Abstract
Background
Diabetic neuropathic pain (DNP) is a serious chronic complication of diabetes mellitus. Oxidative stress and inflammation are considered the central mechanisms of its pathogenesis. Schefflera arboricola (SA) is a Schefflera plant of the Araliaceae family that has the effect of pain relief. In this study, the effect of SA on DNP was first investigated.
Materials and Methods
Diabetic neuropathy in rats was induced by a single injection of streptozotocin (60 mg/kg). Three weeks later, SA (100 mg/kg, 300 mg/kg) or gabapentin (100 mg/kg) was given once a day for a week. Blood glucose, body weight, and thermal withdrawal latency in DNP rats were measured. The morphology of the sciatic nerve was observed by light microscopy and toluidine blue staining. Semiquantitative analysis of myelin basic protein (MBP) of the sciatic nerve was performed by immunohistochemistry. IL-1β, tumor necrosis factor-alpha (TNF-α), and IL-6 were detected by enzyme-linked immunosorbent assay. Commercial reagent kits were used to detect inducible nitric oxide synthase, superoxide dismutase (SOD), and malondialdehyde (MDA). The expression levels of nuclear factor erythroid-2-related factor 2 (Nrf2) and nuclear factor-kappa B (NF-κB) were detected by Western blot.
Results
DNP rat model was successfully constructed. SA and gabapentin could alleviate the thermal sting pain of DNP rats, increase the expression of MBP and SOD, and decrease the expression of proinflammatory agents and MDA. Moreover, SA could reverse the upregulation of NF-κB and the downregulation of Nrf2.
Conclusion
SA has a significant protective effect on DNP, manifesting in the improvement of allodynia and sciatic nerve pathology, inhibition of neuroinflammation, reduction of oxidative stress, enhancement of Nrf2, and inhibition of NF-κB.
Introduction
The pathological conditions of diabetic neuropathy include the accumulation of advanced glycation end-products, neuroinflammation, and cell apoptosis caused by overexpression of poly (ADP-ribose) polymerase (PARP), which can eventually lead to nerve damage and neuralgia. Besides, oxidative stress and inflammation are recognized as the central mechanisms in the pathogenesis of diabetic neuropathy (Vincent et al., 2004). The agents considered first-line analgesics, such as the antiepileptic drug gabapentin (Schmader, 2002), the anticonvulsant pregabalin, the antidepressant duloxetine, and opioids, are still in widespread clinical use. However, these drugs have their limitations and bring about not only many side effects but also unsatisfactory treatment outcomes (Rosenberg & Watson, 2015). Schefflera arboricola (SA) is a plant of the Araliaceae family and has long been used as a folk medicine for the treatment of pain, rheumatoid arthritis, fractures, sprains, and traumatic hemorrhage in the southern provinces of China (Song, 1999). The crude extract of SA has been clinically used for the treatment of sciatica, trigeminal neuralgia, neuropathic headache, gastrointestinal spasm, and so on (Wang et al., 2013; Xu et al., 2006). However, the treatment effects of SA on chronic neuralgia caused by diabetes have not been reported. In this paper, we explored whether SA exerts potent activity against allodynia in a rat model of streptozocin-induced diabetic neuropathy.
Materials and Methods
Animal
The experiments were performed on male Sprague–Dawley rats (Shanghai Shrek Experimental Animal Co., Ltd., China), weighing 200−250 g. A total of five animals were raised in each cage and had free access to food and water. The rodents were kept at a constant room temperature (25°C ± 2°C) with a 12-h light-dark cycle (light time 8:00−20:00 h). All experiments were conducted in accordance with international animal experimental regulations and approved by the Ethics Committee of the Second Affiliated Hospital of Fujian Medical University (Ethical Review No. 2021-504). All behavioral experiments were performed between 9:00 and 17:00 h. Each rat was only used in one experiment and was euthanized at the end of the study.
