Abstract
Background
Objectives
This study aimed to discover the mechanism by which SC treats insomnia through network pharmacology.
Materials and Methods
It was used to study the chemical constituents of SC by ultra-high-performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-QTOF-MS). The SC–Insomnia network and the protein–protein interaction (PPI) network were set up using network pharmacology to research the mechanisms by which SC treats insomnia. The targets found in network pharmacology were proven by enzyme-linked immunosorbent assays (ELISA) and real-time polymerase chain reactions (RT-PCR).
Results
It identified 26 monomer compounds, all of which were lignans. From the SC–Insomnia and PPI networks, seven main active lignans were discovered, which could play an important part in treating insomnia, along with 11 targets that play an important part in the sleep−wake cycle. It was verified by the expression of GABA with ELISA and GABRA1 with RT-PCR.
Conclusion
Based on network pharmacology, the mechanism by which SC treats insomnia is through the sleep–wake cycle. It was a foundation to rationally apply SC for treating insomnia in the clinic.
Introduction
Insomnia is caused by anxiety, excessive fatigue, or neuropathological damage caused by a continuous decline in sleep quality. People suffering from insomnia have difficulty falling asleep, have shallow sleep, are easy to wake up, have more dreams, and so on (Marais & Osuch, 2020). Nowadays, the rhythm of social life is accelerating day by day. With people’s mental and psychological pressure increasing, the population of insomnia is expanding. In recent years, the search for safe and effective methods to prevent and treat insomnia has become the focus of medical attention. With the continuous deepening of the study of traditional Chinese medicine, it has shown good efficacy, less toxicity and side effects, and fewer adverse reactions (Xu et al., 2019).
Network pharmacology interprets the occurrence and development of diseases from the perspective of system biology and the balance of biological networks and understands the interaction between drugs and organisms from the perspective of the holistic view of improving or restoring the balance of biological networks (Li et al., 2022). Because of its holistic and systematic characteristics, it has been widely used in the study of traditional Chinese medicine, which is consistent with the holistic view of traditional Chinese medicine and the principle of treatment based on syndrome differentiation (Xiong et al., 2022).
In this study, the chemical constituents of SC were detected by ultra-high-performance liquid chromatography-quadrupole time-of-flight mass spectrometry (UPLC-QTOF-MS). The mechanism of SC treating insomnia was studied by the SC–Insomnia network and protein–protein interaction (PPI) network. After proving it by enzyme-linked immunosorbent assay (ELISA) and real-time polymerase chain reactions (RT-PCR), it was discovered that SC treats insomnia.
Materials and Methods
Reagents and Materials
Five chemical reference standards, including Schisandrol A (Batch No. 110857-201513), Schisandrin A (Batch No. 110764-201312), Schisantherin A (Batch No. MUST-17032302), Schisandrol B (Batch No. MUST-17040307), and Schisandrin C (Batch No. MUST-17031816). 4-Chloro-dl-phenylalanine (PCPA) was purchased from Sigma (Company Inc., USA). Pentobarbitone sodium was obtained from Merck (Company Inc., Germany). SC was purchased from the Affiliated Hospital of Liaoning University of Traditional Chinese Medicine (Shenyang, China). GABA ELISA Kit was provided by AMEKO (Shanghai, China). Prime® Script RT Reagent Kit with gDNA Eraser and SYBR®Premix Ex Taq TMII (TliRNaseH Plus) were provided by Takara (Japan). Primer sequences were synthesized by Sangong Biotech (Shanghai, China).
Preparation of SC
SC (0.5 g) was extracted with methanol into a 25 mL volumetric flask by ultrasonic treatment (power 250 W, frequency 20 kHz) for 30 min and filtered for chemical constituent analysis. SC was reflux-extracted from 95% ethanol twice. The reflux extracts were combined and concentrated for intragastric administration in rats.
Determination of Chromatographic
Chromatography was conducted on the Agilent 1290 series UPLC system by a 4.6 mm × 100 mm Poroshell SB-C18 column. The column temperature was maintained at 30℃. The gradient mobile phase consisted of solvent A (0.01% formic acid in water) and mobile phase B (acetonitrile) at a flow rate of 0.6 mL/min in positive mode. The gradient elution procedure was 5%–100% B for 0–30 min and 100%–100% B for 30–37 min by a sample injection volume of 0.4 µL.
