Abstract
Background
Pulmonary hypertension (PH) is a pulmonary vascular disease caused by a variety of causes and has a poor prognosis. Hypoxic pulmonary hypertension (HPH) is one of the types of PH. Cordyceps sinensis could treat a variety of lung diseases; its pharmacological active ingredients mainly include cordycepin, nucleosides, cordyceps polysaccharides, sterols, and amino acids. Fermented cordyceps powder (FCP) is an alternative to C. sinensis. In order to investigate the effect of FCP on HPH in rats, we established the HPH rat model.
Materials and Methods
Forty-two male Sprague–Dawley (SD) rats were randomly divided into a control group, hypoxia group, hypoxia + FCP (0.1 g.kg−1.d−1), hypoxia + FCP (0.2 g.kg−1.d−1), hypoxia + FCP (0.4 g.kg−1.d−1), and hypoxia + sildenafil (30 mg.kg−1.d−1) group. Except for the control group, the other five groups of rats were fed in the hypobaric chamber. The corresponding dose was given by intragastric administration, once a day, for 28 days. The mean pulmonary arterial pressure (mPAP) and right ventricle hypertrophy index (RVHI) were measured; hematological parameters white blood cells (WBC), neutrophils (Neu), Lymphocytes (Lym), monocytes (Mon), red blood cells (RBC), hemoglobin (HGB), hematocrit (HCT), and platelets (PLT) levels were detected; serum levels of interleukin-1β (IL-1β) and interleukin-6 (IL-6) were determined by enzyme-linked immunosorbent assay (ELISA); the morphological changes of pulmonary arterioles were observed by hematoxylin and eosin (H&E) staining, and the vascular remodeling indexes, vessel wall thickness as a percentage of vascular outer diameter (WT%), vascular wall area as a percentage of total vessel area (WA%), and lumen area as a percentage of the total vascular area (LA%) were calculated; the protein expression levels of IL-1β, IL-6, p-p38, p38, p-IκBα, IκBα, p-p65, and p65 were determined by western blotting.
Results
FCP could significantly downregulate mPAP, improve pulmonary arteriole remodeling, and reduce the serum levels of IL-1β and IL-6 in HPH rats. In addition, FCP downregulated the protein levels of IL-1β, IL-6, p-p38, p-IκBα, and p-p65.
Conclusion
FCP could alleviate mPAP and vascular remodeling in HPH rats. Its mechanism could be through inhibiting the p38 mitogen-activated protein kinase (MAPK)/nuclear factor-kappa B (NF-κB) signaling pathway and by downregulating the expression of inflammatory factors. Our study might provide a research basis for the therapeutic potential of FCP in HPH.
Introduction
Pulmonary hypertension (PH) is a common pulmonary vascular disease characterized by a persistent increase in mean pulmonary arterial pressure (mPAP ≥ 20 mmHg at rest), followed by increased right ventricular afterload and right ventricular dysfunction, which usually progresses to right heart failure or even death (Mayeux et al., 2021; Simonneau et al., 2019). Hypoxic pulmonary hypertension (HPH) is one of the PH types. Long-term living at high altitudes may contribute to persistent HPH, and HPH incidence increases with altitude (Penaloza & Arias-Stella, 2007).
The pathogenesis of HPH includes hypoxic pulmonary vascular remodeling, abnormal vascular endothelial function, and inflammation (Galiè et al., 2009; Sommer et al., 2016; West, 2012). Among these, the role of inflammatory mechanisms in HPH has been of interest (Pugliese et al., 2015). When alveolar hypoxia occurs, compensatory adaptive constriction of the pulmonary vasculature is required to ensure matching ventilation and blood flow in most areas of the lung and could cause pulmonary vascular remodeling during long-term chronic hypoxia (Wilkins et al., 2015). Hypoxia could also lead to a pulmonary vascular-specific inflammatory response, producing an inflammatory microenvironment. Studies have found that a large number of inflammatory factors accumulated around the remodeled pulmonary vessels and infiltrated into the vessels, producing interleukin-1β (IL-1β), interleukin-6 (IL-6), and chemokine-5. These inflammatory factors promote excessive vascular cell contraction and proliferation, leading to pulmonary vascular remodeling (Scott et al., 2019). Studies have demonstrated that IL-1β and IL-6 levels are correlated with mortality in patients with PH (Cracowski et al., 2014). IL-6 overexpression leads to myelination in distal pulmonary arterioles, promotes the increase of vascular endothelial growth factor (VEGF) and mitogen-activated protein kinase (MAPK), activates the survival of the proliferative factor c-MYC/MAX complex, and produces anti-apoptotic proteins, which collectively cause the proliferation of pulmonary artery smooth muscle cells (PASMC) (Steiner et al., 2009). IL-1β binds to the interleukin receptor (IL-1R1), recruits myeloid differentiation factor 88 (MyD88), and further promotes nuclear factor-kappa B (NF-κB) to stimulate PASMC proliferation. In contrast, the use of IL-1R and IL-6 antagonists attenuated PASMC proliferation in pulmonary arterial hypertension (PAH) rats (Parpaleix et al., 2016; Tamura et al., 2018). Numerous findings suggested that inflammation was closely related to vascular remodeling. Inhibition of specific inflammatory factors might be able to reverse the process of PH (Rabinovitch et al., 2014).
