Abstract
Objectives
Scutellarin, which is obtained from Erigeron breviscapus Hand-Mazz (EBHM), is a flavonoid that has demonstrated the ability to safeguard neural stem cells from hypoxia-induced damage and prevent cell apoptosis. The aim of this study was to investigate the beneficial impacts of Scutellarin on mitophagy in both in vivo and in vitro models of myocardial ischemia-reperfusion (I/R) injury.
Materials and Methods
Prior to inducing models of myocardial I/R injury, mice were administered either Scutellarin (50 mg/kg/day) or a vehicle for seven consecutive days. The mice underwent I/R injury (30 min of left anterior descending (LAD) coronary artery ligation followed by 120 min of reperfusion). Myocardial injury markers were assessed by the enzyme-linked immunosorbent assay (ELISA). The size of the myocardial infarction was measured via 2,3,5-triphenyl tetrazolium chloride triazole staining, and the protein expression of LC3 and caspase-3 was determined through Western blot analysis. In vitro experiments were conducted utilizing cultured H9C2 cells subjected to an oxygen-glucose deprivation/reoxygenation model to investigate the underlying mechanism(s) of Scutellarin’s positive effects (50 µM).
Results
It shows that Scutellarin treatment reduced the size of the myocardial infarctions and decreased the levels of myocardial injury markers. Western blot analysis showed that protein expression of caspase-3 was decreasing and the ratio of LC3Ⅱ to LC3Ⅰ was increasing in the Scutellarin group. In vitro, Scutellarin decreased oxidative stress markers, stabilized mitochondrial membrane potential (∇Ψm), decreased mitochondrial permeability transition pore (mPTP) opening rate, promoted mitochondrial fusion, inhibited mitochondrial fission, and increased adenosine triphosphate (ATP) production and cell viability. Scutellarin increased the commitment of mitophagy by regulating Pink and Parkin, while apoptosis decreased. cAMP-response element-binding protein (CREB) and extracellular signal-regulated kinases 1 and 2 (ERK1/2) phosphorylation were also modulated by Scutellarin.
Conclusion
The myocardial protective effect of Scutellarin may be associated with the phosphorylation of CREB and ERK1/2.
Introduction
Revascularization is aimed at restoring myocardial blood perfusion by opening occluded coronary arteries. Nonetheless, it is also associated with myocardial ischemia-reperfusion injury (I/R). During the reperfusion stage, the restoration of blood and oxygen supply may cause further myocardial damage and trigger additional cardiomyocyte death. The detrimental effects of myocardial I/R injury extend to metabolic processes and myocardial ultrastructure, leading to adverse ventricular remodeling and ultimately contributing to the development of chronic heart failure (Zhang et al., 2019 a, 2019b, 2019c). In light of the adverse consequences of myocardial I/R injury, some patients experience recurrent angina, progressive deterioration of cardiac function, impaired quality of life, and diminished survival rates in the aftermath of revascularization (Liang et al., 2017). Despite previous research highlighting the significance of oxygen stress injury, mitochondrial damage, and microcirculation disorder in the pathogenesis of myocardial I/R injury (Chen et al., 2017; Pachel et al., 2016; Xue et al., 2020), there is still limited understanding regarding the underlying mechanisms of this condition. Furthermore, current available pharmacological agents lack efficacy in protecting the myocardium against myocardial I/R injury.
Autophagy serves as an intracellular mechanism for self-protection and self-renewal, facilitating the removal of long-lived proteins, damaged organelles, and malformed proteins through lysosomal degradation during biosynthesis. Within cardiomyocytes, mitochondria play a crucial role in regulating the balance between cellular survival and apoptosis (Wanget al., 2019). The genes responsible for mitochondrial fission and fusion play a significant role in maintaining mitochondrial homeostasis. A wealth of research has suggested that I/R injury primarily results from a multitude of complex factors that ultimately culminate in disrupting mitochondrial homeostasis (Luongo et al., 2017; Zhou et al., 2018a, 2018b, 2018c). Research on cardiac autophagy has yielded conflicting results. While some researchers have suggested that autophagy could suppress cardiomyocyte apoptosis, others have shown evidence that mitophagy serves as a protective mechanism during myocardial I/R injury (Hamacher-Brady et al., 2006; Liu et al., 2019; Paradies et al., 2018). Nevertheless, some have argued that autophagy exacerbates cardiomyocyte injury (Gao et al., 2018; Xu et al., 2015). Consequently, it is crucial to conduct further investigation into the protective effects of mitophagy on the myocardium.
