Abstract
This study aims to evaluate the cytotoxicity and potential mechanisms of 2,4,6-trichlorophenol (2,4,6-TCP) in mouse embryonic fibroblasts. Our results show that 2,4,6-TCP causes morphological changes and reduces cell viability. The overproduction of reactive oxygen species, the upregulation of nuclear factor-E2-related factor 2 (Nrf2) and heme oxygenase 1 (HMOX1) messenger RNA (mRNA) expressions, and the nuclear translocation of Nrf2 protein demonstrate that 2,4,6-TCP induces oxidative stress, and the Nrf2/HMOX1 pathway might be involved in 2,4,6-TCP-induced antioxidative response. Simultaneously, our data also demonstrate that 2,4,6-TCP upregulates the expressions of binding immunoglobulin protein, inositol-requiring enzyme/endonuclease 1α, and C/EBP homologous protein; stimulates α subunit of eukaryotic translation initiation factor 2 phosphorylation; and induces the splicing of Xbp1 mRNA, suggesting that endoplasmic reticulum (ER) stress is triggered. Moreover, 2,4,6-TCP alters the mitochondrial membrane potential and increases the apoptosis rate, the caspase 3 activity, and the Bax/Bcl-2 ratio, demonstrating that the mitochondrial pathway is involved in the 2,4,6-TCP-induced apoptosis. Thus, these results show that 2,4,6-TCP induces oxidative stress, ER stress, and apoptosis, which together contribute to its cytotoxicity
Introduction
Chlorophenols (CPs) are persistent environmental toxic compounds that are widely used as insecticides 1 and other pharmaceutical intermediates. 2 These pollutants represent serious risks to the animal and/or human health. 3 2,4,6-Trichlorophenol (2,4,6-TCP), one of the most abundant CPs, has been categorized as a potential human carcinogen by the International Agency for Research on Cancers and the World Health Organization. 4 Although there are few studies focusing on the genotoxic effects of 2,4,6-TCP, 5 -7 the exact mechanism for 2,4,6-TCP cytotoxicity is still poorly understood.
The involvement of oxidative stress in the cytotoxicity of many environmental contaminants has been reported in the past. 8 -10 Induced overproduction of reactive oxygen species (ROS) by 2,4,6-TCP 11 suggests that this trichlorophenol may disturb the cellular redox homeostasis. Still, the potential molecular mechanisms and signaling pathways involved in 2,4,6-TCP-induced oxidative stress are not completely understood. Heme oxygenase 1 (HMOX1), a nuclear factor-E2-related factor 2 (Nrf2) target gene, is a ubiquitous stress-inducible gene playing a protective role against oxidative stress in cells and tissues. 12 -15 Under normal conditions, Nrf2 is kept in the cytoplasm by Keap-1 but dissociates from Keap-1 and translocates into the nucleus upon oxidative stress. In the nucleus, Nrf2 binds to the antioxidant response element in the promoter of several antioxidant genes, including HMOX1. 16,17 Hence, the Nrf2/HMOX1 pathway is believed to be an important cellular mechanism to protect against oxidative stress-induced cytotoxicity. 18 -20
The induction of endoplasmic reticulum (ER) stress is regarded as a cellular characteristic of the cytoxicicity response by many pollutants. 21 -23 The ER stress is regulated by the unfolded protein response via the double-stranded RNA-activated protein kinase-like ER kinase (PERK), inositol-requiring enzyme/endonuclease 1α (IRE1α), and activating transcription factor 6 (ATF6) signaling pathways. 24 Upon ER stress, binding immunoglobulin protein (Bip) dissociates from PERK, IRE1α, and ATF6, allowing these 3 proteins to activate downstream signaling molecules, which will result, in turn, in the restoration of a normal ER function. However, if a proper protective response to ER stress fails, apoptosis signals will be activated to induce cell death. 25,26
Apoptosis can rise after cell exposure to both endogenous and exogenous stresses, including exposure to toxic pollutants. 8,23,27 Apoptosis is characterized by cell shrinkage, the decrease in mitochondrial membrane potential (MMP), chromatin condensation, nuclear DNA fragmentation, the release of apoptotic factors, and the activation of caspases. 28 -30 To date, apoptosis has been shown to occur following exposure to 2,4-drichlorophenol, 2,4,5-trichlorophenol, 2,3,5-trichlorophenol, and pentachlorophenol. 31 -34 To the best of our knowledge, only 1 study mentioned 2,4,6-TCP-induced apoptosis in Vero cells. 35 For all these reasons, it is important to better understand the cellular processes involved in both cell toxicity and apoptosis of the widespread pollutant 2,4,6-TCP.