Drugs and Experimental Design
SA tablets were purchased from Quanzhou Zhongqiao (Group) Co., Ltd. Pharmaceutical Company. Gabapentin (98%, Sinopharm Chemical Reagent Co., Ltd., Sinopharm) was included in the behavioral experiments as a positive control. The SA tablets and gabapentin were predissolved in sterile normal saline. Thirty-nine rats were randomly divided into five groups (7−8 rats each). Body weight, blood glucose, and thermal withdrawal latency (TWL) in all groups were recorded. Before streptozocin (STZ, Sigma) injection (baseline), before administration, and 60 min after administration, the paw withdrawal delay time was measured to observe the pain behavior of the rats:
Control: After fasting for 12 h, rats were given a single intraperitoneal injection of vehicle (0.01 M citrate buffer, pH 4.5) at 60 mg/kg, measured with the tail vein blood glucose (Roche) 72 h after the injection, and given saline once a day for 7 consecutive days starting from the 22nd day after the injection of vehicle.
Model of diabetic neuropathic pain (DNP): After fasting for 12 h, rats were given a single intraperitoneal injection of 10% STZ at 60 mg/kg, measured with tail vein blood glucose 72 h after the injection. A blood glucose level greater than 16.7 mmol/L was judged to be successful diabetes induction. A threshold ratio less than 0.8 illustrated the successfully constructed animal model of DNP. The control group with oral gavage of saline followed the same protocol.
SA (100 and 300 mg/kg): Followed the same protocol as the model group with diabetes induction. Starting on the 22nd day after the injection of STZ, two groups were administered SA (100 mg/kg and 300 mg/kg) for 7 consecutive days. The doses of SA were selected from the literature (Sun et al., 2017). The doses of 100 mg/kg and 300 mg/kg are much lower than 5000 mg/kg, which did not cause any death or toxic signs in all rats after 14 consecutive days of treatment (Witthawaskul et al., 2003).
GP (100 mg/kg): Followed the same protocol as the model group with diabetes induction. Starting on the 22nd day after an injection of STZ, rats were injected intraperitoneally with gabapentin (100 mg/kg) for 7 consecutive days.
TWL Test
TWL was measured with the reference of our previous research method (Hargreaves et al., 1988), and the thermal pain threshold of rats was detected at 11 time points: before modeling, on the 7th, 14th, and 21st days after modeling, and 60 min after each dose. Before the test, the rats were placed in the transparent plastic observation box of the pain measuring instrument to adapt for 30 min. When the rat was quiet, the intensity of the heat source was set to 25%, and the stimulation heat source was moved to irradiate the hindlimb sole of the rat to measure the pain point. When the rat retracted the hind paw, the stimulation was automatically stopped, and the time TWL was recorded. TWL was measured three times with a 5-min time interval, and then the mean TWL was obtained.
Histological Evaluation of Light Microscopy
Rats were anesthetized by intraperitoneal injection of 50 mg/kg aldehyde hydrate, and the morphological evaluation was performed immediately after collecting blood from the abdominal aorta. Rats were perfused transcardially with normal saline followed by 4% paraformaldehyde in 0.1 M phosphate-buffered saline (pH 7.4). The sciatic nerve in the middle thigh was quickly resected and fixed in 4% paraformaldehyde, then dehydrated with gradient alcohol, embedded in paraffin and sliced (4 µm thick), repaired with citrate antigen repair solution (pH 6.0), blocked the activity of endogenous peroxidase with 3% hydrogen peroxide solution, incubated with a primary antibody of myelin basic protein (MBP) in 5% bovine serum albumin (BSA) at 4°C for overnight, followed by horseradish peroxidase (HRP)-labeled secondary antibodies incubation at room temperature for 50 min. Subsequently, diaminobenzidine was used for the chromogenic reaction, and hematoxylin was used for counterstaining. The morphology of the sciatic nerve was examined under a light microscope (CK31, OLYMPUS, Japan). The optical density of each image was scanned by Image-ProPlus 6.0 image processing software, and then the optical density value of each image was obtained for semi-quantitative analysis. Another portion of the sciatic nerve slices was processed independently, stained with toluidine blue, and observed under the light microscope. The axonal area was measured by ImageJ image processing software.