At present, the main drugs used in the clinical treatment of HPH include prostacyclin I2 receptor agonists, endothelin receptor antagonists (ERAs), and phosphodiesterase-5 inhibitors (PDE5 inhibitors). The main effect of these drugs is to inhibit pulmonary vasoconstriction but not pulmonary vascular remodeling (Sommer et al., 2021). Recent studies have shown that pulmonary vascular remodeling plays an important role in HPH. Therefore, drugs that inhibit or delay pulmonary vascular remodeling should be developed to treat HPH.
Cordyceps sinensis has anti-inflammatory, antioxidant, and immunomodulatory effects and is commonly used in the treatment of various respiratory diseases (Ng & Wang, 2005; Olatunji et al., 2018). It was shown that ergosterol, the main active ingredient of C. sinensis, could inhibit chronic obstructive pulmonary disease (COPD) inflammation by suppressing the activation of the NF-κB p65 signaling pathway (Sun et al., 2019). Fermented cordyceps powder (FCP) is the mycelium extracted from C. sinensis, which has been proven to have similar active ingredients and pharmacological effects as natural C. sinensis and has been widely used in clinical treatment. Bailing capsule, one of the FCP preparations, has antioxidant and anti-inflammatory effects and could reduce the levels of IL-6, tumor necrosis factor-alpha (TNF-α), transforming growth factor-beta 1 (TGF-β1), and other inflammatory factors in lung injury model rats and inhibit the alveolar inflammatory response and fibrosis (Zhong et al., 2015). The mechanism of FCP on HPH remains unclear and needs further study. In this study, mPAP, RV hypertrophy index (RVHI), hematological parameters, pulmonary vascular remodeling index (vessel wall thickness as a percentage of vascular outer diameter [WT%], vascular wall area as a percentage of total vessel area [WA%], and lumen area as a percentage of total vascular area [LA%]), serum and lung tissue IL-1β, IL-6 levels, and p-p38, p38, p-IκBα, IκBα, p-p65, and p65 protein levels were determined in HPH rats. Our study aimed to investigate the effects of FCP on HPH rats and to elucidate the mechanism of the p38 MAPK/NF-κB pathway.
Materials and Methods
Drugs and Reagents
FCP was purchased from Qinghai Zhufeng Cordyceps Pharmaceutical Co., Ltd., with batch number ZF0421F004; sildenafil (CAS#139755-83-2), uridine standard (CAS#26287-69-4), vernine standard (CAS#118-00-3), inosine standard (CAS#58-63-9), and adenosine standard (CAS#58-61-7) were all purchased from Shanghai Yuanye Biotechnology Co., Ltd.; methanol (CAS#67-56-1; chromatography grade) was purchased from Merck Co., Ltd.
Drug Identification
Uridine, inosine, vernine, and adenosine standards were weighed and dissolved in ultrapure water to get the reference solution, which was filtered by a 0.22-µm stream filter membrane before use. One gram of FCP was weighed, placed in 20 mL of ultrapure water, and sonicated for 30 min. Then, it was centrifuged in a high-speed centrifuge (Sigma 3K15, Germany) at 12,000 r/min (4℃, 15 min). The supernatant was aspirated and filtered through a 0.22-µm stream filter membrane as the test solution. The main components were analyzed by using a high-performance liquid chromatograph (HPLC) (Agilent 1260 Infinity, Germany) with a C18 column (A585V3 SVEA C18 Opal, 250 × 4.6 mm, 5 µm); mobile phase: methanol:water (10:90); sample quantity: 20 µL; the wavelength was detected at 260 nm. The components of uridine, inosine, vernine, and adenosine in FCP were identified by comparing peak areas with corresponding mixed standards. And draw standard curves to calculate the content of four nucleosides.
