Abstract
Background
Paclitaxel administration causes peripheral neuropathy. Auraptene is a natural bioactive monoterpene with anti-inflammatory and anti-neuropathic effects that is widely used.
Objectives
We aimed to study auraptene effects on paclitaxel-induced neuropathy.
Materials and Methods
This study was comprised of two steps of evaluation of the preventive and treatment effects of auraptene in mice. In the first step, mice were randomly allocated into three groups of six animals, including the negative control (NEG CTL): animals injected with paclitaxel (PTX) together with normal saline, PTX (paclitaxel 2 mg/kg given on days 1, 3, 5, and 7), and PREVENTION: PTX + auraptene 100 mg/kg on days 1, 3, 5, and 7. In the second step, animals were allocated into six groups of six: NEG CTL (normal saline), PTX (paclitaxel 10 mg/kg given on days 1, 3, 5, and 7), PTX AUR (PTX + auraptene 50, 75, and 100 mg/kg), and a positive control (PTX-treated animals receiving imipramine 10 mg/kg). Intraperitoneal injection of PTX 2 mg/kg on days 1, 3, 5, and 7 was used in order to induce neuropathy. In both steps of the study, a hot plate test was done on day 7 in order to determine the response to heat. After the scarification of animals, the interleukin-6 (IL-6) level in the sciatic nerve was assessed by western blotting.
Results
In the preventive group, auraptene could reduce hyperalgesia significantly. In the treatment step, the AUR 100 mg/kg compared to NEG CTL groups had significantly increased heat latency. The expression of IL-6 protein in the sciatic nerve was remarkably decreased in both the preventive and treatment groups compared to NEG CTL.
Conclusion
Taken together, our results confirmed that AUR could decrease paclitaxel-induced hyperalgesia in mice and IL-6 protein content in sciatic nerve samples.
Introduction
The most frequent deaths in the world are related to cancer, and chemotherapy is still the most effective treatment against it (Kautio et al., 2008). The increasing rate of cancer deaths is due to chemotherapy and responsiveness in cancerous patients. The increasing use of chemotherapeutics needs more attention to their adverse effects. The adverse effects of these drugs are related to many factors, including their structure, mechanism of action, and individual differences. One of the most frequent adverse effects of anticancer drugs, including vinca alkaloids, platinum derivatives, and taxanes, is peripheral neuropathy (Kautio et al., 2008).
Taxanes, including paclitaxel (PTX), are one of the most effective chemotherapeutics used for many types of cancer, such as ovarian and breast cancers, lung neoplasms, and gastrointestinal tumors (Weaver, 2014). PTX is widely used for ovarian and breast cancer, non-small cell lung cancer, and AIDS-related Kaposi’s sarcoma (Verhulst et al., 2018). Of the PTX-treated patients, about 60%–70% experience peripheral neuropathy that limits drug usage (Staff et al., 2020). Taxanes cause metaphase arrest in mitosis by binding to tubulin (Klein & Lehmann, 2021; Staff et al., 2020). They also bind strongly to axonal microtubules and cause neuropathy, which is one of their main adverse effects (Klein & Lehmann, 2021; Staff et al., 2020).
There is an increasing desire to use herbal medicines because of their safety (Arman et al., 2022; Bibak et al., 2019; Fernández et al., 2021; Ferrarini et al., 2022; Freitas et al., 2021; Rezaee et al., 2019; Shahraki et al., 2020). Auraptene (AUR), (7-geranyloxycoumarin), a coumarin compound found in plants belonging to the Apiaceae and Rutaceae families, is the most abundant prenyloxycoumarin (Derosa et al., 2016). It was isolated from Citrus aurantium and Aegle marmelos, as well as orange, grapefruit, and mandarin (Tayarani-Najaran et al., 2021). AUR has many therapeutic effects, including antioxidative, anti-hypertensive, anti-diabetic, antiprotozoal, anti-genotoxic, anti-inflammatory, immunomodulatory, anti-cancer, and anti-helicobacter (Derosa et al., 2016; Furukawa et al., 2020; Tabrizian et al., 2019; Tabrizian et al., 2015; Tayarani-Najaran et al., 2021). It can inhibit inducible nitric oxide synthase (iNOS) and cyclooxygenase (COX) expression in lipopolysaccharide-induced inflammation and produce anti-inflammatory effects (Genovese & Epifano, 2011). AUR not only poses anti-inflammatory effects peripherally and in the CNS (An et al., 2020). Coumarin derivatives, including AUR, umbelliprenin, and scopoletin, have various degrees of anti-inflammatory effects (Shirani et al., 2021). Umbelliprenin and AUR have similar structures and pharmacological effects (Shirani et al., 2021). Umbelliprenin could decrease hyperalgesia and interleukin-6 (IL-6) levels in a mouse model of PTX-induced neuropathy (Shahraki et al., 2020). AUR neuroprotective effects by reducing microglia and astrocyte hyperactivation were also shown (Furukawa et al., 2020). Moreover, it can increase neurotrophic mediators such as glial cell line-derived neurotrophic factor (GDNF) and brain-derived neurotrophic factor (BDNF) (Furukawa et al., 2020). Both of them have pivotal roles in neuron plasticity and neuronal cell survival (BDNF, 2013; GDNF-dependent neurons, 2013). Mediators such as cytokines, chemokines, growth factors, eicosanoids, and peptides have an effective role in inducing and establishing inflammation and pain (Shirani et al., 2021). IL-6 is a pro-inflammatory cytokine that increases inflammation and pain (Askari et al., 2021; La et al., 2013).
