Abstract
Background
Apigenin effectively inhibits the growth of human gastric carcinoma SGC-7901 cells and induces apoptosis as well. This study investigated the effects of ATF6 and PERK signaling pathways in unfolded protein response (UPR) and endoplasmic reticulum stress (ERS) on the apoptosis induced by apigenin in human gastric carcinoma SGC-7901 cells.
Materials and Methods
SGC-7901 cells were cultured with apigenin and tunicamycin for 48 h or with apigenin for 12–48 h. CCK-8 was used to determine cell viability. The mRNA expression level was detected by RT-qPCR. The expression of related proteins was explored by Western blot. The apoptosis rate of cells and cell-cycle arrest were evaluated by flow cytometer.
Results
The results of CCK-8 confirmed that apigenin could induce apoptosis of SGC-7901 cells. In the apigenin-treated cells group, the protein and mRNA levels of GRP78 and GRP94 dose- and time-dependently increased. Additionally, apigenin-activated UPR components PERK and ATF6. However, apigenin exerted no influence on CHOP expression or JNK activation. Pretreatment with 4-PBA significantly increased the apigenin-triggered apoptosis in SCG-7901 cells (p < 0.05).
Conclusion
The results revealed the protective effect of UPR performance on apigenin-triggered apoptosis in SGC-7901 cells.
Introduction
Gastric carcinoma is one of the major causes of cancer-related fatalities throughout the world (Torre et al., 2015). More than 90% of stomach cancers are caused by the gastric mucosa. Gastric adenocarcinoma incidence exhibits a particular geographic distribution. The highest incidence is reported in East Asia (Kayaalp, 2015). Drug side effects are the most crucial problem in gastric adenocarcinoma chemotherapy. Bioactive ingredients in plant extracts are receiving increasing attention for their promising properties, such as anti-tumor, anti-depressant, and analgesic (Freitas et al., 2021; Mitra et al., 2022; Vieira et al., 2020). Apigenin is obtained from plant extracts, and apigenin chemotherapy is expected to have little or no negative effects; therefore, it may become a promising drug for the chemotherapy of gastric cancer (Qi et al., 2020).
Various cellular stress conditions, such as hypoxia, nutrient deprivation, and dealing with various agents, may cause misfolded and unfolded proteins to aggregate or accumulate in the ER lumen, which causes endoplasmic reticulum stress (ERS) (Chen & Cubillos-Ruiz, 2021). The unfolded protein response (UPR) is an accommodative process during ERS. UPR restores endoplasmic reticulum homeostasis to protect cell survival or contributes to ERS-triggered cell-programmed death (Hetz et al., 2020). It also alleviates ERS through the following mechanisms: the beginning is the reduction of protein synthesis; followed by the induction of ER chaperones to promote protein folding, such as GRP78 and GRP94; and the accumulation of the wrong folding protein is prevented. Low ERS levels are resolved by activating ER sensors such as ATF6, PERK, and IRE1 (Oakes, 2020). Excessive ERS occasions the cytotoxic ERS proteins expression, such as JNK, CHOP, and Caspases. This phenomenon may be transducted during the UPR, ultimately initiating apoptosis.
The current study examined apigenin’s specific antitumor effect on gastric carcinoma cells and elucidated the underlying mechanism. We aimed to identify the occurrence of apigenin-induced ERS and confirm whether ERS contributes to apigenin-triggered apoptosis in SGC-7901 cells.
Materials and Methods
Materials
Apigenin (API), tunicamycin (TM), dimethyl sulfoxide (DMSO), and 4-Phenylbutyric acid (4-PBA) were purchased from Sigma (St. Louis, USA). YOSHI (Wuhan, China) provided the RPMI-1640 and CCK-8. EallBio (Beijing, China) supplied fetal bovine serum (FBS). Becton Dickinson (New Jersey, USA) provided the Annexin V-FITC Apoptosis Detection Kit. DAPI Staining Solution (DAPI), lysis buffer, and propidium iodide (PI) were obtained from Beyotime (Shanghai, China). Cell Signaling Technology (Massachusetts, USA) supplied Rabbit polyclonal antibodies for GRP78, GRP94, and p-JNK. Immunoway (Newark, USA) provided Rabbit polyclonal antibodies for Cleaved-Caspase-3, p-PERK, and β-Actin. Proteintech (Boulder, Colorado, USA) supplied Rabbit polyclonal antibodies for PARP, PERK, JNK, ATF6, and rabbit polyclonal antibody for CHOP was obtained from Affinity Biosciences (Ohio, USA).
