Abstract
Objectives
This study aimed to evaluate the therapeutic effects and mechanism of genistein on dextran sulfate sodium-induced mouse model of ulcerative colitis (UC).
Materials and Methods
A DSS-induced mouse model for UC was used in this study. Mice were then treated with genistein or a combination of genistein and the G protein-coupled estrogen receptor (GPER) antagonist G15. Colon length, disease activity index (DAI), spleen index (SI), histopathological alterations, and integrity of the colonic barrier were evaluated. The expression of inflammatory cytokines and antioxidant capacity were detected. Populations of T helper 17 cells (Th17) and Regulatory T cells (Treg) in the mesenteric lymph nodes (MLN) were analyzed. The expression of a transcription factor for Th17 and Treg in the colonic tissues was detected.
Results
Genistein treatment significantly alleviated DSS-induced colitis, summarized as increased colonic length, decreased DAI, SI, improved histopathological alterations, and colonic barrier integrity. Genistein treatment restrained pro-inflammatory cytokines and oxidative enzyme release while promoting anti-inflammatory and anti-oxidative enzyme release. Flow cytometry indicated that genistein significantly reduced the Th17 population while boosting Treg populations. Furthermore, genistein inhibited the expression of transcription factors associated with Th17 and promoted the expression of transcription factors associated with Treg in the colonic tissue. Intriguingly, these observed effects of genistein were abolished when UC mice were treated with a combination of genistein and GPER antagonist G15.
Conclusion
This study suggests that genistein is potent in protecting against DSS-induced colonic injuries by rebalancing Th17/Treg and that GPER may be a vital target for genistein-mediated immunomodulatory effects in UC.
Introduction
Ulcerative colitis (UC) is a non-specific inflammatory disease of the intestine. The lesion site principally occurs in the mucosal layer of the rectum and colon, and the main pathological manifestation is mucosal ulceration (Kobayashi et al., 2020). A wide variety of factors is involved in the pathogenesis of UC, including environmental factors, lifestyle, genetic factors, and gut microbiota. All these factors can trigger injury of the mucosal barrier and immune barrier in colonic tissues, leading to the accumulation of a large number of immune cells in the intestinal lamina propria (Eisenstein, 2018). Excessive T cell activation in intestinal mucosa plays a vital role in the pathogenesis of UC. T helper 17 (Th17) cells and regulatory T (Treg) cells are both subtypes of T helper cells but perform opposite roles in UC. Specifically, Th17 triggers inflammatory responses and facilitates the activation of the adaptive immune system, while Treg suppresses inflammatory responses and maintains immune homeostasis (Negi et al., 2021; Ueno et al., 2018). Several studies show that an imbalance of Th17/Treg is closely related to the occurrence and development of UC suggesting that harnessing Th17/Treg may be a feasible strategy for the treatment of UC (Long et al., 2020; Tong et al., 2021; Xiao et al., 2024).
Currently, the most commonly used treatment strategies for UC include an immunosuppressant, aminosalicylic acid, and a glucocorticoid. However, the long-term use of these drugs may lead to several serious adverse reactions such as an increased risk of opportunistic infection, bone marrow suppression, and male sterility; thus, these side effects limit their application (Cleveland et al., 2022). Although the therapeutic mesalazine has a few reported adverse reactions, it still has certain shortcomings in the treatment of UC owing to drug resistance resulting from long-term use. Therefore, it is imperative to identify low-cost, low-toxicity, and efficacious therapeutics for the treatment of UC. Prior studies indicate that dietary supplementation of soybean isoflavones is associated with reduced colorectal cancer risk and improved gastrointestinal function (Belobrajdic et al., 2023). Natural soybean isoflavones are principally divided into free aglycone and conjugated glycosides (do Prado et al., 2022). Free aglycones, the primary representative product being genistein, exhibit higher biological properties. However, what role genistein plays in UC and the exact mechanisms behind any potential effect remain largely elusive. As a member of the phytoestrogen family, genistein shares a similar structure with endogenous estrogen. Thus, we imagine that genistein can exert its biological function, at least in part, by binding to endogenous estrogen receptors. There are three subtypes of estrogen receptors, namely, estrogen receptor α, estrogen receptor β, and G protein-coupled estrogen receptor (GPER) (Chen et al., 2022). Among these, GPER impacts profoundly on the immune system. A prior study indicated that GPER deletion is associated with a form of familial immunodeficiency (Sloboda et al., 2019). Accordingly, GPER is expressed in several immune cell subtypes including monocytes/macrophages, neutrophils, eosinophils, B lymphocytes, and T lymphocytes (Notas et al., 2020). Thus, we speculate that GPER may be a potent target for modulating Th17/Treg cell balance.
