Abstract
Introduction
Colorectal cancer is the fifth most common cancer and the fifth leading cause of cancer-related death worldwide. 1 In Taiwan, colorectal cancer has been the most common cancer for numerous years, causing nearly 7000 deaths every year. Although clinical chemotherapy effectiveness has been significantly improved and progressed, the cytotoxicity of existing colorectal cancer chemotherapy drugs has not been resolved, making the cure rate of colorectal cancer still low and the mortality rate still high. 2 This also demonstrates that the development of new, more effective drugs with lower cytotoxicity is urgent.
Punica granatum (pomegranate) belongs to the family Punicaceae, and it is commonly known as pomegranate. Pomegranate is widely used as a traditional medicine, and the leaves of pomegranate have been found to have antioxidant, anti-inflammatory, and anti-cancer effects. 3 Two classes of polyphenol compounds, anthocyanins and hydrolyzable tannins, were found in pomegranate. Furthermore, 90% of the antioxidant activity in pomegranate is caused by these polyphenols, while punicalagin alone accounts for more than 50% of the antioxidant activity. Punicalagin is considered to be the major hydrolyzable tannin compound in pomegranate leaves and bark. 4 Punicalagin has been reported to have several beneficial effects, including antioxidant, anti-inflammatory and anticancer properties. 5 Prior studies have also found that punicalagin has therapeutic effects on cancers, such as leukemia and osteosarcoma.6,7 To the best of our knowledge, the cytotoxicity of punicalagin on colon cancer cells and the underlying mechanism have yet to be addressed.
Apoptosis is an actively regulated cell death mode that includes two activation mechanisms, the intrinsic and extrinsic pathways. 8 Early in embryonic development, apoptosis is activated to remove unwanted cells. The intrinsic pathway causes cell death due to cellular stress and the extrinsic pathway is activated in response to extracellular signals. Both of the pathways activate caspases, which induce cell death by indiscriminately breaking down proteins. Apoptosis also plays a role in preventing cancer. When apoptosis is inhibited, it leads to uncontrolled cell division and eventually cancer.
The epithelial-mesenchymal transition (EMT) is a process in which epithelial cells lose their polarity and tight junctions, and they acquire the ability to migrate and invade. EMT is important in cell-cell adhesion, cell-extracellular matrix adhesion, and wound healing.9,10 In the EMT, epithelial cells alter gene expression profiles, resulting in increased motility and the acquisition of invasive capabilities. Neoplastic epithelial cells promote cell migration and invasion by causing EMT dysregulation. 11
Here, we used three colon cancer cell lines, namely, HCT 116, HT-29 and WiDr, to explore the anticancer effects of punicalagin. To our knowledge, this is the first study to report that punicalagin is cytotoxic to colon cancer cells but not to normal epithelial cells. Punicalagin induced apoptosis by activating caspase-3. Additionally, punicalagin prevented the invasion of colon cancer cells in vitro. Inhibition of MMP-2, MMP-9, Snail, and Slug protein expression also characterized the anti-invasive effects of punicalagin.
Materials and methods
Punicalagin
Punicalagin (2,3-(S)-hexahydroxydiphenoyl-4,6-(S,S)-gallagyl-D-glucose, CAS registry number 65995-63-3, purity ≥98%) was purchased from Sigma-Aldrich (St. Louis, MO, USA), dissolved in dimethylsulfoxide (DMSO; Sigma-Aldrich) to a concentration of 50 mM and stored at -20°C until use. One percent of DMSO was used as solvent control in all experiments.