An Agilent-6550 Q-TOF mass spectrometry was performed with a mass range of
Analysis of the Chemical Constituents of SC by Mass Spectrometry
The Agilent Mass Hunter qualitative analysis was used to analyze the chemical composition by mass spectra of SC in positive ion mode. The characteristic mass spectra peaks were extracted through molecular feature extraction to infer the compound formula with the precise molecular weight and the isotope matching results. The compounds were preliminarily determined by reference standards and reports.
SC–Insomnia Network and PPI Network
Based on the results of SC’s chemical constituents, the pharmacology database and analysis platform of the traditional Chinese medicine system (
Ethics Statement
All experimental procedures have been approved by the Use of Medicine Ethics Review Committee for animal experiments at Liaoning University of Traditional Chinese Medicine.
Animals
All rats (230–270 g) were purchased by the experimental animal center of Liaoning Changsheng Biological Technology Company. They were housed in an environment under the following conditions: a temperature of 22℃ and a humidity of 50%–60%.
Induction of Insomnia and Experimental Groups
Insomnia was induced in rats, followed by intraperitoneal PCPA (0.3 g/kg) for 4 days. All rats were randomly divided into six groups, including normal control, model, diazepam, SC high dose, SC medium dose, and SC low dose groups, with 8 rats in each group. At 5 days after the induction of insomnia, rats were given oral administration with saline in the normal control and model groups. The other groups were given oral administration of high-dose SC (1.944 g/kg), medium-dose SC (0.648 g/kg), low-dose SC (0.216 g/kg), and diazepam (0.1 g/kg) for 14 days.
Sleep Synergistic Experiment by Pentobarbital Sodium
The suprathreshold dose of pentobarbital sodium was 50 mg/kg and the subthreshold dose was 30 mg/kg, according to the pre-experimental results. Six rats in each group were selected for the experiment. After a day of the last given SC, each group was injected intraperitoneally with pentobarbital sodium (30 mg/kg, 50 mg/kg) in subthreshold and suprathreshold dose experiments separately. The number of rats falling asleep in each group was observed and recorded in the subthreshold dose experiment. The disappearance time of the righting reflex (with the disappearance of the righting reflex within 1 min as the standard) and the sleep duration (recovery of the righting reflex as the standard) were observed and recorded in a suprathreshold dose experiment.
ELISA
The levels of γ-aminobutyric acid in rats’ brain samples were from control, model, diazepam, and high-dose, middle-dose, and low-dose groups, with eight in each group. The results were determined by ELISA on the basis of the instructions of the manufacturer.
Molecular Data
RNA from each rat’s brain tissue was extracted by TRIZOL. cDNA was produced by a gDNA removal reaction and a reverse transcription reaction. Differential gene expression was evaluated by qPCR. The results were quantitative in the ratio of GABRA1mRNA to β-actin. The following primers (5′ to 3′ end) were used: for GABRA1mRNA, forward primer (CGTGGTTCCAGAAAAGCCAA) and reverse primer (GCTGGTTGCTGTAGGAGCAT); for β-actin, forward primer (CGCGAGTACAACCTTCTTGC) and reverse primer (CGTCATCCATGGCGAACTGG).
Statistical Analysis
SPSS 19.0 statistical software was used to analyze all quantitative data, which were expressed as the means ± SD as indicated.
