Abstract
Background
Non-alcoholic fatty liver disease (NAFLD) is severely affecting the quality of people’s life. Rhapontigenin (RA) is a stilbene compound isolated from Rhubarb L., which has been reported to have an effect on cholesterol diet-induced hyperlipidemia in rats. This study aims to explore the pharmacodynamics and the mechanism of RA obtained from Rheum franzenbachii Munt. against NAFLD. RA was extracted from the roots of the Rheum L. (Polygonaceae) plant Rheum franzenbachii Munt.
Materials and Methods
RA was extracted by Sephadex-gel column and identified by high-performance liquid chromatography (HPLC)-UV and HR-ESI-MS. The pharmacodynamic indexes of L-02 cells and mice treated with RA were determined by histological staining and ELISA, while the expression of autophagy-related proteins was analyzed by Western blot.
Results
The results in vivo showed that the liver structure of RA-treatment mice was normal, and the organ coefficient was significantly decreased. It also showed that RA could significantly reduce the expression of reactive oxygen species (ROS) in the liver as well as inhibit oxidative stress and inflammatory response. Interestingly, the autophagy inhibitor 3-methyladenine (3-MA) could reverse the effect of RA on NAFLD, which further confirmed that RA plays an anti-NAFLD role through activating lipophagy.
Conclusion
It suggested that RA has an effect on NAFLD by down-regulating the expression of Acyl-CoA oxidase 1 (ACOX1), p-mTOR, and p-Unc-51-like kinase 1 (ULK1), then inhibiting Acetyl-CoA (A-CoA) production, and up-regulating the expression of autophagy protein 5 (Atg5) to promote the lipophagy of lipocytes.
Introduction
Non-alcoholic fatty liver disease (NAFLD) refers to a pathological disease of the liver arising from a combination of factors other than alcohol and other factors that damage the liver, characterized mainly by the accumulation and deposition of excessive fat in the hepatocytes (Chalasani et al., 2018). NAFLD includes non-alcoholic steatohepatitis (NASH), simple fatty liver (SFL), and related cirrhotic diseases. Despite the high prevalence of NAFLD, natural therapies available for NAFLD remain limited. Therefore, it is necessary to discover and study effective drugs for the treatment of NAFLD.
Autophagy is a way in which cells consume and degrade substances in cells to maintain homeostasis, including lipid metabolism (Evans et al., 2017). An increasing amount of evidence suggests an important relationship between autophagy and lipid metabolism, and the pathogenesis of NAFLD is intrinsically linked to autophagy disorder (Zhang et al., 2018; Zhang et al., 2020). Lipophagy means that triglyceride (TG) and total cholesterol (TC) in lipid droplets (LDs) are degraded to free fatty acids (FFAs) by lysosomal hydrolases, and these FFAs are recirculated back into mitochondrial β-oxidation (Cingolani & Czaja, 2016). Phosphatidylethanolamine within autophagy-related gene (Atg) interacts with Unc-51-like kinase 1 (ULK1) and microtubule-associated protein-light 3 to induce lipophagy, leading to the release of FFAs (Foerster et al., 2022). Acyl-CoA oxidase 1 (ACOX1) is the crucial enzyme in the first step of peroxisome β-oxidation; it can increase excess acyl CoA metabolism in cells (Van Veldhoven, 2010). Studies have shown that activation of lipophagy in liver-specific Acox1 knockout (ACOX1-LKO) mice improved fatty degeneration induced by high fat diet (HFD) (He et al., 2020).
Rhapontigenin (RA) (CAS: 500-65-2, PubChem CID: 5320954) is a stilbene compound that could be extracted from most of Rheum L. (Polygonaceae). Rhapontin (CAS: 155-58-8, PubChem CID: 637213) exists in a variety of herbs, including Rheum L., with strong activity and extensive pharmacological effects. Modern pharmacology has shown that Rhapontin could be used to treat osteoporosis (Yang et al., 2021), diabetes, and its complications (Chen et al., 2009). In addition, Rhapontin can effectively inhibit hepatic steatosis (Chen et al., 2009), which has a positive effect on liver protection. Studies have shown that RA can dose-dependently improve the pathological characteristics of degenerative fatty liver in hyperlipidemia rats induced by high cholesterol diet (Jo et al., 2014). However, there are not any reports that RA can effectively treat NAFLD.
In this study, RA was extracted from Rheum franzenbachii Munt. by Sephadex-gel column and its concentration was identified. And then, in vivo and in vitro experiments were used to study the pharmacodynamics and mechanism of RA in the treatment of NAFLD, aiming to provide further selection and a theoretical basis for clinical medication.