Cell culture and treatments
Human normal colon epithelial cells (CCD 841 CoN) and colon cancer cell line (HT-29) (both from the American Type Culture Collection, Manassas, VA, USA) were cultured in DMEM and McCoy’s 5A, respectively. Colon cancer cell lines, HCT 116, and LoVo (from the Bioresource Collection and Research Center; Hsinchu, Taiwan), were cultured in McCoy’s 5A and F-12K, respectively. All cell lines were authenticated by the cell providers. We also checked the ICLAC database to ensure that all cell lines were not misidentified. The mycoplasma test has been performed for all cell lines by the cell providers. All media contained 10% fetal bovine serum (VWR, Radnor, PA, USA),100 units/mL penicillin G, 100 μg/mL streptomycin sulfate, and 250 ng/mL amphotericin B (all from Thermo Fisher Scientific; Waltham, MA, USA). Various concentrations of punicalagin (0–500 μM) and durations of treatment (24, 48 and 72 h) were applied in this study.
Cell viability assay
Cell viability was assessed using a CCK-8 assay kit (Biotools; New Taipei, Taiwan) based on the manufacturer’s instructions. The assay was performed three times in triplicates.
Apoptosis assay
Apoptosis was examined using the Muse Annexin V and Dead Cell Kit (Merck Millipore; Darmstadt, Germany) following the manufacturer’s instructions. The assay was performed in triplicate.
Caspase-3 activity assay
Caspase-3 activity was determined using a caspase-3 assay kit (Sigma-Aldrich). To inhibit caspase-3 activity, the caspase-3 inhibitor N-acetyl-Asp-Glu-Val-Asp-CHO (Ac-DEVD-CHO; Biomol Research Laboratories, Plymouth, PA, USA) was co-treated with punicalagin at a final concentration of 200 μM in a parallel experiment. The assay was conducted three times, independently.
Cell invasion assay
