Abstract
Objectives
The objective of this study was to determine the effects of
Materials and Methods
The T2DM model was established by a high-fat high-sucrose diet (HFSD) for 6 weeks and streptozotocin (STZ) administration. The rats were randomly divided into control, model, metformin (100 mg/kg/day), and
Results
Rats in the model group exhibited significantly higher levels of FPG and 2hPPG but lower SPR than those in the control group. Rats in the Taraxacum group had lower FPG and 2hPPG levels and higher SPR than those of the model group. Moreover, clpP, HSP60, PINK-1, Parkin, P62, Beclin-1, and LC3-II/I proteins were significantly upregulated in the rats of the model group, relative to the control group. UPRmt and mitophagy-related proteins were markedly downregulated in rats of the Taraxacum group, relative to the model group.
Conclusion
T2DM rats exhibited certain levels of anhedonia and depression, which were effectively alleviated by
Keywords
Introduction
Diabetes mellitus (DM) is a metabolic disease typically characterized by chronic hyperglycemia. More than 95% of all patients diagnosed with DM suffer from type 2 diabetes mellitus (T2DM). The prevalence of DM in China is as high as 10.9%, which has become a public health problem. Depression is a mood disorder characterized by a marked and persistent feeling of sadness and loss of interest as the primary symptoms. The global prevalence rate of depression is 2.36 times higher in diabetic patients compared with non-diabetic people (Owusu et al., 2018). DM rats always show depression-like behaviors such as anhedonia and behavioral despair, which are manifested by the decline of sucrose preference rate and the prolongation of forced swimming immobility (Hassan et al., 2019). The underlying factors of diabetes-induced depression mainly include hypothalamic–pituitary–adrenal (HPA) axis dysfunction, 5-hydroxytryptamine (5-HT) dysfunction, inflammatory activation, abnormal neurotrophic factors, and hippocampal damage, among others. Mitochondrial dysfunction is a crucial pathogenesis of DM and associated depression. Mitochondrial unfolded protein response (UPRmt) and mitophagy are the main mechanisms involved in mitochondria-induced self-repair and are activated to restore mitochondrial function when mitochondria are damaged.
Materials and Methods
Animals Used
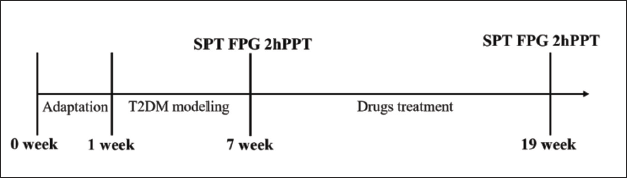
Specific-pathogen free (SPF), Sprague–Dawley (SD) rats, 36 in number (♂, average weight: 200 ± 20 g), were purchased from Beijing HFK Biotechnology Co., Ltd., [Production license number: SCXK (Beijing) 2020-0004]. The Laboratory Animal Ethics Committee of the Medical Department of North China University of Science and Technology scrutinized and approved this experiment. The experimental design is shown in Figure 1.
Reagents Used and Protocols of Drug Preparation
Metformin hydrochloride tablets (0.5 g/tablet) were purchased from Sino-American Shanghai Squibb Pharmaceutical Co., Ltd., Shanghai, China (Batch number: ABS9901). Streptozotocin (STZ) was purchased from Beijing Solarbio Science & Technology Co., Ltd., Beijing, China (CAS: 18883-66-4). Whole herbs of
Superoxide dismutase (SOD) assay and malonaldehyde (MDA) detection kits were provided by Nanjing Jiancheng Bioengineering Institute, Jiangsu, China (Batch numbers: 20210422 and 20210421, respectively). Anti-glyceraldehyde-3-phosphate dehydrogenase (anti-GAPDH), anti-HSP60, anti-clpP, anti-PINK-1, anti-Parkin, anti-P62, anti-Beclin-1, and anti-LC3 antibodies were purchased from ProteinTech Group Inc. (Cat Nos.: 10494-1-AP, 15282-1-AP, 15698-1-AP, 23274-1-AP, 14060-1-AP, 18420-1-AP, 11306-1-AP, and 14600-1-AP, respectively).
