Abstract
Background:
Epilepsy is a chronic neurological condition with various underlying mechanisms. It is known to affect a multitude of people across the globe, regardless of age and gender. Seizures associated with epilepsy involve the participation of stimulatory glutamatergic mechanisms along with inflammation and oxidative damage.
Objectives:
In this investigation, the anti-epileptic effect of scutellarein, a phytochemical compound isolated from Erigeron breviscapus (Vant.), has been evaluated in the pentylenetetrazol (PTZ) kindling epilepsy model in mice.
Materials and Methods:
The experimental mice were categorized into six groups with six animals in each. The first control group was given normal saline. The second group was provided only PTZ through an intraperitoneal route to induce seizures. The third and fourth groups received two oral doses of scutellarein (10 and 20 mg/kg) before 30 min of PTZ induction. Diazepam was intraperitoneally administered to the fifth group as a positive control. The impact of scutellarein on the duration and initiation of clonic and tonic convulsion, mortality, kindling, mobility, and immobility duration in PTZ-induced rodents was estimated. Also, the impact of scutellarein on oxidative stress markers and antioxidant and inflammatory marker levels was also evaluated.
Results:
Scutellarein treatment was able to reduce PTZ-induced seizures in mice. In PTZ animals, scutellarein lowered the seizure severity by suppressing the onset and duration of convulsions. Scutellarein successfully modulated the PTZ-provoked changes in gamma-aminobutyric acid (GABA), glutamate, and dopamine levels, as well as Ca2+ ATPase and Na+ K+ ATPase activity.
Conclusion:
Furthermore, it remarkably reduced the oxidative stress markers and decreased the contents of nuclear factor-kappa B (NF-κB), tumor necrosis factor-α (TNF-α), and interleukin-1β (IL-1β) in PTZ animal brain tissues, confirming its anti-convulsant potential.
Introduction
Epilepsy is a highly common and chronic condition of the nervous system that involves involuntary repetitive seizures, affecting 70 million people, regardless of all ages globally, and contributes to approximately 5% of all the nervous system disorders documented annually across the world (Bell et al., 2015; Paudel et al., 2020; Stafstrom et al., 2015). Multiple processes are implicated in the pathophysiology of epilepsy, including genetic factors, oxidative damage, ion channels, neuroinflammation, and cell damage mediated by the induction of apoptosis (Hung et al., 2015). Convulsions associated with epilepsy demonstrate the involvement of stimulatory glutamatergic processes, inflammation, and free radical damage (Firdous et al., 2021). The usage of seizure models is considered necessary for the identification of promising anti-seizure or anti-epileptic drugs for treating epilepsy. Multiple drugs in use today have been, in fact, identified by employing simple seizure tests such as pentylenetetrazol (PTZ) and maximal electroshock seizure (MES) tests (Löscher, 2017). PTZ-stimulated kindling is a commonly used approach for inducing tonic–clonic as well as myoclonic seizures and thereby identifying novel therapeutic agents with potent anti-convulsant properties (Yuen et al., 2015). PTZ is essentially a gamma-aminobutyric acid (GABA) receptor antagonist and is a widely accepted chemoconvulsant employed for evaluating anti-convulsant drugs. It works by neutralizing its activity while also resulting in oxidative damage due to the overproduction of free radicals and superoxides (Attia et al., 2019). Epileptic seizures reduce the antioxidant defense capacity in the brain and promote the production of reactive oxygen species (ROS), giving rise to oxidative stress (Aguiar et al., 2013; Cardenas-Rodriguez et al., 2013). These increased free radicals, in turn, modify the expression of the inflammatory molecule, nuclear factor-kappa B (NF-κB) (Abdelsalam et al., 2015). It contributes to neuronal hyperexcitability that results in increased seizures. Additionally, another inflammatory cytokine, tumor necrosis factor-α (TNF-α), also possesses neuromodulatory properties and contributes to prolonged seizures (Iori et al., 2016). The levels of interleukin-1β (IL-1β), IL-6, and TNF-α were found to be abnormally high in epilepsy patients, which could contribute greatly to neurodegeneration (Ravizza et al., 2006). Thus, therapeutic agents with potential antioxidant and anti-inflammatory activities could be essential prerequisites for developing drugs for the effective management and treatment of epilepsy.
