Abstract
Background
Previous research has highlighted the regulatory role of miR-182-5p in targeting TLR4 during the pathogenesis of allergic rhinitis. In a different context, TLR4 has been identified as a crucial factor in the development of lung ischemia-reperfusion injury, where its upregulation is believed to initiate the injury process. Additionally, miR-199a-3p has been shown to possess cardioprotective properties in simulated ischemia/reperfusion (I/R) injury models.
Materials and Methods
HE and TUNEL were performed to evaluate the cardiac injury and cellular apoptosis of I/R mice under distinct conditions. Real-time PCR was used to analyze the expression of microRNAs (miRNAs) and mRNAs under differential conditions.
Results
Pretreatment by Shexiang Tongxin Dropping (SXTXD) has been shown to significantly augment the therapeutic efficacy of MSC-derived exosomes (MSC-EXOs) in attenuating cardiac injury in I/R mice. MSC-EXOs effectively restored the repressed miR-182/miR-199a-3p expressions and activated TLR4/CD44 expressions in I/R mice, while SXTXD pretreatment remarkably strengthened the efficiency of MSC-EXOs. Moreover, SXTXD pretreatment notably reinforced the capability of MSC-EXOs to maintain cardiac parameters, including iNOS and interleukin-1β (IL-1β). Furthermore, the luciferase assay indicated that miR-182-5p and miR-199a-3p effectively suppressed the luciferase activities of TLR4 and CD44, respectively, through binding to the 3´ UTR. The overexpression of miR-182-5p and miR-199a-3p significantly suppressed the expression of TLR4 and CD44 in H2C6 and RAW264.7 cells.
Conclusion
In conclusion, our investigation indicates that MSC-derived exosomes, pretreated with SXTXD, hold promise in mitigating cardiac I/R injury by modulating inflammatory responses through the miR-182-5p/TLR4 axis and miR-199a-3p/CD44 axis. These findings suggest potential therapeutic strategies for addressing I/R-related cardiac complications.
Introduction
Myocardial ischemia/reperfusion (I/R) injury is a pathophysiology comprised of a process once the blood supply is restored following myocardial ischemia, which usually happens during or after medical procedures such as angioplasty, coronary artery bypass surgery, or myocardial infarction (Frank et al., 2012; He et al., 2022). Scientists have discovered that I/R injury can initiate a cascade of inflammatory responses, resulting in the release of pro-inflammatory molecules like cytokines and chemokines. These responses can further aggravate tissue damage or disrupt metabolic regulation (Shen et al., 2019). Also, the severity and length of the inflammatory response after the I/R process correlate with heart injury and scar formation (Eltzschig & Eckle, 2011; Frangogiannis, 2012). Therefore, modulation of inflammatory responses has been studied as a potential therapeutic target. Also, other innovative strategies such as stem cell-based therapies (Yu et al., 2017), gene therapies (Lavu et al., 2011), nanotechnology-based interventions (Zhao et al., 2022), and targeted drug delivery systems (George et al., 2022) have been developed.
Exosomes, which are small vesicles rich in nucleic acids, are known to play a key role in intercellular communication and material transport. Recent studies have revealed that exosomes derived from mesenchymal stem cells (MSCs) hold promise as a therapeutic tool for inflammatory diseases, injury repair, degenerative diseases, and tumors (Xu et al., 2020). And microRNAs (miRNAs), which are a type of non-coding RNA made up of only 17–24 nucleotides, have also been found to play a crucial role within exosomes. Valadi et al. (2007) showed that miRNAs are transferred within the cells using exosomes. Previous studies have reported that exosomes are carriers of miRNAs and proteins, which are needed for intracellular communications. Various types of exosomes have been shown to reduce MI/R injury (Chen et al., 2013; Vicencio et al., 2015; Xiao et al., 2017). Moreover, miR-182-5p is shown to be involved in the pathophysiology of cancer, cardiovascular disease, and acute lung injury (Gao et al., 2020; Guzzolino et al., 2020; Zhu et al., 2018). Zhu et al. (2018) revealed that miR-182-5p decreased TLR4-mediated immune responses. Several cardiovascular diseases, such as atrial fibrillation and cardiac failure, showed involvement of miR-199a-5p (Chiang et al., 2015; Haghikia et al., 2011). Hence, therapeutic agents targeting miRNA could be a promising option for healing I/R injuries.