Animals
Male Sprague–Dawley (SD) rats (150 ± 10 g) were obtained from Beijing Vital River Experimental Animal Technology Co., Ltd., and the production license number of experimental animals was SCXK (Beijing) 2021-0011. The rats were kept under conditions of temperature (23 ± 3°C), relative humidity (50 ± 6%), and a light/dark cycle (12 h/12 h). During this period, adequate food and water were given to rats. The animal experimental protocol was approved by Qinghai University’s Institutional Animal Care and Use Committee. Forty-two rats were randomly assigned to six groups: control, hypoxia, H + FCP (0.1 g.kg−1.d−1), H + FCP (0.2 g.kg−1.d−1), H + FCP (0.4 g.kg−1.d−1), and H + sildenafil (30 mg.kg−1.d−1). Except for the control group, the other five groups of rats were placed in a hypobaric chamber (DYC-300, Guizhou Fenglei Oxygen Chamber Co., Ltd., China), which simulated the environment at 5000 m altitude (Nan et al., 2018). The rats in the control and hypoxia groups were given the corresponding volume of 0.5% CMC-Na (Cat No. 8912K, Adamas Reagent Co., Ltd., China) by gavage, the other four groups of rats were given a corresponding dose of drugs by gavage, once a day for 28 days.
Hemodynamic Measurements
After 28 days of hypoxia exposure, the rats were anesthetized using urethane. The external jugular vein was dissected, and a suitable V-shaped incision was made proximal to the vessel. Next, insert the catheter into the V-shaped incision and slowly advance it to the pulmonary artery. A pressure sensor connected to the physiological recorder (BIOPA, USA) was used to monitor vascular pressure waveforms and measure mPAP (Yang et al., 2020).
Blood Routine and ELISA Detection
Rat abdominal aorta blood (2 mL) was collected, and then the hematological parameters such as white blood cell (WBC) counts, neutrophil (Neu) counts, Lymphocyte (Lym) counts, monocyte (Mon) counts, red blood cell (RBC) counts, hemoglobin (HGB) counts, hematocrit (HCT) values, and platelet (PLT) levels were determined by a BC-5000Vet hematology analyzer (Shenzhen Myriad Biomedical Electronic Co., Ltd., China) (Dang et al., 2020). IL-1β and IL-6 in serum levels were detected using the enzyme-linked immunosorbent assay (ELISA) method (Shanghai Yubo, LOT: EDL202206068).
Calculation of RVHI
The heart was washed in saline and covered with filter paper to absorb moisture. The right ventricle (RV), left ventricle (LV), and septum (S) of rats were isolated and weighed separately. Excess tissue, such as atria, blood vessels, and fat, was cut off before weighing. The RV/(LV+S), namely, RVHI, was measured (Yang et al., 2020).
H&E Staining
The upper lobe of the lung of the rat was immersed in a paraformaldehyde (4%) solution for 48 h. Then the tissue was dehydrated, waxed, embedded, and cut into 4-µm paraffin sections of uniform thickness. Next, it was placed in an oven at 37℃, dried for hematoxylin and eosin (H&E) staining, and sealed with neutral resin. The pathological changes of pulmonary arterioles (<100 µm) in each group were observed by microscope. The Case Viewer 2.3 digital section viewing software was used to take screenshots, and the Image Pro Plus 6.0 image software was used to calculate WT%, WA%, and LA% (Chen et al., 2022).
Western Blotting Analysis
A hundred milligrams of lung tissue were weighed, and RIPA assay lysis buffer (Cat No. R0010, Solarbio) was added in proportion. The protein was extracted from each group of rat lung tissue. Additionally, a sodium dodecyl sulfate-polyacrylamide gel was employed to separate the isolated proteins. In the following step, proteins were transported to PVDF (polyvinylidene difluoride) membranes. The PVDF membranes were blocked for 2 h in 10% skim milk and treated with the following antibodies overnight: IL-1β (Cat No. A1112, 1:1000, ABclonal), IL-6 (Cat No. A0286, 1:1000, ABclonal), p38 (Cat No. A4771, 1:800, ABclonal), p-p38 (Cat No. AP0526, 1:800, ABclonal), IκBα (Cat No. ab32518, 1:100000, Abcam), p-IκBα (Cat No. 2859T, 1:1000, CST), p65 (No.10745-1-AP, 1:2000, Proteintech), p-p65 (Cat No. AP0123, 1:1000, ABclonal), β-actin (Cat No.66009-1-Ig, 1:100000, Proteintech), and β-tubulin (Cat No. 66240-1-lg, 1:10000, Proteintech). After being washed (3 × 15 min) with Tris-buffered saline with Tween (TBST), the secondary antibodies were added to the PVDF membranes. And the protein detection was performed using an AI600 Imaging System (GE, USA) with a chemiluminescence image. The gray value was analyzed using ImageJ and the relative expression of the corresponding protein was calculated (Chen et al., 2022).
