginning of the study (week 0), no significant differences (p > 0.05) were observed in body weight among the six groups (Figure 1). Body weight decreased slightly in all STZ-injected groups. This trend in body weight decrease continued in group 2 throughout the experiment, whereas groups 3–6 showed a significant (p < 0.05) regain of body weight as compared to group 2. Metformin-treated rats regained weight during weeks 3–6, but they showed less weight than those in group 1.
SMESE Reduced Blood Glucose
The effect of SMESE on fasting blood sugar (FBS) during the experimental period is shown in Figure 2. Streptozotocin injection caused a significant increase (p < 0.0001) in FBS levels in groups 2–6 compared with the normal group (group 1). In the third week of treatment, groups 4 and 5 showed a significant decrease in FBS values compared to group 2, but the metformin-treated group and group 6 showed a significant decrease starting from the second week of treatment.
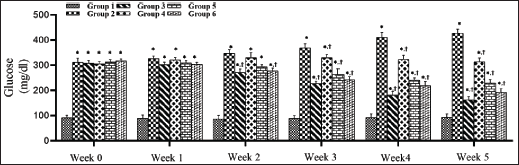
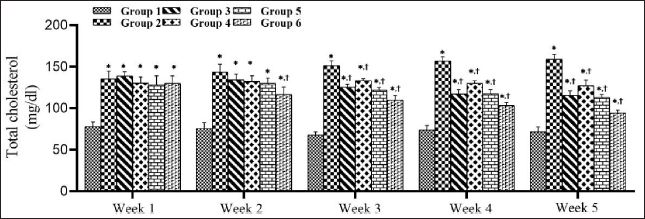
SMESE Reduced Serum Total Cholesterol in STZ-induced Diabetic Rats
The effect of SMESE on the concentration in hyperglycemic rats is shown in Figure 3. STZ caused an elevation in TC concentration in groups 2–6 when compared to normal rats in group 1 (p < 0.0001). By the third week of SMESE administration, TC values started to decrease in groups 3–6 and continued to decrease until the end of the experiment (week 5).
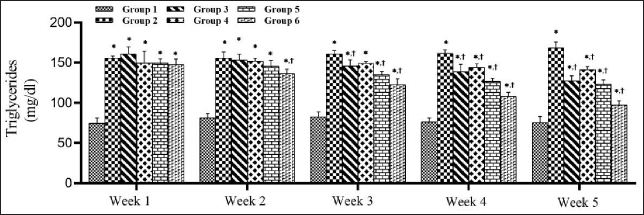
SMESE Reduced Serum Triglycerides Levels in STZ-induced Diabetic Rats
The effect of 30, 100, and 300 mg of SMESE/kg of body weight on serum TG levels in normal and diabetic rats over five weeks of treatment is illustrated in Figure 4. There was a significant decrease in serum TG levels (p < 0.0001) in groups 3–6 compared to that in group 2. This decrease in TG was observed in the second week in group 6 and in the third week in groups 3, 4, and 5 and continued until the end of the experiment.
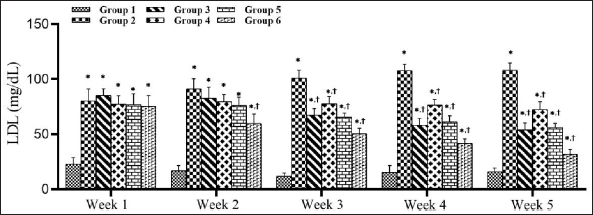
SMESE Decreased Serum Low-density Lipoprotein Levels in STZ-induced Diabetic Rats
Figure 5 illustrates the effect of SMESE at doses of 30, 100, and 300 mg/kg body weight on LDL levels in hyperglycemic rats. A significant decrease in LDL levels (p < 0.0001) was observed in the treated groups (3–6) as compared to the diabetic control (group 2). Beginning in the second week, treatment group 6 showed a significant decrease in LDL value as compared to group 2.
SMESE Decreased Serum Very Low-density Lipoprotein in STZ-induced Diabetic Rats
The effect of SMESE on serum VLDL levels in normal and diabetic rats over five weeks of treatment is shown in Figure 6. There was a significant decrease in serum VLDL levels (p < 0.0001) in groups 3–6 compared to that in group 2. This decrease in VLDL started to appear in the second week in group 6, but in the third week in groups 3, 4, and 5, and continued until the end of the experiment.