Materials and Methods
Chemicals and Reagents
Rheum franzenbachii Munt. was collected from Bozhou Yuelin Pharmaceutical Co. Ltd. The RA (PubChem CID: 5320954; CAS: 500-65-2) reference (PCS1229) was purchased from Chengdu Purechem-Standard Co. Ltd (Chengdu, China). The petroleum ether, ethyl acetate and n-butanol were purchased from Jining Baiyi Chemical Co. Ltd (Jining, China). The 95% ethanol was purchased from Jining Baiyi Chemical Co. Ltd. (Jining, China). The chloroform and methanol were purchased from China National Pharmaceutical Group Chemical Reagent Co. Ltd. The acetonitrile was purchased from Tiandi Co. Ltd. in the United States (Shanghai, China). Thin-layer chromatography (TLC) and column chromatography silica gel were purchased from Qingdao Ocean Chemical Co. Ltd. (Qingdao, China). 3-methyladenine (3-MA, M833793), oleic acid (D875790) and palmitic acid (P815428) were purchased from Macklin (Shanghai, China). Afatating calcium tablets (ACT, H20051407) were purchased from Pfizer Inc. (New York, USA). Zhi-bi-tai capsules (ZBT, Z51022196) were purchased from DIAO Pharmaceutical Group (Chengdu, China). The Hematoxylin and Eosin staining (H&E, PH0516) and Oil red O (PH1738) dye kits were purchased from PHYGENE Biotechnology Co. Ltd (Wuhan, China). TC (A111-1-1), TG (A110-1-1), aspartate aminotransferase (AST) (C010-3-1), alanine aminotransferase (ALT) (C009-3-1), glutathione (GSH) (A005-1-2), malondialdehyde (MDA) (A003-1-2), and superoxide dismutase (SOD) (A001-3-2) were obtained from Jiancheng Biotechnology Co. Ltd. (Nanjing, China). The RIPA lysis buffer (P0013B) and 4′,6-diamidino-2phenylindole (DAPI, C1002) were purchased from Beyotime (Shanghai, China). The BCA protein concentration assay kit (BL521A) was purchased from Biosharp (Hefei, China). The acetyl-CoA (A-CoA) (RX101413H), interleukin-6 (IL-6) (RX203049M), and tumor necrosis factor-α (TNF-α) (RX202412M) ELISA kits were obtained from Quanzhou Ruixin Biological Technology Co. Ltd. (Quanzhou, China,
Extraction and Isolation of RA
The dry roots of R. franzenbachii Munt. are infiltrated with ethanol. Then, extract the matrix with Petroleum ether (v/v), ethyl acetate (v/v), and n-butanol (v/v) in turn to obtain the dry extract. The ethyl acetate portion was separated through a silica gel column. Through 1260 high-performance liquid chromatography (HPLC) and TLC, further separation and purification on the gel column, the purity of the compound was identified by 1260 HPLC. Determination of molecular weight of compounds by mass spectrometry (Yao et al., 2021). (The specific process of extraction, separation, and identification were included in the supplementary document for reference.)
Effect of RA on L-02 Cell Activity
L-02 cells (obtained from the Cell Bank of the Chinese Academy of Sciences) in the logarithmic growth phase were collected, the concentration of the cell suspension was adjusted to 1 × 105 cells/mL, and then, the cells were spread in the 96-well plates. The L-02 cells were divided into solvent blank group, cell control group, and RA treatment group with different concentrations (set as follows: 12.5, 25, 50, and 100 µM RA). After the cell adherence, the culture medium was replaced and continued to cultures. After 24 h, an MTT assay was used to detect the activity of L-02 cells (Li et al., 2018;Ye et al., 2019).
Detection of TC, TG, and the Content of Lipid
L-02 cells in the logarithmic growth phase were collected, the concentration of the cell suspension was adjusted to 5 × 104 cells/mL, and then, the cells were spread in the 6-well plates. After the cell adherence, the media containing 0.3 mM FFA (oleic acid: palmitic acid = 2:1) was placed on the 6-well plates for 24 h, then the RA treatment groups were treated with 12.5, 25, and 50 µM RA.
After 24 h of RA treatment, the culture media was discarded, 200 µL RIPA lysis buffer was added to dissolve the cells in each well, and then, the cell lysate was collected and placed in an ice bath for 15–30 min, and the contents of TC and TG were determined according to the instructions of kit.
After 24 h of RA treatment, the culture media was discarded, and the cells were fixed with 4% paraformaldehyde for 15 min, then washed with phosphate buffered saline (PBS) three times. The oil red O working fluid was added for staining at 37°C for 1.5 h, then differentiated with 75% alcohol for 2 s and washed for 1 min. After 2 min of re-staining with Harris hematoxylin, the sample was washed with tap water, and then, 1% hydrochloric acid alcohol was added for 5 s. Following that, the sample was washed with tap water, ammonia water turned back to blue, and washed with running water. The cells were placed under a light microscope (Olympus IX51, Pennsylvania), and then, the images were captured and analyzed (Zhang et al., 2020 Zhou et al., 2014).
Influence of 3-MA on the Efficacy of RA
3-MA, an autophagy inhibitor, was used to verify whether RA plays an anti-NAFLD role by activating autophagy. The groups were established as the following: control group, model group (FFA), treatment group (FFA + 50 µM RA), and 3-MA group (FFA + 50 µM RA + 5 mM 3-MA). FFA was applied to the cells for 24 h. Culture media containing 50 µM RA only was added to the treatment group, while culture media containing both 50 µM RA and 5 mM 3-MA to the 3-MA group (Yan et al., 2022). After 24 h, the cells were lysed, and the lysate was used to determine the contents of TC and TG (Zhou et al., 2014).