Statistical Analysis
SPSS 27.0 software was used for statistical analysis. The experimental data were expressed as mean ± standard deviation (
Results
Identification of Nucleoside Components of FCP
Nucleosides are commonly used as quality control indicators for FCP (Huo et al., 2017). The contents of four nucleosides in FCP were determined by HPLC to control the quality of FCP. The results showed that the composition of uridine, inosine, guanosine, and adenosine in FCP used in this experiment was 0.0833%, 0.0014%, 0.1148%, and 0.1025% (Cai et al., 2018; Huo et al., 2017). We found that the adenosine content in the FCP we used was much higher than the 0.010% stipulated in the 2020 edition of the Chinese Pharmacopoeia (Figure 1 and Table 1).
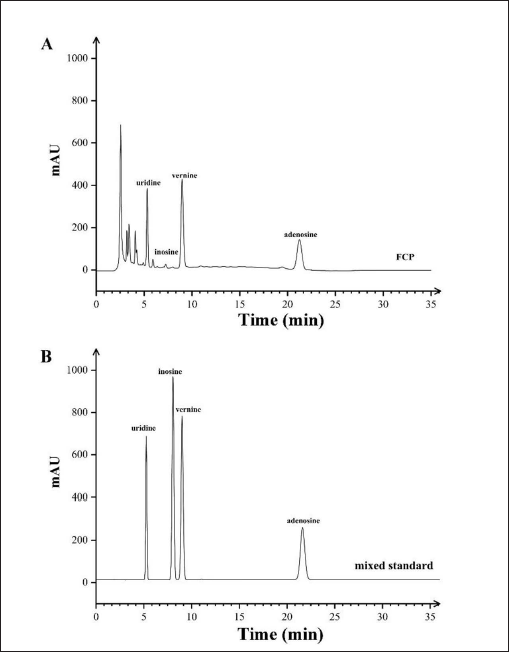
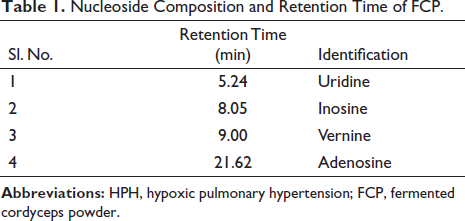
Nucleoside Composition and Retention Time of FCP.
Effect of FCP on mPAP of HPH Rats
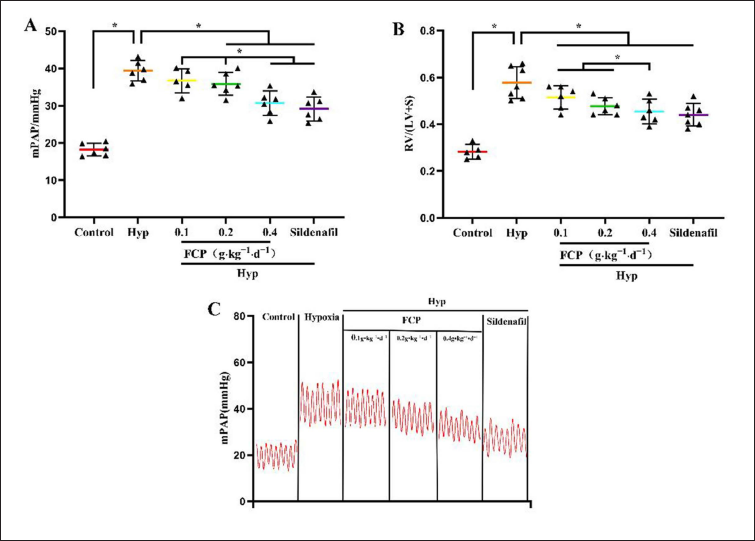
The mPAP of rats in each group was measured after 28 days. In contrast with the control group (18.20 ± 1.70) mmHg, the mPAP was increased in the hypoxia group (39.35 ± 2.74) mmHg, p < 0.05. Compared with the hypoxia group, H + FCP (0.2 g, 35.86 ± 3.09 mmHg), H + FCP (0.4 g, 30.66 ± 3.27 mmHg), and H + sildenafil (30 mg, 30.66 ± 3.27 mmHg) interventions could reduce the mPAP in HPH rats (p < 0.05). H + FCP (0.4 g) intervention group was better than the H + FCP (0.1 g) and H + FCP (0.2 g) intervention groups (p < 0.05). The effect of the H + FCP (0.4 g) intervention was not statistically different when compared with the sildenafil intervention group (p > 0.05). H + FCP (0.1 g) intervention did not reduce mPAP in HPH rats (36.70 ± 3.24 mmHg, p > 0.05) (Figure 2A and C).
Effect of FCP on RVHI of HPH Rats
Compared with the control group (0.30 ± 0.04), the RVHI of rats in the hypoxia group was 0.59 ± 0.06 (p < 0.05). The H + FCP (0.4 g) intervention group (0.46 ± 0.05) had a better effect than the H + FCP (0.1 g) intervention group (0.52 ± 0.05, p < 0.05), which was similar to the effects of the H + sildenafil intervention (0.44 ± 0.05, p > 0.05) (Figure 2B). The results indicated that FCP could reverse RV hypertrophy in HPH rats.