MSCs hold significant promise as a potential source for cancer therapy, mainly through their ability to act as carriers in cancer gene therapy (Sohrabi et al., 2022). The therapeutic effects of MSCs are primarily attributed to the presence of MSC-derived exosomes, which offer a novel cell-free approach to cancer treatment (Ma et al., 2020). In comparison to MSC transplant therapy, these exosomes present several advantages, including enhanced safety, stability, and ease of storage, transport, and administration (Colombo et al., 2014; Mendt et al., 2018; Sun et al., 2010). It is worth noting that unmodified MSC-derived exosomes may exhibit either tumor-promoting or tumor-inhibiting effects. However, modified MSC-derived exosomes have shown potential for suppressing cancer development and progression by delivering a variety of therapeutic molecules, such as miRNAs, anti-miRNAs, suicide gene mRNAs, chemotherapeutic drugs, and specific siRNAs (Sohrabi et al., 2022).
The current study reported for the first time that CD44 is essentially involved in cardiovascular healing and post-ischemic cardiac injury modeling (Cao et al., 2006). During heart muscle infarction, there was a notable upregulation in the levels of CD44. This increased CD44 expression was observed in blood cells infiltrating the injured tissue. Toll-like receptors (TLR) are a group of receptors that function as an initial defense mechanism against various types of pathogens, like bacteria and viruses (Fan et al., 2006; Qureshi et al., 2006). One of the most studied receptors in this family was TLR4, which is hypothesized to be activated in response to bacterial agents like lipopolysaccharide and lipoteichoic acid, along with oxidative stress and necrosis (Jiang et al., 2005; Karikó et al., 2004; Oppenheim & Yang, 2005).
Previous studies have reported that miR-182-5p targets TLR4 expression, contributing to the pathogenesis of allergic rhinitis. Additionally, TLR4 plays a crucial role in lung I/R injury, as its upregulation is believed to be the initial step in I/R injury progression (Dai et al., 2020; Fujiwara et al., 2019; Merry et al., 2015; Zhang & Jin, 2020). Moreover, miR-199a-3p has been shown to exert protective effects on myocardium and cardiomyocytes during simulated I/R injury (Park et al., 2016; Zuo et al., 2016). Based on these findings, we postulate that miR-182-5p and miR-199a-3p might be involved in the regulatory mechanisms of SXTXD-pretreated MSC-derived exosomes in response to I/R injury.
Materials and Methods
Exosome Isolation and Identification
Exosomes were extracted from bone marrow-derived mesenchymal stem cells (BMDMSCs) with and without SXTXD pretreatment. The isolation process involved a series of centrifugation steps after a 24-h culture period in the presence of vesicle-depleted FBS (Wisent Inc., Saint-Jean-Baptiste, QC, Canada), following a method previously described (Wang et al., 2021). The SXTXD pretreatment entailed the supplementation of vesicle-depleted FBS with 1 µM of SXTXD for BMDMSC cell culture. The use of transmission electron microscopy (TEM) (NanoSight Ltd., Amesbury, UK) facilitated the confirmation of successful exosome isolation.
Animal and Treatment
The research protocol for this study was approved by the institutional ethical committee, adhering to the guidelines set by the hospital’s ethics committee and following the protocol procedures of the National Institutes of Health. Male C57BL/6 mice, aged 8 weeks, were used for the experiments and housed individually in clean, plastic containers with controlled lighting conditions and access to food and water. All animal experiments were conducted in compliance with the Institutional Animal Care and Use Committee (IACUC) guidelines. A total of 40 male C57BL/6 mice were divided into five groups, each comprising eight mice:
Sham group: Mice underwent a sham operation without coronary artery ligation. I/R group: Mice underwent a surgical procedure to expose the heart and ligate the left anterior descending coronary artery (LAD) for 60 minutes, followed by reperfusion. I/R+MSC-EXOs (control) group: Mice received intravenous administration of 25 mL of phosphate-buffered saline (PBS) containing 50 µg of MSC-derived exosomes (MSC-EXOs) derived from bone marrow-derived mesenchymal stem cells (BMDMSCs) one day before and one day after the I/R injury. I/R+MSC-EXOs (SXTXD-pretreated) group: Mice received intravenous administration of 25 mL of phosphate-buffered saline (PBS) containing 50 µg of MSC-EXOs derived from SXTXD-pretreated BMDMSCs one day before and one day after the I/R injury.