Western Blotting
After administration, the rats were quickly anesthetized with chloral hydrate, and the sciatic nerve tissue was collected and placed in the tissue lysis buffer containing protease inhibitors. After grinding, the insoluble particles were separated by centrifugation. The bicinchoninic acid assay protein detection kit was used to determine protein concentration. Approximately 20 µg of protein lysates from each sample were loaded into 12% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE) gel and subjected to separation. Then, proteins were transferred onto a polyvinylidene fluoride membrane (Millipore). Afterward, the membrane was blocked with 5% skim milk powder (w/v) for 1 h at room temperature, then incubated overnight with a primary anti-NF-κB or anti-Nrf2 antibodies at 4°C, and then incubated with an HRP-labeled secondary antibody at room temperature for 2 h. After washing the membrane, a chemiluminescence reaction was induced by enhanced chemiluminescence solution (Pierce), and then the samples were exposed to X-ray film for visualization using the Carestream molecular imaging system. Finally, the grayscale value of protein bands was then analyzed using ImageJ software.
Enzyme-Linked Immunosorbent Assay (ELISA)
After the last thermal stimulation pain test, the rat serum and sciatic nerve were collected immediately. Total IL-1β, tumor necrosis factor-alpha (TNF-α), and IL-6 in serum and sciatic nerve homogenate supernatant were measured with an ELISA kit (Rusted Systems, Minneapolis, MN, USA).
Detection of Oxidative Stress Markers and Proinflammatory Mediators
The parameters of oxidative stress were evaluated in tissue (sciatic nerve) homogenate. Superoxide dismutase (SOD), malondialdehyde (MDA), and inducible nitric oxide synthase (iNOS) were detected according to the commercial kit (Nanjing Jiancheng Bioengineering Institute).
Statistical Analysis
All data are represented by the mean ± SEM (standard error of measurement). Analysis of multiple groups was performed by two-way analysis of variance (ANOVA) or one-way ANOVA, followed by Bonferroni or Dunnett post hoc tests. If only two groups are compared, the Student’s t-test is used. A p-value < 0.05 is considered to be statistically significant.
Results
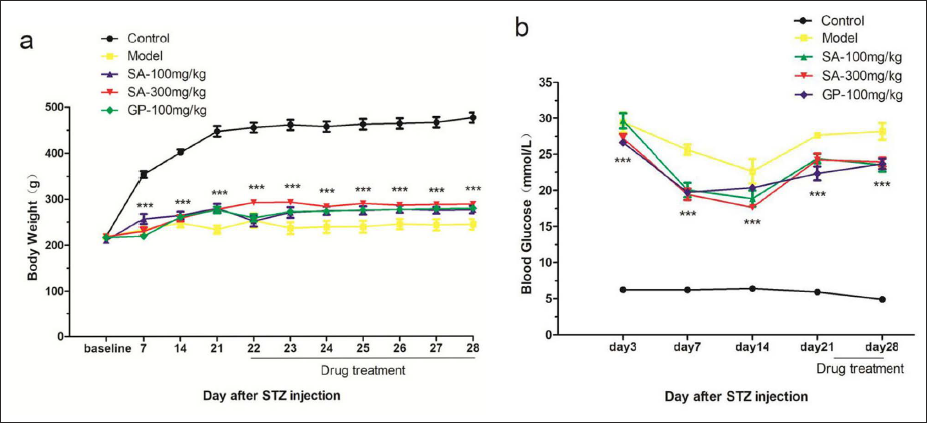
Effects of SA on Body Weight and Blood Glucose
First, we evaluated the effects of SA on body weight and blood glucose in rats. The body weight (Figure 1a) and blood glucose (Figure 1b) were not significantly changed in the treatment group.