M200PR0 microplate reader (Tecan Group Ltd., Männedorf, Switzerland), desktop refrigerated centrifuge (Thermo Fisher Scientific Inc., Massachusetts, US), microscope (Olympus Shinjuku City, Tokyo, Japan), SDS-PAGE unit, Western blot unit, and SDS-PAGE gel imaging system (Bio-Rad, California, US) were used in this study.
Establishment of a T2DM Rat Model and Protocol for Drug Administration
The rats were raised in an SPF room for a week to adapt to the conditions. Water and food were provided
Sucrose Preference Test (SPT)
The SPR in rats of each group was estimated pre- and post-treatment through the SPT. The rats were deprived of food and water for 24 h before SPT. They were then allowed to feed on 100 mL of water and 100 mL of 1% sucrose solution for 1 h. When the SPT was carried out for half an hour, the bottle position of the water and sucrose solution was changed to avoid the interference of the bottle position on the drinking water of rats. The SPR was calculated according to the formula: SPR = weight of sucrose solution intake/weight of total intake.
Enzyme-Linked Immunosorbent Assay (ELISA)
Blood was collected from the abdominal aorta and serum was separated. The levels of SOD protein were determined using an ELISA kit, and MDA levels were ascertained using a detection kit, according to the manufacturer’s instructions.
Western Blot Assay
Hippocampal tissue (100 mg) was collected from each rat, placed in a centrifuge tube, and homogenized in 1 mL of Radio Immunoprecipitation Assay (RIPA) lysis buffer. The homogenate was then incubated on ice for 30 min and centrifuged at 4°C for 15 min at 12,000
Statistical Analysis
The SPSS22.0 software was used for data statistics. Data were presented as mean ± SEM deviation and analyzed by one-way analysis of variance. If the variance is homogeneous, the Bonferroni method is used for the post-test. If the variance is not uniform, the Tamhane T2 method is used for the post-test. Statistical differences between the test groups were considered to be significant when the probability factor
Results
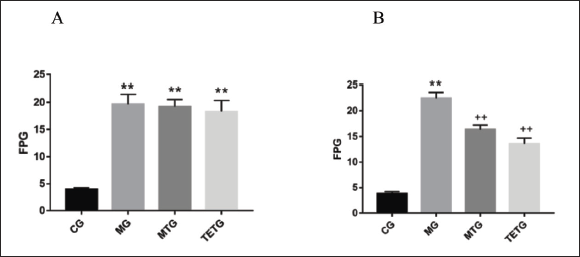
T. officinale Extracts Suppressed FPG Levels in T2DM Rats