The surgical procedure in the I/R group involved exposing the heart and ligating the coronary artery, while the sham group underwent a similar procedure without artery ligation. The administration of MSC-EXOs or SXTXD-pretreated MSC-EXOs was conducted intravenously for both the I/R+MSC-EXOs (control) and I/R+MSC-EXOs (SXTXD-pretreated) groups.
RNA Isolation and Real-Time PCR
In order to examine the effects of exosomes exposed to hypoxic or normoxic conditions, we conducted multiple assays to analyze the expression of specific proteins, including interleukin-1β (IL-1β), Arg1, interleukin-10 (IL-10), interleukin-6 (IL-6), tumor necrosis factor-α (TNF-α), CD206, and tumor growth factor β (TGF-β) mRNA. For each sample, RNA extraction was performed using the RNAiso Plus RNA extraction reagent (Thermo Fisher, MA) following the manufacturer’s instructions to obtain intact RNA. Reverse transcription of the obtained RNA was carried out using the TaqMan Advanced cDNA Synthesis Kit (Abcam, Waltham, MA, USA) and a Reverse Transcription Kit (Qiagen, MD) according to the specific assay procedure provided with the kit. Subsequently, qRT-PCR was performed using a PCR enzyme kit (QIAGEN, Germantown, MD, USA) following the manufacturer’s provided assay protocol. The quantification of gene expressions for IL-1β, Arg1, IL-10, IL-6, TNF-α, CD206, and TGF-β mRNA was analyzed in this study.
Cell Culture and Transfection
H2C6 cells and RAW264.7 cells obtained from Abcam, MA, USA, were cultured in RPMI 1640 medium supplemented with 10% fetal bovine serum and necessary antibiotics. The cells were incubated at 37°C with 5% CO2. Afterward, the cells were divided into four distinct groups:
NC group: cells transfected with 50 nM scramble oligonucleotide; miR-199a-3p group: cells transfected with 50 nM precursor miRNA, miR-199a-3p; miR-182-5p group: cells transfected with 50 nM precursor miRNA, miR-182-5p; and miR-199a-3p+miR-182-5p group: cells transfected with 50 nM of both miR-199a-3p miRNA precursor and miR-182-5p miRNA precursor.
After transfection, the cells were treated for 48 h.
Luciferase Assay
To investigate the regulatory network of miR-182/TLR4 and miR-199a-5p/CD44, we performed a luciferase assay. Two separate luciferase analyses were conducted: the first analysis involved sequence analysis to predict the binding site of miR-182 on TLR4. Wild-type (WT) and mutant TLR4 luciferase-containing vectors were generated and transfected into cells along with miR-182. The second analysis focused on sequence analysis to predict the binding site of miR-199a-5p on CD44. This led to the creation of WT and mutant CD44 luciferase vectors, which were then transfected into cells along with miR-199a-5p. In the luciferase assay protocol, the growth medium was removed from the plates, and cells were rinsed with a PBS solution while taking care not to dislodge the cells. The PBS solution was gently removed, and approximately 20 µL/well of lysis reagent was added. On the plate containing the cell lysate, 100 µL of Luciferase Assay Reagent was added to each well. The emitted illumination was then quantified, and data were collected for analysis.
Apoptosis Analysis
Upon reaching confluence, the cells were seeded into 10-cm dishes at a concentration of 1 x 106 cells. Following trypsinization, the cells were subjected to a phosphate-buffered saline solution wash, as well as fixation and permeabilization with 100 µL of BD Cytofix/Cytoperm buffer (BD Biosciences) for 30 min at room temperature. To execute the RPA2 staining, non-chromatin bound to RPA2 was extracted according to protocol. The cells underwent a PBS solution wash containing BSA, followed by fixation and permeabilization. After being washed with 0.5 mL of 1 × BD Perm/Wash buffer (Sigma, Waltham, MA, USA), the cells were resuspended in 50 µL of the same buffer solution along with specific primary antibodies. The cells were incubated for at least 1 h at room temperature and then washed with the same buffer. The resultant pellets after centrifugation were subjected to redissolution in the aforementioned buffer, which was supplemented with secondary antibodies of particular specificity. The samples were then left to incubate for a duration of 30 min at ambient temperature in the absence of light.