Effect of SA on Thermal Hyperalgesia
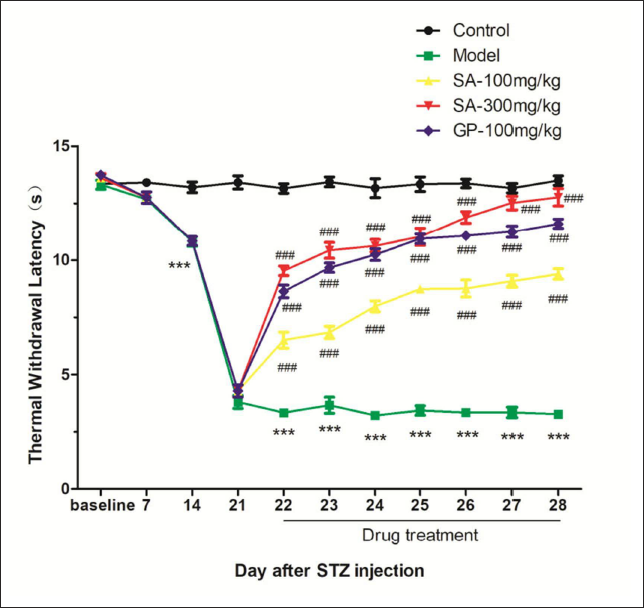
The diabetic models induced by STZ were used to evaluate the effect of SA on DNP. TWL was measured before STZ injection (baseline), 7, 14, and 21 days after STZ injection, and 1 h after each dose (22−28 days after STZ injection). As shown in Figure 2, the TWL decreased significantly (p < 0.001 vs. the normal control group), and thermal hyperalgesia lasted for the whole observation period (Figure 2). After the first dose administered (22nd day), SA (100 mg/kg, 300 mg/kg) and gabapentin (100 mg/kg) significantly reversed the thermal hyperalgesia (p < 0.001 vs. saline; Figure 2). On the 6th and 7th days, the TWL of the 300 mg/kg SA group was significantly higher than that of gabapentin at 100 mg/kg as a positive control (p < 0.05).
Effect of SA on The Morphology of Sciatic Nerve
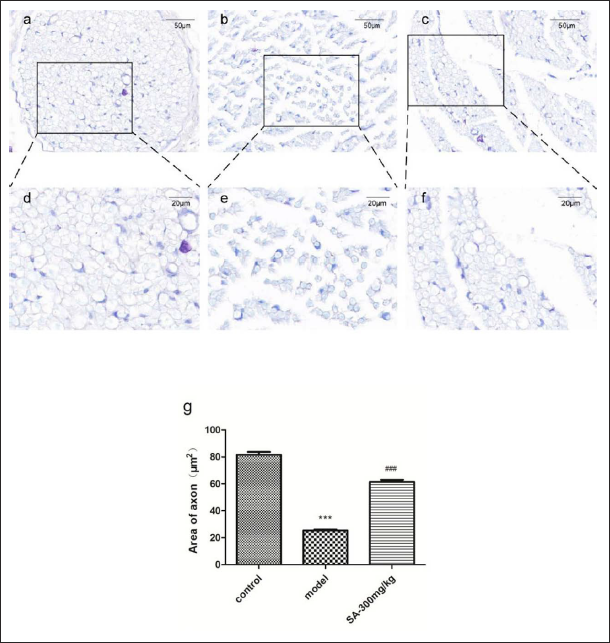
In order to directly evaluate the effect of SA on the morphology of the sciatic nerve, the sciatic nerves of three rats in each group were randomly selected for immunohistochemical experiments with toluidine blue staining. Three images were randomly collected from each specimen, and the morphological structure of the sciatic nerve was photographed under a light microscope (Figure 3). Twenty myelinated nerve fibers were randomly selected from each image. The axonal area of myelinated nerve fibers in each group was measured by ImageJ image processing software, and the axonal area was compared quantitatively. The black box area (Figure 3a, d) showed that the nerve fibers in the control group were plump, clear, and large in size, and there was almost no dense substance. Compared with the control group, the nerve fibers in the model group (Figure 3b, e) were relatively sparse, dense substance appeared in some axons, and the axonal area became smaller (p < 0.001). In the 300 mg/kg treatment group (Figure 3c, f), most of the nerve fibers were fuller, only a few dense substances existed in the axons, and the axonal area became larger (p < 0.001 vs. model group).