Content of A-CoA and Western Blot Analysis
The content of autophagy regulator A-CoA in the cells was determined according to the instructions of the ELISA. The protein concentration of each group above was adjusted to the same, and then, an appropriate amount of 5 × Loading was added, boiled the sample for 10 min. The protein samples were segregated using 8−10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis (SDS-PAGE gel); then, they were transferred to a NC filter membrane. After blocking, use 5% skim milk for 2 h. Following blocking, the membrane was incubated with the primary antibody at 4°C overnight, and then, incubated with the anti-rabbit secondary antibody for 2 h. The protein bands were imaged using an Enhanced Chemiluminescence (ECL reagent) and photographed using a Fluor Chem FC3 gel imager (Protein Simple, California) (He et al., 2020; Mariño et al., 2014; Son et al., 2019).
Animal Treatment
Specific pathogen Free (SPF) male C57BL/6 mice (20 ± 5 g) were provided by the Experimental Animal Center of the Three Gorges University (Yichang, China), with license number: SCXK (E) 2017-0012. Mice were kept at a temperature of 24 ± 2°C, a humidity of 50 ± 5%, and 12 h light/dark cycles. All the mice were provided with unlimited water and food. All the animal protocols were approved by the Experimental Animal Ethics Committee of the Hubei University of Chinese Medicine (approval number: 00291056). This study was carried out in accordance with the recommendations of the Animal Care and Use Committee of the Institute of Materia Medica, China. All the methods were reported in accordance with ARRIVE guidelines for the reporting of animal experiments.
Further, 90 mice were equally divided into nine groups, the treatment was as follows: the control group mice were given a basal diet, the RA group mice were given a basal diet and 8 mg/kg RA daily by gavage, the HFD group mice were given a HFD, the RA + HFD group mice were given at different doses of 2, 4, and 8 mg/kg RA daily by gavage (according to the literature), the positive drug groups were given 5 mg/kg ACT daily (according to the dose of an adult daily) or 60 mg/kg ZBT daily (according to the dose of an adult daily) by gavage, and RA + 3-MA + HFD group was given 8 mg/kg RA daily by gavage, with 30 mg/kg 3-MA (Bi et al., 2023; Chen et al., 2023) dissolved in physiological saline and injected intraperitoneally three times a week. All the other groups were fed a HFD for 8 weeks except the control and RA groups. The mice were given medicine for 4 weeks and sacrificed by 2% pentobarbital sodium after 12 h from the final administration. Blood was collected and centrifuged, the serum and liver tissues were stored at −80℃.
Histopathological Examination
The liver tissues were sectioned after fixation and embedding. The slices were first dehydrated before being stained with Oil red O solution and then differentiated with 1% hydrochloric acid alcohol. Following that, the sections were stained with hematoxylin for 1−3 min. Finally, the slices were treated with alcohol and xylene until they were transparent and then sealed with neutral gum. These samples were reviewed by a light microscope and photographed for analysis (Li et al., 2021 Zheng et al., 2018).
Determination of ROS in Frozen Sections
The frozen liver tissue sections were incubated in the ROS dye for 30 min, then decolorized 2−3 times in PBS, and finally incubated in the DAPI dye for 10 min. After that, washed the dye away, and the tablets were sealed with an anti-fluorescence quenching sealing agent. The sections were observed and photographed under a fluorescent microscope for analysis (Yasuda et al., 2017; Zhang et al., 2007).
Determination of Biochemical and Inflammatory Factors
The liver tissues were mixed with physiological saline at 1:9, and the supernatant was obtained by centrifugation (4000 rpm, 4℃) after homogenization. The contents of TC, TG, GSH, MDA, and SOD in the supernatant and ALT and AST in serum were determined by kits. The expression of IL-6 and TNF-α in the supernatant was detected by ELISA.
Results
Results of the MTT Assay
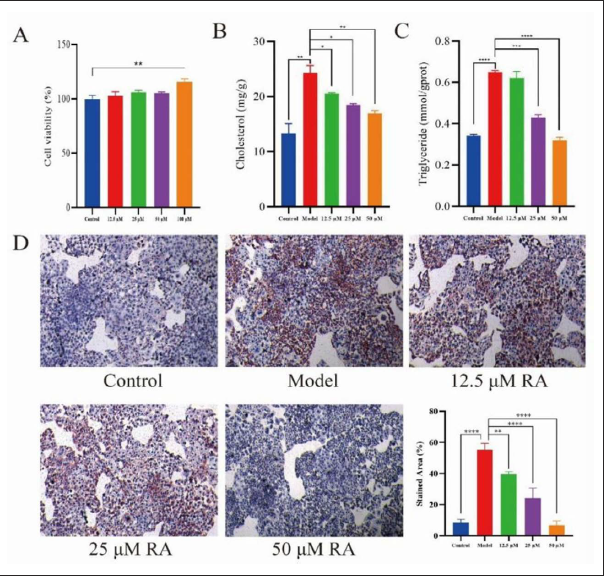
Figure 1A showed that when the concentration of RA was 12.5 to 50 µM, and the cells were not significantly affected; however, when the concentration of RA was 100 µM, it had a significant effect on L-02 cells. Therefore, the concentration of 12.5, 25, and 50 µM RA was selected for cell experiments.