Analysis of Flow Cytometry Samples
The cell samples were treated according to the above-mentioned procedures using the Bio-Rad Flow Cytometer. 4′,6-diamidino-2-phenylindole (DAPI) was used to excite with a 405 nm laser, and emissions were detected with a filter. A laser of 488 nm wavelength was used to detect the Alexa 488 fluorochrome, and the emitted light was collected via a 530/40 filter. Gradually, all the fluorochromes were excited, and no baseline correction was needed.
Immunohistochemistry
The murine cerebral cortex was immobilized with 4% paraformaldehyde, and the cerebral sections were obstructed with 10% normal goat serum. Subsequently, the sections were incubated with TLR4 and CD44 antibodies overnight and then washed with PBS buffer. The images were captured using an upright fluorescence microscope (Nikon).
H&E Staining
The biological tissue examples were fixed with formalin and integrated with paraffin for hematoxylin and eosin (H&E) staining. The sections were scrutinized with the assistance of a fluorescence microscope.
TUNEL Assay
The cells were procured from tissue samples of cohorts of animals and assessed for their apoptosis state using a TUNEL assay kit (Abcam, Waltham, MA, USA) as stipulated by the manufacturer’s assay protocol.
Statistical Analysis
The study’s outcomes were presented as a mean accompanied by the standard error (SD) of the mean. In order to assess discrepancies between results obtained from animal studies, the one-way ANOVA was employed, and the post hoc test of Tukey was employed. In the study, p-values less than 0.05 for all of the distinct assays conducted were regarded as statistically significant.
Results
SXTXD-Pretreated MSC-EXOs is Associated with Decreased Cardiac Injury of I/R Mice
As indicated by Figure 1A, we used an electron microscope to check the EXOs collected from the supernatant of BMCMSCs that had been cultured with or without the SXTXD pretreatment. MSC-EXOs showed remarkable efficiency in restoring the decreased ejection fraction (Figure 1B) and fractional shortening (Figure 1C), while SXTXD-pretreated MSC-EXOs showed a stronger capability than MSC-EXOs. H&E staining and TUNEL assays indicated that SXTXD pretreatment effectively strengthened the effect of MSC-EXOs on attenuating the increased cardiac injury (Figure 1D) and cardiac cellular apoptosis (Figure 1E) of I/R mice.
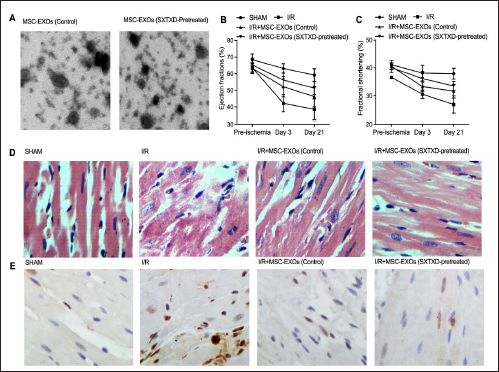
SXTXD-Pretreated MSC-EXOs is Associated with the Restored Expressions of miR-182, miR-199a-3p, TLR4, and CD44 in I/R Mice
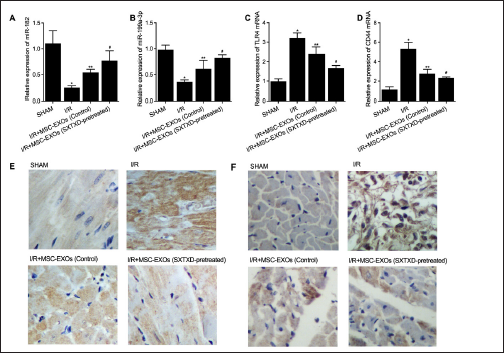
In accordance with the data presented in Figure 2, a noticeable suppression of miR-182 (Figure 2A) and miR-199a-3p (Figure 2B) was observed in I/R mice, in contrast to the sham-operated mice. However, the administration of SXTXD pretreatment effectively enhanced the efficacy of MSC-EXOs in restoring the repressed expression of miR-182 (Figure 2A) and miR-199a-3p (Figure 2B) in I/R mice. Conversely, the upregulation of TLR4 mRNA (Figure 2C) and CD44 mRNA (Figure 2D) was closely associated with MSC-EXOs in I/R mice. Nonetheless, the application of SXTXD pretreatment significantly improved the effect of MSC-EXOs in rectifying the disrupted TLR4 mRNA (Figure 2C) and CD44 mRNA (Figure 2D) in I/R mice. Furthermore, the IHC outcomes also substantiated that SXTXD pretreatment remarkably reinforced the ability of MSC-EXOs to decrease the enhanced expression of TLR4 protein (Figure 2E) and CD44 protein (Figure 2F) in I/R mice.