In this study, we sought to investigate the therapeutic effects of genistein in a DSS-induced experimental model of acute UC. Based on our findings, we speculate that GPER-induced Th17/Treg cell rebalance may play an essential role in this pathobiological process.
Materials and Methods
Animals and Treatment
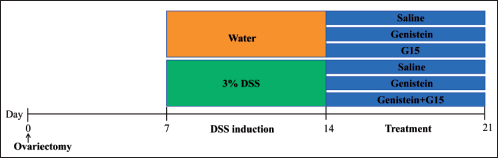
In this study, 6-8 week-old female C57BL/6 mice were maintained with ad libitum access to food and water with a 12-h light/dark cycle. All animals were subjected to ovariectomy (OVX) to eliminate possible effects stemming from circulatory estrogen. On day 7, mice were supplied with 3% dextran sulfate sodium salt diluted in drinking water for 7 consecutive days to induce an experimental mouse model of acute colitis. G15 was used as an antagonist for GPER. Mice were randomly assigned to the control, genistein (Gavage, 40 mg/kg per day), G15 (intraperitoneal injection, 150 µg/kg per day), DSS, DSS+Genistein, and DSS+Genistein+G15 groups. After treatment for seven consecutive days, mice were anesthetized with a mixture of xylazine (10 mg/kg, i.p.) and ketamine (90 mg/kg, i.p.), and mice were subsequently sacrificed by decapitation, and indicated samples were collected for further analysis (See time schedule in Figure 1).
Diagram Outlining the Experimental Procedure and Treatment Grouping.
Evaluation of DAI Score and SI
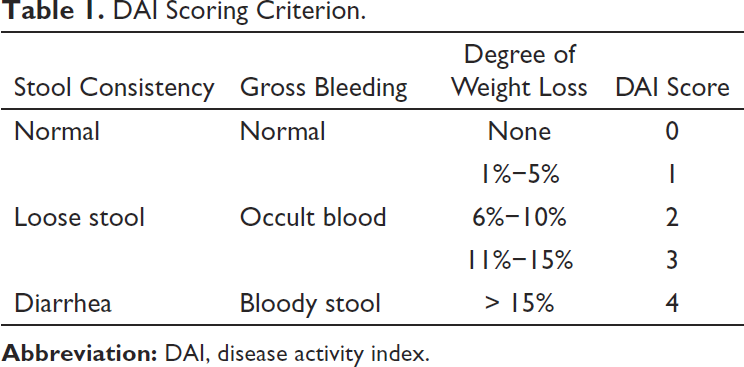
Disease activity index (DAI) was determined by evaluating stool characteristics, occult blood, or bloody stools as well as a degree of weight loss. The scoring criteria are shown in Table 1. Spleen index (SI) represents the functions of splenic immune function, and SI was calculated as spleen weight/body weight.
DAI Scoring Criterion.
HE staining and immunohistochemistry
For hematoxylin-eosin (HE) staining, distal colonic tissues were fixed in 10% formalin for 48 h. Tissues were subsequently embedded in paraffin and cut into 5-µm-thick sections. HE staining was conducted following standard procedures. Following this, stained sections were examined for histopathological alteration, and the severity and extent of inflammation, crypt damage, and percentage of the colon involved were evaluated, the evaluation criterion of which was described elsewhere (Kihara et al., 2003). For immunohistochemical staining, distal colonic tissues were collected and fixed overnight in 4% paraformaldehyde (Blumcke et al., 2017). Samples were then transferred to a 30% sucrose solution until they completely sank into the solution. Samples were subsequently embedded in paraffin and cut into 5-µm-thick sections. Sections were subsequently incubated with anti-occludin (1:200; Abcam, UK, catalog: ab216327) primary antibody for 48 h, followed by HRP-conjugated anti-rabbit IgG (1:50; Beyotime, China) secondary antibody for 4 h. Tissue sections were observed and photographed using an inverted microscope (Leica, Germany).