Effect of RA on the Content of Lipid
The results showed that compared to the control group, the contents of TC and TG in the model group were significantly increased (p < 0.01), as shown in Figure 1B and C, indicating that FFA successfully induced the model in vitro. In the RA treatment groups, the contents of TC and TG were lower than those in the model group in a dose-dependent manner (p < 0.05). Figure 1D showed that FFA induced a large amount of lipid accumulation in L-02 cells, and in the RA treatment group, the number of red LDs was significantly reduced in a dose-dependent manner.
Influence of 3-MA on the Efficacy of RA
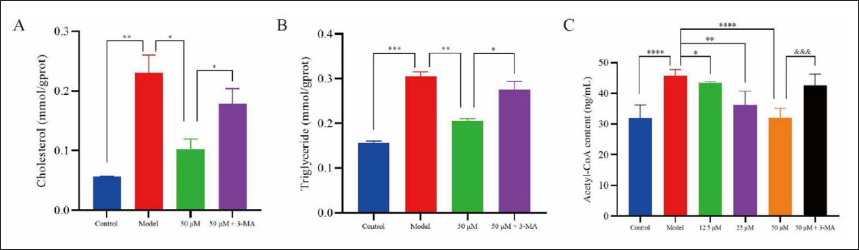
Figure 2A and B showed that the contents of TC and TG in the L-02 cells were significantly decreased in the RA treatment groups (p < 0.05). Compared with the RA treatment group, the contents of TC and TG were significantly higher in the 3-MA group (p < 0.05), which showed that the autophagy inhibitor 3-MA inhibited the action of RA on the lipid accumulation of L-02 cells.
Content of A-CoA
ELISA was used to determine the content of autophagy regulator A-CoA, and the results were shown in Figure 2C. It showed that the content of A-CoA in the model group was significantly higher than that in the control group (p < 0.001), and the content of A-CoA in RA treatment groups was significantly lower than that in the model group with a dose-dependent manner (p < 0.05), while the addition of 3-MA reversed the effect of RA (p < 0.005).
Effect of RA on ACOX1 and Autophagy-related Proteins
The expression of ACOX1, p-mTOR, p-ULK1, and Atg5 was analyzed by Western Blot, as shown in Figure 3. The results showed that the expression of ACOX1, p-mTOR, and p-ULK1 was significantly increased (p < 0.005), while the expression of Atg5 was significantly decreased (p < 0.05). After the cells were treated with RA, the expression of ACOX1, p-mTOR, and p-ULK1 was down-regulated in a dose-dependent manner(p < 0.05), while the expression of Atg5 protein was significantly up-regulated in a dose-dependent manner (p < 0.05). However, the addition of 3-MA significantly reversed the effect of RA on the expression of ACOX1 and Atg5 (p < 0.01).
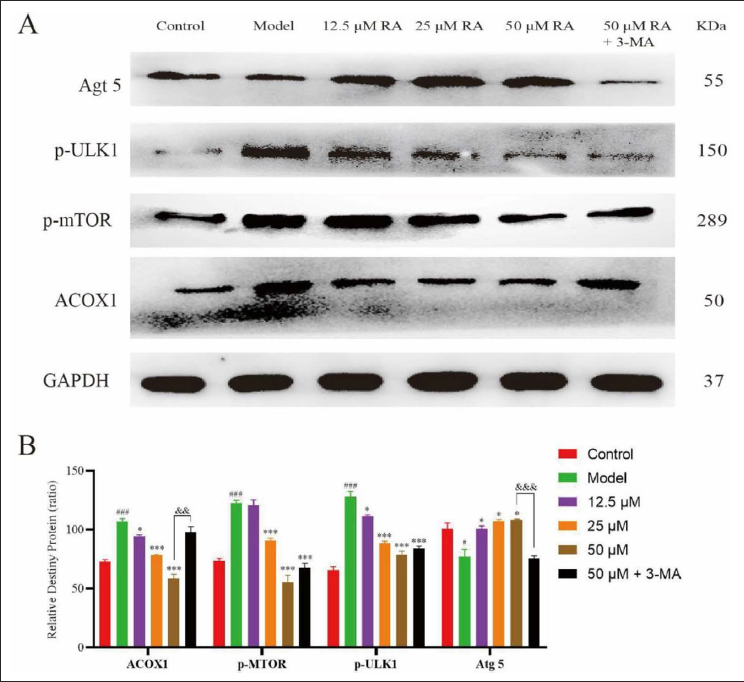
The results of Western Blot suggested that RA might improve NAFLD via regulating the ACOX1 and mTOR/ULK1 pathways.