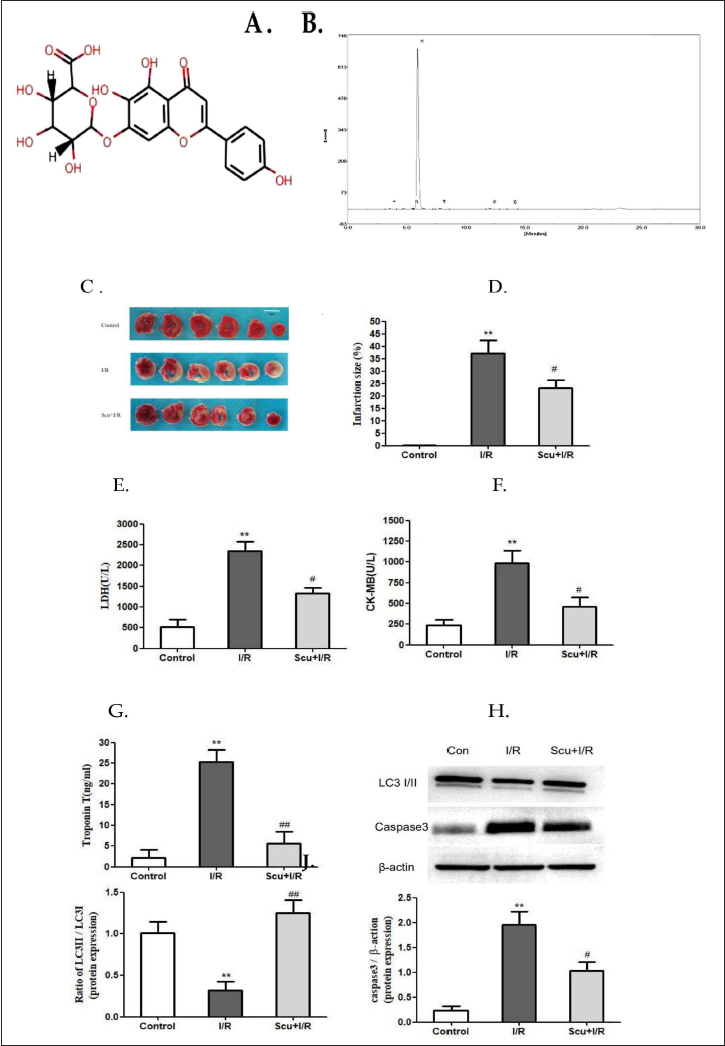
Erigeron breviscapus (Vant.) Hand-Mazz. (EBHM), a traditional Chinese medicinal plant, is a dicotyledonous plant in the Compositae chrysanthemum family. EBHM is primarily found in southwestern China, including Hunan, Guangxi, and Yunnan provinces, and has been used to reduce toxicity, eliminate dampness, promote circulation, alleviate inflammation, and alleviate pain (Fan et al., 2021). Scutellarin is a monomeric flavonoid compound that can be found in the herbal medicine EBHM (Figure 1A). Studies on Scutellarin in the past three decades have demonstrated the effectiveness of the herbal medication in treating cerebrovascular and cardiovascular diseases, especially ischemic stroke and coronary heart disease (Chledzik et al., 2018; Lin et al., 2007; Wang & Ma, 2018; Wang et al., 2016). Scutellarin has the ability to permeate through the bloodstream and distribute extensively throughout the brain and heart tissue. When present in myocardial tissue, Scutellarin can significantly mitigate the harm caused by various forms of injury (Jia et al., 2008; Wang et al., 2015; Zhi et al., 2018). Mechanistic investigations have demonstrated that Scutellarin has the capacity to decrease vascular resistance, enhance cardiac function, suppress interstitial fibrosis, and consequently exert a safeguarding effect in cases of myocardial ischemic disorders (Pan et al., 2011; Wang et al., 2018). Drawing upon previous investigations, it has been reported that Scutellarin demonstrates the potential to alleviate cognitive deficits in mouse models of hypoxia by invoking apoptosis inhibition. Given this discovery, it is postulated that Scutellarin may hold the potential to shield cardiomyocytes from I/R injury by evoking apoptosis inhibition (Wang et al., 2017). Prior research has indicated that Scutellarin may safeguard astrocytes from hypoxia/reoxygenation damage by potentially stimulating the production and secretion of neurotrophins (Chai et al., 2013).
In the present investigation, we scrutinized the conceivable impacts of Scutellarin on myocardial I/R injury by utilizing a murine model and culturing H9C2 cells. We hypothesize that Scutellarin can protect against myocardial I/R injury by promoting mitophagy and mitigating oxidative stress. This cardioprotective effect may be mediated by regulating mitochondrial homeostasis and mitigating mitochondrial dysfunction. Furthermore, we propose that Scutellarin’s actions may involve the activation of the extracellular signal-regulated kinases 1 and 2 (ERK1/2)-cAMP response element-binding protein (CREB) signaling pathway.
Materials and Methods
Antibodies and Reagents
Scutellarin (purity: 98.6%; Figure 1B displays the high-performance liquid chromatograms that were provided by the supplier of Scutellarin) was purchased from Shanghai Yuanye Bio-Technology Co. Ltd. (Cat. number YY90017, Shanghai, China). Antibodies for T-ERK1/2 (Cat. number #4695), P-ERK1/2 (Cat. number #4370), U0126 (Cat. number #9903), microtubule-associated protein one light chain 3 (LC3)Ⅰ (Cat. number #4599), LC3Ⅱ (Cat. number #3868), P62 (Cat. number #3912), Parkin (Cat. number #2132), Pink (Cat. number #6946), caspase-3 (Cat. number #9662), CREB (Cat. number #9197), and Cyt c (Cat. number #4280) were from Cell Signaling Technology (MA, USA). Phosphatase inhibitor cocktail tablets (Cat. number PhosSTOP) were from Roche Diagnostics (Baden-Wuerttemberg, Germany). Trizol reagent (Cat. number T9424), 2,3,5-triphenyl tetrazolium chloride triazole (TTC, Cat. number 17779), 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT, Cat. number M2128), JC-1 (Cat. number T4069), dihydroethidium (Cat. number 37291), radioimmunoprecipitation assay buffer (RIPA) buffer (Cat. number R0278), and KG-501 (Cat. number 70485) were from Sigma Aldrich (MO, USA). Dulbecco’s modified eagle medium (DMEM) high-glucose medium (Cat. number 10938025), DMEM medium (Cat. number 11966025), 2-mercaptoethanol (Cat. number 31350010), penicillin and streptomycin (Cat. number 10378016), fetal bovine serum (FBS, Cat. number 16140063), GlutaMAX (Cat. number 35050061), and Accutase (Cat. number A11105-01) were from Gibco (CA, USA). Lactate dehydrogenase (LDH, Cat. number im-E20034), troponin T (Cat. number DL-TNT-mu), malondialdehyde (MDA, Cat. number im-E20347), and superoxide dismutase (SOD, Cat. number im-E20348) were from KALLANG Biological Technology Co., Ltd. (Shanghai, China). Creatinine kinase-MB (CK-MB, Cat. number 96TF10306) and adenosine triphosphate (ATP, Cat. number tg0074) were from Westing Biological Technology Co., Ltd. (Shanghai, China). RevertAid First Strand cDNA Synthesis Kit (Cat. number K1621) was from Invitrogen (CA, USA). SYBR® Select Master Mix (Cat. number 4472919) was from applied biosystems (CA, USA).