Cell Culture and Treatments
Human gastric cancer SGC-7901 cells were purchased from the Chinese Academy of Science’s Cell Bank and grown in RPMI-1640 with FBS (10%) and were kept at 37°C and 5% CO2. When experimenting, the FBS proportion was lessened to 2%, to keep the cell alive minimally but not allow it to grow. Cells in the exponential growth phase were cultivated for 24 h in the presence of apigenin at 20, 40, 60, or 80 µmol/L, or cultivated in 60 µmol/L apigenin for 12, 24, and 48 h (Each group contained 0.1% DMSO). An equivalent volume of DMSO served as solvent control. DMSO was used to dissolve TM, which was used as a positive control at a final concentration of 5 mg/mL.
Cell Viability Measurement
Cell viability was assessed by the CCK-8 test. Cells were seeded into 96-well plates and incubation overnight, then the cells were treated with apigenin (0, 20, 40, 60, or 80 µmol/L) for 12, 24, and 48 h. Subsequently, 10 µL of CCK-8 reagent was added to each well, and the cells were cultured at 37 °C for 4 h. The optical density of each well was tested with a Benchmark Plus microplate reader (Bio-Rad, California, USA) at 450 nm, and then calculated the cell viability.
Cell-cycle Arrest Analysis
Apigenin was used to treat the cells for 24 h or 48 h at the indicated concentrations of 0, 20, 40, 60, and 80 µmol/L. After treatment, the cells were collected and centrifuged after being washed in ice-cold PBS. After fixation with 75% alcohol, cells were incubated with propidium iodide staining solution containing RNase A for 30 min and then the flow cytometry detected the data (Becton Dickinson, New Jersey, USA), and ModFitLT software was used to analyze cell cycle data.
Quantification of Apoptosis
Cell apoptosis measurements were performed following the recommendation provided by the Annexin V-FITC Apoptosis Detection Kit manufacturer. Briefly, the cells were twice washed in cold PBS before being collected by centrifugation. Re-suspended in binding buffer, the cells were then dyed with PI and Annexin V-FITC at 25°C for 15 min while being shielded from light. Using Cell Quest software and flow cytometry (BD, New Jersey, USA), cells were analyzed.
Apoptosis Assessment
To observe the effect of apigenin combined with ERS inhibitor on cell nuclear morphology, cells were cultured in 24-well plates. After pretreatment with 4-PBA for 2 h, cells were incubated with 60 µmol/L apigenin for 24 h. Following DAPI staining for 15 min and PBS washing, the cells were observed by laser confocal microscope (Nikon, Tokyo, Japan).
RT-qPCR Assay
Total RNA extracted from apigenin-treated cells was inverted into cDNA. Following the instructions of the reagent manufacturer, the gene expression of GRP78 and GRP94 was measured by the 2x Realtime PCR Supermix (Mei5bio, Beijing, China). Using the 2-∇∇ct method, GRP78 and GRP94 relative expression levels were calculated. Specific primer sequences are presented in Table 1.
Primer Sequences.

Western Blot Assay
Cells were collected and resolved in the lysis buffer to extract total protein. SDS-PAGE gels were used to analyze samples before being transferred onto the PVDF membrane. After the GRP78, GRP94, CHOP, PERK, p-PERK, JNK, p-JNK, ATF6, β-Actin, Caspase-3, and PARP primary antibody, and secondary antibody incubation, the ChemiImager 4000 was used to analyze the bands (Alpha Innotech, California, USA).