Thus, since AUR could decrease inflammation, in the current study, AUR effects on PTX-induced neuropathy were evaluated in mice.
Materials and Methods
Animals
Fifty-four inbred strains of male albino mice, 30–35 g and 4 weeks of age, were housed in cages in a 12 h/12 h day/night cycle at 22°C and had food and water ad libitum. All animal experiments prepared by the National Academy of Sciences and published by the National Institutes of Health (NIH publication no. 85-23, revised 1996) are in conformity with EU Directive 2010/63/EU for animal experiments.
Reagents
PTX was purchased from Felaxis/multi-pharma (Giza, Egypt), and AUR (C19H22O3, Mm: 298.376 g/mol, >95% purity) was purchased from Sigma-Aldrich Germany. IL-6, anti-b-actin antibody, and secondary rabbit antibody were obtained from Cell Signaling (Beverly, MA, USA). Coomassie (Bradford) Protein Assay Kit was obtained from Thermo Scientific (Rockford, IL, USA).
Experimental Groups and Study Design
In two steps of the present study, 54 mice were allocated into nine groups, with six animals in each group. In the first step, mice were randomly allocated into three groups of six animals, including NEG CTL: animals injected PTX together with normal saline (subcutaneously), PTX (paclitaxel 2 mg/kg intraperitoneally given on days 1, 3, 5, and 7), and PREVENTION: PTX + auraptene (100 mg/kg on days 1, 3, 5, and 7, SC).
In the second step, there were a negative control (PTX-treated mice receiving normal saline), three groups of AUR (which received single-dose AUR at 50, 74, and 100 on day 7), a prevention group (PTX-treated mice receiving AUR 100 mg/kg on days 1, 3, 5, and 7), and a positive control group (PTX-treated mice receiving a single dose of imipramine 10 mg/kg on day 7). In order to induce neuropathy, all doses and the method of injection of AUR were based on previous studies (Arabi et al., 2021; Etemad et al., 2019).
(a) Hot-Plate Test
For hot-plate tests, Ugo Basil (Gemonio, Italy) was used. In this assay, animals were put on the hot plate device in order to calculate the time they start licking, lifting paws, or jumping from the surface (i.e., response latency). The cut-off time (the maximum time that can put animals on the hot plate device) was considered 45 s (Shahraki et al., 2020). Latencies were measured at 30, 60, 90, and 120 min after AUR and imipramine 10 mg/kg i.p. (Rezaee et al., 2019; Zarrindast et al., 2000).
(b) IL-6 Protein Expression Assessment
Animal anesthesia was induced by using ketamine/xylazine (100/10 mg/kg, respectively) (Karimi et al., 2010), then they were decapitated. The sciatic nerve samples were collected and stored at −80°C for the calculation of the IL-6 protein level using western blotting. Firstly, the samples were homogenized (using a mechanical homogenizer) in a buffer solution [50 mmol/L Tris–HCl; pH 7.5; 150 mmol/L NaCl; 1% SDS/protease inhibitor cocktail (Sigma-Aldrich)], followed by centrifugation (1,000 rpm for 10 min at −4°C), the supernatants were collected, and the total protein content was calculated. Secondly, 200 mg of supernatant was loaded on 10% SDS-PAGE and separated. Then, transformation was done to the polyvinylidene difluoride (PVDF) membranes, which were then blocked by skim milk (5% non-fat dry milk). Finally, after the time of blocking (1 h at room temperature), the samples were adjugated with the primary antibody (IL-6 with a dilution of 1:1000 overnight in the shaker at 4°C) and the secondary rabbit HRP-conjugated antibody (1:3000) for 1 h at room temperature. Then the chemiluminescence blots were used for image analysis with Syngene ChemiDoc. Eventually, the calculation of blots was done by GeneTools software (Syngene, Frederick, MD, USA).
Statistical Analysis
Data were analyzed using SPSS version 16 (SPSS, Inc., Chicago, IL, USA). Continuous variables were compared using a two-way ANOVA. Statistically significant differences were considered at p < 0.05.