SA Decreased the Level of MBP in Sciatic Nerve
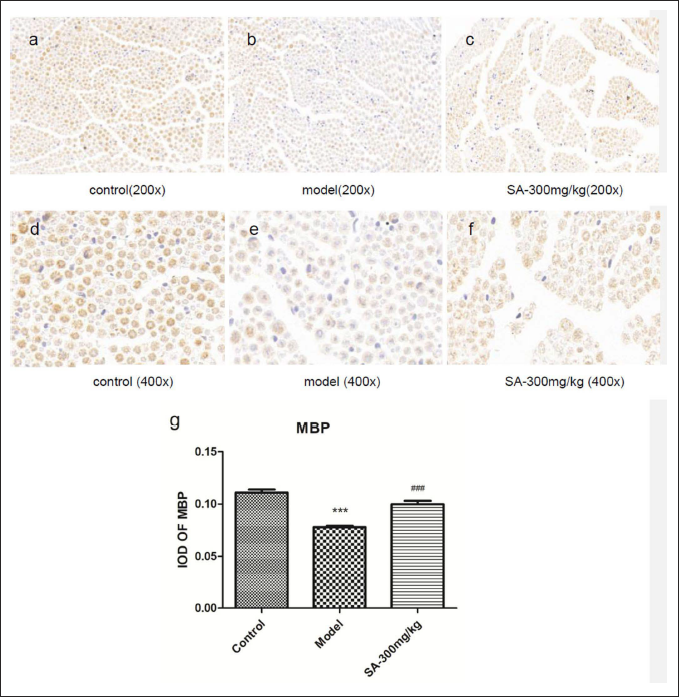
Five rats in each group were randomly selected for the immunohistochemical experiment. Three images were randomly collected from each specimen, and the structure of the sciatic nerve was photographed under a light microscope. The optical density of each image was scanned by Image-ProPlus 6.0 image processing software, and MBP was semi-quantitatively analyzed. The myelin sheath structure of a single nerve fiber in the normal control group could be seen at high magnification (400×); the color and thickness of the myelin sheath were uniform; the MBP positive expression area was brown and dark; and the nerve fibers were plump (Figure 4a, d). Compared with the control group, the staining color of the model group was obviously lighter, the outline of most of the myelin sheath was less clear (Figure 4b, e), the optical density (Figure 4g) decreased significantly (p < 0.001), and the positive expression of MBP was weaker. Compared with the model group, the color of myelin sheath in the 300 mg/kg treatment group became noticeably darker (Figure 4c, f), the optical density of MBP (Figure 4g) increased significantly (p < 0.001), and the expression of MBP was upregulated. Our results indicated that SA could up-regulate the expression of MBP in the sciatic nerve of DNP rats.
SA Attenuates Proinflammatory Mediators in Serum and Sciatic Nerve
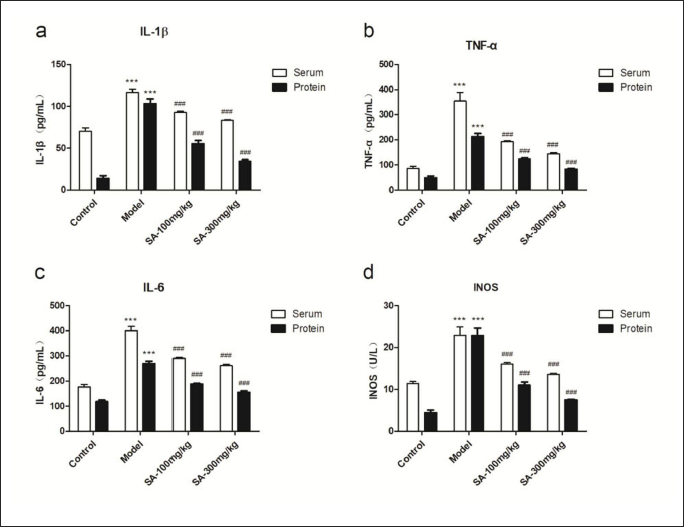
Compared with the normal control group, the levels of total IL-1β, TNF-α, IL-6, and iNOS in the supernatant of sciatic nerve homogenate and serum from DNP rats were significantly increased on the 28th day. Both low and high doses of SA could significantly decrease the levels of IL-1β, TNF-α, IL-6, and iNOS, either in the sciatic nerve or in the serum (Figure 5).