Currently, available anti-epileptic medications, which are essentially used to manage epileptic conditions, only alleviate seizure symptoms but do not eliminate seizure-induced neuronal damage. Furthermore, the usage of these synthetically produced drugs is associated with serious consequences such as depression, anxiety, ischemia, cognitive disability, and physical impairments (Bagheri et al., 2019; Ramalingam et al., 2013). Natural compounds derived from medicinal herbs have significantly aided in the exploration of alternative therapies and can very well serve as a potential replacement for anti-epileptic drugs with active components and enhanced safety and effectiveness (Zhu et al., 2014). Phytomedicines are traditionally employed to treat different forms of epilepsies and their effectiveness in treating the disease lies in their high antioxidant capacities (Okoye et al., 2010).
Erigeron breviscapus (Vant.) is a prominent Chinese medicinal plant with well-documented antioxidant and neuroprotective properties. Its therapeutic effect in neurological disorders can be linked to the suppression of γ-aminobutyric acid transaminase (GABA-T) (Gu et al., 2017). Scutellarein (4′,5,6,7-tetrahydroxyflavone or 6-hydroxy apigenin) is a potent phytophenol isolated from the plant that is known to possess anti-inflammatory, anti-cancer, neuroprotective, and antioxidative properties (Lin et al., 2019; Tang et al., 2014). The phenolic compound is employed successfully for the treatment of inflammation, diabetes mellitus, neurological disorders, asthma, digestive complications, and urinary infections (Sung et al., 2015). Because of its phenolic composition and flavonoid-derived backbone, scutellarein may have a modulating role in the treatment of neurological diseases, as flavonoids can impair cellular oxidative reactions in the CNS (Sung et al., 2015). The primary objective of the current investigation is to assess the anti-convulsant effects of Scutellarein in a PTZ kindling epileptic model in mice by reduction of neuroinflammation and free radical damage. The duration and initiation of tonic and clonic convulsions, locomotor activity, levels of GABA, dopamine, and glutamate, and activities of Na+ K+ ATPase and Ca2+ ATPase, antioxidant markers, and NF-κB, TNF-α, and IL-1β levels were measured in the kindled mice along with histopathological analysis of brain tissues.
Materials and Methods
Chemicals
Scutellarein (≥98%), PTZ, sodium chloride (≥99.0%), and various other reagents were procured from Sigma Aldrich, USA. The bioassay kits required for ELISA analysis of respective biomarkers were acquired from Thermo Fisher Scientific, USA, and Biocompare, USA, respectively.
Experimental Animals
Male Swiss mice (30 g and 60 days old) were procured from the central animal establishment, housed in polyacrylic enclosures, and maintained at 22 ± 2°C with 55%–65% humidity and 12-h/12-h light and dark alternating cycle. The mice were fed a regular diet along with purified drinking water. All the experiments were carried out after receiving an ethical clearance certificate from the animal ethical committee.
Experimental Setup
The rodents were categorized into five groups with six mice in each. The first control group was given normal saline (0.9% Sodium chloride). The second group received only PTZ via an IP route (in 0.9% Sodium chloride) to induce seizures (PTZ group without treatment). The third and fourth groups received two oral doses of scutellarein (10 and 20 mg/kg) before 30 min of PTZ induction. Diazepam (5 mg/kg) was administered intraperitoneally to the fifth group, 30 min before inducing with PTZ. After the treatment and PTZ induction, the mice were attentively supervised for a duration of 30 min to observe any signs of convulsion initiation, clonic and tonic convulsion period, total rodents exhibiting convulsions, and the mortality count. The results were recorded and noted.