Quantitative Real-Time Polymerase Chain Reaction
Total RNA was extracted from distal colonic tissues using Trizol buffer. Following this, RNA was reverse-transcribed into cDNA using a commercially available kit. Quantitative RT-PCR was conducted using an SYBR green kit on a 7300 Plus Real-time PCR System (Thermo Fisher, USA) following the manufacturer’s manual. A total of 20 µL reaction mixture was used, and this included 5 pmol of each primer, 10 µL PCR master mixture, 1 µL of cDNA template, and water. The thermocycling protocol used was as follows: initial 95℃ for 30 s, followed by 40 cycles at 95℃ for 10 s, and 58℃ for 30 s. The relative expression of target genes was normalized to GAPDH, and data were analyzed using the 2-∇∇Ct method. Primer sequences used in this study are shown in Table 2.
PCR Primers Used in this Study.

Oxidative/Anti-oxidative Enzyme Activity and Inflammation Cytokines Levels in Serum
Colonic tissues were homogenized and centrifuged, and the supernatant was collected for the measurement of myeloperoxidase (MPO) activity following the manufacturer’s instruction (Abcam, UK). The abundance of malondialdehyde (MDA), a marker of oxidative stress, as well as cellular antioxidants glutathione and superoxide dismutase (SOD) were evaluated using commercially available kits (Abcam, UK). Blood samples were collected from retro-orbital bleeds, and the serum levels of indicated inflammatory cytokines IL1-β, IL-6, TNF-α, and IL-10 were detected using commercially available ELISA kits per the manufacturer’s instruction (Beyotime, China). Data were obtained using a microplate reader.
Flow Cytometry Analysis
Mesenteric lymph nodes (MLN) were lysed into a cell suspension medium. Red blood cells were removed using an RBC lysis buffer. Subsequently, the cell suspensions were incubated with an equal volume of lymphocyte separation solutions (Solarbio, China, catalog: P8620) to obtain an MLN lymphocyte preparation. MLN lymphocytes were subsequently resuspended in buffer solutions for further flow cytometry analysis. For the analysis of the Th17 subset, cell suspensions were labeled with CD4 and IL-17A (EbioScience, USA). For the analysis of the Treg subset, cell suspensions were labeled with CD4, CD25, and FOXP3 (EbioScience, USA). The flow cytometer (CytoFLEX, Beckman, USA) was gated on CD4+ T cells for the analysis of Th17 and Treg cells.
Statistical Analyses
All data are expressed as mean ± SEM. Data were analyzed using one-way ANOVA, followed by Tukey’s post-test for subgroup comparisons. P < 0.05 was considered statistically significant.
Results
Genistein Treatment Decreases the DAI and SI Score in DSS-induced Acute Colitis
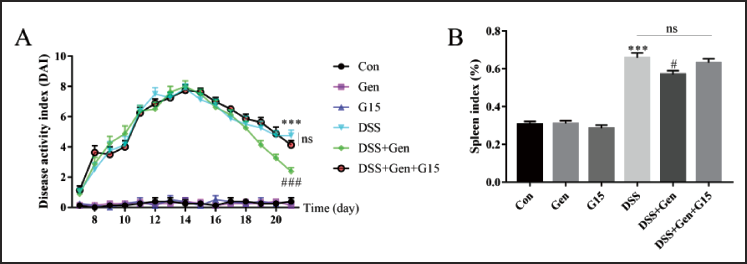
DAI and SI scores represent the severity of colitis and splenic immune function, respectively. DSS induction resulted in increased DAI (p < 0.001, Figure 2A) and SI (p < 0.001, Figure 2B) scores compared to controls, while genistein treatment in the UC model remarkably decreased the DAI (p < 0.001, Figure 2A) and SI (p = 0.028, Figure 2B) scores. The treatment of the UC mouse model with a combination of genistein and GPER antagonist G15 diminished these effects. No differences were observed among the control, genistein, and G15-only groups. These findings suggest that genistein can potently alleviate the severity of colitis and improve splenic immune functions in this UC model by targeting GPER.