In this study, mouse embryonic fibroblasts (MEFs) were used as a model to study the cytotoxic effects and molecular mechanisms of 2,4,6-TCP in oxidative stress, ER stress, and apoptosis. Changes in ROS levels as well as the expression of both Nrf2 and HMOX1 were examined to better understand the effect of 2,4,6-TCP on oxidative stress. The ER stress response to 2,4,6-TCP was monitored by detecting expressions of ER stress related-genes and ER stress related-protein levels. The impact of 2,4,6-TCP on cell death was examined by assessing apoptosis levels, caspase 3 activity, MMP, and the Bax/Bcl-2 ratio.
Materials and Methods
Cell Culture
The MEFs were from Dr William Bonner’s laboratory (National Institutes of Health [NIH], Maryland, USA). Cells were cultured in high glucose Dulbecco’s modified Eagle's medium (DMEM) medium (Gibco, New York, USA) supplemented with 10% fetal bovine serum at 37°C in a humidified atmosphere containing 5% CO2. Experiments were performed using 70% to 80% of cell confluency with viability ≥ 90% determined by the trypan blue staining.
Reagents and Antibodies
2,4,6-Trichlorophenol (purity > 98%), carbonyl cyanide m-chlorophenyl hydrazone (purity > 97%), tunicamycin (Tu, purity > 95%), and dimethyl sulfoxide (DMSO, purity > 99.9%) were obtained from Sigma Aldrich (St Louis, Missouri). A stock solution of 2,4,6-TCP 1.0 mol/L was prepared using DMSO. 2,4,6-Trichlorophenol was further diluted to 0.5, 0.75, and 1.0 mmol/L in DMEM. The DMSO concentration in all cell cultures was kept below 0.1%, with no detectable effects on cell in our preliminary experiments. The Nrf2 and β-actin antibodies were obtained from Santa Cruz Biotechnology (California, USA). The Bip, phosphorylation of α subunit of eukaryotic translation initiation factor 2 (p-eIF2α), and C/EBP homologous protein (CHOP) antibodies were purchased from Cell Signaling (Beverly, Massachusetts). All other chemicals used in our study were of analytical grade and were purchased from local companies.
Cell Viability-3-(4,5-Dimethylthiazol-2-yl)-2,5-diphenyl Tetrazolium Bromide Assay
Cell viability was detected by the colorimetric 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay. Briefly, MEFs seeded into 96-well plates with a density of 8000 cells per well were allowed to attach overnight. After exposure of MEFs to 2,4,6-TCP (0, 0.25, 0.5, 0.75, and 1.0 mmol/L) for 24 hours, the media were removed, and MTT was added to each well at a final concentration of 0.5 mg/mL and further incubated for 4 hours at 37°C. The media were then removed, and DMSO (150 μL) was added to dissolve the formazan crystals. The absorbance was detected at 490 nm using a microplate reader (Sunrise; Tecan, Austria). Each assay involved 8 wells per condition and was performed in triplicate (n = 3).
Annexin V-Fluorescein Isothiocyanate and Propidium Iodide Staining
Apoptosis assay was performed with annexin V (AV)-fluorescein isothiocyanate (FITC)/propidium iodide (PI) Apoptosis Detection Kit (BestBio, China) following the manufacturer’s instructions after cells were treated with 2,4,6-TCP (0, 0.5, 0.75, and 1.0 mmol/L) for 24 hours. In brief, cells were collected, washed with cold phosphate-buffered saline (PBS), resuspended in 500 μL 1 × AV-FITC binding buffer containing 5 μL AV-FITC and 5 μL PI, and incubated for 15 minutes at room temperature in the dark. Samples were subjected to flow cytometric analysis using a BD LSRFortessa (BD Bioscience, New Jersey, USA). This experiment was repeated at least 3 times. The results were expressed as mean ± standard deviation (SD) in the present study (n = 3).