Mouse Model of Myocardial I/R Injury
The study protocol was approved by the Institutional Animal Care and Use Committee and the Ethics Committee of Kunming Medical University with approval numbers Kmmu20021365, in accordance with NIH guidelines. Adult male C57BL/6 mice (12 weeks of age; Peking Weitonglihua Laboratory Animal Center, Beijing, China) were fed according to our previous study. In accordance with the ethical guidelines and the principle of minimal animal usage, a total of 30 mice were utilized for the purpose of establishing the experimental model. The experimental mice were divided into three groups: the control group, the myocardial I/R injury group, and the Scutellarin + myocardial I/R injury group (Scu + I/R). Fifteen murine hearts were subjected to TTC staining (n = 5/group), whereas an additional set of 15 was collected for Western blot analysis subsequent to blood extraction (n = 5/group). The mice underwent cardiac I/R injury as previously described (Zhou et al., 2018a, 2018b, 2018c). An intraperitoneal injection of 1% sodium pentobarbital (50 mg/kg) was used to anesthetize mice. Following intubation, the mice were connected to a ventilator and secured onto a circulating warming surgical table. A standard lead electrocardiogram was attached, and the surgical area was aseptically prepared. The heart was exposed via a left thoracotomy between the fourth and fifth ribs. A 6.0 suture needle was used to pass through the left anterior descending (LAD) artery from the lower edge of the left atrial appendage. Two suture loops were passed through the ligation site, and the LAD was ligated. The left ventricular anterior wall gradually turned pale, and ST-segment elevation was shown in the electrocardiogram, indicating successful ligation (this resulted in extensive anterior wall ischemic injury). The suture loops were placed on each side of the incision and left outside of the chest cavity. After 30 min of ischemia, both suture loops were slowly pulled out horizontally to restore blood flow, resulting in ST-segment elevation resolution. Samples were harvested after 120 min of reperfusion. Mice were maintained under 3% isoflurane anesthesia during the procedure, and heart rate and respiration were monitored throughout. Sham-operated mice underwent the same procedure without occlusion of the LAD. Before models of I/R injury were performed, mice received either Scutellarin (50 mg/kg/d) or vehicle (0.9% normal saline, solvent for scutellarin) intraperitoneally (i.p.) for seven consecutive days (n = 5 per group). The dosage and treatment duration were based on previous findings (Lin et al., 2007). After the myocardial I/R injury, blood from mice was analyzed for CK-MB (ng/mL), troponin T (ng/mL), and LDH (KU/L) levels using enzyme-linked immunosorbent assay (ELISA). Each mouse sample underwent individual testing with three replicate wells for each sample. All procedures were according to the manufacturer’s instructions for commercial kits (all assays were repeated three times).
Myocardial Infarct Size
The size of the myocardial infarction was determined through staining with TTC. Briefly, the hearts of mice were rapidly removed at the end of reperfusion, washed three times with phosphate buffer solution (PBS, pH 7.4), and then frozen at –20°C for 30 min. The left ventricle of the frozen myocardial tissue was sliced into six sections with a thickness of 2 mm. Subsequently, the mouse heart was incubated with 2% TTC at 37°C for 20 min and fixed in a 10% formalin solution (pH 7.4) for 12 h. The infarct area appeared as an off-white color, while the area at risk appeared red in color. The images were digitally analyzed using the Alpha Ease FC Imaging System. Myocardial infarct size was expressed as a percentage of infarct area/(infarct area + the area at risk) × 100% (Zhouet al., 2017a, 2017b), n = 5/group.
Cell Model of Myocardial I/R Injury
H9C2 cells (Wuhan Punuosai Technology Company, China) were cultured in normal medium (DMEM high-glucose medium containing: 2-mM GlutaMAX, 100 U/mL of penicillin and 100 µg/mL streptomycin (P/S), 0.1 mM 2-mercaptoethanol, and 10% FBS) at 37℃ in a 5% CO2 incubator. The cells were randomly divided into the control group and the experimental group. For the MTT assay, cells in the experimental group were cultured in different concentrations of Scutellarin (10, 30, 50, 100 µM) for 24 or 48 h. To perform the I/R model, cells were incubated in a serum-free DMEM medium after removing the complete medium, followed by exposure in a 5% CO2, 1% O2 incubator at 37℃ for 2 h, and normal reperfusion for 6 h. In the Scutellarin treatment group, Scutellarin (50 µM) was added in a normal medium before I/R injury, while cells in the control group were routinely cultured. Then, cells and medium were harvested after a series of processes for further analysis. LDH (KU/L), MDA (µmol/L), SOD ((U/mL), and ATP (mmol/gprot) were assayed via ELISA. All procedures were according to the manufacturer’s instructions for commercial kits (all assays were repeated three times).
H 2 O 2 Assay in vitro
Retrieve cells from the centrifuge tube, centrifuge to remove supernatant, add 1 mL of original per tube based on 106 cells (power of 20%, 3 s ultrasound, 10 s amplitude, repeated 30 times), centrifuge at 8000g and 4℃ for 10 min, and collect the pellet. Utilize commercially available kits (Solarbio, Beijing, China) as previously described to quantify oxidative stress markers, specifically hydrogen peroxide (H2O2) (Yang et al., 2013).
Mitochondrial Membrane Potential and Mitochondrial Permeability
Mitochondrial membrane potential (∇Ψm) was evaluated using JC-1 staining. Cells were first rinsed with PBS, after which they were incubated in a JC-1 solution (2.5 g/ml) for a duration of 30 min at 37°C. Following this, the cells were washed with binding buffer and imaged. Fluorescence intensity was then quantified using ImageJ software. The ratio of aggregate to monomer (red/green) was subsequently determined for analysis; each group was tested in triplicate (Zhang et al., 2019a, 2019b, 2019c).
The opening of the mitochondrial permeability transition pore (mPTP) was measured using tetramethylrhodamine ethyl ester (TMRE). Specifically, in accordance with previous research, the arbitrary mPTP opening time was defined as the point at which TMRE fluorescence intensity decreased by half between the initial and residual fluorescence intensity readings and each group was tested in triplicate (Zhou et al., 2017a, 2017b).