Statistical Analyses
Experiments were conducted at least thrice. Results were expressed as mean ± standard deviation (SD). Using Levene’s method, the homogeneity of variance was examined. The One-way ANOVA and LSD t-test were used for statistical analysis. In all analyses, p < 0.05 was set as statistically significant.
Results
Apigenin-reduced Cell Viability in SGC-7901 Cells and Caused G2/M Phase Cell Cycle Arrest
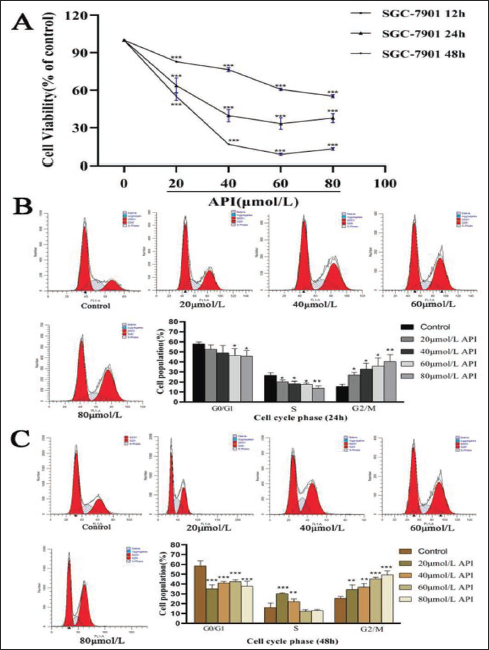
To prove our previous finding indicating that apigenin can induce cell suppression in gastric cancer cells, the cells were exposed to diverse concentrations of apigenin for 12, 24, or 48 h. Cell viability was assessed by CCK-8 kit. As illustrated in Figure 1A, the viability of the cells was dramatically decreased with increasing concentration and time after being exposed to 20–80 µmol/L apigenin for 48 h. This effect peaked when the cells were exposed to 60 µmol/L apigenin for 48 h. The cell viabilities of diverse dose apigenin were 82.87% ± 0.45, 76.49% ± 1.63, 60.68% ± 0.98, and 55.2% ± 1.33 for 12 h (p < 0.001), while the cells treated with apigenin after 24 h, the cell viabilities were 63.78% ± 6.07, 39.69% ± 4.98, 33.28% ± 4.65, and 37.61% ± 3.62 (p < 0.001). In addition, 48 h of induction by the same apigenin concentration, the corresponding cell viability was 55.07% ± 3.24, 16.66% ± 0.37, 8.63% ± 0.9, and 13.07% ± 1.09 (p < 0.001). Further, flow cytometry analysis revealed that apigenin was found to cause a significant increase in the percentage of the SGC-7901 cell population at the G2/M phase of the cell cycle indicating cell cycle arrest (Figure 1).
Effect of Apigenin on Cell Viability Inhibition in Human Gastric Cancer SGC-7901 Cells. (A) Human Gastric Cancer SGC-7901 Cells were Exposed to Apigenin at 20, 40, 60, and 80 μmol/L for 12h, 24 h, or 48h. Cell Viability was Assessed By the CCK-8 Test. Data were from Three Independent Experiments (Vs. Control, *** p < 0.001). (B&C) SGC-7901 Cells were Exposed to Apigenin at 20, 40, 60, and 80 μmol/L for 24 h or 48 h Followed By PI Staining; Cell Cycle Distribution Analysis was Analyzed By ModFitLT.