Effect of RA on Liver Appearance and Organ Coefficient
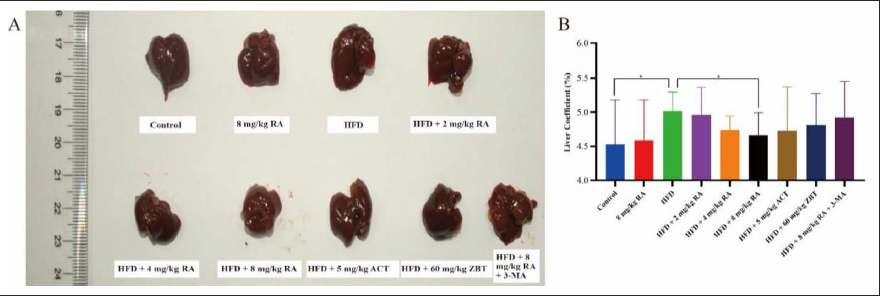
Figure 4A showed that in the control group, the liver was light red to red, while the liver capsule was complete, smooth, and elastic. Furthermore, the liver edge was sharp and there was no greasy feeling in the section. In the HFD group, it was obvious that the liver was swollen, with a dark color. In addition, the liver capsule was tight, the surface was rough and lusterless, the liver edge was blunted, and there was a greasy feeling in the section. Meanwhile, the symptoms of liver injury in RA treatment groups were improved and the livers had swollen in a dose-dependent manner. The liver surface was smooth and elastic, the liver edge was sharp, and the section was not greasy. Moreover, due to the long-term HFD, the organ index of HFD group mice was higher (p < 0.05), indicating that the mice in the HFD group developed NAFLD, as shown in Figure 4B. However, the organ coefficient of mice was improved in the 8 mg/kg RA treatment group.
Effect of RA on Lipid and Pathological Tissues
The lipid was stained by oil red O, as shown in Figure 5. Compared with the control group, red fat droplets stained by oil red O was observed in the liver tissues of the HFD group. In the RA treatment groups, the red fat droplets in the mice liver tissues gradually decreased in a dose-dependent manner, which demonstrated the lipid-lowering effect of RA. The lipid-lowering effect of RA was similar to the ACT group but better than the ZBT group. Compared with the RA treatment groups, the number of red fat drops in the liver tissues of the 3-MA group was higher.
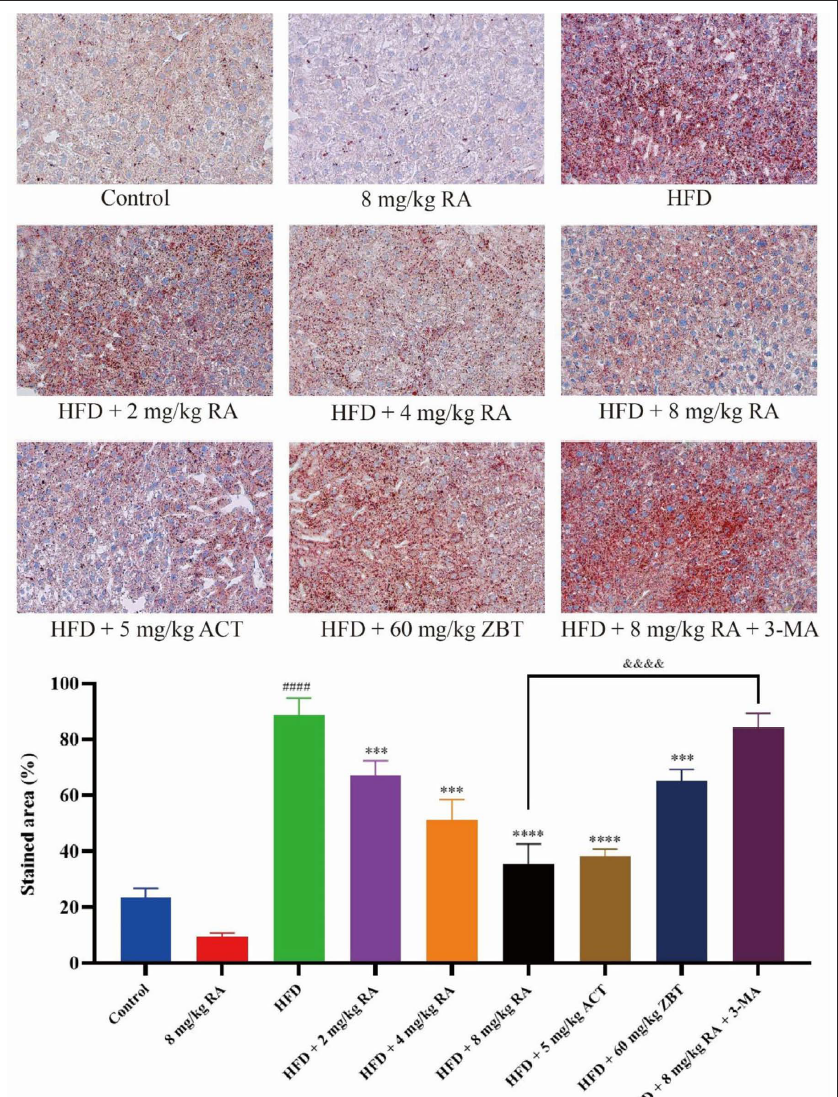
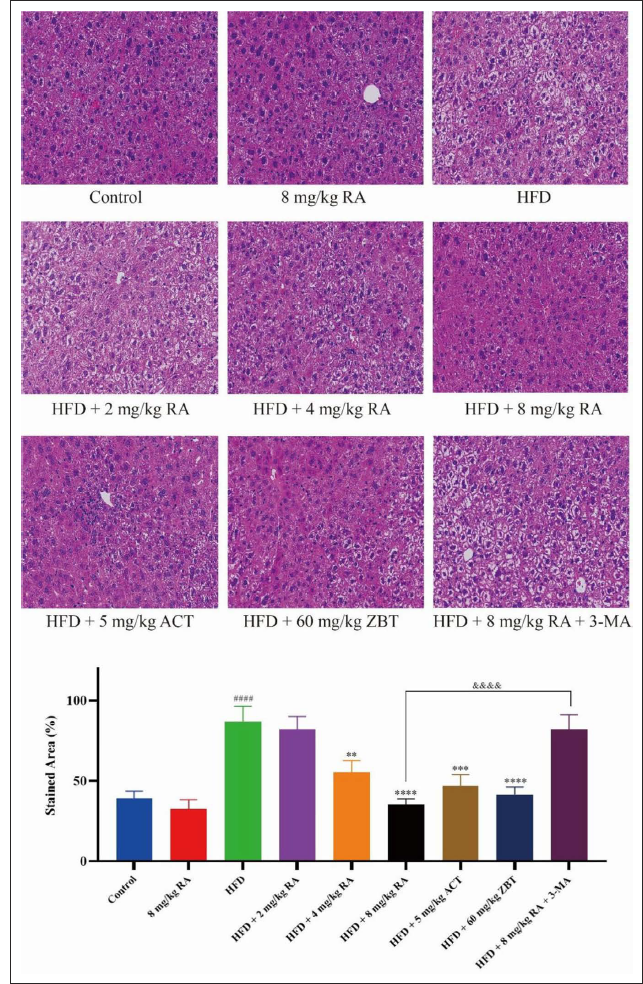