Western Blots
Western blots were operated as previously described (Wang et al., 2017). In brief, mouse heart tissue and H9C2 cell samples were homogenized using RIPA buffer containing a cocktail of phosphatase inhibitors, then centrifuged at 12,000g for 10 min to collect the supernatant. Heat-denatured samples were separated using 8% SDS-PAGE, transferred to polyvinylidene fluoride membranes, and subsequently blocked with 5% bovine serum albumin (BSA). Overnight incubation of each sample was performed using a primary antibody targeting one of the following specific targets: LC3 (1:1000 dilution; rabbit monoclonal), P62 (1:1500 dilution; rabbit monoclonal), caspase-3 (1:2000 dilution; rabbit monoclonal), mito-cytc (1:1000 dilution; rabbit monoclonal), cyto-cytc (1:1000 dilution; rabbit monoclonal), Parkin (1:1000 dilution; rabbit polyclonal), Pink (1:1000 dilution; rabbit monoclonal), P-CREB (1:1000 dilution; rabbit monoclonal), T-CREB (1:1000 dilution; rabbit monoclonal), T-ERK1/2 (1:1000 dilution; rabbit polyclonal), P-ERK1/2 (1:2000 dilution; rabbit monoclonal), and anti-β-actin (1: 1000 dilution; mouse monoclonal). Subsequently, the membranes were incubated with a horseradish peroxidase-conjugated secondary antibody at a dilution of 1:2000, and visualization was accomplished utilizing a standard electrochemiluminescence method. The intensity of each band was quantified using Image J software (NIH, Bethesda, MD, USA), with normalization relative to β-actin. Protein concentration was determined via the bicinchoninic acid assay method.
Reverse Transcription Polymerase Chain Reaction (RT-PCR)
Cells were subjected to RNA isolation utilizing Trizol reagent, subsequent to which total mRNA was utilized to produce cDNA using the Revert Aid First Strand cDNA Synthesis Kit. SYBR® Select Master Mix was utilized to implement a RT-PCR for quantification of mRNA expression, with the process being carried out on an ABI7300 thermocycler for a total of 40 cycles. The 2–△△CT relative quantification method was utilized, along with glyceraldehyde 3-phosphate dehydrogenase (GAPDH) utilized as the internal control, and the mRNA expressions of Beclin1, mitofusions 1 (Mfn1), mitofusions 2 (Mfn2), optic atrophy 1 (Opa1), Fission 1 (Fis1), dynamin-related protein (Drp1), mitochondrial fission factor (Mff), and peroxisome proliferator-activated receptor gamma coactivator-1α (PGC-1α) were determined. Primers were designed using the software NetPrimer (
Statistical Analysis
The data was presented as mean ± standard deviation and analyzed using the Student’s t-test or one-way analysis of variance, followed by the least significant difference test for pairwise comparisons. Statistical significance was established at p < 0.05. Each testing group included at least three replicates. In vitro experiments were performed with at least three replicates for each experimental condition to ensure reproducibility and reliability. The same experimental condition was repeated three times to assure consistency and validity.
Results
In vivo Experiments
Scutellarin reduces myocardial infarction size in a mouse model of I/R injury.
The myocardial infarction area was shown by TTC staining (n = 5/group). Consistent with the results of the previous study, LAD coronary artery occlusion might induce significant myocardial infarction in the I/R group (Figure 1C). Scutellarin reduced the infarct size significantly (p < 0.05, Figure 1D).
Scutellarin reduces the expression of cardiac injury markers in a mouse model of I/R injury.
Compared with the control group, the activity of LDH was significantly higher in the I/R group. The activity of LDH was reduced with the treatment of Scutellarin (p < 0.01 or p < 0.05, Figure 1E). CK-MB was rapidly increased in the reperfused myocardial tissue, and Scutellarin treatment reduced the levels of CK-MB (p < 0.05, Figure 1F). Myocardial injury induced a significant increase in troponin T, then it decreased in the group treated with Scutellarin, p < 0.01 (Figure 1G).
Scutellarin increases the ratio of LC3Ⅱ to LC3Ⅰ and reduces the expression of caspase-3 in the myocardium of I/R-injured mice.
The radio of LC3Ⅱ to LC3Ⅰ is downregulated in I/R-injured mice while upregulated in Scutellarin-treated mice (Figure 1H and I, p < 0.01). What is more, increased expression of caspase-3 was noticed in the mice after I/R injury. The levels of caspase-3 were decreased in the Scutellarin treatment group, p < 0.05 (Figure 1H and J).
In vitro Experiments
Scutellarin relieves oxidative stress, inhibits apoptosis, and promotes autophagy in I/R injury.
The effects of different concentrations of Scutellarin (0, 10, 30, 50, and 100 µM) on cell viability were assessed by MTT in 24 and 48 h. The doses (10, 30, and 50 µM) of Scutellarin raised the cell viability in comparison with the vehicle group (0 µM). Among them, cells cultured in 50 µM Scutellarin for 48 h might bring about the optimal effect on cell viability (Figure 2A). Therefore, 50 µM Scutellarin (cultured cells for 48 h) was used in the subsequent experiments. Compared with the I/R-treated group, Scutellarin treatment increased cell viability (p < 0.05, Figure 2B).