Effect of FCP on Pulmonary Arterioles Pathological Remodeling
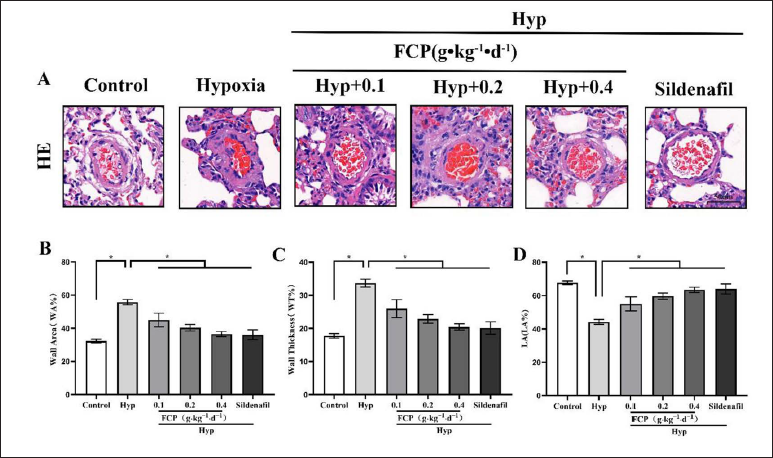
The intima of pulmonary arterioles was intact in the control group, with a dense and neat distribution of cells, thin and uniform walls, and no thickening of the muscular layer. The walls of pulmonary arterioles in hypoxia group rats were significantly thickened, with hyperplasia of the mid-membrane PASMCs and narrowing of the lumen (Figure 3A). The pulmonary arterioles remodeling index (WT%, WA%, and LA%) was measured by Image Pro Plus 6.0. The results showed that WA% and WT% in the hypoxia group were 55.78 ± 1.55% and 33.67 ± 1.19%, respectively, which were significantly higher in contrast with the control group (WA%: 32.27 ± 1.14%, WT%: 17.77 ± 0.69%, p < 0.05). The LA% in the hypoxia group was 44.22 ± 1.55%, which was lower than 67.73 ± 1.14% in the control group (p < 0.05). In addition, in contrast with the hypoxia group, WT% and WA% were reduced and LA% was increased in the FCP and sildenafil intervention groups (p < 0.05; Figure 3B–D). Therefore, FCP could reverse pulmonary arteriole remodeling in HPH rats.
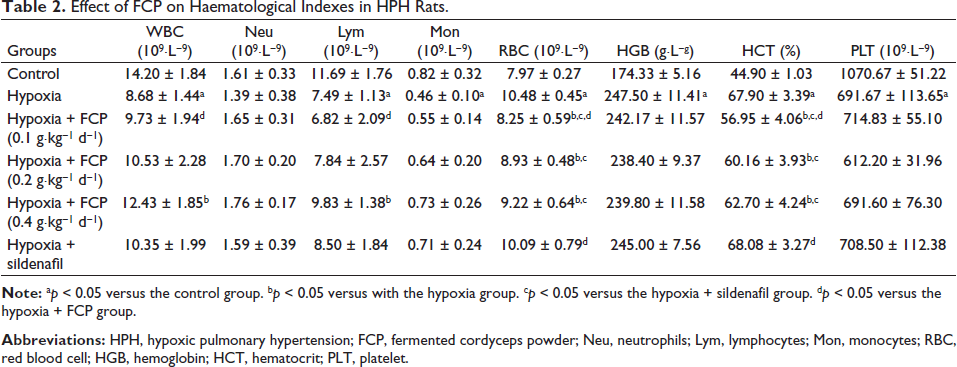
Effect of FCP on Routine Blood Indicators
In contrast with the control group, hematological indicators showed that RBC, HGB, and HCT were increased in the hypoxia group, while the levels of WBC, Lym, and PLT were reduced (p < 0.05), and the concentrations of Neu and Mon did not change (p > 0.05). Compared with the hypoxia group, the levels of WBC and Lym in the FCP (0.4 g) intervention were increased, and those of RBC and HCT in the FCP (0.1 g), FCP (0.2), and FCP (0.4 g) interventions were reduced (p < 0.05). However, HGB and PLT concentrations were not changed after FCP and sildenafil treatment (p > 0.05, Table 2).
Effect of FCP on Haematological Indexes in HPH Rats.