SXTXD-Pretreatment Remarkably Enhanced the Capability of MSC-EXOs on Regulating the Expression of iNOS, IL-1β, Arg1, IL-10, IL-6, TNF-α, CD206, and TGF-β in I/R Mice
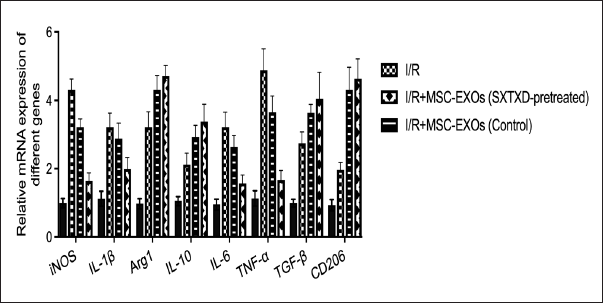
In Figure 3, we conducted a comparative analysis of iNOS, IL-1β, Arg1, IL-10, IL-6, TNF-α, CD206, and TGF-β expression among different groups of mice. Our results revealed that, compared to Sham-operated mice, I/R mice exhibited significantly elevated mRNA expression levels of iNOS, IL-1β, Arg1, IL-10, IL-6, TNF-α, CD206, and TGF-β in cardiac cells. Additionally, we observed that SXTXD pretreatment effectively enhanced the impact of MSC-EXOs in reducing the increased mRNA expression levels of iNOS, IL-1β, IL-6, and TNF-α in the cardiac cells of I/R mice, as compared to the I/R + MSC-EXOs (control) group. However, in I/R mice, MSC-EXOs increased the mRNA expression levels of Arg1, IL-10, CD206, and TGF-β, and SXTXD pretreatment further amplified the activating efficiency of MSC-EXOs in this regard.
The Expression of TLR4 and CD44 were Decreased due to the Binding of miR-182-5p and miR-199a-3p, Respectively on the 3´ UTR Site
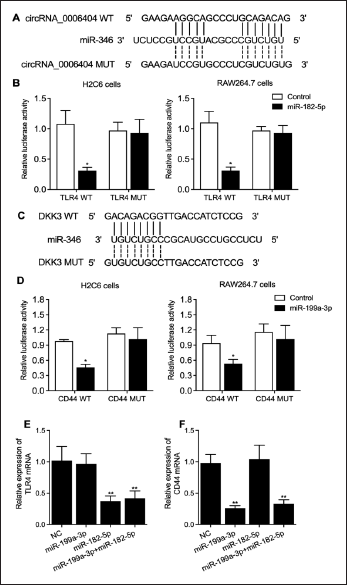
In Figure 4A, we identified a specific binding site for miR-182-5p on TLR4 within the 3′ UTR. Subsequently, when co-transfecting H2C6 and RAW264.7 cells with WT TLR4 and miR-182-5p mimics, we observed a significant suppression of luciferase activities, as depicted in Figure 4B. In Figure 4C, we identified another binding site for miR-199a-3p on CD44 within the 3′ UTR. Similarly, co-transfection of H2C6 and RAW264.7 cells with wild-type CD44 and miR-199a-3p mimics led to a notable reduction in luciferase activities, as demonstrated in Figure 4D. Moreover, we assessed the impact of miR-182-5p and miR-199a-3p+miR-182-5p on the expression of TLR4 mRNA in H2C6 and RAW264.7 cells, as shown in Figure 4E. Our findings revealed significant suppression of TLR4 mRNA expression by miR-182-5p and miR-199a-3p+miR-182-5p. Similarly, in Figure 4F, we investigated the effects of miR-199a-3p and miR-199a-3p+miR-182-5p on CD44 mRNA expression in H2C6 and RAW264.7 cells, demonstrating substantial suppression of CD44 mRNA levels by these miRNAs.