Caspase 3 Activity Determination
The activity of caspase 3 was determined according to Caspase 3 Activity Assay Kit (Beyotime, China) to assess the effect of 2,4,6-TCP on apoptosis. Following exposure of MEFs to 1.0 mmol/L 2,4,6-TCP for different time point, approximately 2 × 106 cells were lysed in ice-cold lysis buffer for 15 minutes in an ice bath. The reaction mixture contained assay buffer, samples, and Ac-DEVD-pNA (acetyl-Asp-Glu-Val-Asp p-nitroanilide; 2 mmol/L) as substrate. After incubation, the enzymatic activity of caspase 3 was determined at 405 nm using a microplate reader (Sunrise; Tecan). The results of each treatment were pooled from 3 replicate samples derived from 3 independent experiments.
Reactive Oxygen Species and MMP Detection
The ROS and MMP were monitored with dichlorodihydrofluorescein diacetate (DCFH-DA) and 5,5,6,6-Tetrachloro-1,1,3,3-tetraethylbenzimidazolylcarbocyanine iodide (JC-1; Beyotime, China), respectively. In brief, following exposure to 2,4,6-TCP (1.0 mmol/L for 1, 2, 4 and 6 hours; 0.5, 0.75, and 1.0 mmol/L for 24 hours), the cells were harvested, washed, and incubated with JC-1 and DCFH-DA stain buffer, respectively, at 37°C for 30 minutes in darkness. After washing of cells with PBS 3 times, JC-1 and DCFH-DA fluorescence intensity were assessed by flow cytometry. The data were recorded for 10 000 cellular events per sample. The results of each sample were pooled from 3 replicate samples derived from 3 independent experiments.
Reverse Transcription-Polymerase Chain Reaction Analysis of Xbp1 messenger RNA Splicing
After exposure of MEFs to 2,4,6-TCP (0, 0.5, 0.75, and 1.0 mmol/L) for 12 hours, total RNA was isolated using RNAiso Plus (TaKaRa, Japan) according to the manufacturer’s protocol. The splicing of Xbp1 messenger RNA (mRNA) was examined by reverse transcription-polymerase chain reaction (RT-PCR) analysis as described previously. 36 The PCR amplification of Xbp1 mRNA was performed using Premix Taq (TaKaRa Taq Version 2.0 plus dye; TaKaRa) according to the manufacturer’s instructions (98°C 5 minutes, 98°C 30 seconds-55°C 30 seconds-72°C 30 seconds 35 cycles, and 72°C 5 minutes). The primer pair of Xbp1 is shown in Table 1. The PCR products were separated by electrophoresis on a 3% agarose gel (Invitrogen, California, USA), visualized by ethidium bromide staining, and the density of each band was semiquantified using ImageJ 1.45s (NIH). The data from replicate pools were depicted as mean ± SD (n = 3).