Stimulation of Kindling
PTZ (35 mg/kg) was supplemented intraperitoneally to the rodents on a regular basis for a total of 15 times. After every PTZ supplementation, the animals were attentively supervised for a period of 30 min. The timing for the seizure to appear following the PTZ challenge (latency) along with the period of clonic and tonic convulsions was noted. The following seizure stages were studied using the scale proposed by Schroder et al. (1993). Stage 0 implies nil response, stage 1 signifies jerks in the face and mouth, stage 2 denotes axial convulsive signals through the body, stage 3 implies rearing and myoclonic jerks, stage 4 implies clonic seizures with animals collapsing on their side, and stage 5 implies a once appearing tonic and clonic convulsions. The intensity of the convulsions was recorded during PTZ induction. After the seizures subsided, the mobility of the mice was measured using an actophotometer. Shortly after, each mouse was placed on the actophotometer separately, and the overall activity was recorded for 5 min and represented as counts/5 min. Afterward, a forced swimming test was carried out in order to evaluate their depressive conduct (Patil et al., 2012). The rodents were placed in a cylinder (25 cm × 12 cm × 25 cm) for 5 min with water level up to 15 cm, and the total duration of immobility was recorded.
Brain Tissue Sample Preparation
Following the behavioral tests, mice were subjected to anesthesia with an intraperitoneal injection of sodium pentobarbital (50 mg/kg b.wt) and euthanized by cephalectomy. The brain tissue of each rodent was removed and treated with cold saline before being homogenized with a phosphate buffer (10% w/v, 0.1 M, pH 7.4). The homogenization process was performed in a 1.5-mL safe-lock polypropylene tube for 5 min at 4°C. Centrifugation of the resultant solution was carried out (3,000 rpm, 15 min), and the uppermost solution was used for further testing. Histological investigations were performed on a section of brain tissues.
Assessment of the Levels of GABA, Glutamate and Dopamine and Activities of Na+ K+ ATPase and Ca+ ATPase in the Brain Tissue Samples
The GABA (Cat No. LS-F4121), glutamate (Cat No. MBS756400), and dopamine (Cat No. MBS733582) levels in the brain tissue samples of control and treated animals were measured using assay kits. The activities of Ca+ ATPase and Na+ K+ ATPase in the brain tissue samples were measured using testing kits as per the manufacturer’s guidelines (MyBioSource, USA).
Quantification of Antioxidant Markers and Oxidative Stress Markers in the Brain Tissues
The presence of malondialdehyde (MDA) in the tissue samples of the experimental animals was determined using the Niehius and Samuelson (1968) method. Absorbance at 530 nm was measured. The reduced glutathione (GSH) level was determined using the Jollow et al. (1974) method. The absorbance at 412 nm was measured, and the results were reported as GSH/mg/mg of protein. The technique described by Sun et al. (1988) was employed to measure superoxide dismutase (SOD) activity, and the results were reported as SOD/U/mg of protein. The nitric oxide (NO) level was investigated using the Green et al. (1982) approach, and the results were recorded in mg/mL.
Quantification of Inflammatory Biomarkers Level
The levels of inflammatory biomarkers such as NF-κB (Cat No. E-EL-R0674), TNF-α (Cat No. E-EL-M3063), and IL-1β (Cat No. E-EL-M0037) in the control and treated animals were quantified using respective assay kits according to the protocols described by the manufacturer (Elabscience Biotechnology Inc., USA).
Histopathological Analysis of Brain Tissue Samples
The brain hippocampus tissues were fixated in formalin (10%) for 1 day before being treated with xylene and subsequently with isopropyl alcohol for 12 h. Integration of tissues in paraffin wax was carried out and sliced to achieve a size of 5 mm. Hematoxylin and eosin (H&E) were employed to stain the cut sections. Finally, the examination of tissues under a light microscope followed by obtaining microphotographs at a 40× magnification.
Statistical Analysis
The obtained results are presented as the mean ± SD of triplicates. The data were analyzed by the GraphPad Prism software (GraphPad Software, Inc., version 6.02, USA), using one-way ANOVA followed by Tukey’s post hoc test. The significance level was fixed at p < 0.05.