Genistein Treatment Improves the Pathological Changes Observed in Colonic Tissues in the DSS-induced Acute Colitis Model
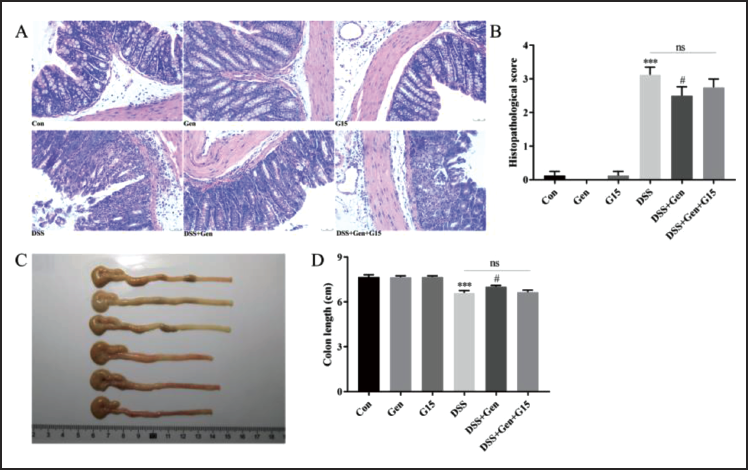
Next, we analyzed the pathomorphological changes in distal colonic tissue by measuring histological alterations (Figure 3A) and colon length (Figure 3C). Genistein significantly blunted DSS-induced colonic injury (p = 0.018, Figure 3B) and colon shortening (p = 0.021, Figure 3D). The combined treatment of genistein and G15 counteracted the effects of genistein-only treatment in this acute colitis model, and no differences were observed among the control, genistein, and G15-only groups. These results suggest that genistein may improve the pathomorphological changes in colonic tissues in this acute colitis model by targeting GPER.
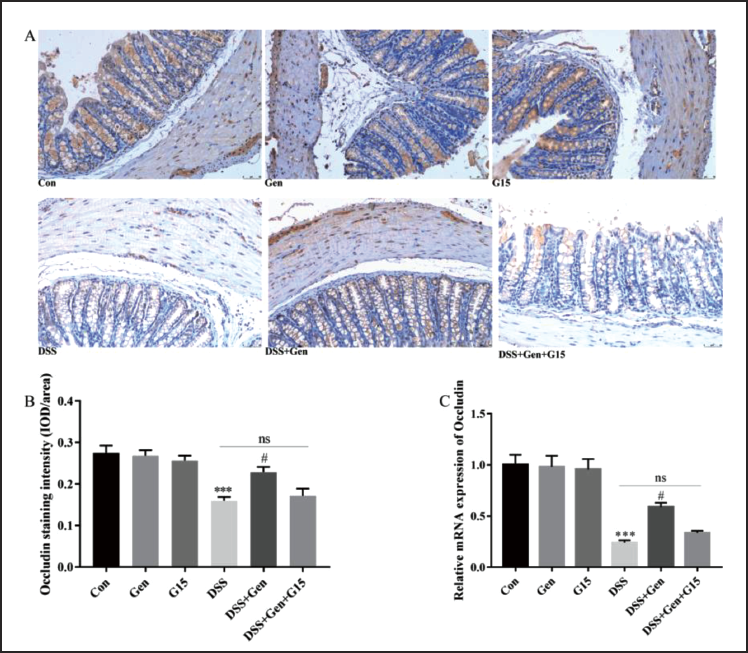
Genistein Treatment Improves the Integrity of the Intestinal Barrier in DSS-induced Acute Colitis
The impaired intestinal barrier is one of the primary pathological hallmarks in UC, and tight junction proteins such as occludin play an essential role in maintaining the integrity of the intestinal barrier. Thus, we evaluated occludin expression in distal colonic tissues to evaluate the integrity of the intestinal barrier using immunohistochemical staining (Figure 4A) and qRT-PCR (Figure 4C). Compared to controls, DSS exposure resulted in decreased expression of occludin as observed in both immunohistochemical staining (p < 0.001, Figure 4B) and qRT-PCR data (p < 0.001, Figure 4C). Genistein limited DSS-induced impairment of the intestinal barrier as evidenced by an increased mean density of occludin immunohistochemical staining (p = 0.017, Figure 4B) and increased occludin mRNA abundance using qRT-PCR (p = 0.033, Figure 4C). Similarly, treatment using the combination of genistein and G15 had no effects on occludin expression in the UC model animals. Furthermore, no differences were observed among the control, genistein, and G15-only groups. These data suggest that genistein, by targeting GPER, improves the integrity of the intestinal barrier normally seen in the DSS-induced acute colitis model.