Cell invasion was analyzed using the Cell Invasion Assay Kit (Merck Millipore). The methods are described in detail in our prior paper. 12
Western blotting
Denatured protein samples were subjected to 15% SDS-PAGE and proteins were transferred to nitrocellulose membranes. Blocked blots were incubated overnight at 4°C with the primary antibodies: active caspase-3 (cat. no. AB3623, Merck Millipore), MMP-2 (cat. no. NB200-114, Novus Biologicals), MMP-9 (cat. no. NBP2-13173, Novus Biologicals), Snail (cat. no. NBP2-27184, Novus Biologicals), and Slug (cat. no. ARG54888, Arigo Biolaboratories). After washing, membranes were incubated with peroxidase-conjugated secondary antibodies (cat. no. A4416/A6154, Sigma-Aldrich) for 1 h. Anti-β-actin (cat. no. A5441, Sigma-Aldrich) was used as a control for equal protein loading. Protein signals were detected using enhanced chemiluminescence reagents (Thermo Fisher Scientific). Band intensities were analyzed using GeneTools software (Syngene, Cambridge, UK).
Statistical analysis
Unpaired Student’s t test was used to identify significantly different means (p < .05).
Results
Punicalagin induced the death of colon cancer cells in vitro
First, a CCK-8 assay was used to detect the cytotoxicity of punicalagin in HCT 116, HT-29, and LoVo colon cancer cells. Treatment of HCT 116 cells with different concentrations of punicalagin (100, 200, 300, 400, and 500 μM) for 24, 48 and 72 h resulted in a dose- and time-dependent decrease in cell viability relative to control cells (Figure 1(b)). Treatment with punicalagin did not cause a significant decrease in the survival rate of CCD 841 CoN cells. (Figure 1(a)). The IC50 value of punicalagin treatment for 48 h was between 100 and 200 μM. Similar results were obtained