Results
Impact of Scutellarein on the Duration and Initiation of Clonic and Tonic Convulsion and Mortality Counts in the PTZ-induced Rodents
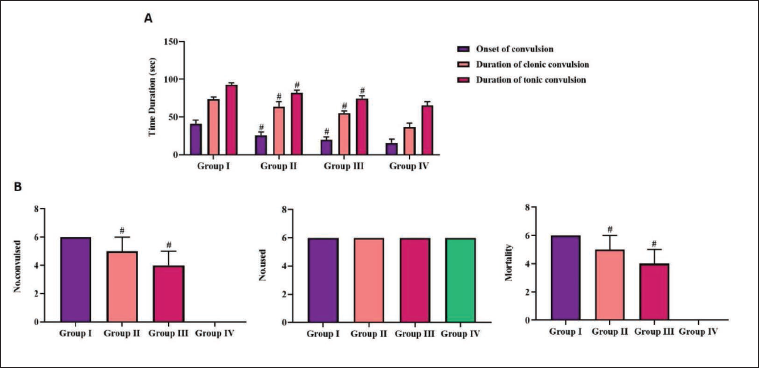
PTZ-induced mice had a faster initiation of seizures and longer time span of clonic and tonic convulsions than scutellarein or diazepam-treated mice. PTZ also exhibited the highest mortality rate in mice. Conversely, both the scutellarein doses revealed appreciable (p < 0.05) mitigative effects in a dose-related manner. In PTZ-administered rodents, scutellarein reduced the start of convulsions and the duration of clonic and tonic convulsions. Scutellarein administration also significantly (p < 0.05) decreased the mortality rate in PTZ-induced rodents (Figure 1).
Effect of Scutellarein on PTZ-induced Locomotor Activity, Immobility, and Kindling Period in Kindled Mice
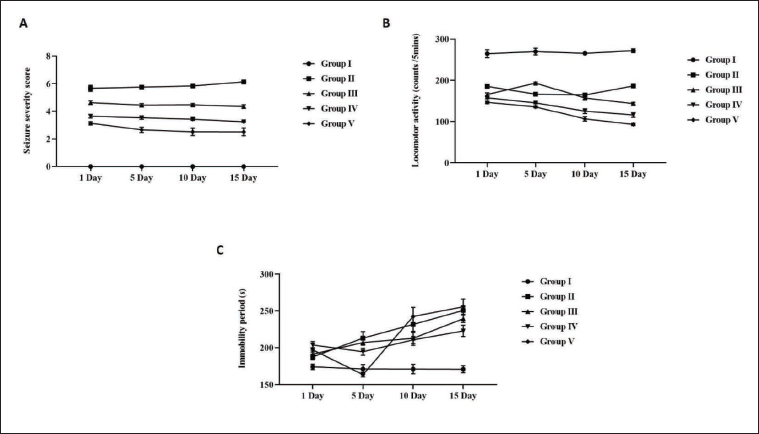
PTZ-administered animals had more severe seizures in comparison with the control mice. Interestingly, the administration of scutellarein to PTZ-triggered mice led to a marked decrease in seizure severity on the 10th and 15th day, as opposed to the PTZ-alone exposed rodents. The PTZ administration also resulted in a significant (p < 0.05) decrease in locomotor activity and an increase in the immobility duration of the animals (Figure 2). In this study, both the scutellarein doses revealed no major variations from the PTZ challenge. Scutellarein had no effect on locomotor activity or immobility time in PTZ mice. Diazepam administration also reduced the degree of seizures in PTZ-challenged rodents. Diazepam treatment, like scutellarein, exhibited no differences in the mobility and immobility time of the PTZ-induced rodents.