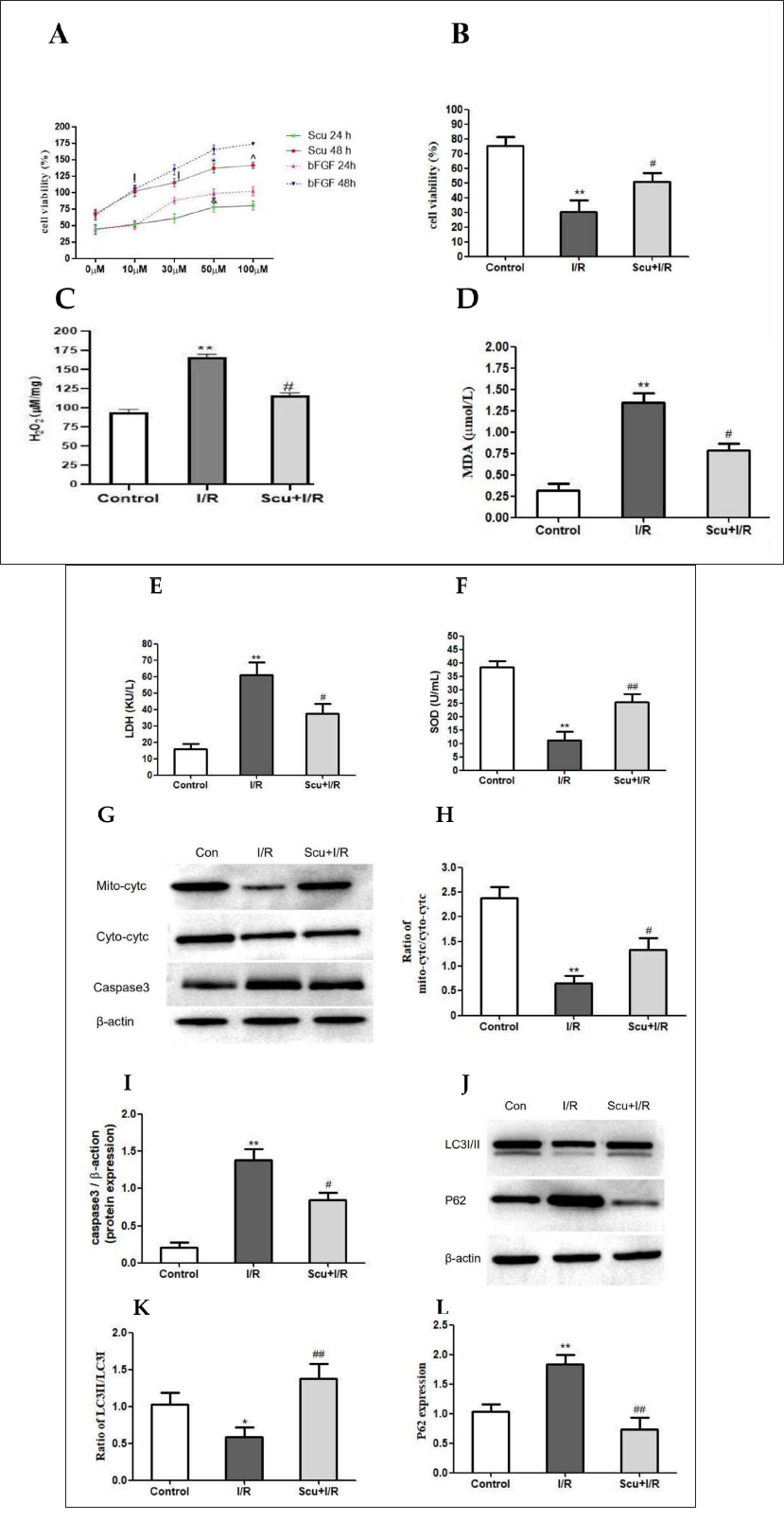
(A) H9C2 cells were incubated with Scutellarin of varying concentrations for 24 h and 48 h. H9C2 cell viability was assessed by MTT. bFGF worked as a positive control (n = 3/group. ! vs. 0 µM group, p < 0.05; * vs. 30 µM group, p < 0.05; ^ vs. 50 µM group, p > 0.05; & vs. 50 µM in 48-h group, p < 0.05). (B) H9C2 cells were exposed to I/R injury and then treated with Scutellarin (50 µM) for 48 h. H9C2 cell viability was assessed by MTT (n = 3/group. ** vs. control group, p < 0.01; # vs. I/R group, p < 0.05). (C–E) Quantitative analysis of oxidative stress markers, including H2O2, MDA, and LDH (n = 3/group. ** vs. control group, p < 0.01; # vs. I/R group, p < 0.05). (F) Quantitative analysis of antioxidative stress markers SOD (n = 3/group. ** vs. control group, p < 0.01; ## vs. I/R group, p < 0.01). (G) Western blot with antibodies against mito-cyt c, cyto-cyt c, and caspase-3. (H) Cyt c Western blot analysis (n = 5/group. ** vs. control group, p < 0.01; # vs. I/R group, p < 0.05). (I) Caspase-3 Western blot analysis (n = 5/group. ** vs. control group, p < 0.01; # vs. I/R group, p < 0.05). (J) Western blot with antibodies against LC3I/II and P62. (K) LC3Ⅱ/LC3Ⅰ Western blot analysis. The value was normalized to the control group (n = 5/group. ** vs. control group, p < 0.01; # vs. I/R group, p < 0.05). (L) P62 Western blot analysis. The value was normalized to the control group (n = 5/group. ** vs. control group, p < 0.01; ## vs. I/R group, p < 0.01).
The levels of H2O2 in the I/R group were higher than in the control group (p < 0.01, Figure 2C). Scutellarin treatment significantly decreased the contents of H2O2 (p < 0.01, Figure 2C). I/R injury significantly increased the levels of MDA and LDH compared with the control group (p < 0.01, Figure 2D and E). When I/R-injured cells were treated with Scutellarin, the levels of MDA and LDH were decreased (p < 0.05, Figure 2D and E). As shown in Figure 2F, compared with the I/R group, Scutellarin significantly increased the expression of SOD (p < 0.01).
As shown in Figure 2G–H, I/R injury resulted in a decreased ratio of mito-cyt c to cyto-cyt c (p < 0.01). Scutellarin treatment limited the cyt-c leakage, and the ratio increased (p < 0.05). Meanwhile, caspase-3 was increased in I/R-treated cells but reduced in cells treated with Scutellarin (p < 0.05 or p < 0.01, Figure 2G and I).
The ratio of LC3Ⅱ to LC3Ⅰ was reduced in the I/R group (p < 0.05) but increased after Scutellarin treatment (p < 0.01, Figure 2J and K). At the same time, compared with the control group, the level of P62 was increasing in the I/R group (p < 0.01). Scutellarin treatment decreased the level of P62 (p < 0.01, Figure 2L).
Scutellarin promotes autophagy and relieves cardiomyocyte injury via the ERK1/2-CREB signaling pathway.