Discussion
The cardiac I/R injury is composed of both ischemia and reperfusion processes. During ischemia, the blood supply to the heart muscle is reduced or interrupted, and the deprivation of oxygen and nutrients may cause cell death and tissue damage (Carlucci et al., 2008). And during reperfusion, the restored blood flow with the sudden influx of oxygen can also cause heart tissue damage, inflammation, and oxidative stress (Marin et al., 2021). Cardiac I/R injury can be life-threatening, depending on the severity of the injury and the individual’s overall health (Hausenloy & Yellon, 2013). For example, severe cardia I/R cases can lead to arrhythmias, myocardial stunning, microvascular obstruction, heart failure, and other complications, which decrease life quality and increase death risk (Hausenloy & Yellon, 2013). Managing the symptoms of I/R injury and reducing the risk of related complications can be achieved through a combination of cardiac rehabilitation and medication treatment, while in severe injury cases, invasive treatments such as coronary artery bypass surgery or heart transplantation are necessary (Ferdinandy et al., 2023; Sánchez-Hernández et al., 2020).
Prior research has also highlighted the effectiveness of exosome treatment in mitigating cardiac I/R injury. For instance, Tang et al. (2020) conducted a recent study showing that MSC-EXOs could protect the myocardium against I/R injury by inhibiting pyroptosis, a critical step in I/R injury treatment. Pyroptosis, a form of programmed cell death associated with inflammatory responses, has been implicated in the pathogenesis of various diseases, including neurodegenerative diseases (Fricker et al., 2018) and atherosclerosis (Duewell et al., 2010). Pyroptosis is mediated primarily by caspase-1 (Chen et al., 1996), and the main signaling pathway of pyroptosis is the NLRP3 inflammatory pathway. Activation of NLRP3 leads to the formation of the NLRP3 inflammasome, which in turn cleaves procaspase-1 into mature caspase-1, promoting inflammatory responses and exacerbating cell damage (Franchi et al., 2009; Kelley et al., 2019). These biological processes significantly modulate the downstream signaling pathways and thus promote inflammatory responses, leading to more cell damage (Schroder & Tschopp, 2010). During early reperfusion, caspase-1 activation has been shown to induce cardiomyocyte pyroptosis (Audia et al., 2018). Tang et al. (2020) demonstrated that MSC-EXOs can downregulate NLRP3 and caspase-1 expressions in I/R-injured SD rats. Moreover, other recent studies have investigated the therapeutic potential of MSC-EXOs in the treatment of I/R injuries. For example, Zhao et al. (2015) reported that MSC-EXOs could promote the repair of ischemic-injured hearts by inhibiting myocardial cell apoptosis, promoting cell proliferation, and inducing angiogenesis through the regulation of Bcl-2 family proteins. Additionally, Zhao et al. (2019) demonstrated that MSC-EXOs could reduce cardiac I/R injury infarct size and alleviate inflammation levels in mice. Furthermore, they found that MSC-EXOs could modify the polarization of M1 macrophages to M2 macrophages by regulating the signaling between miR-182 and TLR4 (Zhao et al., 2019). Overall, these studies collectively confirm the cardioprotective role of MSC-EXOs in the treatment of cardiac I/R injury, which is consistent with the findings of our study.