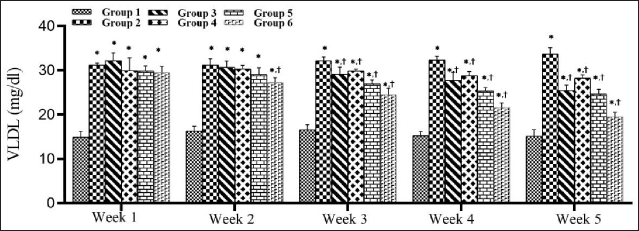
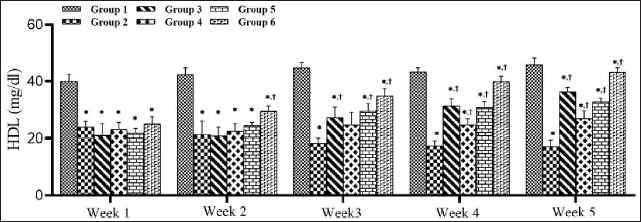
STZ Decreased High-density Lipoprotein Levels in Diabetic Rats and SMESE Reversed this Decrease
Figure 7 shows the HDL levels in the control and experimental groups during the five-week experimental period. Oral administration of SMESE at doses of 30, 100, or 300 mg/kg body weight to diabetic rats significantly reversed the decrease in HDL induced by STZ (p < 0.0001). At the end of the experiment, SMESE administration and metformin treatment caused a progressive increase in HDL levels.
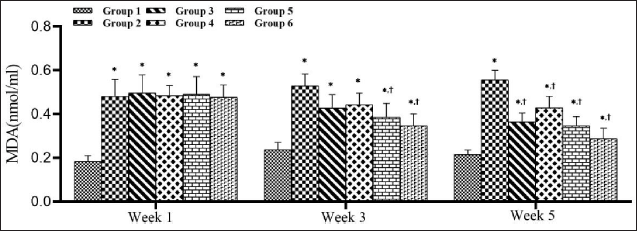
STZ Increased Malondialdehyde Levels in Diabetic Rats and SMESE Reversed This Increase
As shown in Figure 8, STZ injection caused a significant increase (p < 0.0001) in MDA levels in groups 2–6 compared with the normal group (group 1). Beginning with the third week, groups 5 and 6 showed a significant decrease in MDA value compared with group 2.
Discussion
About 60% of the world’s population uses folklore medicines derived from medicinal plants (Modak et al., 2007). S. marianum has been reported as one of the most important medicinal herbs used in the present times (Karaiskou et al., 2021). Phytochemicals play a significant role in the treatment of different diseases and are used in both traditional and modern systems of medication (Ali et al., 2020). One of the objectives of this study was to characterize the phytochemical and antioxidant profiles of S. marianum seed extracts by measuring their total phenols, flavonoids, tannins, alkaloids, saponins, and antioxidant capacity.
Consistent with many other studies (Aziz et al., 2021; Pereira et al., 2014), SMESE was found to contain significant amounts of polyphenols, flavonoids, alkaloids, tannins, and saponins. Polyphenols act as primary antioxidants or free radical terminators, and DPPH analysis of the ethanol extract of S. marianum seeds demonstrated that SMESE has free radical scavenging activity (Supplementary Figure 3). Antioxidants neutralize the harmful effects of free radicals on cells, and the modulation by antioxidants of certain diseases continues to drive research on the antioxidant and antiradical properties of the active ingredients contained in various medicinal plants and in dietary supplements.
S. marianum is a rich source of phytochemical compounds that play functional roles in plant metabolism and have nutraceutical effects on human health. 80% ethanol extracted the highest amount of phytochemicals from S. marianum seeds, and our results are in line with the report by Anwar et al. (2012), who showed that 80% ethanol extracted significant amounts of TFC and TPC compared to methanol and other solvents. In the current study, the proximate analysis of S. marianum seeds (Supplementary Table 1) was in agreement with that of other studies (Dabbour et al., 2014).
α-Amylase is an enzyme that catalyzes the hydrolysis of α-linked polysaccharides into α-anomeric products, and it serves as the active enzyme in pancreatic juice and saliva (Suvd et al., 2001). Inhibition of α-amylase is an efficacious way to stabilize blood glucose levels within the physiological range, which is critical in human pathologies like diabetes mellitus (Bhuyan et al., 2015). α-Amylase inhibitors or starch blockers reduce the utilization of starch by the body, mainly by blocking the hydrolysis of 1,4-glycosidic linkages of starch and other oligosaccharides into simple sugars such as maltose and maltriose (Dineshkumar et al., 2010). Medicinal plants synthesize several groups of natural α-amylase inhibitors, including phenols, flavonoids, catechins, hydrolyzable tannins, and proteins (Lo Piparo et al., 2008). SMESE, which contains high amounts of phenols and flavonoids, has demonstrated the capacity to inhibit α-amylase activity by 76.89% of the control (Supplementary Figure 1). The α-amylase inhibitory activity of SMESE may be attributed to phenolic compounds, such as phenolic acids and flavonoids, which bind covalently to α-amylase and modify its activity, since they have the ability to form quinones or lactones that react with nucleophilic groups on the enzyme molecule (Wahyuni et al., 2020).