Primers of Detected Genes.
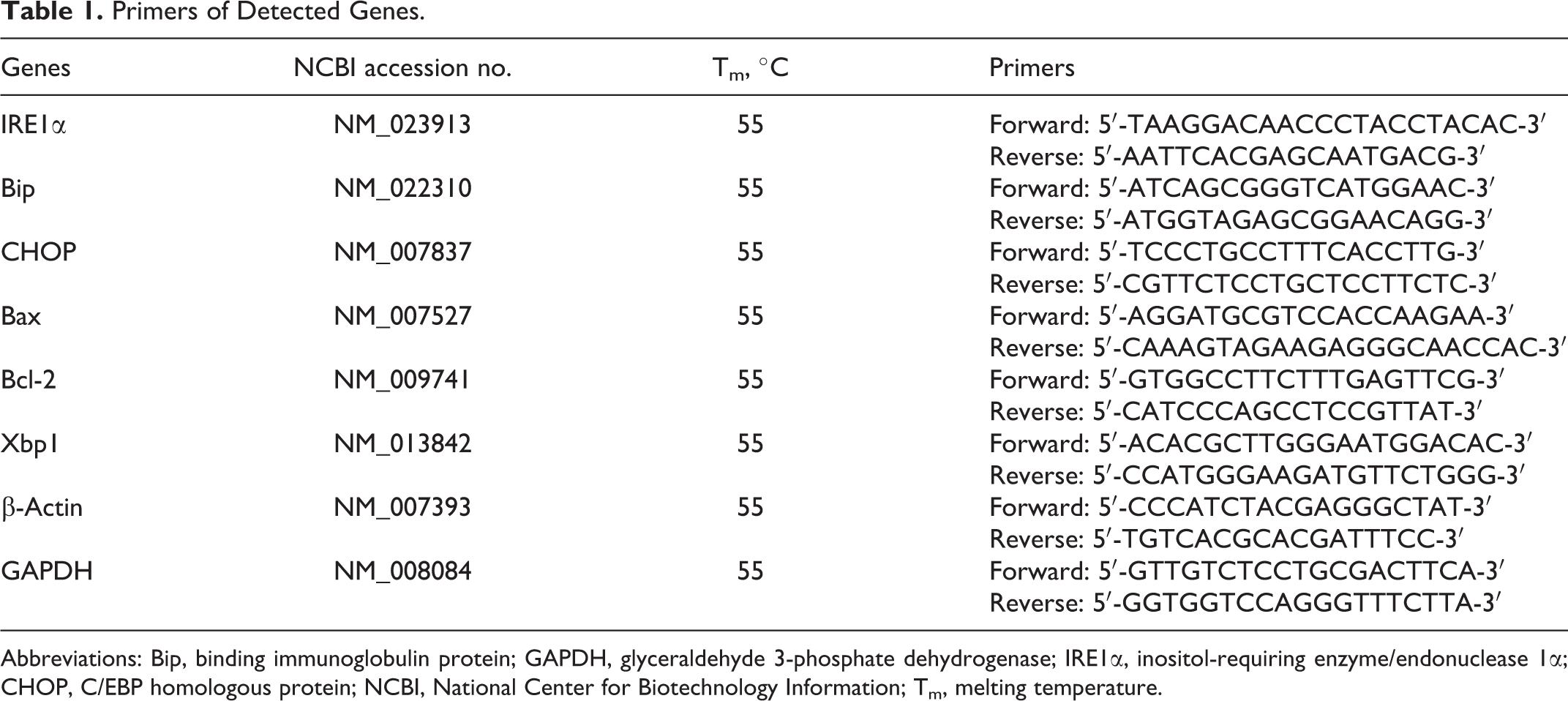
Abbreviations: Bip, binding immunoglobulin protein; GAPDH, glyceraldehyde 3-phosphate dehydrogenase; IRE1α, inositol-requiring enzyme/endonuclease 1α; CHOP, C/EBP homologous protein; NCBI, National Center for Biotechnology Information; Tm, melting temperature.
Real Time Quantitative PCR Assay
Following exposure of MEFs to 2,4,6-TCP (0, 0.5, 0.75, and 1.0 mmol/L) for 12 hours, quantitative PCR (qPCR) was performed with the CFX96 real time fluorescent qPCR instrument (Bio-Rad Laboratories, California, USA) using SYBR Premix DimerEraser (TaKaRa) according to the manufacturer’s instructions. The expression levels of β-actin and glyceraldehyde 3-phosphate dehydrogenase were used as internal controls to normalize the related gene expression levels. A melting curve was added in order to check the specificity of PCR products. Each sample was run 3 replicates, and the qPCR assay experiment was repeated at least 3 times (n = 3) to obtain a good reproducibility. All primers were purchased from GENEWIZ, Inc (Suzhou, China). The primer sequences used for qPCR are shown in Table 1.
Immunofluorescence Staining
The nuclear translocation of Nrf2 was immunocytochemically detected in the cells that were first attached to the coverslips. The MEFs were treated with 0.5 mmol/L 2,4,6-TCP for 12 hours, and then the medium was aspirated. Then the cells were washed with PBS and fixed in ice-cold 100% methanol for 15 minutes at −20°C. After washing with PBS 3 times, the cells were incubated with blocking butter (PBS containing 5% BSA and 0.3% Triton-100) for 60 minutes at room temperature. The cells were first treated with anti-Nrf2 antibody (1:200) overnight at 4°C and subsequently incubated with FITC-labeled goat antirabbit immunoglobulin G (Invitrogen) for 60 minutes at 25°C. Then the fluorescence intensity was monitored with an Axio Observer Z1 fluorescence microscopy (Zeiss, Germany). The experiment was repeated at least 3 times.