by treating HT-29 and LoVo cells with punicalagin for 24, 48 or 72 h (Figures 1(c) and 1(d)). Because it can cause obvious cell death, 200 μM punicalagin was selected as the concentration for subsequent experiments. Effect of punicalagin on the viability of human colon epithelial cells and colon cancer cell lines. The cells were incubated with different concentrations of punicalagin for 24, 48, and 72 h. Absorbance measured using a CCK-8 assay relative to the untreated subgroup in (a) colon epithelial cells, (b) HCT 116, (c) HT-29, and (d) LoVo cells. Data are the results of three independent experiments and presented as means ± S.D. (*p < .05, **p < .01).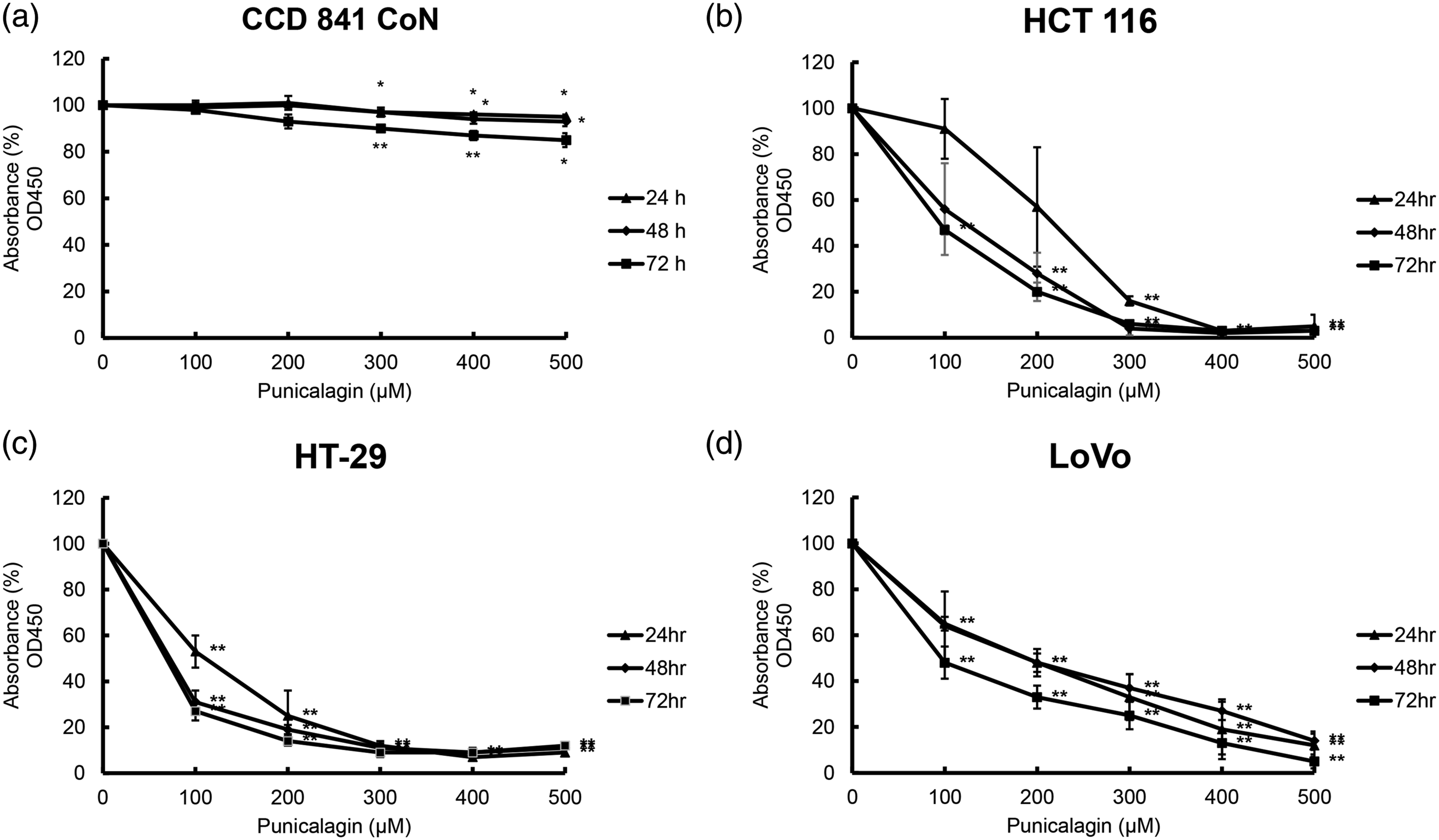
Apoptosis induced by punicalagin in HCT 116, HT-29, and LoVo cells
Early in apoptosis, cell membrane asymmetry disappears, and cell membrane-embedded phosphatidylserine residues become externalized. To further understand whether the cell cytotoxicity caused by punicalagin treatment is caused by apoptosis, the percentage of early and late apoptotic HCT 116, HT-29, and LoVo cells was detected by annexin V/PI double staining. Exposure to 200 μM punicalagin for 48 h increased the percentage of apoptotic cells in the three colon cancer cell lines (Figure 2). In contrast, 200 μM punicalagin did not induce apoptosis of CCD 841 CoN cells. These data demonstrated that punicalagin induced the death of HCT 116, HT-29, and LoVo cells through the activation of apoptosis. Effect of punicalagin on the apoptosis of human normal colon epithelial cells and colon cancer cell lines. Cells were treated with 100 μM punicalagin for 48 h, then analyzed using annexin V/PI double staining. Punicalagin treatment induced consistent changes seen as increased percentage of apoptotic cells in (a) CCD 841 CoN, (b) HCT 116, (c) HT-29, and (d) LoVo cells. The representative diagrams of flow cytometry analyses were displayed. Data are presented as the mean ± S.D. of three independent experiments (**p < .01).