SA Relieved Oxidative Stress
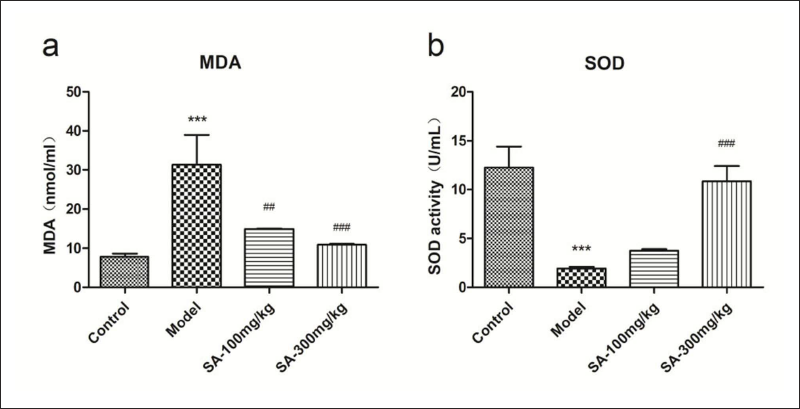
Compared with the normal control group, the levels of MDA in the supernatant of sciatic nerve homogenate from DNP rats were significantly increased on the 28th day (p < 0.001). Both low and high doses of SA could reduce the MDA level (Figure 6a). The activity of SOD decreased significantly in DNP rats, while only the high dose (300 mg/kg) of SA could significantly increase the activity of SOD (p < 0.001) (Figure 6b).
Effects of SA on NF-κB and Nrf2 of Sciatic Nerve
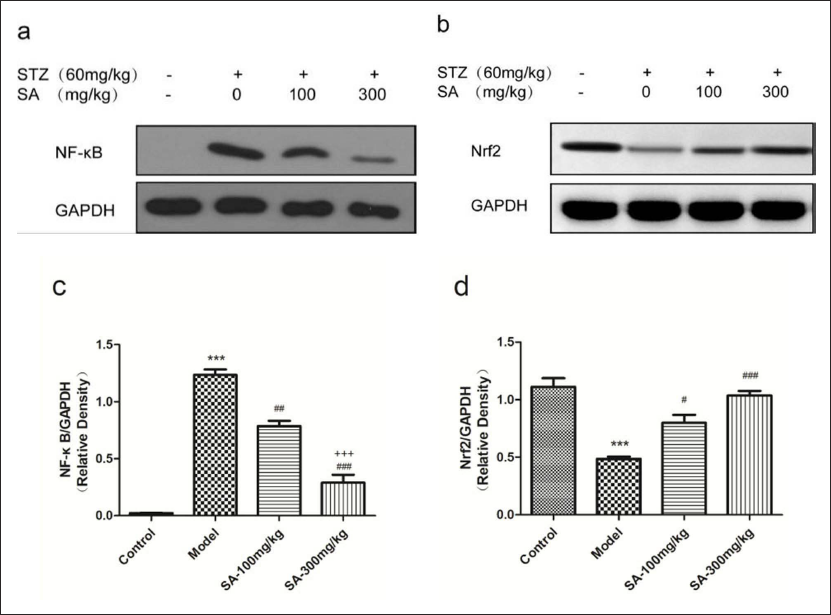
Diabetic neuropathy is a disease with an altered expression pattern of Nrf2 and NF-κB. The data showed that the expression of NF-κB in the DNP model group was increased and was significantly higher than that in the normal control group (Figure 7a, c) (p < 0.001). After treatment with SA (100 mg/kg or 300 mg/kg) for 7 days, the expression levels of NF-κB were decreased significantly and in a dose-dependent manner (Figure 7b). The central regulator of the antioxidant stress response is the Nrf2 signal pathway, and Nrf2 can downregulate the expression of NF-κB. The results showed that the expression levels of Nrf2 were decreased in DNP rats (Figure 7b, d) (p < 0.05), while both doses (100 mg/kg and 300 mg/kg) of SA could enhance the expression of Nrf2 (Figure 7c).
Discussion
The therapeutic effect of diabetic neuropathy is still limited due to the complex pathogenesis of diabetic peripheral neuropathy. Medicinal plants and their active ingredients are increasingly used to treat diabetes and its complications (Elbanna et al., 2017; Salimifar et al., 2013; Smith & Clinard, 2014). SA prolonged the pain threshold in DNP rats and alleviated neuropathic pain in diabetic rats. The effect of SA on tactile sensation was dose-dependent, with the earliest appearance after the first gavage. After continuous intragastric administration, the efficacy of the high-dose group was higher than that of gabapentin on the 6th and 7th days. The intragastric administration is close to the clinical administration route of SA. In addition, the trend of the thermal pain threshold curve of the SA treatment group showed that high efficacy could be achieved with sequential administration of SA at different doses. In a previous acute toxicity test, the SA aqueous extract at a dose of 5000 mg/kg did not cause death in all rats after 14 consecutive days of treatment, and the rats did not show any sign of toxicity or changes in general behavior or other physiological activities (Witthawaskul et al., 2003). In our study, no obvious toxic effects could be observed during the treatment period, and the blood glucose and body weight of rats were not significantly affected. These findings might promote a novel insight into the treatment of peripheral neuropathy with SA and its extracts.