H&E staining was used to demonstrate histopathological analysis of mice liver tissues, as shown in Figure 6. Compared with the control group, the liver tissues in the HFD group had an unclear liver impeller profile, the liver cells were disordered near the central vein, and there were medium and large bubble fatty lesions in the lobule of the liver. In addition, some balloon-like changes and point necrosis were also observed. Compared with the control group, both RA treatment groups and positive drug treatment groups had an effect on the pathological tissues, and the cell structure of the liver tissue of mice was normal and vacuoles were reduced. Only in the 8 mg/kg RA treatment group, the structure and outline of the liver lobules were complete and clear, no vacuolar LDs were observed in the tissues, and the morphology and structure of the tissues were normal. The hepatocytes were irregular polygons, with round nucleus placed in the center of the cells, arranged radially, and no obvious pathological changes were found. Meanwhile, the addition of the autophagy inhibitor 3-MA reversed the effects of RA.
Effect of RA on Oxidative Stress
ROS staining was used to analyze whether RA had an effect on oxidative stress, as shown in Figure 7. The results showed that the red fluorescence was the weakest in the control group and the strongest in the HFD group, indicating that a long-term HFD could cause oxidative stress in the liver tissues and increased the expression of ROS. The decreased fluorescence intensity in the RA treatment groups further indicated that RA could remove excessive ROS in a dose-dependent manner and had a greater antioxidant effect. The fluorescence intensity of the ZBT group was similar to that of the 4 mg/kg RA treatment group, while the intensity of the ACT group was similar to the 8 mg/kg RA treatment group. However, the intensity of 3-MA was similar to the HFD group, which indicated the addition of 3-MA reversed the effects of RA.

In DHE group, the stronger the red fluorescence was, the higher the content of ROS was. The DAPI group was used to demonstrate cell apoptosis. The weaker the blue fluorescence, the more cell apoptosis. MERGE groups are used to highlight colors.