Results
Effects of AUR on IL-6 Protein Expression
Sciatic nerve analysis of IL-6 protein content showed that AUR reduced IL-6 protein expression. As shown in Figure 1, the negative control and AUR administered in prevention groups differed significantly in this regard (p < 0.05). Furthermore, AUR 100 mg/kg significantly reduced IL-6 protein in comparison to the negative control group (p < 0.01).
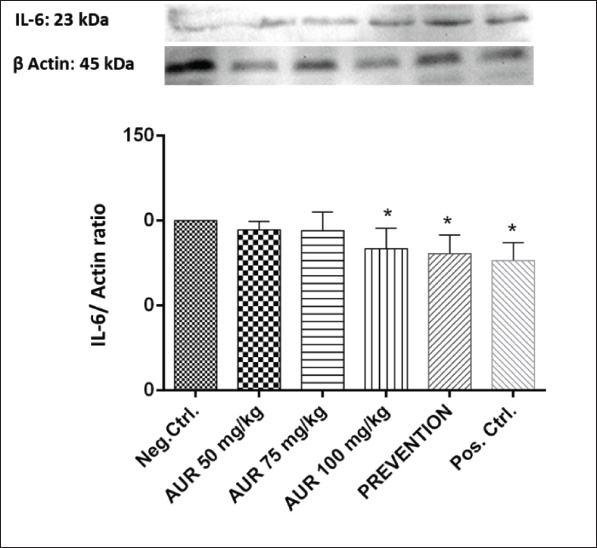
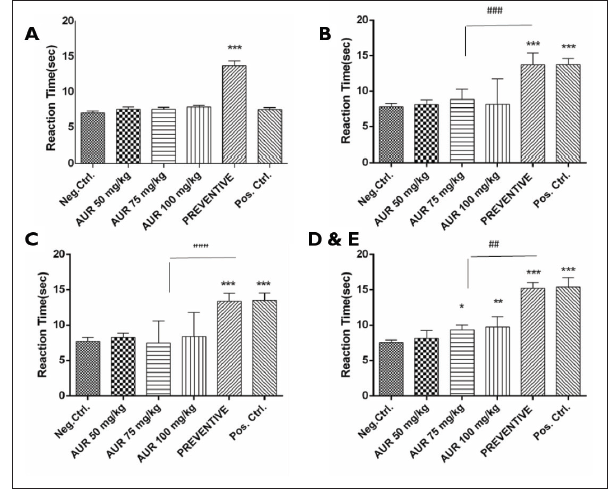
Effects of AUR on Neuropathic Pain Induced by PTX: Hot Plate Test
Hyperalgesia was measured by the hot plate at the different time points (0, 30, 60, 90, and 120 min) following AUR administration. The prevention group, which received AUR along with PTX on days 1, 3, 5, and 7, had significant differences (***p < 0.001) in heat response compared to the NEG CTL group (Figure 2). At 30 and 60 min, differences were significant between the preventive and positive control (i.e., imipramine) in this parameter. After 90 and 120 min following a single dose injection of AUR, differences were significant at 75 and 100 mg/kg of AUR (*p < 0.05 and **p < 0.01 for AUR 75 and 100 mg/kg, respectively) compared to NEG CTL (Figure 2). No significant differences between the AUR 50 mg/kg single dose and the negative control were found (p > 0.05).
Discussion
Cancer treatment adverse effects are still a major concern for clinicians. Neuropathic pain induced by anticancer drugs, including taxanes, influences the quality of life of patients. Our data confirmed that AUR could significantly reduce hyperalgesia in the treatment and preventive groups compared to the negative control. Moreover, the results confirmed that AUR could reduce IL-6 protein levels in sciatic nerve samples harvested from animals. Therefore, our results showed anti-hyperalgesia in an animal model using a hot plate test in single and multiple doses. Another finding confirmed the effects of AUR only in the IL-6 (proinflammatory cytokine) in sciatic nerve samples, and for a more elucidated mechanism of action, it needed to do more evolution in other possible mechanisms. Tricyclic antidepressants such as amitriptyline and imipramine are used to alleviate peripheral neuropathy induced by chemotherapeutic drugs (Akhtaruzzaman et al., 2021).
For alleviating PTX-induced neuropathy, other potential therapies reported in the literature, including some natural products and drugs (carbamazepine, pregabalin, N-acetylcysteine, gabapentin, nimodipine, minocycline, metformin, oxcarbazepine venlafaxine, recombinant human leukemia inhibitory factor) and some non-pharmacological methods including acupuncture, cryotherapy, exercise therapy, and Vitamin B or Vitamin E, have been tried (Burgess et al., 2021; Klein & Lehmann, 2021; Staff et al., 2020). However, for the aforementioned compound, there are some limitation, including adverse drug reactions and serious drug interactions. These are the reasons for the investigation of new compounds, including AUR. In past studies, it was confirmed that AUR didn’t have significant adverse drug reactions; it could possibly be one valuable alternative for PTX-induced hyperalgesia.