Results
The Chemical Constituents of SC
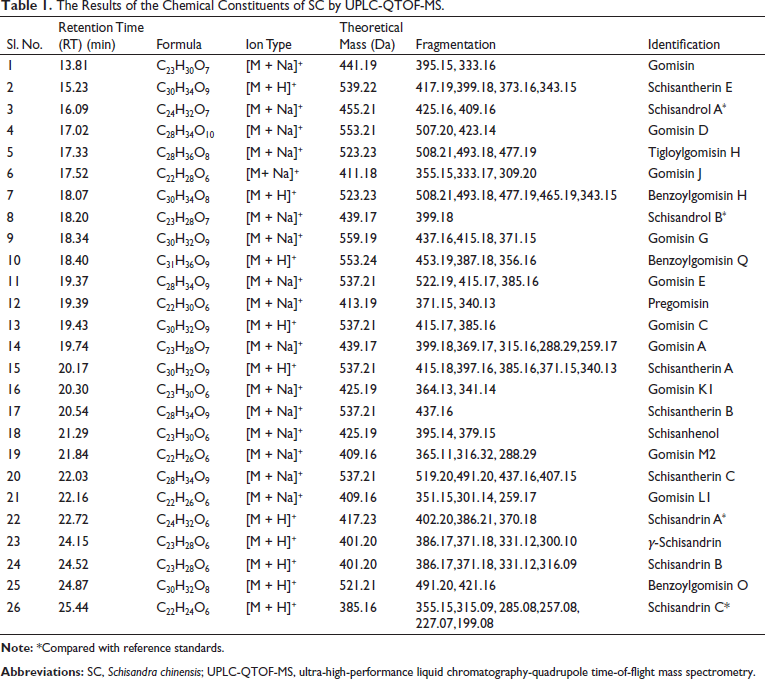
The Base Peak Chromatogram (BPC) was observed in Figure 1 by using the UPLC-QTOF-MS condition described above. In positive-ion mode, 26 monomer compounds were identified (Gao et al., 2019; Guo et al., 2021; He et al., 2013; Huang et al., 2008; Lee et al., 2013; Lei et al., 2020; Li et al., 2016, 2018; Liu et al., 2013, 2017; Onay et al., 2020; Shi et al., 2009; Sun et al., 2019; Yang et al., 2017; Zhang et al., 2017; Zhou et al., 2011), all of which were lignans in Table 1. The specific secondary cracking process of the representative compound is shown in Figure 2.
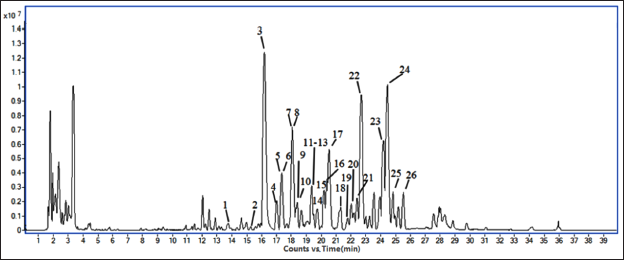
The Results of the Chemical Constituents of SC by UPLC-QTOF-MS.
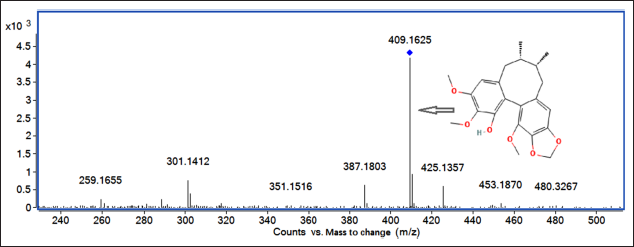
Quasi-molecular ion peak was 409.16 [M + Na]+; the elemental composition of the compound was C22H26O6, and secondary ion fragments information of the compound were 351.15, 301.14, and 259.17.
SC–Insomnia Network and PPI Network
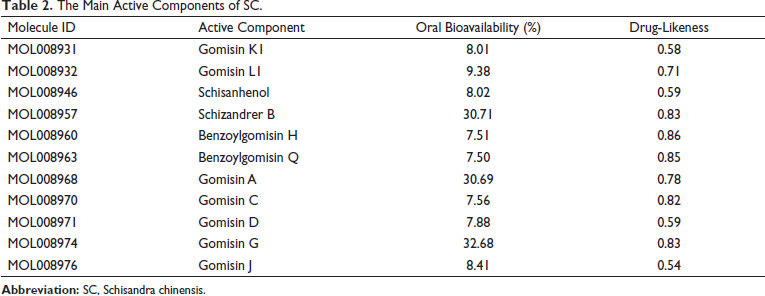
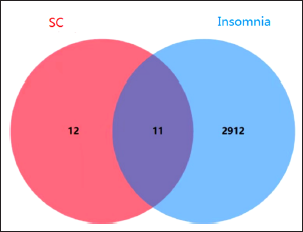
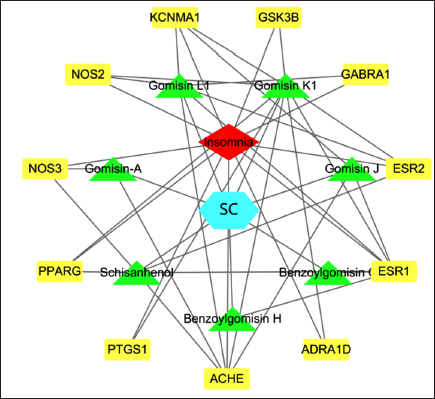
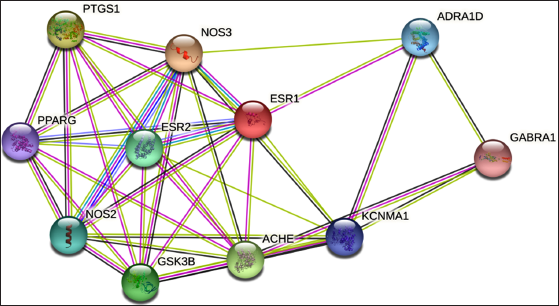
The 11 active constituents or targets of SC were found with oral bioavailability ≥30% or drug-likeness ≥0.18 from the TCMSP database (Table 2). Through Venn analysis, 11 common targets of SC–Insomnia were illustrated, as shown in Figure 3. The SC–Insomnia network was visualized by Cytoscape software in Figure 4. The seven main active components could play an important part in treating insomnia by targeting the following targets: calcium-activated potassium channel subunit α1 (KCNMA1), Glycogen synthase kinase-3 β (GSK3B), GABRA1, estrogen