In contrast to the previously mentioned studies, our research focused primarily on the impact of SXTXD-pretreated MSC-EXOs in the treatment of cardiac I/R injury. SXTXD is a traditional Chinese medicine widely used for treating ischemic heart disease, and prior investigations have explored its mechanism and therapeutic effects in myocardial ischemia (Lin et al., 2017; Qi et al., 2017). For instance, Qi et al. (2017) reported that SXTXD could protect against isoproterenol-induced myocardial ischemia by regulating the ERK1/2 signaling pathway, while Lin et al. (2017) demonstrated that SXTXD promoted the expression of Bcl-1 in rat myocardial tissues. Given that one of the components of SXTXD, muskelin, has been utilized in the treatment of exosomes in other studies, we aimed to compare the therapeutic effects of SXTXD-pretreated MSC-EXOs with regular MSC-EXOs in our investigation. We discovered that SXTXD pretreatment enhanced the effects of MSC-EXOs. Inflammatory responses and related cytokines have been implicated in the pathogenesis of cardiac I/R injury, and the protective effects of MSC-EXOs have been attributed to their ability to suppress activated inflammatory responses in I/R-injured cardiomyocytes. In our study, we assessed various parameters in the cardiac tissue of I/R mice under different conditions. SXTXD pretreatment significantly augmented the ability of MSC-EXOs to regulate the expression of iNOS, IL-1β, Arg1, IL-10, IL-6, TNFα, CD206, and TGFβ in I/R mice. These findings align with previous reports and shed light on the underlying mechanisms of the therapeutic effects of exosomes in treating cardiac I/R injury.
In a previous study that aimed to examine the protective impacts of SXTXD on hypoxia-reoxygenation injury induced by Na2S2O4, SXTXD pretreatment was found to exhibit a notable impact on H9c2 cardiomyoblast cells exposed to Na2S2O4, resulting in increased cell viability and the prevention of abnormal morphological changes. Furthermore, SXTXD pretreatment mitigated the hypoxic damage caused by Na2S2O4, leading to a decrease in pro-apoptotic Bax expression and an increase in anti-apoptotic Bcl-2 expression in H9c2 cells (Lin et al., 2019). In contrast, in this study, we treated I/R mice with SXTXD-pretreated MSC-EXOs. SXTXD-pretreated MSC-EXOs significantly decreased the cardiac injury of I/R mice. Furthermore, we assessed the efficacy of MSC-EXOs (control) and MSC-EXOs (SXTXD-pretreated) in modulating the expression of miR-182, miR-199a-3p, TLR4, and CD44 in I/R mice under different conditions. SXTXD-pretreated MSC-EXOs demonstrated significant capacity for preserving the proper regulation of miR-182, miR-199a-3p, TLR4, and CD44 expression in I/R mice.
The immune cells called macrophages are key mediators of the initiation and characterization of cardiac inflammation. In the current study, the authors used a model named the macrophage depletion model and showed that the effects of exosomes are heavily dependent on their interaction with macrophages. A number of significant pieces of evidence have suggested that macrophage stem cells have the capability to transform into the anti-inflammatory M2 phenotype (Kudlik et al., 2016). In our research, we evaluated various parameters in the cardiac tissue of I/R mice under different conditions. Notably, SXTXD pretreated MSC-EXOs exhibited a remarkable enhancement in their capability to regulate the expression of iNOS, IL-1β, Arg1, IL-10, IL-6, TNF-α, CD206, and TGFβ in I/R mice. These findings align with previous reports and provide insights into the mechanism underlying the therapeutic effects of exosomes in treating cardiac I/R injury.
The role of miR-199a-5p in cardiovascular injury development has been well established in research. Transfection with miR-199a-5p mimics led to reduced levels of GSK-3β, while miR-199a-5p inhibitors increased GSK-3β levels. Studies using various in vivo models of myocardial infarction and ischemic reperfusion have demonstrated the cardioprotective effects of miR-199a-3p and miR-214. In this study, we investigated the regulatory function of miR-182-5p and miR-199a-3p on TLR4 and CD44 using luciferase assays. Both miR-182-5p and miR-199a-3p targeted the 3′ UTR, effectively suppressing TLR4 and CD44 expression, respectively. Past reports showed that downregulation of miR-182-5p protected against cardiac I/R injury via suppressing the production of ROS (Li & Jin, 2022), and this is opposite to the results of our study, which showed that upregulation of miR-182-5p is protective by inhibiting expression of TLR4. We reasoned that such a discrepancy could have resulted from distinct signaling pathways and different downstream effectors that miR-182-5p is involved in at different spatial and temporal points.