Impact of Scutellarein on GABA, Dopamine, and Glutamate Levels, and Ca2+ ATPase and Na+ K+ ATPase Activities in the PTZ-induced Mice
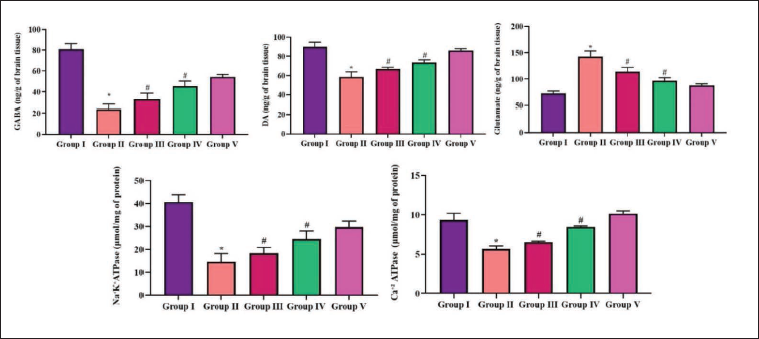
GABA and dopamine levels were discovered to be repressed, whereas glutamate levels were shown to be rapidly raised in the brain tissue samples of PTZ-induced animals. The PTZ stimulus also significantly (p < 0.05) reduced the Ca2+ ATPase and Na+ K+ ATPase activities. The administration of scutellarein to PTZ-challenged rodents remarkably (p < 0.05) enhanced GABA and dopamine levels while decreasing glutamate levels (Figure 3). Scutellarein administration also significantly increased the activities of Ca2+ ATPase and Na+ K+ ATPase in PTZ-triggered mice.
Impact of Scutellarein on the Antioxidant Markers and Oxidative Stress Indicators in the Brain Tissue Samples of PTZ-administered Mice
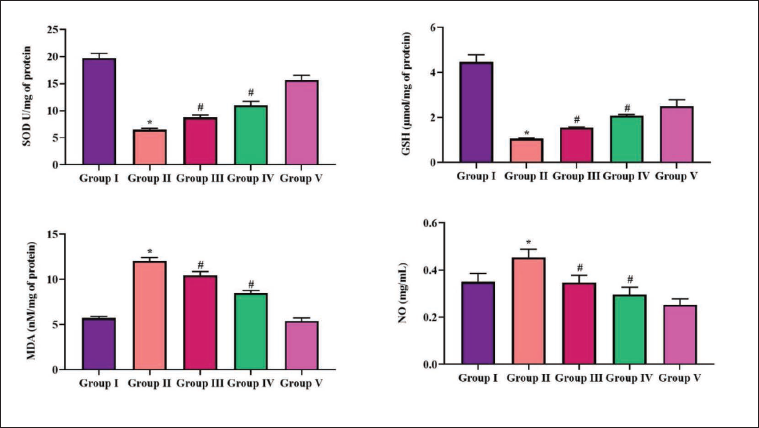
The impact of scutellarein on the antioxidant markers and oxidative stress markers in the brain tissue samples of PTZ-induced mice was assessed, and the results are depicted in Figure 4. PTZ rodents exhibited a remarkable decrease in the antioxidants, SOD and GSH, while increasing the MDA and NO levels. Contrastingly, the levels of NO and MDA were significantly (p < 0.05) reduced in PTZ-triggered mice that received 10 and 20 mg/kg of scutellarein. The phytocompound also increased the antioxidant status of GSH and SOD. Diazepam administration significantly (p < 0.05) reduced NO and MDA levels while increasing GSH and SOD levels.
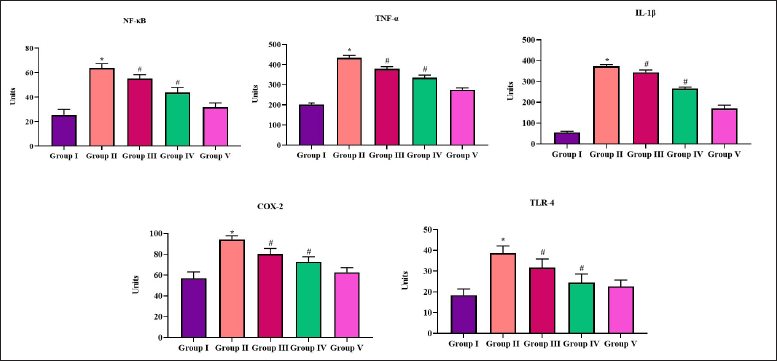
Impact of Scutellarein on the Inflammatory Marker Levels in the Brain Tissue Samples of PTZ-induced Rodents
The effects of scutellarein on the contents of inflammatory mediators were examined with the help of marker-specific assay kits. The levels of NF-κB, TNF-α, and IL-1β were found to be up-regulated in the PTZ-triggered mice, as shown in Figure 5. The scutellarein administration, on the contrary, substantially (p < 0.05) suppressed the contents of NF-κB, TNF-α, and IL-1β in the PTZ mice. Diazepam supplementation decreased the levels of all the inflammatory markers in the PTZ rodents.