Apigenin Promotes Apoptosis of SGC-7901 Cells
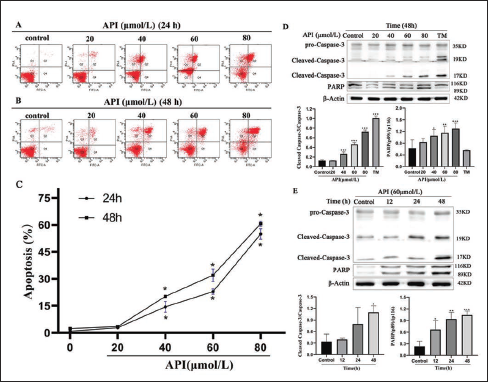
After the inhibitory effect of apigenin on the proliferation of gastric cancer cells was confirmed, flow cytometry was used to investigate whether apigenin-attracted cell suppression occurs through apoptosis. The results revealed that the apoptosis rates of different concentrations of apigenin were 2.83% ± 0.21, 14.43% ± 3.02, 22.93% ± 1.50, and 54.97% ± 2.93 for 24 h (p < 0.05), respectively. Moreover, the corresponding cell apoptosis rates were 3.5% ± 0.26, 20.2% ± 0.26, 32.03% ± 3.44, and 60.67% ± 1.47 (p < 0.05) as induced by the same apigenin concentrations for 48 h (Figure 2). In addition, the western blot assay illustrated that Caspase-3 and PARP upregulated after apigenin treatment in SGC-7901 cells (Figures 2 D & E). These data confirmed apigenin promotes apoptosis of SGC-7901 cells.
Apigenin-induced Apoptosis of Human Gastric Cancer SGC-7901 Cells. (A&B&C) SGC-7901 Cells were Exposed to Apigenin at 20, 40, 60, and 80 μmol/L for 24 h or 48h; The Apoptosis Rate was Determined By Flow Cytometry; Data were from Three Independent Experiments (Vs. Control, *p < 0.05). (D) SGC-7901 Cells were Treated with Apigenin at 20, 40, 60, and 80 μmol/L and 3 μg/ mL TM for 48 h; Cleaved-Caspase-3 and PARP Expression was Detected by Western Blotting. (E) SGC-7901 Cells were Treated with Apigenin at 60 μmol/L for 12, 24, and 48 h and were Subjected to Western Blot Analysis. Columns: Mean of Triplicate Treatments; Bars: ± SD. (Vs. Control, *p < 0.05, ** p < 0.01, *** p < 0.001).
Apigenin Activates ERS in SGC-7901 Cells
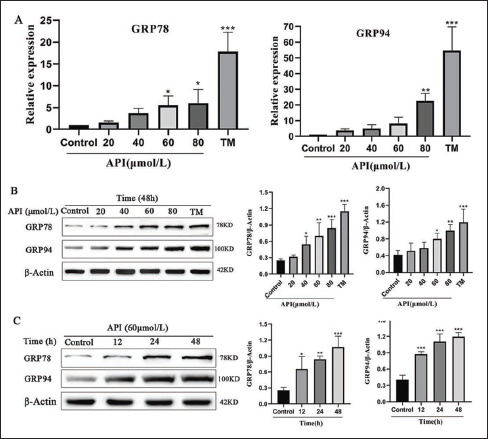
Activated ERS in tumor cells may induce apoptosis or promote cell survival. To explore whether apigenin activates ERS in SGC-7901 cells, the amounts of GRP78 and GRP94 protein expression were assessed to verify ERS. The glycosylation inhibitor tunicamycin (TM), which triggers UPR and ERS, acted as a positive control. As shown in Figure 3A, RT-qPCR results revealed that GRP78 and GRP94 mRNA expression increased gradually with increasing doses of apigenin. Subsequently, the Western blot test examined that the exposure of SGC-7901 cells to apigenin upregulated the protein expression of GRP78 dependent upon both dose and time (Figure 3). The alteration of ER chaperone GRP94 was in accord with that of GRP78 (Figure 3).
Upregulation of ER Molecular Chaperones was Induced by Apigenin. (A) SGC-7901 Cells were Treated with Apigenin at 20, 40, 60, and 80 μmol/L and 3 μg/mL TM for 48 h; The Expression of GRP78 and GRP94 was Examined by Real-time Quantitative PCR (B) SGC-7901 Cells were Treated with Apigenin at 20, 40, 60, and 80 μmol/L and 3 μg/mL TM for 48 h; The Expression of GRP78 and GRP94 was Examined by Western Blot. (C) SGC-7901 Cells were Treated with Apigenin at 60 μmol/L for 12, 24, and 48 h and were Subjected to Western Blot Analysis. Columns: Mean of Triplicate Treatments; Bars: ± SD. (Vs. Control, *p < 0.05, ** p < 0.01, *** p < 0.001).