One of the most widely used methods for evaluating hyperalgesia in rodents is the hot plate test. The paw withdrawal threshold is a factor that is measured in the hot plate test. In the current study, it was confirmed that AUR in the preventive and treatment groups reduced hyperalgesia by increasing this threshold. In a study, the injection of recombinant IL-6 into the red nucleus could increase this threshold in rats (Ding et al., 2018). They confirmed that IL-6 has its effects on the induction of neuropathy by activating the JAK/STAT and ERK signaling pathways (Burgess et al., 2021). Consistently, another study showed that AUR increased GDNF and BDNF in neuroblastoma Neuro2a cell lines cultured with AUR (Furukawa et al., 2019; Furukawa et al., 2020). In agreement with these results, our study confirmed that AUR could decrease IL-6 protein levels in preventive and treatment groups. However, studying IL-6 levels in sciatic nerve samples could not completely elucidate the mechanism of AUR action.
AUR and umbelliprenin belong to the prenyloxy umbelliferon derivatives that are found in the Rutaceae family, especially Citrus spp. (Fiorito et al., 2022). Neuroprotective and anti-inflammatory effects of AUR in mice were confirmed (Okuyama et al., 2013). In mice with global ischemia, AUR could reduce inflammation as well as apoptosis in their hippocampus (Furukawa et al., 2012). They confirmed that another mechanism of anti-inflammatory effects relies on suppression of neuronal inflammation (Okuyama et al., 2013). IL-6 is involved both in chronic and acute phases of inflammation (Hirano, 2020). In our study, it was confirmed that IL-6 levels decreased, and the aforementioned study is in line with ours.
Furthermore, past studies confirmed that AUR could decrease IL-6, matrix metalloproteinase 2, and other key inflammatory cytokines, including IL-8, in oral epithelial cells stimulated with lipopolysaccharides (LPS) (La et al., 2013).
iNOS could increase pro-inflammatory cytokines such as TNF-α, interlukin-1β, and interlukin-6, as well as COX-2 production (Hsia et al., 2021; Raykova et al., 2003). It was reported that AUR could decrease iNOS and pro-inflammatory mediators in an animal model of lung injury (Tayarani-Najaran et al., 2021).
In the peripheral nerves, it has been demonstrated that PTX-induced neuropathy increases inflammation and oxidative stress. It leads to mitochondrial dysfunction in peripheral nerves. The reduction of inflammation alleviates neuropathic symptoms, including tingling, pain, and numbness in the feet (Wu & Chen, 2019). Another experiment confirmed that AUR inhibited inflammation in LPS-treated RAW 264.7 cells. As already mentioned, they confirmed that AUR subsides inflammation by decreasing the production of prostaglandin E2 and COX-2 transcription (Yan et al., 2013). Subsequent inflammation is another possible mechanism for decreasing hyperalgesia following PTX-induced neuropathy (Wang et al., 2018; Wu & Chen, 2019).
Conclusion
Taken together, the results of this study showed that PTX induces neuropathy and hyperalgesia. Both single-dose and four-dose AUR given accompanied by PTX reduced hyperalgesia. Furthermore, our results showed that AUR could reduce the IL-6 protein level in sciatic nerves harvested from mice. Study design with a wider range of doses and routes of admiration may be useful for more detailed results. Study limitations are related to elucidating more mechanisms of anti-hyperalgesia in AUR. It needed to evaluate mechanistic pathways, including the calculation of other cytokines, iNOS, and COX-2 levels in samples obtained from the blood and sciatic nerve.
Abbreviations
AUR: Auraptene; PTX: Paclitaxel; IL-6: Interleukin 6; AIDS: Acquired immunodeficiency syndrome; iNOS: Inducible nitric oxide synthase deficiency; COX: Celecoxib; GDNF: Glial cell derived-neurotrophic factor; BDNF: Brain-derived neurotrophic factor; SDS-PAGE: Sodium dodecyl-sulfate polyacrylamide gel electrophoresis; PVDF: Polyvinylidene fluoride; HRP: Horseradish peroxidase; JAK/STAT: Janus kinase/signal transducers and activators of transcription; ERK: Extracellular signal-regulated kinases.
Footnotes
Acknowledgments
The authors would like to thank the Zabol University of Medical Sciences for their support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this study.
Statement of Informed Consent and Ethical Approval
This study has been approved with the ethical approval No. ir.zbmu.aec.1401.12.