Genistein Treatment Improves the Antioxidant and Anti-inflammation Activity in DSS-induced Colitis
We next examined the potential anti-oxidative and anti-inflammatory effects of genistein in this acute colitis model. As shown in Figure 5A, DSS induction resulted in increased release of lipid peroxidase (MDA) (p < 0.001) and oxidative stress marker enzyme MPO (p < 0.001). In contrast, DSS decreased the abundance of antioxidant enzymes GSH (p < 0.001) and SOD (p < 0.001) in the distal colonic tissues. We noted that genistein treatment in this colitis model significantly decreased the relative abundance of MDA (p = 0.004) and MPO (p = 0.017) while increasing the levels of GSH (p = 0.022) and SOD (p = 0.002) in the colon tissues. In addition, ELISA (Figure 5B) showed that genistein treatment in this colitis model limited the DSS-induced pro-inflammatory microenvironment, as evidenced by decreased pro-inflammatory cytokines IL1-β (p = 0.012), IL-6 (p < 0.001), and TNF-α (p = 0.038). Furthermore, genistein treatment in this colitis model significantly increased the levels of anti-inflammatory cytokine IL-10 (p = 0.030) in serum.
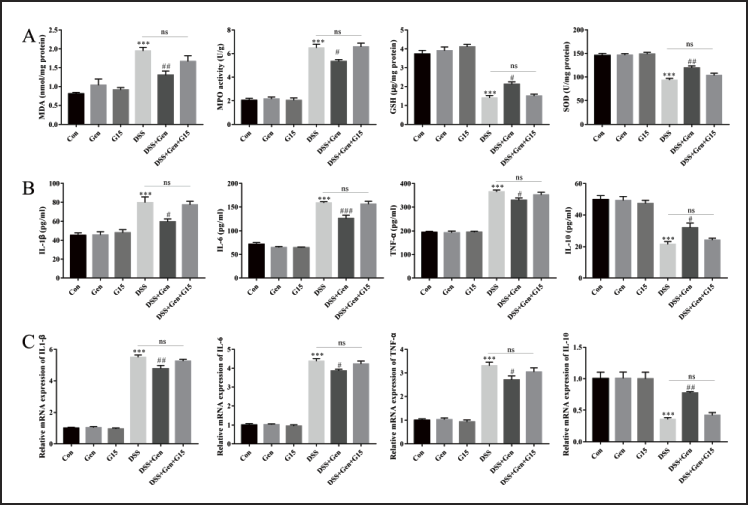
qRT-PCR (Figure 5C) further verified the effects of genistein in reducing inflammatory effects in the DSS-induced acute colitis model. Specifically, qRT-PCR data revealed the decreased mRNA abundance of IL1-β (p = 0.002), IL-6 (p = 0.016), and TNF-α (p = 0.03) and an increased expression of IL-10 (p = 0.006) in the distal colonic tissues. Of note, the combination of genistein and G15 counteracted the anti-oxidative and anti-inflammatory effects of genistein in the UC model. No differences were observed among the control, genistein, and G15-only groups. These data provide evidence that genistein exerts anti-oxidative and anti-inflammatory effects in the DSS-induced acute colitis model by targeting GPER.
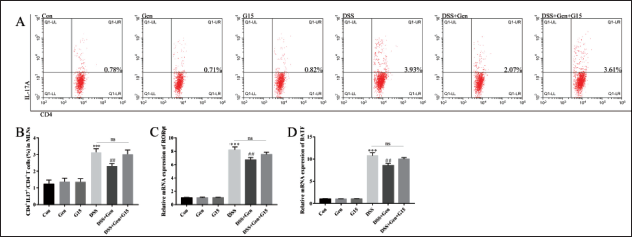
Genistein Treatment Represses Th17 Expansion in DSS-induced Colitis
The Th17/Treg cell imbalance, which favors the pro-inflammatory and destructive Th17 subpopulation, plays a vital role in the onset and progression of UC. Flow cytometry analysis (Figure 6A) demonstrated that, compared to the control group, DSS exposure resulted in an increase in the Th17 population in MLN (p < 0.001, Figure 6B). In contrast, genistein treatment in this UC model inhibited increased levels of Th17 in MLN (p = 0.007, Figure 6B). Furthermore, qRT-PCR demonstrated that genistein treatment in this UC model significantly inhibited Th17 expansion, as evidenced by decreased expression of the key transcription factors RORγt (p = 0.007, Figure 6C) and BATF (p = 0.003, Figure 6D) when compared to the DSS-only group. The combined treatment of genistein and G15 counteracted the effects of genistein-only treatment in this acute colitis model. No differences were observed among the control, genistein, and G15 groups. These data suggest that genistein restrains Th17 cell expansion in the DSS-induced acute colitis model by targeting GPER.