Activation of UPR Signaling by Apigenin in SGC-7901 Cells
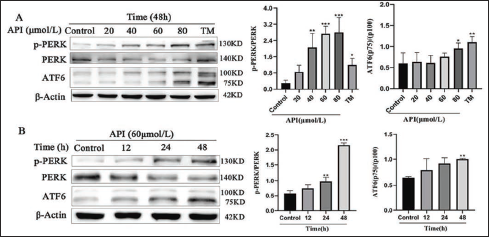
Furthermore, we detected two main signals of UPR downstream of ERS, namely, the ATF6 and PERK pathways, to investigate the effects of apigenin on UPR. Western blot analysis presented that apigenin dose- and time-dependently induced PERK phosphorylation and p50 ATF6 generation in SGC-7901 cells (Figure 4).
Apigenin-activated UPR Pathways in SGC-7901 Cells. (A) SGC-7901 Cells were Treated with Apigenin at 20, 40, 60, and 80 μmol/L or 3 μg/mL TM for 48 h; UPR Signaling was Examined by Western Blot. (B) SGC-7901 Cells were Treated with Apigenin at 60 μmol/L for 12, 24, and 48 h and were Subjected to Western Blot Analysis. Columns: Mean of Triplicate Treatments; Bars: ±SD. (Vs. Control, *p < 0.05, ** p < 0.01, *** p <.001).
UPR Induced by Apigenin Failed to Activate the ERS-associated Apoptosis Pathway
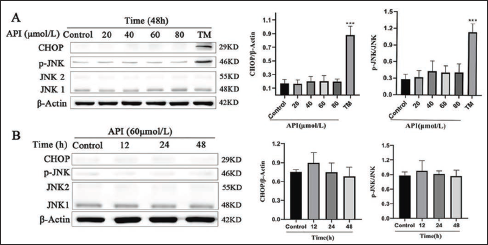
To determine whether apigenin-induced apoptosis is regulated by ERS, we investigated whether apigenin regulates the expression of ERS-associated apoptotic proteins. The Western blot analysis was used to investigate the levels of ER-associated apoptotic proteins. Results showed neither a dose- nor time-dependent manner of CHOP and the JNK protein was phosphorylated when the cells were exposed to apigenin (Figure 5).
Apigenin Failed to Activate the ER-stress-associated Apoptosis Pathway in SGC-7901 Cells. (A) SGC-7901 Cells were Treated with Apigenin at 20, 40, 60, and 80 μmol/L or 3 μg/mL TM for 48 h; The ER-stress-associated Apoptosis Pathway was Examined by Western Blot. (B) SGC-7901 Cells were Treated with Apigenin at 60 μmol/L for 12, 24, and 48 h and were Subjected to Western Blot Analysis. Columns: Mean of Triplicate Treatments; Bars: ±SD. (Vs. Control, *P < 0.05, ** P < 0.01, *** P < 0.001).
ERS Reduced Apigenin-induced Apoptosis in SGC-7901 Cells
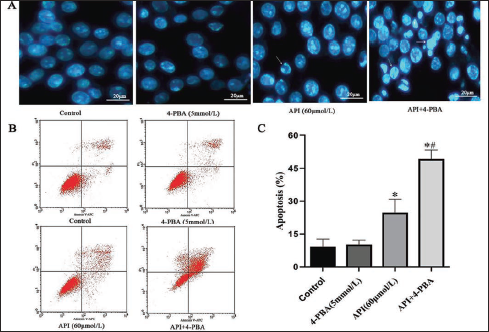
To further investigate the role of ERS in apigenin-triggered cell apoptosis, SGC-7901 cells were pretreated with 5 mmol/L 4-PBA for 2 h before apigenin (60 µmol/L) treatment. Results indicated pretreatment with the ERS inhibitor 4-PBA significantly enhanced the apigenin-induced apoptosis. There were more cells in the 4-PBA and apigenin combined group than those in the control or 4-PBA group that represented apoptosis-related morphologic alterations, such as cell chromatin condensation and crescentic change (Figure 6A). Flow cytometric analysis results also indicated that exposure to 4-PBA (5 mmol/L) or apigenin (60 µmol/L) induced apoptosis in 10.12% ± 2.08 or 24.62% ± 6.21 (p < 0.05) of the cells, correspondingly. The combination of 4-PBA and apigenin triggered apoptosis in 49.23% ± 4.08 (p < 0.05) of the cells (Figure 6). Apoptosis was more effectively activated by apigenin when 4-PBA and apigenin were combined than when each agent was used alone. The above findings suggest that ERS and ATF6/PERK signaling protect human gastric cancer cells from apigenin-induced apoptosis.