Toll-like receptors (TLRs) are a group of antigen-identifying receptors that play a crucial role in initiating immune responses upon exposure to pathogens. In humans, a total of approximately 10 TLRs have been identified. TLRs 3, 4, 5, and 9 have been specifically associated with cardiac I/R injury (Chen et al., 2014; Favre et al., 2007; Oyama et al., 2004; Parapanov et al., 2015). Inhibition of TLR4 by Eritoran has been shown to significantly reduce the size of cardiac infarcts in C57Bl/6 mice (Shimamoto et al., 2006). Moreover, increased expression of TLR4 within monocytes has been observed in patients with acute myocardial infarction (Ishikawa et al., 2008; Kashiwagi et al., 2012). TLR4 serves as a target gene for miR-760 and miR-182-5p, and it is involved in both innate and adaptive immune responses (Wang et al., 2011). Several studies have demonstrated elevated levels of TLR4 expression in the nasal mucosa of I/R patients (Radman et al., 2015).
The surface glycoprotein called CD44 controls the fusion of macrophages and plays a crucial role in the innate immune response in the lungs. In CD44-knockout mice, there was a reduced accumulation and expression of macrophages and anti-inflammatory cytokines (Hollingsworth et al., 2007). The glycoprotein is the key activator of leukocytes and parenchymal cells during the inflammation process. The results of this investigation validate the functional involvement of TLR4 and CD44 in the pathological progression while highlighting the therapeutic efficacy of SXTXD-pretreated MSC-derived exosomes in cardiac I/R injury. This efficacy may be attributed to the exosomes’ capacity to attenuate the activated inflammatory response by modulating the expression of TLR4 and CD44.
Conclusion
Our study not only introduces a novel therapeutic approach using SXTXD-pretreated MSC-derived exosomes for treating cardiac I/R injury, which could be used as an alternative or complementary option to traditional treatments, but also offers valuable targets for future research and drug development by identifying the miR-182-5p/TLR4 axis and miR-199a-3p/CD44 axis as key players in the inflammatory response. Moreover, our research also highlights the potential of stem cell-derived exosomes as powerful therapeutic agents. This field has the potential to revolutionize medical treatments for various diseases and injuries. Finally, this study also supported the assumption that similar inflammatory pathways and exosome-based therapies could be relevant in other disease models that involve tissue damage and inflammation. In summary, our investigation has evinced that SXTXD-pretreated MSC-derived exosomes are associated with attenuated cardiac I/R injury. Also, by establishing the miR-182-5p/TLR4 axis and the miR-199a-3p/CD44 axis, which mediated inflammatory responses, we found that the administration of SXTXD-pretreated MSC-derived exosomes is associated with the regulation of miR-182-5p and miR-199a-3p, as well as the suppressed inflammatory responses mediated by these miRNAs.
However, the present study has certain limitations that must be acknowledged. The sample size employed in the animal study is relatively small, and we think a larger sample size would further support our findings in this study. Also, it is imperative to underscore that the outcomes of this study necessitate further validation of human samples based on clinical observations. More importantly, as SXTXD is usually used for short-term control of coronary heart disease, we did not focus on its long-term effect on cardiac I/R injury. Therefore, it is crucial that future studies delve into the potential long-term protective effect of SXTXD on cardiac I/R injury.
Footnotes
Abbreviations
I/R: Ischemia/reperfusion; SXTXD: Shexiang Tongxin Dropping; EXOs: Exosomes; MSCs: Mesenchymal stem cells; miRNA: microRNA; TLR: Toll-like receptors; BMDMSCs: Bone marrow-derived mesenchymal stem cells.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Budget Project of Shanghai University of Traditional Chinese Medicine (2021LK054), Shanghai Putuo District Health System Science and Technology Innovation Project (ptkwws202209), Hospital Level Project of Putuo Hospital (2020301A), Hundred People Plan of Putuo Hospital (2022-RCJC-01), Shanghai Wu Mengchao Medical Science and Technology Fund Project (JJHXM-2021007), Medical Innovation Research Project of “Scientific and Technological Innovation Action Plan” (20Y11910100, 21Y11909600, and 22Y11909600) and Clinical Advantage Disciplines of Health System in Putuo District of Shanghai (2023ysxk01).
Statement of Informed Consent and Ethical Approval
The institutional ethical committee has approved the protocol of this study. All the animal experiments were performed according to the guidelines of the ethics committee of the hospital. The experiment protocols were also in accordance with the guidelines of the National Institutes of Health.