Effect of FCP on IL-1β and IL-6 Levels of HPH Rats
To investigate the mechanism of FCP, we measured the expression of inflammatory cytokines in HPH rats. ELISA analysis indicated that serum IL-1β levels were higher in the hypoxia group (52.34 ± 2.89 ng L−1) than in the control group (31.15 ± 1.57 ng L−1; p < 0.05). In contrast with the hypoxia group, IL-1β in H + FCP and H + sildenafil groups was significantly decreased [FCP (0.1 g): 45.35 ± 1.79 ng L−1, FCP (0.2 g): 41.97 ± 2.60 ng L−1, and FCP (0.4 g): 36.22 ± 2.19 ng L−1] and sildenafil 33.39 ± 1.54 ng L−1, p < 0.05 (Figure 4A). Western blotting analysis showed that IL-1β levels were higher in the hypoxia group (1.35 ± 0.41) than in the control group (0.41 ± 0.17). IL-1β levels were significantly lower in the H + FCP (0.1 g), H + FCP (0.2 g), H + FCP (0.4 g), and H + sildenafil groups (p < 0.05) (Figure 4C and D).
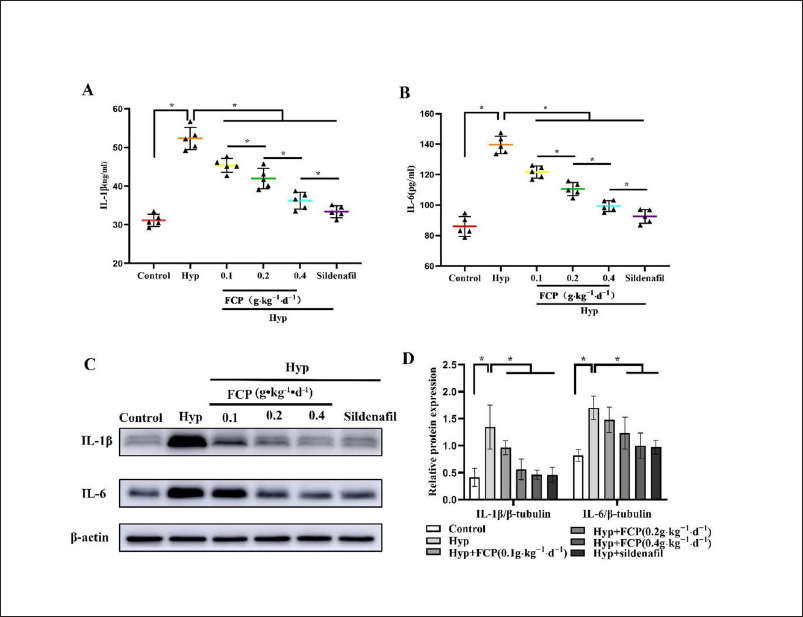
In contrast with the control group (85.95 ± 6.47 pg.mL−1), IL-6 levels in serum were increased in the hypoxia group (139.54 ± 5.79 pg.mL−1, p < 0.05). IL-6 levels in H + FCP and H + sildenafil groups were significantly decreased, which were 121.65 ± 3.91 pg mL−1 (FCP-0.1 g), 110.50 ± 4.34 pg mL−1 (FCP-0.2 g), 99.32 ± 3.61 pg mL− (FCP-0.4 g), and 92.52 ± 4.53 pg mL−1 (sildenafil) (p < 0.05 vs. hypoxia group, Figure 4B). Western blotting analysis indicated that the relative expression of IL-6 was higher in the hypoxia group (1.77 ± 0.33) than in the control group (0.85 ± 0.25) (p < 0.05). In contrast with the hypoxia group, H + FCP (0.2 g), H + FCP (0.4 g), and H + sildenafil intervention groups reduced IL-6 protein expression in HPH rats (H + FCP 0.2 g: 0.96 ± 0.06, H + FCP-0.4 g: 0.93 ± 0.16, H + sildenafil: 1.11 ± 0.26, p < 0.05). However, the H + FCP (0.1 g) intervention did not reduce IL-6 protein expression significantly (H + FCP-0.1 g: 1.48 ± 0.24, p > 0.05, Figure 4C and D).