Rats of MG exhibited significantly higher FPG levels than those of the CG (
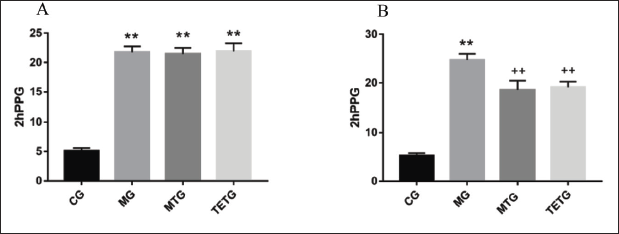
T. officinale Extracts Suppressed 2hPPG Levels in T2DM Rats
Rats in the other three groups exhibited higher 2hPPG levels than those in the CG (
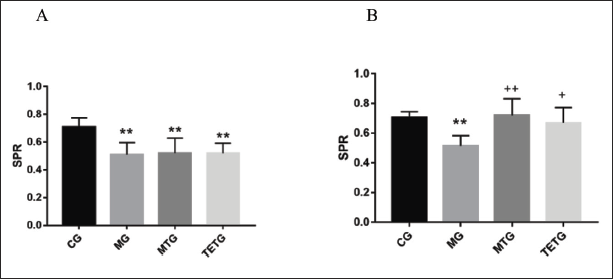
T. officinale Extracts Elevated SPR in T2DM Rats
Rats in the other three groups exhibited lower SPR compared with those in the CG (
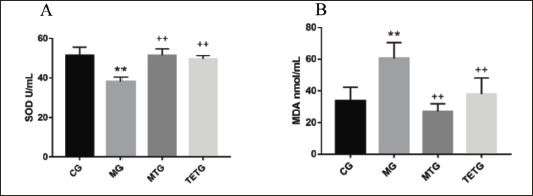
T. officinale Extracts Modulated Serum SOD and MDA Levels in T2DM Rats
Rats of the MG exhibited significantly lower SOD and higher MDA levels, while those of the TETG demonstrated elevated SOD but suppressed MDA levels when compared with those from MG (
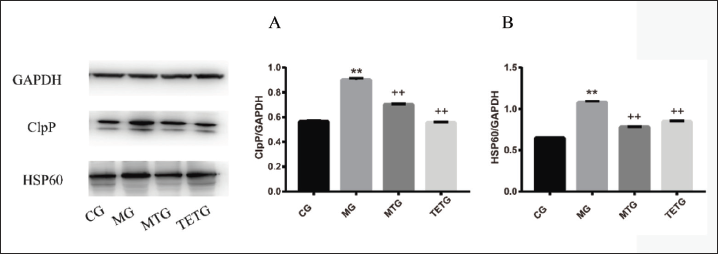
T. officinale Extracts Downregulated Expression of UPRmt-related Proteins in the Hippocampi of T2DM Rats
UPRmt-related proteins, clpP, and HSP60 were markedly upregulated in the hippocampi of rats from the CG, compared with those from the MG (
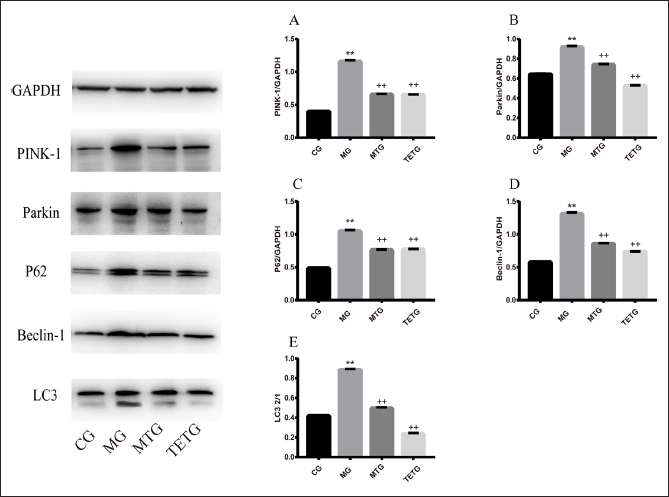
T. officinale Extracts Suppressed the Levels of Mitophagy-related Proteins in the Hippocampi of T2DM Rats
Mitophagy-related proteins: PINK-1, Parkin, P62, Beclin-1, and LC3 I/II were significantly upregulated in the hippocampi of rats from the MG compared with those in the CG (
Discussion
Current research trends on the pathogenesis of DM have begun to focus on the role of mitochondrial dysfunction. This model of DM pathogenesis is characterized by damage to mitochondrial morphology and mitochondrial (mt) DNA, as well as inhibition of respiratory chain functions (Rowley et al., 2017). The pathogenesis of depression involves hippocampal neurons exhibiting varying stages of impairment in mitochondrial function, reduced mitochondrial numbers, morphological and/or oxidative damage in mitochondria, variations in mtDNA sequence, lowered energy metabolism, and respiratory dysfunction (Klinedinst & Regenold, 2015; Allen et al., 2018). Therefore, improving the mitochondrial quantity, quality, and function may be a potential therapy method for managing diabetes-induced depression.