ERS Inhibition Enhances Apigenin-induced Apoptosis in SGC-7901 Cells. (A) SGC-7901 Cells were Exposed to 4-PBA or Apigenin Alone or in the Presence of 5 mmol/L 4-PBA and 60 μmol/L Apigenin for 24 h; The Cells were Stained with DAPI and Then Visualized Under a Confocal Microscope (400×). (B) SGC-7901 Cells were Treated as in (A); The Cells were Stained with Annexin V-FITC/PI and Then Detected by Flow Cytometry Analysis. (C) Columns: Mean of Triplicate Treatments; Bars: ± SD. (Vs. Control, *p < 0.05; Vs. Apigenin Group, # p < 0.05).
Discussion
Plant extracts are remarkable sources for drug development and discovery, and several new chemotherapeutic agents are discovered and designed using plant-derived compounds (Ağagündüz et al., 2022; Küpeli Akkol et al., 2020). Apigenin is a nontoxic dietary flavonoid that exists in many herbs, including chamomile, parsley, peppermint, thyme, and horsetail herb (Kim et al., 2019). Apigenin reportedly exerts antioxidant, anti-inflammatory, and antidepressant activities, it also exerts antitumor effects by multiple pathways (Salehi et al., 2019). However, apigenin’s effects on apoptosis have received conflicting reports. Apigenin induces apoptosis of human gastric cancer cells by activating the mitochondrial signaling pathway (Chen et al., 2014). But some studies have found that apigenin restores cell viability in PC12 cell apoptosis mediated by 4-HNE (Wu et al., 2015). This suggests that apigenin has an antioxidant effect and can reduce the antioxidant damage of some cells to reduce cell apoptosis. That means apigenin has broad prospects and is particularly attractive in the exploitation of emerging drugs for cancer prevention and treatment. In the present study, we assessed the potential cancer-fighting ability of apigenin and explored its underlying mechanism of action in human gastric cancer SGC-7901 cells.
To determine whether apigenin inhibits the viability of human gastric cancer cells, the CCK-8 assay was performed, which demonstrated that apigenin markedly reduced the viability of the cells with exposure time and concentration (Figure 1A). The viability inhibition effect was more than 90% when the cells were exposed to 60 µmol/L apigenin for 48 h. Other apigenin concentrations also suppressed cell viability to different degrees. Our research on cell cycle arrest shows apigenin-induced cycle arrest in cells at the G2/M phase. Similar results were obtained in AGS human gastric cancer cells (Kim et al., 2020). These illustrate that apigenin can prevent cell proliferation by causing G2/M phase arrest in human gastric carcinoma cells.
We provided evidence that apigenin kills the SGC-7901 cells through apoptosis, as documented via flow cytometry (Figure 2). Fluorescence-assisted cell sorting analysis and immunoblotting analysis revealed that apigenin promoted the increase of apoptotic cells and up-regulated the expression of apoptotic proteins with exposure time and concentration (Figure 2). The results indicate that apigenin induces gastric cancer cells apoptosis.
Major cellular organelles participate in apoptosis signal pathways. The significance of ER in cell death processes has been underlined by recent research (Chen et al., 2020). In the current study, the involvement of UPR and ERS in apigenin-triggered apoptosis of human gastric carcinoma SGC-7901 cells was investigated.