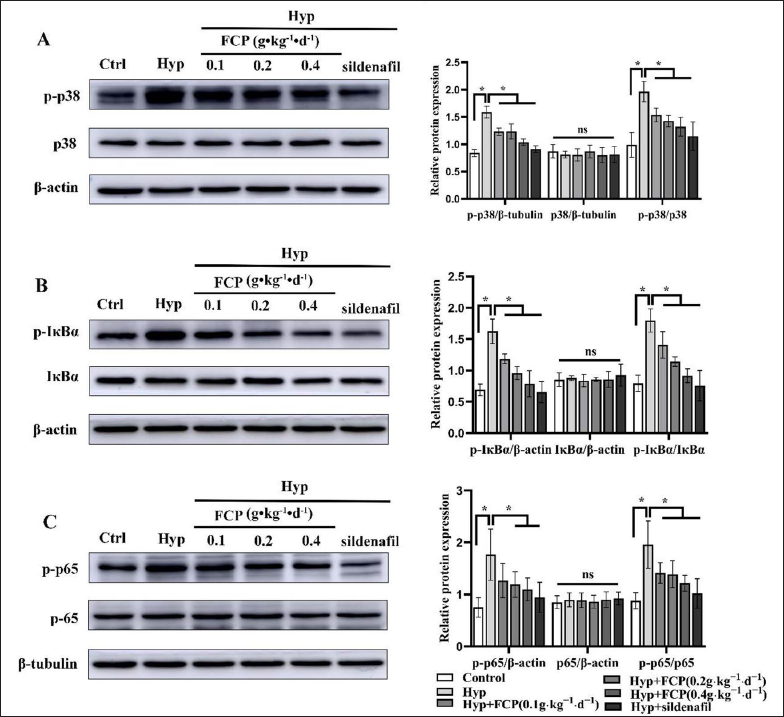
Effect of FCP on p38 MAPK/NF-κB Signaling Pathway in Lung Tissue
The expression levels of phosphorylated p38 (1.96 ± 0.19), phosphorylated IκBα (1.80 ± 0.18), and phosphorylated NF-κB p65 (1.96 ± 0.45) proteins in the hypoxia group were higher than those in the control group (0.99 ± 0.23, 0.79 ± 0.13, 0.88 ± 0.16, p < 0.05). In contrast with the hypoxia group, FCP and sildenafil intervention reduced the levels of phosphorylated p38, phosphorylated Iκbα and phosphorylated NF-κB p65 proteins in lung tissue of HPH rats (p < 0.05, Figure 5).
Discussion
C. sinensis has anti-inflammatory, antitumor, and immunomodulatory effects. Its pharmacological active ingredients mainly include cordycepin, nucleosides, cordyceps polysaccharides, sterols, and amino acids (Olatunji et al., 2018). However, the production of natural C. sinensis is limited to meet the increasing demand. Artificially cultured cordyceps mycelium has been proven to contain similar active ingredients and effects as C. sinensis (Nxumalo et al., 2020). Studies have shown that FCP could treat a variety of lung diseases (Xu & Li, 2010), improve lung function, improve airway conditions and reduce the complication rate in COPD patients (Na, 2019). However, the effects of FCP on HPH and its mechanism are still unclear.
Studies have confirmed that sildenafil could improve PH by inhibiting phosphodiesterase and exerting vasodilation, which has been widely used in clinical practice. Therefore, sildenafil was used as a positive control drug. In this study, mPAP ≥25 mmHg was used as the criterion for successful modeling in rats, RVHI was used as the evaluation index of right ventricular hypertrophy, the pathological morphological changes of pulmonary arterioles were observed by HE staining, and WT% and WA% were calculated to evaluate the remodeling of pulmonary arterioles. Compared with the control group, mPAP, RVHI, WA%, and WT% of rats in the hypoxia group were increased, while LA% was decreased. In our study, the rat HPH model was successfully established. FCP intervention could effectively reduce mPAP, alleviate the hypertrophy of the RV, and remodel the pulmonary arterioles in HPH rats. And the mechanism of FCP action might be related to the inhibition of NF-κB-dependent activation of pro-inflammatory factors IL-1β and IL-6 and the downregulation of the p38 MAPK/NF-κB signaling pathway.
In contrast with the control group, WA% and WT% were higher and LA% was lower in HPH rats (Figure 3B–D), while WA% and WT% were lower and LA% was higher in the FCP intervention groups. These results indicated that FCP could improve vascular remodeling in HPH rats. Studies have also found that C. sinensis extract and cordycepin could inhibit the proliferation of PASMC isolated from idiopathic pulmonary arterial hypertension patients. And C. sinensis extract also significantly reversed hypoxia-induced pulmonary vasoconstriction and reduced mPAP in mice (Luitel et al., 2020).
The inflammatory response was considered a central link in the development of HPH, which was manifested by the inflammatory factors accumulated around pulmonary vessels and could lead to hypoxic pulmonary vascular remodeling (Frid et al., 2006; Stenmark et al., 2005). Hypoxia could induce the elevation of IL-6 and C-reactive protein, which induce inflammation and promote the proliferation, migration, and generation of vascular cells (Eltzschig & Carmeliet, 2011; Whiteford et al., 2016). Our study also found IL-1β and IL-6 levels in HPH rats were increased, and they decreased after FCP intervention. Studies (Sun et al., 2018) have also shown that nucleosides, the active component of C. sinensis, could reduce levels of IL-1β and TL-6 in serum, downregulate the expression of NF-κB/p65 in mice, and suppress the inflammatory response.