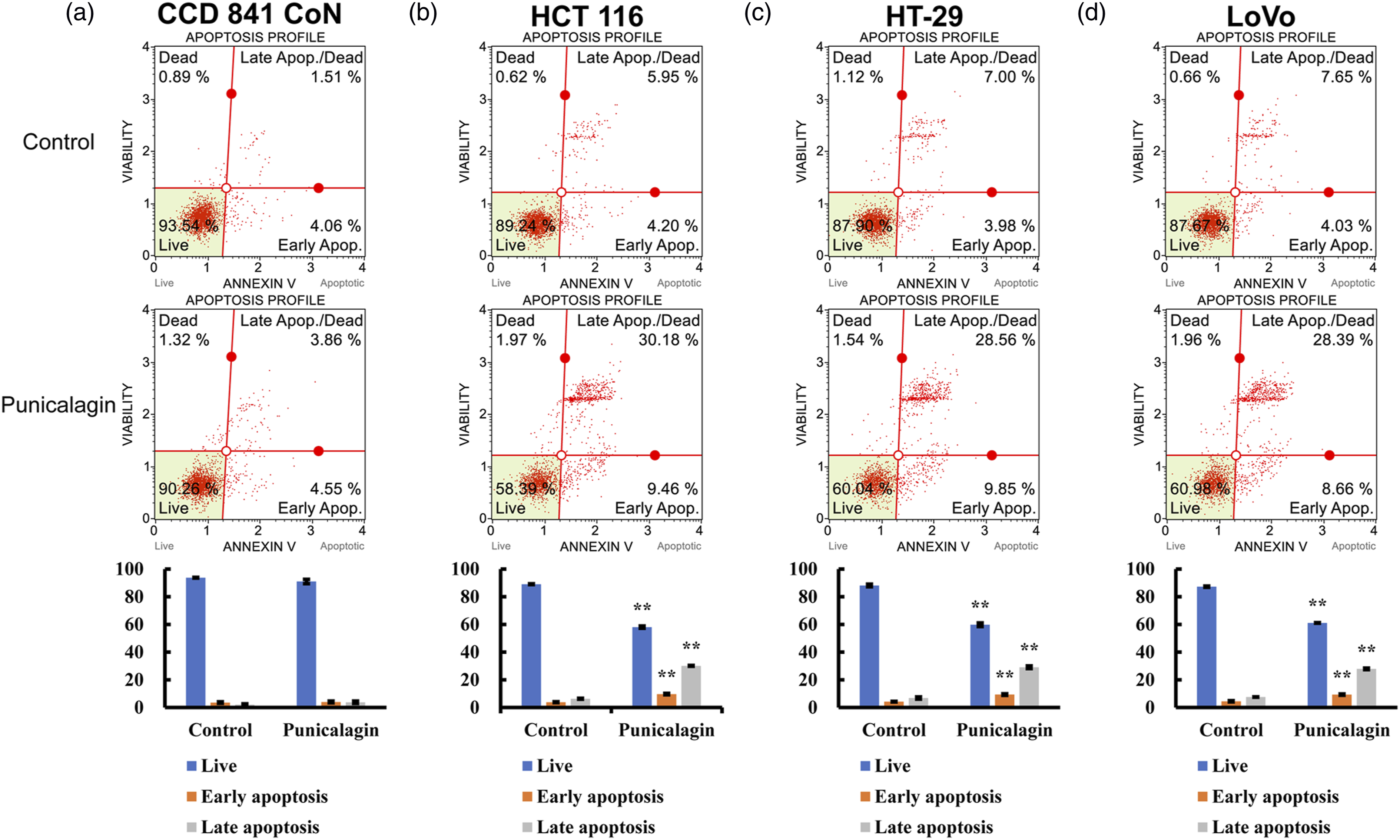
Punicalagin activated caspase-3 in HCT 116, HT-29, and LoVo cells
Next, the role of caspase-3 in punicalagin-induced apoptosis in HCT 116, HT-29, and LoVo cells was explored. The results showed that exposure to punicalagin for 24 h significantly enhanced caspase-3 activity in all three of the cell lines (Figure 3(a)). The effect of caspase-3 inhibitor Ac-DEVD-CHO was examined to investigate whether caspase-3 activation was necessary for punicalagin-induced apoptosis. When the cells were co-treated with Ac-DEVD-CHO, punicalagin-induced caspase-3 activation was suppressed in AGS cells (Figure 3(a)). Western blotting using an anti-active caspase-3 antibody was used to verify enzyme activity. Following incubation with punicalagin for 24 h, the amount of active caspase-3 increased, suggesting that punicalagin induced apoptosis via the activation of caspase-3 (Figure 3(b)). In contrast, 200 μM punicalagin did not activate caspase-3 in CCD 841 CoN cells. These results indicated that the activation of caspase-3 is critical for punicalagin to induce apoptosis in these three cancer cell lines. Punicalagin activates caspase-3 in human normal colon epithelial cells and colon cancer cell lines. (a) Cells were incubated with or without 100 μM punicalagin for 48 h. To inhibit caspase-3 activity, a caspase-3 inhibitor Ac-DEVD-CHO, co-treated with punicalagin, was added to the cells at a final concentration of 200 μM. Each bar is expressed as a value of caspase-3 activity by measuring the release of p-nitroaniline (OD405). Data are the results of three independent experiments and presented as means ± S.D. (*p < .05, **p < .01). (b) Total cell lysates were collected and subjected to western blotting analysis using anti-active caspase-3 and β-actin.