UPRmt and mitophagy are the main cellular mechanisms involved in the repair of mitochondrial damage. Specifically, UPRmt is a stress response in which transcriptional activation of mitochondrial heat shock proteins and proteases, as well as other nuclear-encoded genes is initiated to maintain mitochondrial protein homeostasis (Jovaisaite et al., 2014). Notably, abnormal protein structure resulting from mitochondrial dysfunction causes activation of UPRmt. HSP60, a molecular chaperone, plays a crucial role in facilitating proper protein assembly and correct protein folding in mitochondria (Richards et al., 2023). clpP assists in protein degradation. HSP60 and clpP were upregulated in the mitochondria of skeletal muscles indicating that UPRmt was activated (Wang et al., 2022).
Mitophagy is target-specific autophagy in which cells selectively degrade damaged or dysfunctional mitochondria. It is mediated by the PINK-1/Parkin signaling pathway, which is a classic mechanism for studying mitophagy in mammalian cells. PTEN-induced kinase 1 (PINK-1) is a mitochondrial serine/threonine-protein kinase. Under normal conditions, PINK-1 is located in the inner mitochondrial membrane. Upon mitochondrial damage, with the help of translocase of the outer mitochondrial membrane (TOMM) and translocase of the inner mitochondrial membrane (TIMM), it enters the cytoplasm, where it is degraded to minuscule levels. PINK-1 identifies damaged mitochondria by constantly checking for the oxidative state of mitochondria and sends signals for the recruitment and activation of Parkin (Nguyen et al., 2016). Parkin, an E3 ubiquitin ligase, promotes the ubiquitination of proteins located on the mitochondrial membrane of damaged mitochondria. Autophagic receptor proteins, such as P62, OPTN, and NDP52 interact with the ubiquitinated proteins on the mitochondrial membrane. The microtubule-associated protein 1 light chain 3 (LC3), binds with the autophagic receptor protein located on the damaged mitochondria (Eiyama & Okamoto, 2015). These mitochondria are then coated with the autophagosome membrane, a phenomenon that activates the autophagic lysosome system, which degrades these mitochondria (Tan et al., 2018).
The potential use of
Conclusion
Summary
In this experiment, we studied the effect and mechanism of
Abbreviations
CG: Control group; 2hPPG: 2-hour postprandial plasma glucose; 5-HT: 5-hydroxytryptamine; DM: Diabetes mellitus; ECL: Enhanced chemiluminescence; ELISA: Enzyme-linked immunosorbent assay; FPG: Fasting plasma glucose; HFSD: High-fat high-sucrose diet; HPLC: High-performance liquid chromatography; HPA: Hypothalamic–pituitary–adrenal; MDA: Malonaldehyde; MTG: Metformin treatment group; LC3: Microtubule-associated protein 1 light chain 3; UPRmt: Mitochondrial unfolded protein response; MG: Model group; PINK-1: PTEN-induced kinase 1; RIPA: Radio immunoprecipitation assay; STZ: Streptozotocin; SPT: Sucrose preference test; SPR: Sucrose preference rates; SOD: Superoxide dismutase: SPF: Specific-pathogen free; SD: Sprague–Dawley;
Authors’ Contributions
All authors have agreed to the submission of this manuscript for publication. JL and XW designed the experimental scheme. MW performed the experiments, analyzed the data, and interpreted the results. ZG and LM performed the animal experiments and behavior tests. ELISA and Western blot were performed by MW and ZL. The figures and manuscript were written by MW and ZG.
Footnotes
Acknowledgments
The authors thank the Diabetes Integrated Traditional Chinese and Western Medicine Prevention and Treatment Team of Tangshan City.
Declaration of Conflicting Interests
The authors indicated no conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the National Natural Science Foundation of China (82205066), the Ministry of Science and Technology Assistance Project for Developing Countries (KY201904005), and the Research Program Project of Hebei Provincial Administration of TCM (2022139).