Western blot analysis demonstrated CREB phosphorylation repressed by I/R injury (Figure 3A and B), as indicated by a decreased ratio of P-CREB versus T-CREB (p < 0.01). Interestingly, Scutellarin treatment upregulated the ratio of P-CREB versus T-CREB (p < 0.01). After KG-501 (an inhibitor of CREB) was added to the Scutellarin-treated group, the mRNA expression of Beclin1 was decreased in the I/R-treated cells, increased with Scutellarin treatment, and reduced in the group that added KG-501 (p < 0.05 or p < 0.01, Figure 3D). Similarly, the I/R-induced increase in LDH was also suppressed by Scutellarin, and this beneficial effect was not noted after the blockade of the CREB pathway (p < 0.05, Figure 3E). As shown in Figire 3F, I/R-inhibited cell viability could be reversed by Scutellarin, and this effect was invalid following treatment with KG-501 (p < 0.05).
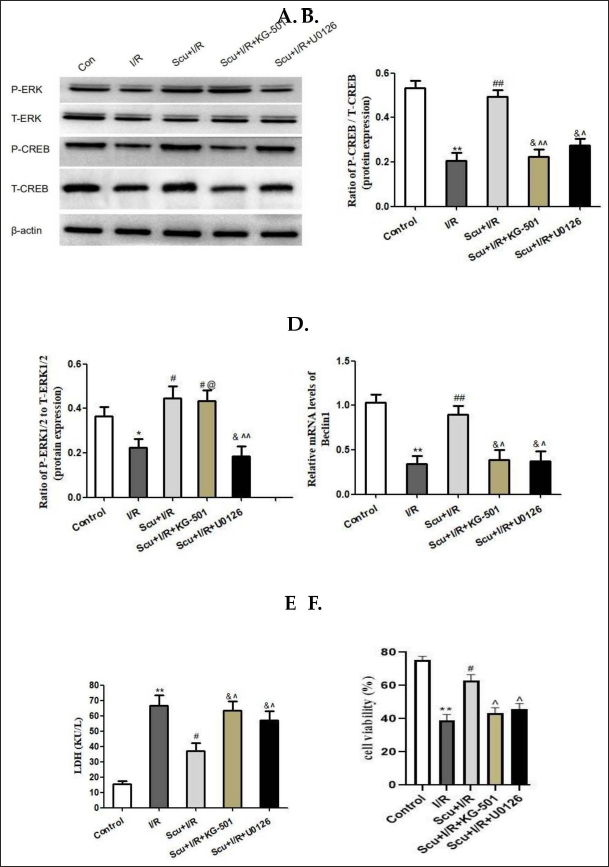
Western blot analysis demonstrated that ERK1/2 was repressed by I/R injury (Figure 3A and C), as indicated by a decreased ratio of P-ERK1/2 versus T-ERK1/2 (p < 0.05). Scutellarin treatment upregulated the ratio of P-ERK1/2 versus T-ERK1/2 (p < 0.05). CREB had the same trend of changing as ERK1/2. However, when U0126 (an inhibitor of ERK1/2) was used, a decline in the ratio of P-CREB versus T-CREB was observed (p < 0.05, Figure 3A and C), and the protective effects of Scutellarin were suppressed (Figure 3D–F).
We propose that the regulation of ERK1/2-CREB pathway phosphorylation by Scutellarin promotes the upregulation of autophagy-related proteins, which in turn leads to the inhibition of I/R injury through mitophagy.
Scutellarin regulates CREB phosphorylation to repair myocardial I/R injury by alleviating mitochondrial dysfunction and inducing mitophagy.
I/R injury impaired the ∇Ψm, and Scutellarin treatment reversed the stability of ∇Ψm (p < 0.01or p < 0.05, Figure 4A–D). The mPTP opening rate was increased after I/R injury (p < 0.05) and decreased in the Scutellarin treatment group (p < 0.05, Figure 4A–D).
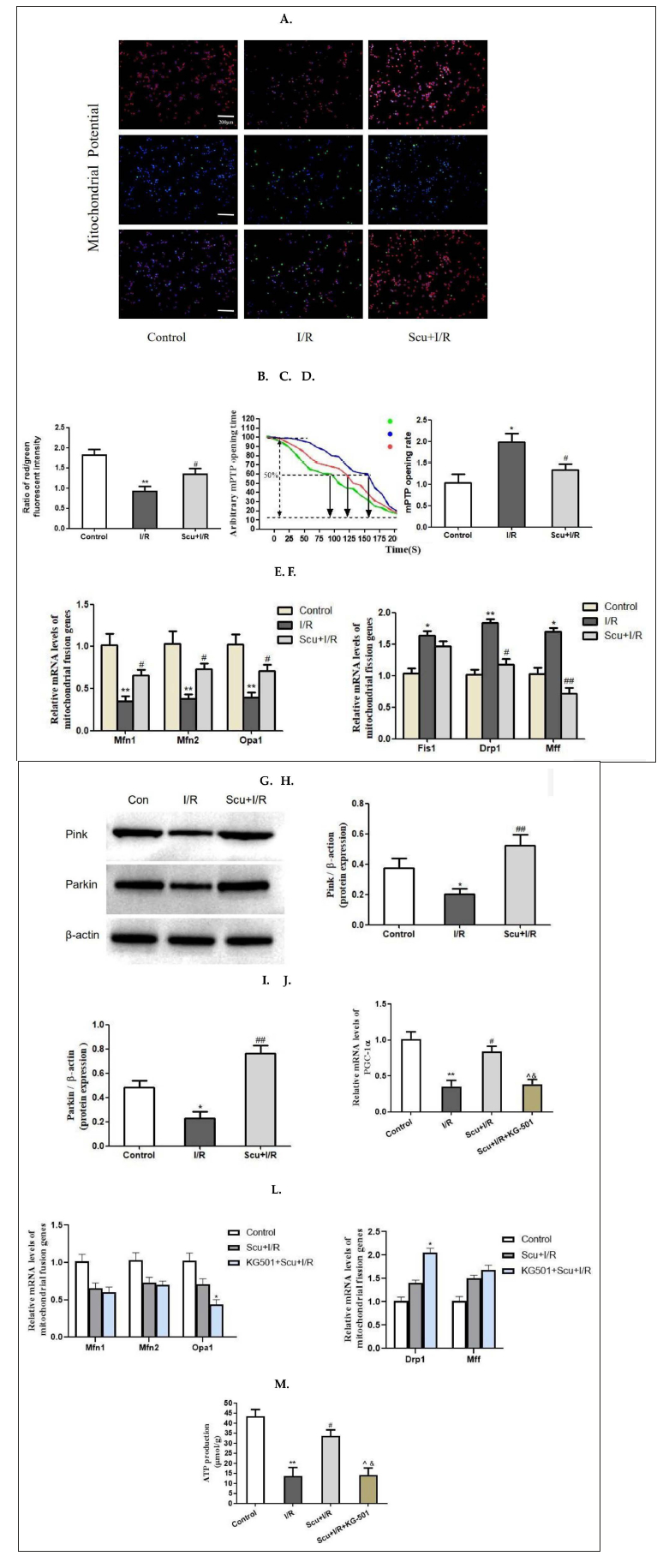
Fusion-related genes, such as Mfn1, Mfn2, and Opa1, were reduced in the I/R injury group, and this reduction was ameliorated in the Scutellarin group. The levels of fission-related genes, such as Fis1, Drp1, and Mff, are shown in Figure 4F. Notably, fission-related genes were increasing in the I/R injury group. Scutellarin treatment decreased the levels of Drp1 (p < 0.05) and Mff (p < 0.01) (Figure 4E). There was no statistical difference between the I/R group and the Scutellarin treatment group among the levels of Fis1.