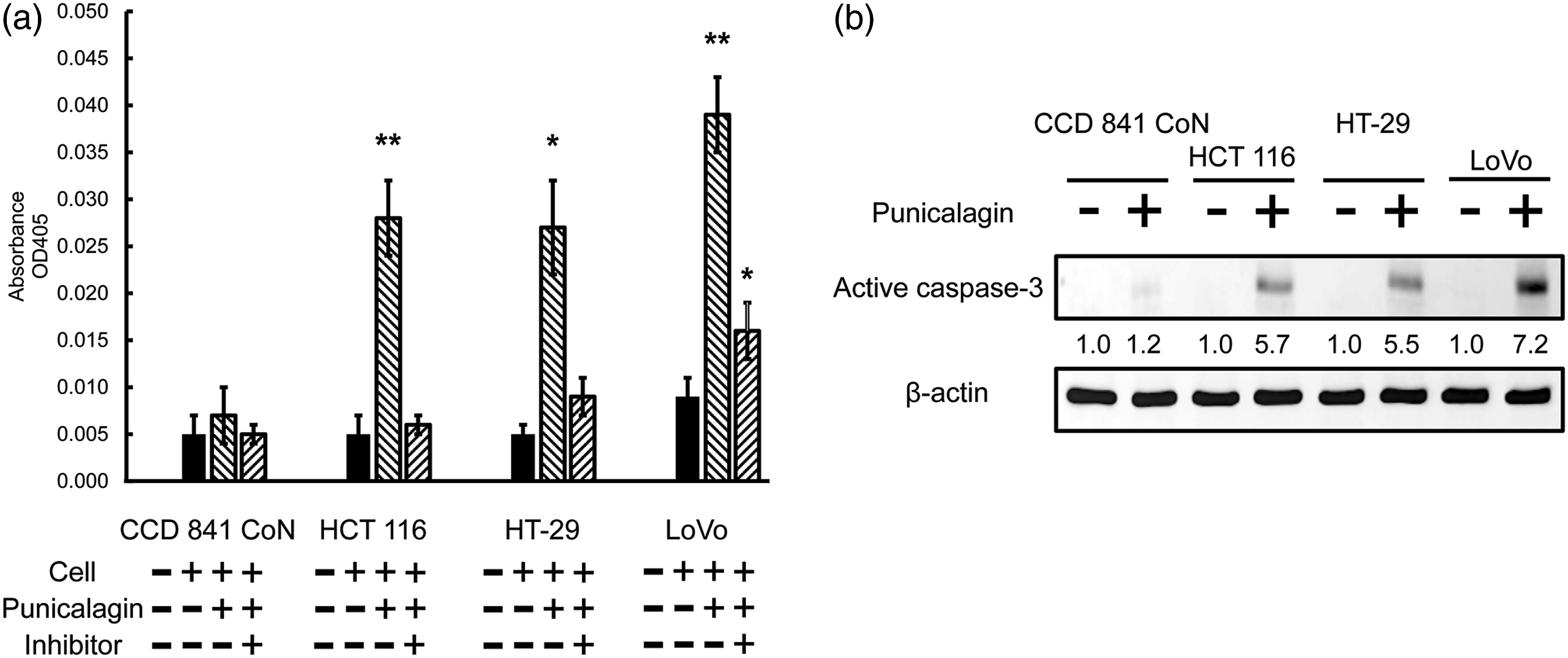
Punicalagin inhibited invasion of colon cancer cells
A cell invasion assay was used to study the effect of punicalagin on HCT 116, HT-29, and LoVo cell invasion. The results showed that after 48 h of treatment with punicalagin, compared with the control group, there were fewer cells invading. This result indicated that cell invasion was suppressed after treatment with punicalagin (Figure 4(a)). MMP-2 and MMP-9 play important roles in promoting the metastasis of cells. Western blotting using anti-MMP-2 and MMP-9 antibodies was performed to study the effect of punicalagin on these cell invasion proteins. Here, reduced expression of MMP-2 and MMP-9 implied that exposure to 100 μM punicalagin for 48 h inhibited the invasion of colon cancer cells (Figure 4(b)). Next, whether there was a correlation between punicalagin and the EMT in colon cancer cells was examined. Western blotting using anti-Snail and anti-Slug antibodies was performed to study the effect of punicalagin on these EMT-related proteins. The decreased expression of Snail and Slug suggested that a 48 h exposure to punicalagin suppressed the EMT in colon cancer cells (Figure 4(b)). These results showed that punicalagin inhibits the invasion of colon cancer cells by reducing the expression of proteins involved in cell invasion. Effect of punicalagin on the invasion of human colon cancer cell lines. (a) Cells were treated with or without 100 μM punicalagin for 48 h, then analyzed using a Cell Invasion Assay kit. A typical result from three independent experiments is shown. (b) Total cell lysates were collected and subjected to western blotting analysis using anti-MMP-2, MMP-9, Snail, Slug, and β-actin.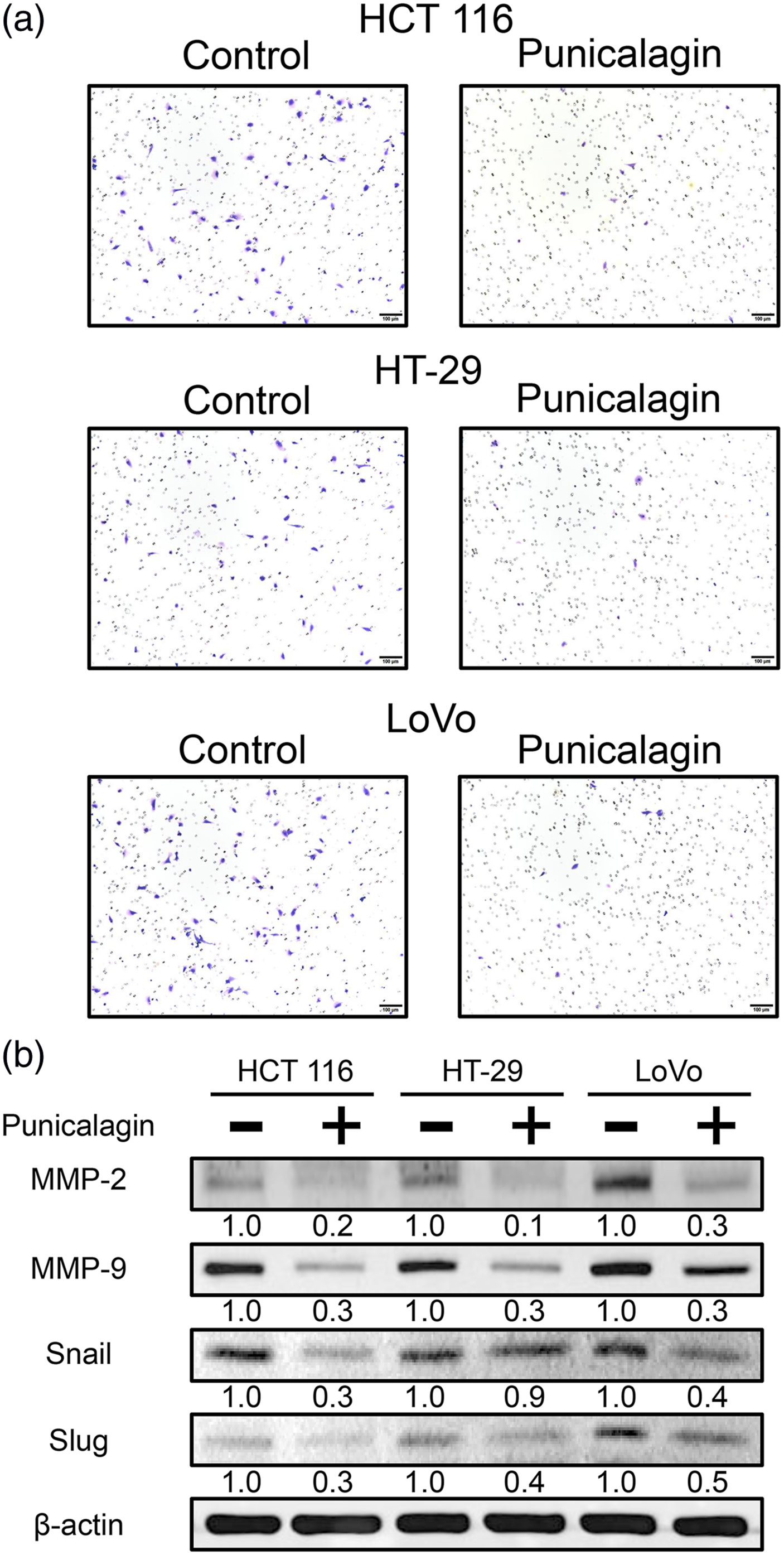
Discussion
Colorectal cancer, a cancer with a high incidence and high mortality, is the leading cause of cancer-related deaths worldwide (according to WHO statistics on cancer, more than one million people worldwide suffer from colorectal cancer every year, twice as many men as women, and is considered an important global health issue). Colorectal cancer remains an important health burden attributable to several factors, including the lack of understanding of the process of colorectal carcinogenesis, and the lack of appropriate prognostic biomarkers. Chemotherapy for colorectal cancer has been limited due to its high toxicity and low specificity. As a result, new, less toxic drugs for colorectal cancer are needed more today than in the past. The tannin punicalagin has been reported to have therapeutic effects in several different cancers. A study using leukemia cells indicated that punicalagin showed cytotoxic activity against NB4 and MOLT-4 leukemia cells but not against normal cells. 6 Berköz et al. pointed out that punicalagin acted as an anti-proliferative substance in A549 lung cancer cells, but not in normal lung cells. 13 The cytotoxicity of punicalagin against colorectal cancer cells is still unknown. In this study, we found that punicalagin had anticancer activity against HCT 116, HT-29, and LoVo colorectal cancer cells, but not normal colorectal epithelial cells. These results reveal that punicalagin may be a potential anticancer agent in the treatment of colorectal cancer.