Glycation is one of the most detrimental processes that take place in the body. It is a multi-step process in which reducing sugars react with the amino groups of proteins and other macromolecules non-enzymatically, altering the structure and function of these compounds and producing a variety of abnormal compounds called advanced glycation end products (AGEs), which have many adverse effects (Hadid et al., 2022; Trougakos et al., 2013). AGEs cause many deleterious physiological effects, such as inducing oxidative and carbonyl stress and interfering with different signal transduction pathways, thereby mediating the pathophysiology of many glycation-induced ailments (Singh et al., 2014).
Plant-derived ingredients are the most sought-after class of antiglycative candidate compounds. Phenolic compounds are among the most efficacious agents studied. Vlassopoulos et al. (2015) reviewed studies investigating the polyphenols’ anti-glycating role and concluded that pretreatment of protein with phenolic acids can regulate the subsequent glycation in physiological systems. This was also demonstrated by Hadid et al. (2022) using in vitro and in vivo models. In the present study, we evaluated the anti-glycating effect of SMESE and found it to have a high antiglycation activity (72.42% against BSA glycation) when SMESE was used at 100 µg/mL. This is likely due to the fact that SMESE has the most total phenols and the highest antioxidant activity (Supplementary Figure 3). To the best of our knowledge, there were no published studies on the antiglycation activity of S. marianum seed extract against BSA glycation.
Many studies have used STZ routinely to induce diabetes in rats, such as Sprague Dawley and Wistar strains, by injecting intraperitoneal or intravenous doses of STZ ranging from 40 to 100 mg/kg (Iskender et al., 2017). The findings from those studies are consistent with that of our study, which showed increased levels of serum glucose.
In this study, the STZ-treated groups, as expected, showed reduced body weight compared with the normal group, which is consistent with many previous studies (Hossein-Nia et al., 2018). Body weight loss in diabetic rats has been explained as a result of the degradation of structural proteins due to the unavailability of carbohydrates as an energy source; thus, protein wasting and fatty acid store depletion induced by lack of insulin might lead to a reduction in muscle mass and a reduction in body weight (Hassan et al., 2015).
Oral administration of SMESE reversed this decrease in body weight, as rats treated with SMESE started to regain body weight. SMESE seems to have improved the management of blood glucose in rats, and our results are broadly in line with those of many previous studies (Sekiou et al., 2021).
Several studies have revealed the potential role of polyphenolic compounds, particularly flavonoids, in the treatment of diabetes. By activating the cAMP/PKA signaling cascade, flavonoids boost the production and release of pancreatic insulin from beta cells, besides the fact that they are potent inhibitors of α-glucosidase, which reduces glucose absorption in the small intestine (Zeka et al., 2017).
The anti-hyperglycemic potency of SMESE may be due to the polyphenol-rich S. marianum seed extract reported by many authors using other polyphenol-rich extracts (Sun et al., 2016). However, this is the first time that SMESE has been shown to decrease glucose levels in diabetic rats. The present findings are consistent with those of Rozeno Pessoa et al. (2015), who found that STZ-induced diabetic rats have elevated glucose; increased cholesterol, LDL, and triglycerides; and decreased HDL levels, and the hyperglycemia is usually accompanied by hypercholesterolemia; an increase in triglycerides, LDL, and VLDL; and a decrease in HDL. Activation of the hormone-sensitive lipase during insulin deficiency usually accompanies an increased release of free fatty acids from the fat tissue. Thus, excess fatty acids in the plasma produced by STZ-induced diabetes enhance the conversion of excess fatty acids into phospholipids and cholesterol, which, along with the excess triglycerides formed in the liver, may be discharged into the blood in the form of lipoproteins.