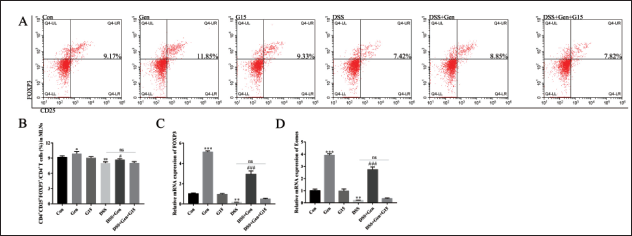
Genistein Treatment Boosts Treg Expansion in DSS-induced Acute Colitis
Our flow cytometry analysis (Figure 7A) indicated that DSS treatment resulted in a decreased population of Treg in MLN compared to the control group (p = 0.0078, Figure 7B). In contrast, genistein treatment in this UC model mice boosted the Treg population when compared to the DSS-only group (p = 0.026, Figure 7B). In addition, genistein-only treatment showed an increase in Treg populations when compared to the control animals (p = 0.021, Figure 7B). qRT-PCR analysis further indicated that genistein treatment in the DSS group dramatically resulted in increased abundance of Treg-associated transcription factors FOXP3 (p = 0.001, Figure 7C) and EOMES (p < 0.001, Figure 7). Moreover, the genistein group exhibited an increased expression of FOXP3 (p < 0.001, Figure 7C) and EOMES (p < 0.001, Figure 7D) compared to the control group. Consistent with previous results, the combination of genistein and G15 counteracted these observed effects of genistein-only treatment in the UC model. No differences were observed between the control and G15-only groups. These data indicate that genistein may promote Treg cell expansion in the DSS-induced acute colitis model by targeting GPER.
Discussion
In this report, we explored the therapeutic effects of genistein in a DSS-induced experimental mice model of acute colitis. Genistein treatment remarkably alleviated colonic injury as evidenced by improved severity of disease, histopathological alterations, anti-inflammatory and anti-oxidative properties, splenic immune function, and intestinal barrier integrity. Mechanistically, we posit that by targeting GPER, genistein exerted a potent role in modulating Th17/Treg cell balance, resulting in a resetting of the intestinal immune microenvironment and alleviation of inflammatory signatures.
Adaptive immunity plays a particularly important role in the pathogenesis of UC, and T helper (Th) cells are the principal regulators of the immune response in this disease. The principal subtypes of Th cells are Th1, Th2, Treg, and Th17 (Mousset et al., 2019). Under normal conditions, Th17 secretes chemokines and inflammatory factors that defend against bacterial and fungal infections. In contrast, Treg plays an immunosuppressive role, resulting in immune tolerance. The imbalance of Th17/Treg caused by either dysregulated activity and over-proliferation of Th17 cells or, in contrast, Treg deficiency is a key factor in the pathogenesis of UC. Therefore, it is crucial to understand the factors that affect the balance of Th17/Treg cells to better understand and effectively treat UC. Th17/Treg balance is regulated by several factors, including co-stimulatory signals, cytokines with immunomodulatory properties, metabolic status, gut microbiota, and receptors expressed by CD4+ T cells (Zhang et al., 2021). Many phytochemicals have been proven to regulate Th17/Treg cell balance to alleviate UC. Such phytochemicals like curcumin, gingerol, and ginsenosides have been investigated, and as their side effects are limited, these compounds have promising prospects for their clinical application (Sheng et al., 2020; Xiao et al., 2022; Zhang et al., 2022). We found that genistein significantly alleviated DAI scores and pathological changes within the intestine and promoted increased integrity of the intestinal barrier. The disruption of the intestinal barrier is hallmarked by UC; tight junction protein such as occludin maintains the intestinal barrier by protecting the intestinal mucosa from potentially toxic substances. IHC staining and mRNA expression for occludin suggested that genistein ameliorated DSS-induced colonic injury, indicating the potential protective effect of genistein on the intestinal barrier. Previous studies determined that genistein has anti-inflammatory and antioxidant effects, and although the underlying mechanism responsible for this effect is unclear (Al-Khayri et al., 2022; Weng et al., 2019), we speculate that genistein’s therapeutic effect on UC may be related to its anti-inflammatory properties and this led us to examine the intestinal inflammatory status in UC mice. The results showed that genistein significantly improved the inflammatory microenvironment of UC mice, as shown by a limited release of pro-inflammatory factors such as IL1-β, TNF-α, and IL-6 in response to DSS and promoting the release of the anti-inflammatory factor IL-10. Considering the importance of Th17/Treg balance in UC-associated inflammation, we further measured Th17 and Treg abundance in MLN by flow cytometry. We observed that the number of Th17 cells as well as the expression of their transcription factors RORγt and BATF were significantly increased. In contrast, the number of Treg and the expression of transcription factors FOXP3 and EOMES were significantly reduced. These findings suggest that genistein can enhance the anti-inflammatory response in UC mice by regulating Th17/Treg cell balance.