Western Blotting Analysis
After exposure of MEFs to 2,4,6-TCP (0.5, 0.75, and 1.0 mmol/L), the cells were harvested from the culture medium and lysed in radioimmunoprecipitation assay buffer containing 1 mmol/L PMSF (Beyotime, China). The isolated proteins were subjected to sodium dodecyl sulfate polyacrylamide gel electrophoresis and transferred to a polyvinylidene difluoride membrane. The membrane was blocked in 5% nonfat milk or BSA containing Tris-buffered saline and Tween 20 buffer at room temperature for 1 to 2 hours, probed using the specific primary antibody (anti-Bip, anti-CHOP, or antiphospho-eIF2α, respectively) overnight at 4°C and then incubated with the secondary antibody at room temperature for 1 hour. β-Actin was used as a loading control. Western blot detections were achieved by chemiluminescence (Thermo Scientific, Rockford, Illinois), and quantitative analyses were performed using the Quantity One software (Bio-Rad). The Western blots shown are representative of 3 independent experiments.
Statistical Analysis
Statistical significance was determined by 1-way analysis of variance followed by least significant difference multiple comparison tests (SPSS Statistics, Version 19.0). A probability of value of
Results
2,4,6-Trichlorophenol Decreased Cell Viability
2,4,6-Trichlorophenol toxicity to MEFs was tested by MTT assay after cells were treated with increasing doses of the compound (0.25-1.0 mmol/L) for 24 hours. Exposure to 2,4,6-TCP lowered cell viability in a dose-dependent manner (Figure 1A). Cell viability was decreased by 13.88%, 21.93%, and 31.01% following a 24-hour treatment with 0.5, 0.75, and 1.0 mmol/L, respectively. Furthermore, as shown in Figure 1B, cells showed significant morphological changes, including cellular shrinkage and cell rounding after being exposed to 2,4,6-TCP for 24 hours.
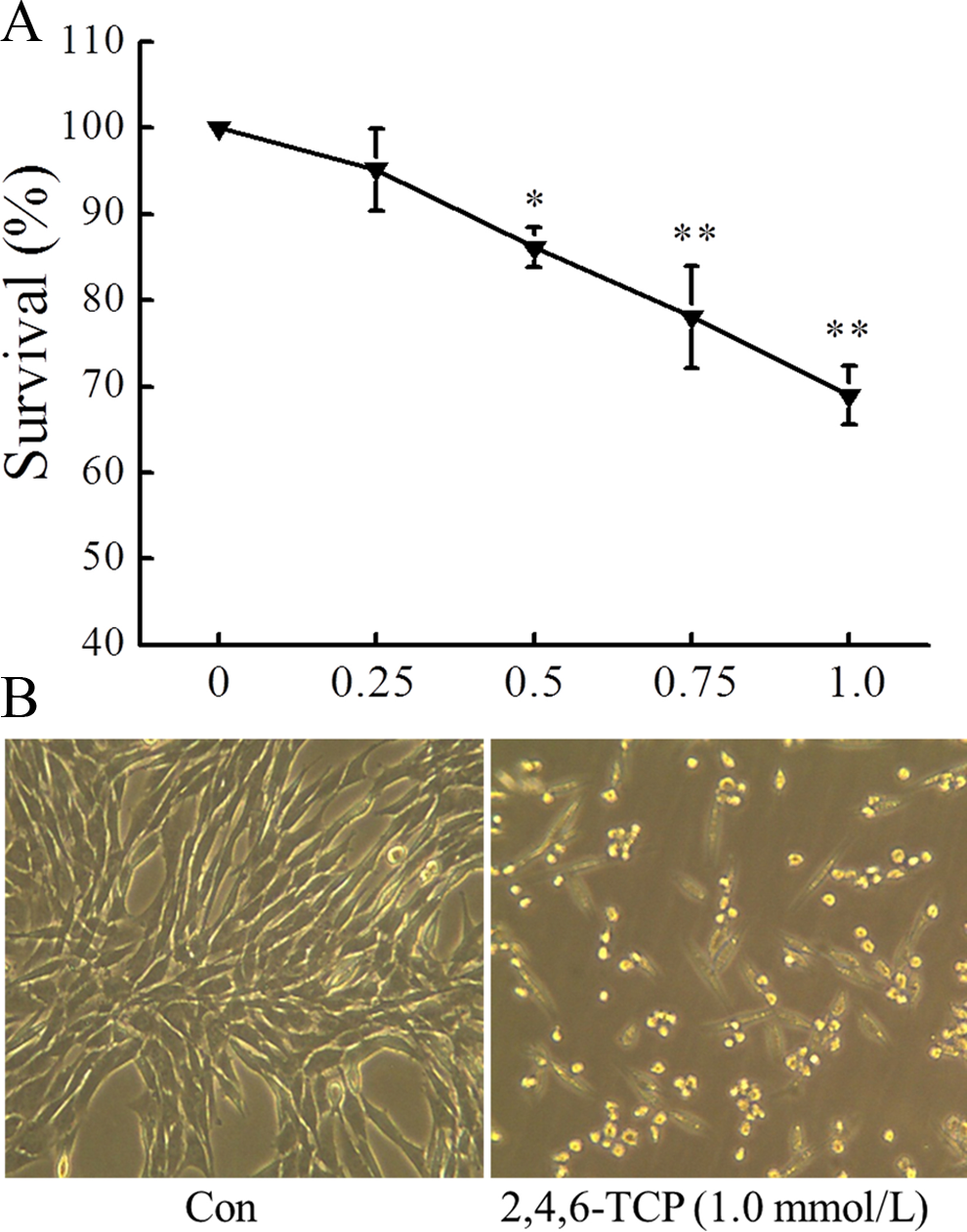
Effect of 2,4,6-TCP on the viability and morphology of MEFs. A, Cell viability was determined by the 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl tetrazolium bromide (MTT) assay after cells were exposed to 2,4,6-TCP for 24 hours. Cell viability was expressed as percentage compared to the control with the viability of control cells designated as 100%. B, Morphological changes in MEFs were observed under microscope after 2,4,6-TCP treatment for 24 hours. Data were presented as mean ± standard deviation (SD; n = 3). *
2,4,6-Trichlorophenol-Induced Oxidative Stress
The MEFs exposure to 2,4,6-TCP (1.0 mmol/L) induced a biphasic ROS production, with an early ROS burst occurring within 1 hour of exposure followed a transient drop within 1 to 2 hours, and then, a second ROS burst occurring within 2 to 5 hours (Figure 2A). As shown in Figure 2B and 2C, 2,4,6-TCP significantly increased HMOX1 and Nrf2 mRNA expressions (
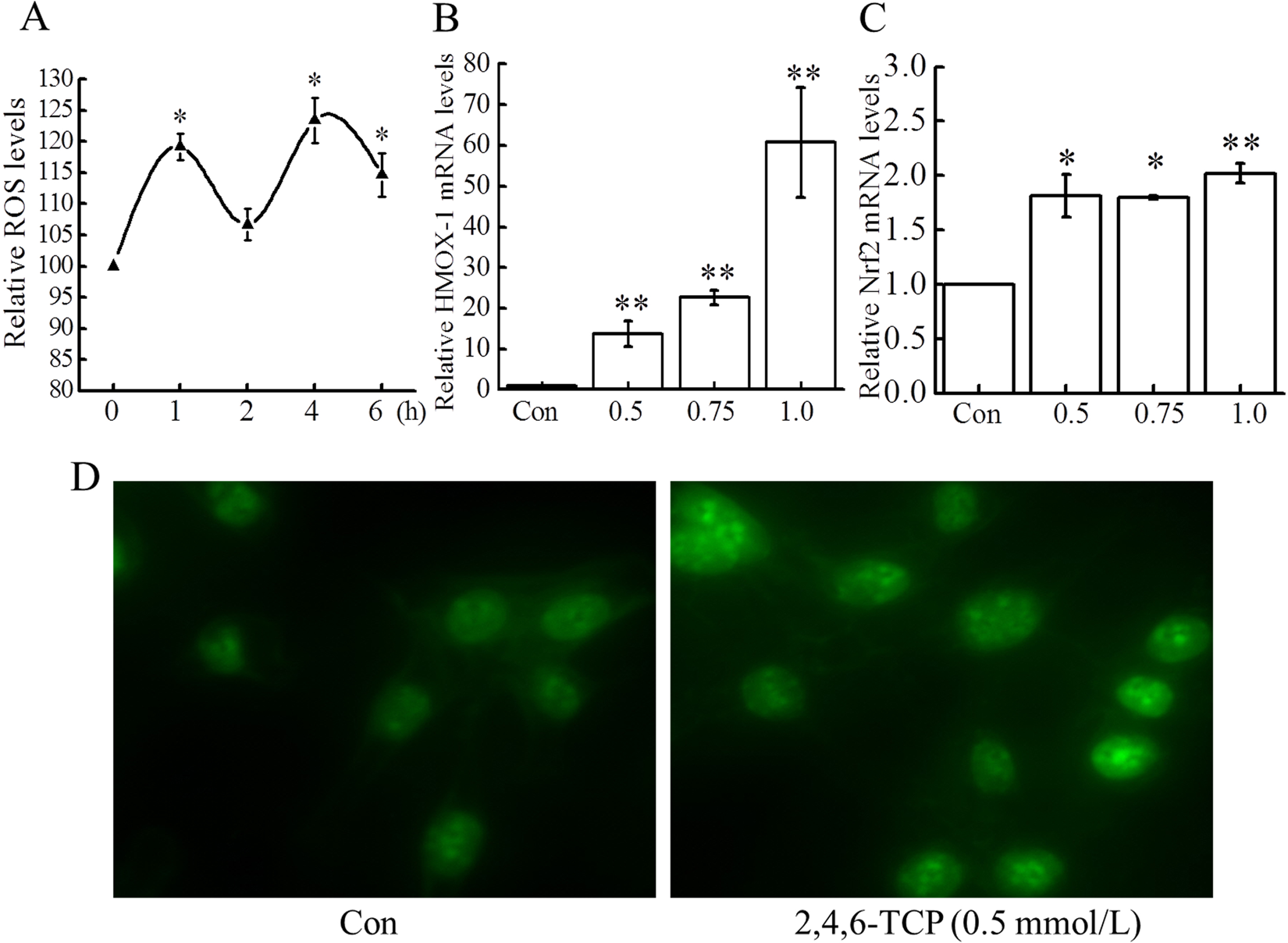