receptor 2 (ESR2), ESR1, adrenergic receptor α1D (ADRA1D), acetylcholinesterase (ACHE), prostaglandin-endoperoxide synthase 1 (PTGS1), recombinant human peroxidase proliferator-activated receptor γ (PPARG), endothelial nitric oxide synthase (NOS3), and induced nitric oxide synthase (NOS2). The SC–Insomnia PPI network was obtained by the String platform, importing the aforementioned 11 targets,
The Main Active Components of SC.
Sleep Synergistic Experiment Results by Pentobarbital Sodium
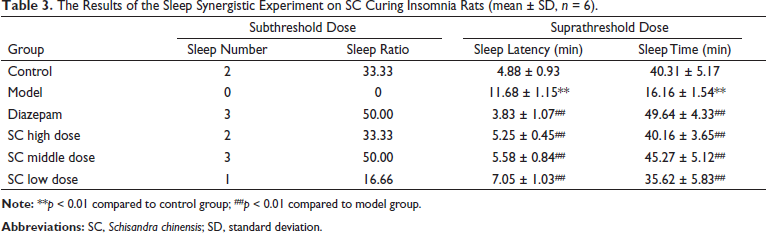
It was found in Table 3 that the sleep latency and sleep time were sharply shortened in the model group compared with the control group (
The Results of the Sleep Synergistic Experiment on SC Curing Insomnia Rats (mean ± SD, n = 6).
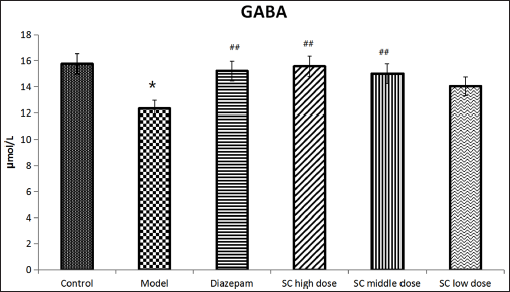
Effect of SC on Level of GABA
The results are displayed in Figure 6. In comparison with the control group (15.76 ± 2.60), the level of GABA was down distinctly in the model group (12.33 ± 1.67) (
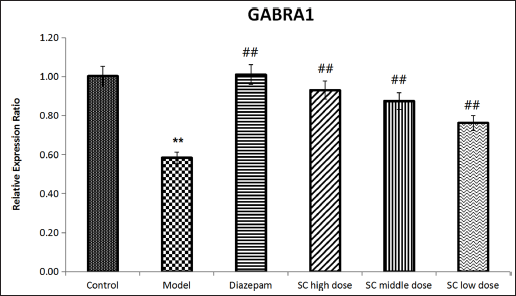
Determination of GABRA1 mRNA Level
The results of the determination of GABRA1 mRNA levels are shown in Figure 7. Compared with the control group (1.00 ± 0.06), GABRA1 mRNA levels were significantly reduced in the model group (0.58 ± 0.03) (
Discussion
Sleep is an important part of the sleep–wake cycle. The continuous reduction of sleep time caused by various reasons often leads to many physiological disorders, especially abnormal brain function. The sleep-wake cycle is controlled by two major systems: the sleep-promoting neuron and the arousal neuron (Li, 2008). It was demonstrated in Figure 8 that SC was effective in treating insomnia through the sleep–wake cycle.