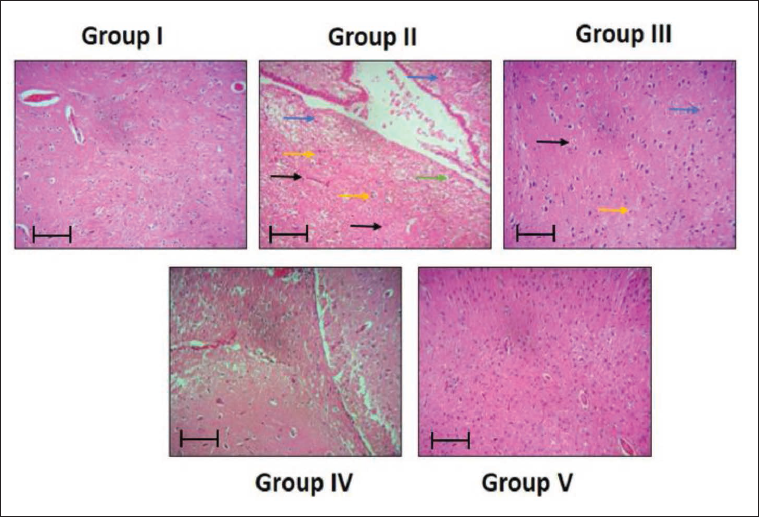
Effect of Scutellarein on the Histopathological Modifications in the Brain of PTZ-triggered Mice
The PTZ-challenged mice exhibit considerable histological modifications in brain tissues, such as exaggerated inflammatory cellular penetration rates, pyknosis, and neuronal death. The brain tissues of the control mice revealed normal morphology with no evidence of inflammation. The scutellarein treatment significantly reduced the PTZ-induced histological changes. PTZ-challenged mice treated with scutellarein had fewer inflammatory symptoms, pyknosis, and necrosis in their brain tissues. Diazepam administration also reduced the histological alterations in brain tissues caused by PTZ (Figure 6).
Discussion
Oxidative stress is the most notable mechanism in the onset and advancement of epilepsy (Ramalingam et al., 2013). Previous research has linked PTZ-provoked kindling to elevated glutamate activity and the resultant formation of free radicals in neurons. In the kindling epileptic model, oxidative stress is a major contributor to brain tissue damage following convulsions. In earlier studies, PTZ-triggered kindling has been reported to be associated with higher lipid peroxidation and a decline in antioxidant enzymes (Hussein et al., 2016). GSH is an endogenous antioxidant whose reduced form combines with free radicals and protects the body from oxidative toxic damage (Samudrala et al., 2015). Damage to polyunsaturated sites in neuronal membranes results in excessive lipid peroxidation, which is reflected through increased MDA levels (Kiasalari et al., 2013). Significant amounts of NO generated during seizures suppress mitochondrial oxidative phosphorylation, resulting in mitochondrial resistance failure and, as a direct consequence, neuronal apoptosis (Tawfik et al., 2018). In this study, the oxidative stress indicators NO and MDA levels were significantly reduced, whereas the antioxidant status of SOD and GSH was enhanced in PTZ-triggered mice that received scutellarein. Previously, the stronger antioxidant and neuroprotective effect of scutellarein against oxidative stress has been studied which aligns with the observed results in this study (Shi et al., 2015). Inflammatory processes in the brain are key contributors to seizure initiation and progression (Maroso et al., 2010). The inflammatory mediator molecules are important participants in the inflammatory and immunological systems. Such regulators contribute to epileptic pathology and involve IL-1β, IL-6, and TNF-α (Zhang et al., 2017). Furthermore, a prior study using a kindling model of epilepsy established a link between TNF-α and epileptogenesis, with TNF-α treated rats experiencing longer seizures (Firdous et al., 2021). The role of NF-kB in seizure advancement is well recognized, and decreasing the level of NF-κB significantly suppresses seizure incidences (Zhang et al., 2017). Owing to the involvement of inflammatory mediators such as NF-κB, TNF-α, and IL-1β in the epileptic pathogenesis, their contents in PTZ-induced rodents upon scutellarein treatment have been evaluated. The scutellarein administration substantially suppressed the levels of the inflammatory markers, indicating their anti-convulsant property.