Effect of RA on Biochemical and Inflammatory Factors
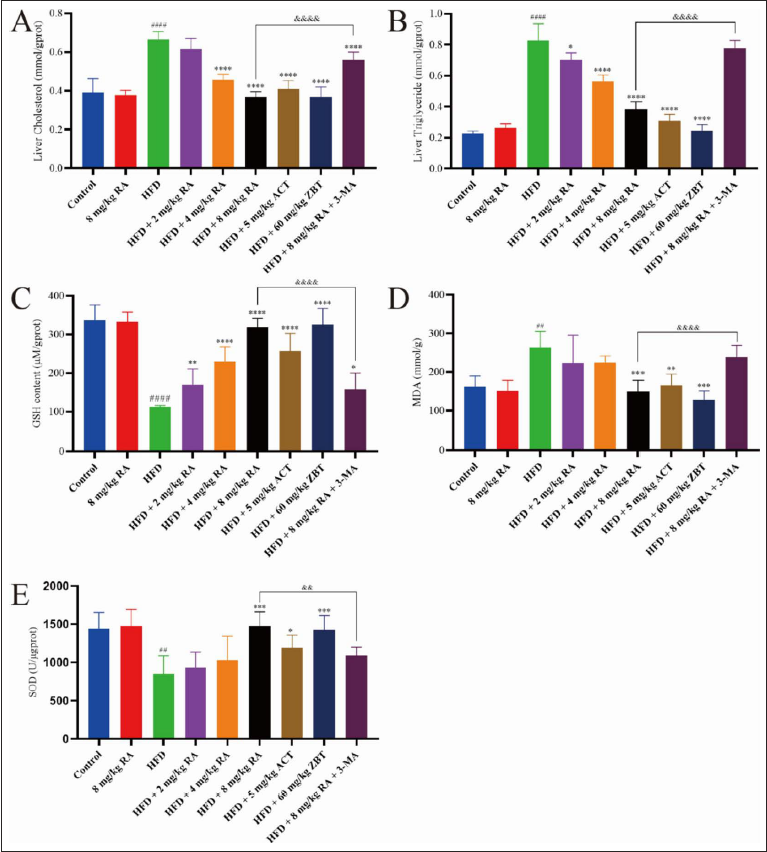
The contents of TC, TG, MDA, AST, and ALT (TC, TG, and MDA were in the supernatant of mice liver tissues, AST and ALT were in the serum) of the HFD group were significantly increased (p < 0.01), and the contents of GSH and SOD in the supernatant of mice liver tissues of the HFD group were significantly decreased (p < 0.01), as shown in Figures 8 and 9A-B. Compared with the model group, the contents of TC, TG, MDA, AST, and ALT of RA treatment groups were significantly reduced in a dose-dependent manner as well as the contents of GSH and SOD were increased in a dose-dependent manner. The contents of TC, TG, MDA, AST, and ALT of the positive drug treatment groups were also significantly decreased (p < 0.01), while the contents of GSH and SOD of the positive drug treatment groups were also significantly increased (p < 0.05). The effects of RA on TC, GSH, MDA, SOD, and ALT were better than ACT, but poorer on TG and AST than ACT. The effects of RA on TC, GSH, SOD, and AST were similar to ZBT, and poorer on TG, MDA, and ALT than ZBT. Meanwhile, the addition of the 3-MA reversed the effects of RA on TC, TG, GSH, MDA, SOD, and ALT (p < 0.01).
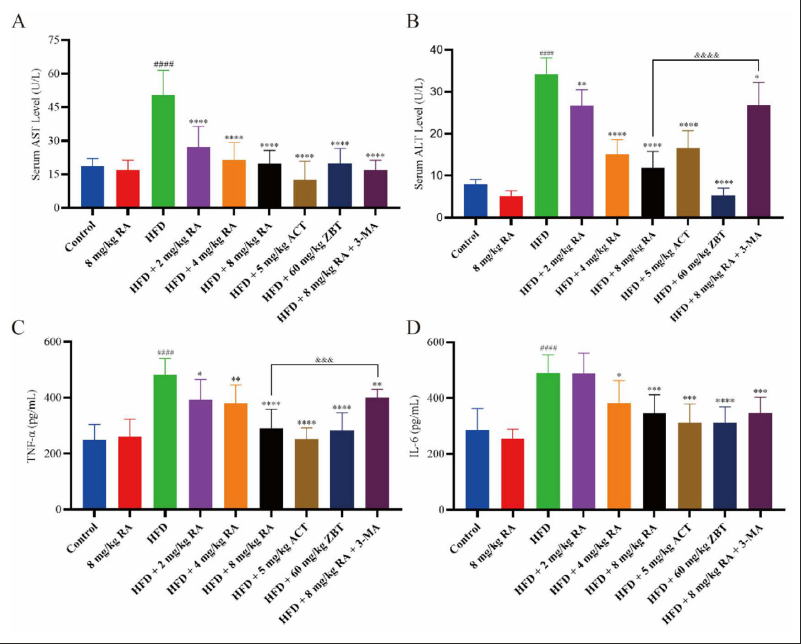
ELISA was used to analyze the expression of inflammatory factors, as shown in Figure 9C and D. The results showed that the expression of TNF-α and IL-6 in the supernatant of mice liver tissues was significantly increased (p < 0.001), which indicated that a long-term HFD caused liver inflammation. The contents of TNF-α and IL-6 were significantly decreased in a dose-dependent manner of the RA treatment groups. Positive drug treatment groups were also able to significantly inhibit the increase of the above factors (p < 0.005), and the effects were greater than that of the 8 mg/kg RA treatment group. The addition of 3-MA significantly reversed the inhibitory effect of RA on the expression of TNF-α (p < 0.005), indicating that autophagy played a key role of RA in the treatment of NAFLD.
Statistical Analysis
All the data was presented as mean ± SD. The differences between groups were analyzed by ANOVA (one-way analysis of variance). p < 0.05 represented a statistically significant difference.