The influence of the menstrual cycle on the course of inflammatory bowel disease (IBD) has been identified for decades (Kane et al., 1998). Women with bowel disease often display worse symptoms during menstruation, and older women are more likely to develop IBD than younger women (Greuter et al., 2020). These findings suggest that sex hormones, such as estrogen, likely play an important role in IBD. As a phytoestrogen, we speculated that the therapeutic effect of genistein on UC mice may be related to estrogen receptor activation and this led us to explore its relationship with GPER. The results showed that genistein, when combined with the GPER inhibitor G15, did not significantly improve the pathological changes in intestinal morphology and inflammatory microenvironment displayed in UC model mice. We further examined the expansion of Th17/Treg cells, and, as predicted, genistein treatment boosted Treg abundance and inhibited Th17 expansion in the DSS-induced UC model. Furthermore, the combination of genistein and G15 diminished these effects. These data indicate that genistein can ameliorate UC by targeting GPER.
Consistent with our findings, a prior pre-clinical experimental animal study also indicated that GPER may be a therapeutic target for UC (Wang et al., 2021). We further found that the therapeutic effects of GPER may be associated with its potential to modulate Th17/Treg cell balance. GPER is a G protein-coupled receptor with seven transmembrane domains, binds estradiol with high affinity, and activates downstream signaling pathways. Unlike the classical estrogen receptors α and β, GPER mediates a series of rapid biological functions (Jacenik et al., 2019). Many studies have shown that GPER has a marked effect on the function and expansion of Th17 and Treg cells and a variety of mechanisms may be involved in this effect. Previous studies have shown that GPER activation can lead to thymic atrophy, suggesting that GPER may act as an immunosuppressive agent (Wang et al., 2008). An in vitro study also showed that the GPER activator G1 promoted FOXP3 expression and subsequent Treg expansion in the condition of Th17 cell polarization (Brunsing et al., 2013). In addition, G1 can also boost Treg expansion by promoting the expression of IL-10, and this effect was abolished by ERK inhibitors, but not by p38 or Jun N-terminal kinase inhibitors, suggesting that GPER regulates IL-10 production through ERK (Brunsing et al., 2011).
We note some limitations in this study. First, purified CD4+ T cells that lack GPER should be used to determine the effects of genistein on the differentiation and expansion of Th17/Treg. Second, the exact mechanism by which GPER regulates Th17/Treg balance requires further exploration. Finally, this study was conducted on mice, so it is unclear whether the findings can be directly applied to humans. Future studies should investigate the effects of genistein on human patients with UC.
Conclusion
Genistein robustly attenuated the pathologies observed in the DSS-induced acute UC mice model by regulating Th17/Treg cell balance. The effect target of genistein in UC may, at least in part, be through targeting GPER.
Abbreviations
DSS: Dextran sulfate sodium; Th17: T helper cell 17; Treg: Regulatory t cell; UC: Ulcerative colitis; GPER: G protein coupled estrogen receptor; DAI: Disease activity index; SI: Spleen index; HE: Hematoxylin-eosin; MPO: Myeloperoxidase; MDA: Malondialdehyde; SOD: Superoxide dismutase; MLN: Mesenteric lymph nodes; FOXP3: Forkhead box P3; EOMES: Eomesodermin.
Footnotes
Acknowledgements
We appreciate Ejear-editing for preparing this manuscript.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Provincial Natural Science Foundation of Hunan (2020JJ8002).
Statement of Ethical Approval and Informed Consent
This study was approved the the Ethics Committee of The Central Hospital of Hengyang City.