Epilepsy is thought to occur because of an imbalance that occurs between the stimulatory and inhibitory components of CNS, which is generated by a decline in GABAergic and/or glutamatergic transmission (Mante et al., 2017). In the human brain, GABA is the most significant inhibitory neurotransmitter. Anti-epileptic agents target GABA receptors, which are broadly diffused in the human brain (Grosso et al., 2013). Numerous phytoconstituents have been demonstrated to exhibit an anti-convulsant effect in rodent seizure models, which has been ascribed to the effect of flavonoids, terpenoids, and furanocoumarins on GABA receptors and gated ion channels (Schachter et al., 2015). In this study, the phytocompound scutellarein was able to augment the GABA levels in the brain tissue samples of the PTZ-induced rodents.
PTZ-triggered kindling is linked to increased activity of a smaller group of glutamatergic mechanisms that use N-methyl-
Oxidative stress has also been associated with changes in the biological functions of calcium, which result in mitochondrial inefficiency and, consequently, neuronal damage (Martinc et al., 2012). Other studies have found that, in epilepsy models, there is a significant suppression in the activities of ion channels such as Na+/K+-ATPase, as well as abnormalities in ion channels and glutamatergic synapses (Kobylarek et al., 2019). Scutellarein administration was able to significantly increase the activities of Ca2+ ATPase and Na+ K+ ATPase in PTZ-triggered mice. Based on the findings, it is reasonable to speculate that the plant-derived extract provided protection against PTZ-induced seizures via stimulating GABA neurotransmission in the CNS to prevent glutamatergic neurotransmission mediated by the NMDA receptor. The results offer scientific support to the conventional usage of scutellarein as an anti-epileptic.
Conclusion
The findings from this study revealed that scutellarein reduced PTZ-induced seizures in mice. In PTZ animals, scutellarein lowered seizure severity by suppressing the onset and duration of convulsions. Scutellarein successfully modulated the PTZ-triggered changes in GABA, glutamate, and dopamine levels, as well as Ca2+ ATPase and Na+ K+ ATPase activities. Furthermore, it remarkably reduced the oxidative stress indicators and decreased the contents of NF-κB, TNF-α, and IL-1β in PTZ animal brain tissues, confirming its antioxidant and anti-inflammatory properties. These findings demonstrated that scutellarein successfully alleviated PTZ-induced convulsive seizures in mice, though our present studies failed to determine the concentration of scutellarein in the brain tissues of the animals to assess the blood–brain barrier crossing and the toxicity of scutellarein to other organs of the body. Furthermore, more research is needed in the years to come to uncover the mitigative effect of scutellarein against epileptic convulsion and its molecular processes.
Footnotes
Summary
Epilepsy is a highly common and chronic condition of the nervous system that involves involuntary repetitive seizures, affecting 70 million people, regardless of all ages globally.
In PTZ animals, scutellarein lowered seizure severity by suppressing the onset and duration of convulsions.
Scutellarein successfully modulated the PTZ-triggered changes in GABA, glutamate, and dopamine levels, as well as Ca2+ ATPase and Na+ K+ ATPase activities.
Abbreviations
PTZ: Pentylenetetrazol; MES: Maximal electroshock seizure; GABA: Gamma-aminobutyric acid; ROS: Reactive oxygen species; MDA: Malondialdehyde; SOD: Superoxide dismutase; GSH: Reduced glutathione; NO: Nitric oxide.
Author Contributions
JD and YL: Hypothesis, experiment design, and data validation. JW and DW: Experiments, data curation, and review. QZ and YL: Manuscript writing, editing, and review.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Department of Pediatrics, The Bishan Hospital of Chongqing, Chongqing, China.