In eukaryotic cells, secretory proteins and transmembrane proteins are synthesized and folded predominantly in ER. The protein flow in ER may change significantly according to physiological state and environmental conditions. Unfolded proteins store up in the ER lumen and cause ERS when lots of proteins flow into the ER (Bao & Bassham, 2020). GRP78 and GRP94 are vital proteins associated with the ERS response and are also markers of ERS (Zhang et al., 2019). When the function and homeostasis of the ER were disrupted by the physiological stress, the GRP78 and GRP94 expression increases, thereby protecting the organ or tissue from damage, they can also protect cells against death (Zhu & Lee, 2015). Our experiment uncovered that GRP78 and GRP94 were translationally induced by apigenin (Figure 3). These results provided evidence that apigenin possibly triggers ERS in SGC-7901 cells.
Cells have developed UPR signaling pathways to maintain ER homeostasis. Signal transductions during ERS are commonly mainly conducted by ATF6, IRE1, and PERK, they are ERS sensors. PERK activation can phosphorylate the eIF2α, attenuating cell translation levels (Zhou et al., 2020). Moreover, When the ERS occurs, ATF6 will be transported to the Golgi apparatus, in which ATF6 is sequentially cleaved. The cytoplasmic domain is released following this cleavage, and it moves to the nucleus, where it activates genes containing an ERS response element (Yuan et al., 2020). After performing Western blotting, we revealed that apigenin exposure caused PERK phosphorylation and ATF6 activation and that their expression increased with exposure time and concentration (Figure 4). The findings suggested that apigenin may cause at least two UPR pathways to activate in SGC-7901 cells.
As a signal pathway to alleviate ERS, UPR can protect cells. When considerably harsh and drawn-out ERS occurs, apoptosis is triggered to obliterate the injured cells. Nevertheless, cell survival under mild stimuli maintains normal physiological functions by inducing UPR (Chen & Cubillos-Ruiz, 2021). CHOP has been identified as a pivotal regulator of ERS-triggered apoptosis, which is activated under the induction of ATF6 and PERK (Hu et al., 2018; Li et al., 2020). The most typical apoptotic pathway of ERS is the CHOP signaling pathway. Activation of the JNK pathway is another apoptotic mechanism in ERS that may conduce to apoptosis in answer to other stimuli (Park et al., 2020). Our results showed that apigenin failed to enhance CHOP activation and had no effect on JNK activation as well (Figure 5). Results manifested that the ERS activation of apoptosis pathways did not participate in the apigenin-triggered apoptosis of SGC-7901 cells.
Furthermore, we used 4-PBA, a particular ERS inhibitor, to examine the characteristics of ERS in apigenin against SGC-7901 cells. Results showed that 4-PBA exerted no influence on the apigenin-triggered apoptosis of SGC-7901 cells (Figure 6). Under the in vitro application of apigenin, ERS blockage by 4-PBA enhanced SGC-7901 cell apoptosis (Figure 6). The augmentation of tumor cell death by 4-PBA treatment correlated with the capabilities of this reagent to abolish the process of ERS. All these suggested that apigenin-induced ERS and UPR were used as adaptations in SGC-7901 cells. It is suggested that the combination of 4-PBA and apigenin would be a potential chemotherapeutic strategy against gastric cancer. Recent studies have also revealed that some flavonoids alone show slightly lower antitumor activity, but in combination with chemotherapy drugs exhibit synergistic effects (Fernández et al., 2021).
Conclusion
To summarize, our research showed that apigenin triggered ERS and UPR in human gastric cancer cells. Such phenomena counteracted the anti-cancer effects of apigenin. This study afforded new opinions into the apigenin-induced cell death mechanism and further evidence for using combined apigenin and ERS inhibitors as a promising cancer chemotherapy strategy.
Footnotes
Abbreviations
CCK-8: Cell counting kit 8; RT-qPCR: Real-time quantitative polymerase chain reaction.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Funding
This work was supported by the Youth Talent Promotion Program of School of Public Health, North China University of Science and Technology (QNRC202305) to Liying Hou.