In this study, we found that SMESE caused a significant reduction in blood glucose, total cholesterol, triglyceride, and LDL and an increased level of plasma HDL, in addition to significant improvement in body weight in STZ-induced diabetic rats, and this is consistent with many previous studies (Saadh et al., 2020). These authors found that oral administration of milk thistle and fenugreek seeds for two weeks resulted in significant improvements in body weight, blood glucose, glycosylated hemoglobin (HbA1c), cholesterol, and triglyceride levels in alloxan-induced diabetic rats. HDL is known to have a strong protective effect against atherosclerosis. An important mechanism underlying this protection is the role of HDL in the removal of excess cholesterol from peripheral tissues, where cholesterol is degraded into bile acids. This pathway plays an important role in reducing cholesterol levels in the blood and peripheral tissues and inhibiting atherosclerotic plaque formation in the aorta.
One of the consequences of increased blood glucose levels in diabetes is the production of free radicals and the induction of oxidative stress. As described by King and Loeken (2004), excess glucose may activate protein kinase C (PKC) via several mechanisms, including the activation of phospholipase C, the induction of diacylglycerol (DAG) synthesis, and the inhibition of DAG kinase. PKC increases oxidative stress by activating mitochondrial NADPH oxidase, leading to impaired insulin passage through the endothelial wall; therefore, hormone delivery to tissues is limited (Bertelsen et al., 2001). Oxidative stress decreases the activities of catalase, SOD, and glutathione peroxidase in the liver tissue and elevates the levels of MDA (Pradeep et al., 2007). Our results are consistent with the findings reported by others showing that diabetic rats expressed an increase in MDA levels compared to the normal rat group (Ghanbari et al., 2015).
MDA, the end product of lipid peroxidation, is considered as a specific oxidative stress indicator (Arican & Kurutas, 2008). Our results showed that MDA levels in diabetic rats were significantly higher than those in the controls (Figure 8), and rats treated with SMESE showed reduced levels of MDA compared to diabetic rats. SMESE treatment reduced MDA levels to normal values. Our findings are in agreement with pre-clinical studies that concluded that silymarin from S. marianum decreased MDA levels (Alabdan, 2015). It can be concluded that SMESE decreased MDA levels and inhibited lipid peroxidation.
The effects on MDA may be attributed to the antioxidant effects of SMESE since it was shown that the methanol extract of seeds and leaves of S. marianum has high antioxidant activity (Ganbaatar et al., 2014), whereas in Egypt, the ethanol extract of S. marianum seeds showed strong antioxidant activity with a DPPH assay of 91.68% at 200 µg/mL (Ali et al., 2020). This antioxidant activity was attributed to the presence of silymarin, which is a complex mixture of six major flavonolignans: silybin A and B, isosilybins A and B, silydianin, and silychristin, in addition to other minor polyphenolic compounds (Aziz et al., 2021). One limitation of the current work is that it cannot specify whether the observed beneficial effects of the ethanol seed extract are due to silymarin, any of its components, or even some different substances. This is because the solvent ethanol extracts many active ingredients from the seed powder. It is worth mentioning that we also found that the methanol extract shows equally significant free radical scavenging activity as the ethanol extract (Supplementary Figure 3).
Conclusion
SMESE has a high content of phenols, flavonoids, tannins, alkaloids, and saponins and has significant antioxidant activity. This demonstrated antioxidant activity seems to impact SMESE’s inhibition of BSA glycation and α-amylase activity, as well as its in vivo effect on streptozotocin-induced diabetes in rats. SMESE reversed to a significant degree all the ill effects induced by STZ, including body weight, blood glucose, total cholesterol, triglycerides, LDL, VLDL, and HDL, as well as the lipid peroxidation product MDA. These beneficial effects of SMESE call for further work to develop alternative treatments for hyperglycemia and hyperlipidemia using natural resources.
Authors’ Contribution
S.A. and M.H. conceptualization; M.S.M. methodology, data curation, software and illustrations, writing the first draft; S.A. validation; S.A. and M.H. editing and review of the manuscript.
Data Availability Statement
All data generated or analysed during this study are included in this article or in the supplementary material appended to it. Further inquiries can be directed to the corresponding author.
Footnotes
Acknowledgements
The authors wish to thank Miss Amani Harb for the excellent technical assistance in the data processing while using GraphPad Prism software.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval and Informed Consent
The protocol of the experiments was performed in accordance with the guidelines of the Committee for the Purpose of Control and Supervision on Experiments on Animals and with the guidelines and regulations of the University of Jordan. An Internal Review Board approval (#105, dated October 7, 2021) was obtained.
Funding
This research received a Grant from the Deanship of Research, The University of Jordan (#19/2020/882, dated September 24, 2020).
Institutional Review Board Statement
The animal study protocol was approved by the Institutional Review Board at the University of Jordan (decision # 105, dated 7 October 2021).
Supplemental Material
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.