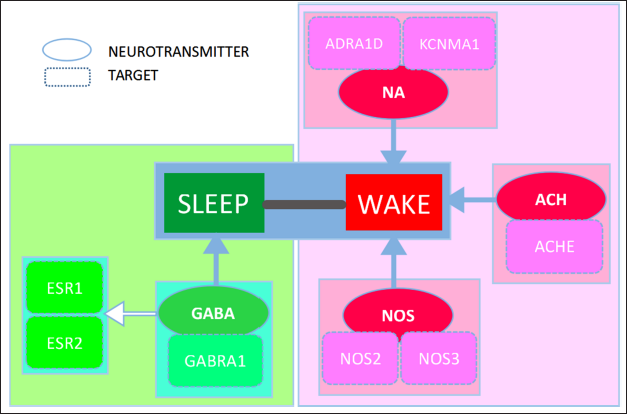
In the sleep-promoting neuronal system, GABA, as an inhibitory neurotransmitter affecting sleep, is the neurotransmitter in most of the major neurons. The specific transmembrane receptors (GABAAR, GABABR, and GABACR) are bonded to GABA in the plasma membrane through both presynaptic and postsynaptic neurons in the nervous system. This binding causes the opening of ion channels to allow either the flow of negatively charged chloride ions into the cell or positively charged potassium ions out of the cell. GABAAR is commonly used in the study of insomnia, and GABAAR acts as a GABRA1 receptor (Liang & Chen, 2015; Liu et al., 2011; Zhong et al., 2021). The results showed that the contents of GABA and GABRA1mRNA in rats’ brains in the model group were decreased (
In the sleep–wake cycle, the other factor is the arousal neuron system, which is much more complex. It is composed of multiple neuronal systems that use different neurotransmitters, including glutamate, (-)-noradrenaline, dopamine, 5-hydroxytryptamine, histamine, nitric oxide, acetylcholine, and so on. The following factors could affect the arousal neuron. ADRA1D has receptors that respond to epinephrine and (-)-noradrenaline in the organs that receive sympathetic postganglionic nerve fibers (Orand et al., 2015). By bonding calcium channels, some adrenergic receptors are regulated by reducing activity and release. KCNMA1, regulating calcium channel activation, might influence sleep (Contet et al., 2016). Nitric oxide synthase belongs to the nervous system, and NOS2 and NOS3 are its isoforms (González-Castro et al., 2019). ACHE is a key enzyme in biological nerve conduction. It degrades acetylcholine at the cholinergic synapse and stops the excitatory effect of neurotransmitters on the postsynaptic membrane to ensure the normal transmission of nerve signals in the body (Liu et al., 2022). PPARG is a group of nuclear proteins involved in the transcription of factors that regulate a variety of biological functions, including gene expression, inflammatory processes, and related pathology. PPAR α, PPAR γ, and PPAR β are included in the PPAR family subtypes. Some scholars have found that activation of PPAR α could strongly enhance wakefulness while reducing sleep (Montagner et al., 2011; Murillo-Rodriguez, 2017).
In addition, PTGS1 cyclooxygenase1, which is involved in and exacerbates inflammation, enhances sedative-hypnotic effects by reducing the level of PTGS1 and other inflammatory factors (Xie et al., 2015).GSK3B is a central nervous system serine-threonine kinase that is involved in mood regulation and circadian rhythms. It has been shown that GSK3B can indirectly affect the expression of
Conclusion
The research identified 26 monomer compounds by UPLC-QTOF-MS. Based on network pharmacology, seven main active components and eleven targets were found. The mechanism by which SC treats insomnia was through the sleep–wake cycle. It was a foundation to rationally apply SC for treating insomnia in the clinic.
Summary
Through network pharmacology, this study discovered the mechanism of SC in curing insomnia. It could be indicated that SC treats insomnia by affecting the sleep−wake cycle, which laid a foundation to rationally apply to treat insomnia of SC in the clinic.
Footnotes
Abbreviations
SC:
Acknowledgments
The authors are grateful to the Department of Science and Technology of Liaoning Province for partial financial support.
Declaration of Conflicting Interest
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by grants from the guiding project of the Natural Science Foundation of the Department of Science and Technology of Liaoning Province (No.2019-ZD-0442).
Statement of Informed Consent and Ethical Approval
The animal experimental procedures have been approved by the Use of Medicine Ethics Review Committee for animal experiments at Liaoning University of Traditional Chinese Medicine.