Compared with the control group, protein expressions of Pink1 and Parkin were decreasing in the I/R group, and Scutellarin treatment increased the protein expressions of Pink1 (p < 0.01) and Parkin (p < 0.01) as shown in Figure 4G–I.
The mRNA level of PGC-1α decreased in the I/R group, while Scutellarin treatment increased the mRNA level of PGC-1α (p < 0.01 or p < 0.05, Figure 4J). However, this beneficial effect was not noted after inhibiting CREB phosphorylation (p < 0.05, Figure 4J). Concurrently, when CREB phosphorylation was inhibited, the mitochondrial fusion-related gene Opa1 was reduced and the mitochondrial fission-related gene Drp1 was increased (Figure 4K–L).
I/R injury resulted in decreased mitochondrial ATP production (p < 0.01, Figure 4M), while it increased in the Scutellarin group (p < 0.05, Figure 4M). The beneficial effect of Scutellarin was not noted after inhibiting CREB phosphorylation (p < 0.05, Figure 4M).
The above data suggests that Scutellarin is effective in promoting mitophagy and restoring mitochondrial structure and function by regulating CREB phosphorylation.
Discussion
Mitochondrial oxidative stress, dysfunction, and imbalanced homeostasis play critical roles in myocardial I/R injury (Zhang et al., 2016). Such damage leads to decreased energy output, excessive production of reactive oxygen species, injuring cardiomyocytes, and inducing programmed cell death through the release of proapoptotic factors (Fuhrmann & Brüne, 2017; Yu et al., 2019). Thus, it is crucial to mitigate mitochondrial oxidative stress, restore mitochondrial function, and maintain mitochondrial homeostasis to minimize the incidence of myocardial I/R injury. Earlier research has indicated that Scutellarin has the potential to safeguard human umbilical vein endothelial cells from oxidative stress (Mo et al., 2018), ease apoptosis induced by
Mitochondria play a critical role in ATP synthesis and oxidative phosphorylation within cardiomyocytes. During I/R, mitochondrial damage occurs and leads to the production of excessive ROS, resulting in oxidative stress. Likewise, oxidative stress exacerbates the damage to mitochondria and triggers cellular death (Hoek et al., 1997). The present study has demonstrated that Scutellarin may effectively attenuate the overproduction of ROS, suppress MDA and LDH expression, promote SOD expression, and elevate ATP content. These findings suggest that Scutellarin serves to safeguard cardiomyocytes against oxidative stress induced by I/R.
Oxidative stress induced by myocardial I/R injury may induce mitochondrial membrane permeabilization, resulting in the release of apoptotic proteins. The proper regulation of mitochondrial oxidative stress and biogenesis is dependent on PGC-1α. However, during myocardial I/R injury, the expression of PGC-1α decreases, leading to mitochondrial dysfunction (Yu et al., 2017). The structure and function of mitochondria are governed by fusion and fission genes. An imbalance between these processes may suggest impaired mitochondrial function and structural damage. The fusion of mitochondria is regulated by Mfn1, Mfn2, and OPA1, whereas Fis1, Drp1, and other genes participate in mitochondrial fission (Aravamudan et al., 2017). Mitochondrial fission disrupts mitochondrial homeostasis and facilitates the generation of fragmented mitochondria. Conversely, mitochondrial fusion fosters inter-mitochondrial communication, facilitates the exchange of contents, and promotes the maintenance of mitochondrial network integrity (Vásquez-Trincado et al., 2016). Mitophagy promotes the removal of fragmented mitochondria (Maneechote et al., 2017). The protective roles of mitochondrial fusion and mitophagy have been detected in vivo and in vitro myocardial I/R injury models (Fuhrmann & Brüne, 2017; Rossello et al., 2017; Zhu et al., 2018). According to the findings of this research, Scutellarin is capable of upregulating PGC-1α expression, downregulating the expression of genes involved in mitochondrial fission, and upregulating the expression of genes involved in mitochondrial fusion and mitophagy, thereby reducing mitochondrial membrane permeabilization, decreasing the release of apoptotic proteins, and inhibiting cardiomyocyte apoptosis.