In our present study, hematological parameters, including WBC, Neu, Lym, Mon, RBC, HGB, HCT, and PLT, were detected in each group. It was reported that chronic hypoxia increased blood viscosity (Bradford, 2007). Our experimental data showed that RBC, HGB, and HCT counts were increased in the blood of HPH rats, which might be due to the control of the HIF-1-erythropoietin-RBC system (Semenza, 2004). We found that FCP intervention reduced RBC and HCT counts in HPH rats (Table 2). Studies have shown that WBC counts in HPH rats are significantly lower than those in normoxic rats (Orth et al., 2005). Our results showed that FCP intervention could partially reverse the levels of WBC and Lym counts (Table 2). In addition, PLT counts in the hypoxia group were lower than those in the control group, which might be caused by increased PLT consumption under inflammatory conditions (Bradford, 2007; Gaertner & Massberg, 2019; Otaka et al., 2020). However, FCP intervention did not alter PLT counts in HPH rats (Table 2).
Inflammatory factors activate p38 MAPK, which further promotes the activation of the downstream NF-κB signaling pathway, thus promoting the occurrence of PAHs in rats (Cuadrado & Nebreda, 2010; Hu et al., 2019; Kumar et al., 2019). p38 MAPK could regulate cell proliferation and intracellular and extracellular signal transduction (Huang et al., 2010). p38 MAPK is inactive in the non-phosphorylated state and is activated by double phosphorylation of the Thr-Gly-Tyr motif within the regulatory loop located between subdomains VII and VIII when stimulated by stress such as hypoxia (Kumar et al., 2003; Raman et al., 2007). Phosphorylated p38 MAPK activates substrates such as transcription factors and protein kinases that mediate inflammatory responses, which mediate inflammation (Coulthard et al., 2009; He et al., 2018). NF-κB is the main downstream pathway factor of p38 MAPK, and phosphorylated p38 MAPK leads to phosphorylation and ubiquitination of NF-κB inhibitor alpha (IκBα), leading to activation of NF-κB (Je et al., 2004). Under normal physiological conditions, the NF-κB dimer binds to IκBα and localizes to the cytoplasm without transcriptional activity. When stimulated by cytokines, viral infection, oxygen free radicals, and other stress or inflammation, inhibitor of kappa B kinase (IKK) is phosphorylated and ubiquitinated, triggering the degradation of IκBα. Then, the degraded IκBα translocates free NF-κB p65 to the nucleus and binds to the corresponding DNA sites, initiating target gene transcription (Baker et al., 2011; Mitchell & Carmody, 2018), and further promoting the increase of the IL-1β and IL-6 levels. While NF-κB inhibitors alleviate the inflammatory response and reverse the progression of PAH (Fan et al., 2016; Hosokawa et al., 2013; Sawada et al., 2007).
Consistent with reported studies (Hu et al., 2019; Patel et al., 2017), our study also showed that the p38 MAPK/NF-κB signaling pathway was activated in HPH rats under chronic hypoxia conditions. Compared with the hypoxia group, FCP intervention could inhibit the p38 MAPK/NF-κB signaling pathway in the lung tissue of HPH rats (Figure 5).
Conclusion
FCP could reduce mPAP and alleviate pulmonary arteriole remodeling in HPH rats. Its mechanism could be through inhibiting the p38 MAPK/NF-κB signaling pathway expression and by downregulating the level of inflammatory factors. Our study provides a research basis for the therapeutic potential of FCP in HPH. Further mechanistic studies in depth are ongoing.
Summary
HPH is a serious respiratory disease with a high mortality rate. FCP could reduce mPAP, inhibit the expression of the p38 MAPK/NF-κB signaling pathway, and reduce the level of inflammatory factors in HPH rats.
Abbreviations
FCP: fermented cordyceps powder; HPH: hypoxic pulmonary hypertension; IL-1β: interleukin-1β; IL-6: interleukin-6; HGB: hemoglobin; HCT: hematocrit; Lym: lymphocyte; MAPK: mitogen-activated protein kinase; Mon: monocyte; mPAP: mean pulmonary arterial pressure; NF-κB: nuclear factor kappa-B; Neu: neutrophil; PH: pulmonary hypertension; PAH: pulmonary arterial hypertension; PASMC: pulmonary artery smooth muscle cell; PLT: platelets; RBC: red blood cell; WBC: white blood cell.
Footnotes
Acknowledgments
The authors would like to thank Quanyu Yang and Gaqin for their technical assistance in operating the hypobaric chamber.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval and Consent to Participate
This animal study protocol was approved by the Animal Conservation and Utilization Committee of Qinghai University.
Funding
This work was supported by the National Natural Science Foundation of China (82060786, 81860768), Natural Science Foundation of Qinghai province (2021-ZJ-907), Qinghai Province “Kunlun Talents, High-end Innovative and Entrepreneurial Talents” Project, West Light Foundation of The Chinese Academy of Sciences.