Apart from the influence on behavior, the histological examination of the sciatic nerve showed that SA could also attenuate nerve injury in DNP rats. The main changes in neuronal morphology caused by DNP were the thickening of the basement membrane, decrease of myelinated fibers with active axonal degeneration, loss and cavitation in the axons of unmyelinated nerve fibers, and so on (Qiu et al., 2003). The results of toluidine blue staining showed that the gap of nerve fibers in DNP rats was enlarged, the outline of the myelin sheath was unclear, dense substances in some axons were observed, and the axonal area was smaller than that of the normal control. Peripheral MBP plays an important role in myelination and maintaining the correct structure of myelin. When the peripheral nerve is damaged, the structure and function of the myelin sheath of myelinated nerve fibers are changed, which makes the expression of protective MBP abnormal (Brettschneider et al., 2015; Shi et al., 2013). Therefore, we used the immunohistochemical method to semi-quantitatively analyze the MBP of the sciatic nerve to evaluate the protective effect of SA on the myelin sheath, and the results showed that the expression of MBP in the myelin sheath was weaker. The decrease in pain threshold, the downregulation of expression of MBP, and the morphological changes in the sciatic nerve suggested that the myelin sheath and axon of the sciatic nerve in DNP rats had been damaged. After the treatment with SA, the nerve structure was improved; most of the nerve fibers were plump; the gap between nerve fibers was narrowed; the axonal area was enlarged; dense substances were observed in only a few axons; and the expression of MBP was enhanced, indicating that SA can have a certain protective effect on the myelin sheath and axons.
Due to the fact that SA cannot restore blood glucose in DNP rats, its anti-neuropathic effects seem to be independent of blood glucose control. Oxidative stress is a key pathological process of diabetes-induced nerve injury, which can eventually lead to vascular dysfunction and subsequent intimal hypoxia-induced sensory (thermal hyperalgesia) neurological impairment (Kasznicki et al., 2012; Sandireddy et al., 2014). A large number of studies have reported that SA exerts anti-inflammatory and antioxidant effects in multiple different animal models and can reduce the levels of oxidative stress indexes and proinflammatory cytokines (El-Hagrassi et al., 2022; Zhang et al., 2019). Therefore, we first discuss the influence of SA on the oxidative stress indexes. Our results showed that the MDA of lipid peroxidation in the model group increased significantly while the SOD decreased. After treatment with SA, the MDA intensity of lipid peroxidation decreased significantly, while the activity of the antioxidant enzyme SOD increased significantly. These findings all suggest that SA may be involved in the inhibition of lipid peroxidation.
Constantly high glucose is considered to be the basis of the inflammatory response and nerve injury that give rise to DNP. All characteristic classical pathways, such as the polyol pathway and the protein kinase C (PKC) pathway, can directly or indirectly induce a proinflammatory response (Singh et al., 2014). The central mechanism of the inflammatory response to DNP is focused on the cascade activation of NF-κB (Cameron & Cotter, 2008). The activated NF-κB can transfer into the nucleus and initiate the expression of proinflammatory cytokines TNF-α, IL-1β, and IL-6 (Gutierrez & Hoyo-Vadillo, 2017; Williams & Nadler, 2007), inflammation-related protein iNOS, and cyclooxygenase (Cameron & Cotter, 2008). In turn, these cytokines are inducers of NF-κB. The two are interrelated and mutually potentiating, eventually resulting in progressing neuroinflammation and oxidative stress (Karin et al., 2004; Tan et al., 201). Compared with the normal control group, the levels of TNF-α, IL-6, and iNOS in serum and sciatic nerve tissue of the model group were higher (Figure 5). High levels of inflammatory factors and iNOS are risk factors for diabetic neuropathy (Purwata, 2011; Satoh et al., 2003). Blocking the production or inhibiting the activity of TNF-α can delay or halt the development and aggravation of diabetic complications (Zhang et al., 2015). After treatment with SA, the levels of TNF-α, IL-1β, IL-6, and iNOS in serum and sciatic nerve tissue of DNP rats decreased, suggesting that SA may be involved in anti-inflammatory effects on neuropathic pain. However, this study is just a preliminary observation of the influence of SA on the inflammatory response. Rescue experiments will be conducted to further verify our results in our future studies.