Autophagy is a metabolic process that is highly conserved across different organisms and plays an essential role in the development of myocardial injury resulting from I/R. The formation of the Beclin1 complex is instrumental in promoting the assembly and elongation of the autophagosomal membrane (Zhang et al., 2019a, 2019b, 2019c). Autophagy begins with the formation of the autophagic membrane, which is followed by the formation of autophagosomes. The incorporation of LC3II, which is converted from LC3I, into the autophagic membrane signifies the maturation of the process (Zhao & Zhang, 2019). The autophagosomes that have matured will undergo degradation in the autolysosomes. In the final step, p62 binds to the LC3-interacting region and completes the degradation process within the interior of the autophagosome (Ha et al., 2014). In the autophagy flux, there is an increase in the expression of Beclin1 and the LC3II to LC3Ⅰ ratio, while the expression of p62 decreases. Autophagy is a vital physiological metabolic process that allows for the maintenance of intracellular homeostasis and the preservation of heart function. However, during myocardial I/R injury, excessive autophagy may exacerbate damage to cardiomyocytes. Thus, the regulation of autophagy could be a promising therapeutic target for preventing cell apoptosis and providing protection against myocardial I/R injury (Lampert et al., 2019). According to this study, the administration of Scutellarin may reduce p62 expression, enhance Beclin1 expression, and increase the LC3II to LC3Ⅰ ratio. Consequently, Scutellarin might promote moderate autophagy in response to myocardial I/R injury. Furthermore, mitophagy serves as a protective mechanism against different cellular insults by eliminating dysfunctional, senescent, and damaged mitochondria. Different mitophagy pathways have been identified at the molecular level, including PINK1-Parkin, BNIP3-NIX, and FUNDC1 (Zhu et al., 2019). It is important to note that various mitophagy pathways have distinct effects on mitophagy function. For instance, mitophagy mediated by BNIP3-NIX can be stimulated during lipid metabolism and can subsequently decelerate the progression of cirrhosis in the liver (Zhou et al., 2018a, 2018b, 2018c). FUNDC1 is of significant importance in the development of severe obesity and insulin resistance in a mouse model subjected to a high-fat diet (Wu et al., 2019). The Pink1-Parkin-mediated pathway has been shown to provide protective effects in myocardial I/R injury by alleviating oxidative stress, inhibiting mitochondrial fission, promoting ATP production, and suppressing apoptosis (Andres et al., 2014; Sun et al., 2019). Our current investigation reveals that Scutellarin enhances mitophagy through the Pink-Parkin pathway. Furthermore, our findings support previous research demonstrating that mitophagy via the Pink-Parkin pathway is associated with increased ATP production, decreased oxidative stress, improved mitochondrial structure and function, balanced mitochondrial homeostasis, and inhibited apoptosis.
To investigate the molecular mechanism underlying the cardioprotective effects of Scutellarin, we analyzed its impact on the MAPK-ERK-CREB signaling pathway. Previous studies have shown that the MAPK-ERK-CREB signaling pathway regulates mitophagy in several disease models. For instance, the ERK-CREB pathway has been identified as a crucial regulator of mitophagy activation and management in neuroinflammation and cerebral I/R injury (Merjaneh et al., 2017; Zhang & Yu, 2018). Our experimental findings revealed that the administration of Scutellarin led to an increase in the P-ERK1/2/T-ERK1/2 ratio and the P-CREB/T-CREB ratio and facilitated autophagy while also reversing mitochondrial dysfunction. Additionally, the inhibition of U0126/KG-501 hindered both of these changes, indicating that Scutellarin mediates its protective effects by activating the ERK1/2-CREB signaling pathways.
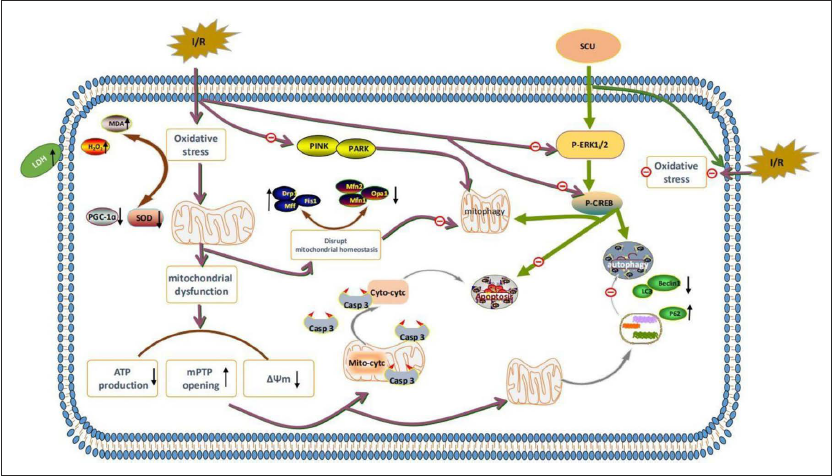
Our collective findings suggest that Scutellarin may alleviate oxidative stress, suppress apoptosis, and stimulate mitophagy. Moreover, via the regulation of ERK1/2-CREB phosphorylation, Scutellarin appears to exhibit a cardioprotective effect against myocardial I/R injury (Figure 5).
Footnotes
Abbreviations
SCU: scutellarin; I/R: ischemia-reperfusion; CREB: cAMP response element-binding protein; ERK: extracellular signal-regulated kinase; TTC: 2,3,5-triphenyl tetrazolium chloride triazole; LC3: microtubule-associated protein one light chain3; MTT: 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide; RIPA: radioimmunoprecipitation assay buffer; DMEM: dulbecco’s modified eagle medium; FBS: fetal bovine serum; LDH: lactate dehydrogenase; MDA: malondialdehyde; SOD: superoxide dismutase; CK-MB:. creatinine kinase-MB; ATP: adenosine triphosphate; mPTP: mitochondrial permeability transition pore.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethics Approval
The study was approved by the Institutional Animal Care and Use Committee and the Ethics Committee of Kunming Medical University with approval numbers Kmmu20021365, in accordance with NIH guidelines.
Funding
This work was supported by the Project of the National Nature Science Foundation of China (No. 82160269; No. 82360275), the Project of the Yunnan Province Science and Technology Foundation (No. 202301AY070001-020; No. 202201AY070001-126), the Project of the Yunnan Provincial Department of Education (2023Y0640), the Project of the Second Affiliated Hospital of Kunming Medical University Fundamental Research (No. 2021yk007), and the Project of Kunming Medical University Fundamental Research (No. 2021-JY-073). Supplemented: the Yunnan provincial platform for research and development of new clinical medical key technologies in the field of cardio-cerebral treatment (No. 202203AC100007).