Discussion
Overexpression of TC and TG in liver cells belongs to lipid abnormal accumulation. When the lipid content in the liver exceeds the capacity of liver metabolism, it will lead to abnormal lipid metabolism, which will further develop into hepatic steatosis. The results of in vitro experiments showed that the content of TC and TG and the number of LDs were significantly reduced under the treatment of RA, which indicated that RA had a lipid-lowering effect.
Studies have shown that the depletion of A-CoA can induce autophagy (Mariño et al., 2014). A-CoA in the cytoplasm is mainly produced from citric acid by the lipogenic enzyme ACLY. The expression of ACLY in the liver will be significantly suppressed in the liver upon HFD, then the ACOX1-mediated fatty acid oxidation is the main source of A-CoA in liver cells in this condition. The A-CoA produced by peroxisomal could be continuously acetylated by Raptor, leading to the activation of mTORC1 and inhibition of ULK1 phosphorylation, thereby limiting the lipophagy of LDs. ACOX1 is a key enzyme in the first step of peroxisomal β-oxidation. Deficiency of ACOX1 in the liver can activate lipophagy, thereby preventing fatty liver caused by HFD. mTORC1 is a major regulator of cell growth and metabolism, and autophagy could be inhibited by mTORC1 (Noda, 2017). Recent studies have shown that A-CoA produced by leucine catabolism promotes the activation of mTORC1 after acetylation by Raptor (Son et al., 2019). The results showed that the addition of 3-MA could reverse the effect of RA on reducing the content of TC and TG, indicating that the lipid-lowering effect of RA may be related to the activation of lipophagy. The detection results showed that the addition of 3-MA could reverse the effect of RA on reducing the content of A-CoA, indicating that RA might play a lipid-lowering effect by inhibiting the expression of A-CoA in cells, thereby activating lipophagy. It could be speculated from the results of the Western Blot experiment that RA could down-regulate the expression of ACOX1, p-mTOR, and p-ULK1 and then inhibit A-CoA production and upregulate the expression of Atg5 to promote the lipophagy of lipocytes.
The pharmacological activity of RA was investigated through a mouse model of HFD-induced chronic NAFLD. In vivo experimental results indicate that RA can effectively improve HFD-induced liver injury and lipid accumulation. Further analysis revealed that RA was able to reduce the levels of ALT and AST in serum, indicating that RA could improve long-term HFD damage to hepatocyte membranes and had a hepatoprotective effect.
Studies have indicated that oxidative damage and dysfunction of mitochondria can promote the development of NAFLD to NASH, and mitochondrial damage is reported that it can lead to incomplete oxidation of fatty acids, excessive ROS, and toxic lipid intermediates. The development of inflammation in the liver is an important indicator to evaluate the development of NAFLD. Reducing inflammation is crucial in the treatment of NAFLD because persistent inflammation can lead to fibrosis and ultimately cirrhosis. The content of ROS in mice livers was detected by frozen section immunohistochemistry, and the content of ROS was significantly decreased after treatment with RA. Meanwhile, RA could also inhibit HFD-induced the reduction of SOD, GSH, and the production of MDA in mice livers. In vivo experiments, RA reduced the expression of HFD-induced increase of IL-6 and TNF-α in mice livers. It is suggested that RA can exert its therapeutic effect on NAFLD through its antioxidant and anti-inflammatory effects. This study explored the effects of RA on NAFLD from the aspects of lipophagy, antioxidation, and anti-inflammation, but not limited to this. We will continue to explore the mechanism of RA on NAFLD from other aspects in the future.
Conclusion
In general, our results showed that RA from Rheum L. has a great therapeutic effect on NAFLD by inhibiting the expression of ACOX1, p-mTOR, and p-ULK1, and then inhibiting the production of A-CoA, and up-regulating the expression of Atg5 to promote the lipophagy of lipocytes. We believe that the results of this study will provide further theoretical support for the pharmacodynamics and the mechanism of Rheum L. in the treatment of NAFLD.
Abbreviations
3-MA: 3-methyladenine; A-CoA: Acetyl-CoA; ACOX1:Acyl-CoA oxidase 1; ACT: Afatating calcium tablets; ALT:Alanine aminotransferase; AST: Aspartate aminotransferase;Atg5: Autophagy protein 5; GSH: Glutathione; HFD: Highfat diet; LDs: Lipid droplets; IL-6: Interleukin-6; MDA:Malondialdehyde; NAFLD: Non-alcoholic fatty liver disease;PBS: Phosphate buffered saline; RA: Rhapontigenin; ROS:Reactive oxygen species; SOD: Superoxide dismutase; TC:Total cholesterol; TG: Triglyceride; TLC: Thin-layerchromatography; TNF-α: Tumor necrosis factor α; ZBT: Zhibi-tai capsules.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
All animal experimental procedures and animal care were approved by the Hubei University of Chinese Medicine Ethics Committee and were conducted in accordance with the guidelines of the National Institutes of Health on the care and use of animals. Approval No. SCXK2017-0012.