Oxidative stress and neuroinflammation do not play separate roles in the development of DNP. Excessive oxidative stress caused by persistent hyperglycemia can result in the activation of Nrf2 and NF-κB, which are sensitive to redox environments. The imbalance of the Nrf2-NF-κB axis manifesting as continuous upregulation of NF-κB and downregulation of Nrf2 can promote the aggravation of neuroinflammation and oxidative stress, further leading to peripheral nerve fiber damage, a decrease in nerve blood perfusion, and the release of pain-producing substances such as bradykinin and prostacyclin (Yerra et al., 2013). Pharmacological modulators regulating two transcription factors at the same time can alleviate the therapeutic deficit of diabetic neuropathy by inhibiting the NF-κB signaling pathway while activating the Nrf2 signaling pathway (Kumar & Sharma, 2010; Negi et al., 2011a, b). What is worse, the pharmacologic agents, only targeting Nrf2 or NF-κB or possessing antioxidant or anti-inflammatory activity, were not very efficient in treating DNP (Yerra et al., 2013). In our study, the decrease of Nrf2 and the increase of NF-κB appeared in the model group on the 28th day after injection of STZ, while the NF-κB could be effectively decreased and Nrf2 could be upregulated by SA in the sciatic nerve of diabetic rats (Figure 7). However, whether the beneficial effect of SA on diabetic neuropathy is attributed to its balance on the Nrf2-NF-κB axis remains to be further studied.
Conclusion
In our study, SA showed a significant protective effect on DNP, manifesting in the improvement of allodynia and sciatic nerve pathology, inhibition of neuroinflammatory processes, and reduction of oxidative stress. Meanwhile, SA did not affect blood glucose or body weight. Further, SA could enhance Nrf2 and inhibit NF-κB. In conclusion, SA has therapeutic potential for DNP, which requires more in-depth investigations, especially more evidence from clinical observation.
Abbreviations
DNP: diabetic neuropathic pain; SA: Schefflera arboricola; MBP: myelin basic protein; MDA: malondialdehyde; SOD: superoxide dismutase; NF-κB: nuclear factor kappa-B; Nrf2: nuclear factor erythroid-2-related factor 2; IL-1β: interleukin-1β; TNF-α: tumor necrosis factor-α; IL-6: interleukin-6; iNOS: inducible nitric oxide synthase; STZ: streptozocin; TWL: thermal withdrawal latency.
Footnotes
Acknowledgment
The authors would like to thank all the supporting staff in the Department of Pharmacy of Quanzhou Medical College, the Pharmaceutical Department of the Second Affiliated Hospital of Fujian Medical University, the Pharmaceutical Department of Quanzhou First Hospital Affiliated to Fujian Medical University, and the Quanzhou Hospital of Traditional Chinese Medicine for their technical assistance in this research project.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Statement of Ethical Approval and Informed Consent
This animal study protocol was approved by the Ethics Committee of the Second Affiliated Hospital of Fujian Medical University (Ethical Review No. 2021-504).
Funding
Quanzhou Medical College research project, Quanzhou Medical College (Grant number: XJK1920B); Startup Fund for scientific research, Fujian Medical University (Grant number: 2020QH1105); Fujian educational and scientific research project for young and middle-aged teachers (science and technology), education of Fujian Province (Grant number: JAT220721).
Summary
SA treatment of DNP rats decreased neuropathic pain behavior. Only SA at a 300 mg/kg concentration was more effective than gabapentin (100 mg/kg) after 6 days of continuous administration.
SA significantly reduced the injury of sciatic nerve axons and myelin sheaths in DNP rats, increased MBP, and did not affect body weight or blood glucose.
SA significantly inhibited inflammation and antioxidant stress, reduced proinflammatory mediators and MDA, and increased SOD.
SA reversed the upregulation of NF-κB and downregulation of Nrf2 in the sciatic nerve of DNP rats.