One of the strategies to treat cancer is to induce apoptosis of cancer cells. 14 A study using Caco-2 colorectal cancer cells showed that punicalagin induced the intrinsic pathway of apoptosis by activating caspase-3 and caspase-9 and suppressing Bcl-XL. Another study using PC-3 prostate cancer cells described an extrinsic pathway in which punialagin triggered apoptosis by activating caspase-3 and caspase-8.15,16 Punicalagin also induced apoptosis in ME-180 and HeLa cells by suppressing NF-κB and β-catenin signaling pathways respectively.17,18 Previous studies also mentioned that punicalagin can induce autophagy. Cheng et al. reported that punicalagin induced autophagy in BCPAP papillary thyroid carcinoma cells through the expression of beclin-1, the degradation of p62, and the activation of the LCII-3 conversion. 19 It was also found that punicalagin treatment induced apoptosis and autophagy in U87MG glioma cells. 20 However, the mechanism by which punicalagin induces cell death in colorectal cancer remains known. In this study, we found that punicalagin induces apoptosis by activating caspse-3, and then induces the death of colorectal cancer cells through apoptosis.
EMT is a reversible process by which epithelial cells transition between epithelial and mesenchymal states and is essential for normal development and tissue regeneration. Dysregulated induction of the EMT is involved in promoting migration and invasion, as well as enhancing resistance to chemotherapy, and is associated with malignant features of cancer cells during cancer progression and metastasis.21,22 Furthermore, MMPs play an important role in normal development through cellular regeneration, programmed cell death, angiogenesis, and many other essential physiological processes, in addition to being involved in many pathological processes such as the EMT. Several studies have described the effects of punicalagin on cell migration and invasion. Punicalagin was shown to inhibit ovarian cell migration by increasing TIMP-2 and TIMP-3 expression and decreasing MMP-2 and MMP-9 activity. 23 Punicalagin also inhibited HeLa cell migration by downregulating MMP-2 and MMP-9 and upregulating TIMP-2 and TIMP-3. 17 Pan et al. elucidated that punicalagin inhibited the migration and invasion of MCF-7 and MDA-MB-231 breast cancer cells by increasing the expression of E-cadherin and decreasing the expression of MMP-2, MMP-9, and N-cadherin. 24 Punicalagin inhibited proliferation, invasion, and angiogenesis of osteosarcoma cells by blocking NF-κB signaling. 7 The results of this study are consistent with previous studies, demonstrating that punicalagin suppresses the invasion of colorectal cancer cells by inhibiting MMP-2, MMP-9, Snail, and Slug.
In conclusion, this study demonstrates that punicalagin inhibits cancer growth by inhibiting proliferation and invasion. Further studies are needed to identify therapeutic targets involved in punicalagin-induced cytotoxicity and inhibition of invasive activity. Nevertheless, the results of this study provide preliminary evidence supporting the use of punicalagin, a phytochemical used in traditional medicine, as a new potent candidate for colorectal cancer chemotherapy.
Footnotes
Acknowledgments
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was support by the research grants (No. CMFHR10918 and No. 110CM-TMU-12) from Chi Mei Medical Center, Tainan, Taiwan.