Effect of 2,4,6-TCP on oxidative stress. A, Effect of 2,4,6-TCP on ROS. Following mouse embryonic fibroblasts (MEFs) exposure to 2,4,6-TCP (1.0 mmol/L) for different times, ROS was monitored with dichlorodihydrofluorescein diacetate (DCFH-DA) by flow cytometry. B and C, Effect of 2,4,6-TCP on the mRNA expressions of HMOX1 and Nrf2. After 12 hours of incubation with 2,4,6-TCP (0.5, 0.75, and 1.0 mmol/L), RNA was prepared and the mRNA expressions of Nrf2 and HMOX1 were measured by quantitative polymerase chain reaction (qPCR) and normalized with β-actin and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). D, Effect of 2,4,6-TCP on nuclear translocation of Nrf2 protein. Nrf2 protein expression was detected by immunofluorescence staining as described in Reverse Transcription-Polymerase Chain Reaction Analysis of Xbp1 messenger RNA Splicing section. Nrf2 protein expression was increased in the nucleus, which indicated nuclear translocation of Nrf2 after exposure of cells to 2,4,6-TCP (0.5 mmol/L) for 12 hours. All experiments were repeated at least 3 times. Data were presented as mean ± standard deviation (SD; n = 3). *
2,4,6-Trichlorophenol-Induced ER Stress
To uncover whether 2,4,6-TCP could induce ER stress, mRNA expressions of ER stress-related genes (IRE1α, Bip, and CHOP) were determined by qPCR analysis. As shown in Figure 3, IRE1α, Bip, and CHOP mRNA levels dramatically increased after 12 hours treatment with 2,4,6-TCP. In addition, spliced Xbp1 mRNA, an indicator for IRE1α activation, was found to be increased drastically in cells treated with 2,4,6-TCP for 12 hours. As shown in Figure 3D, Xpb1u mRNA was significantly cut to Xbp1s mRNA in a concentration-dependent manner (
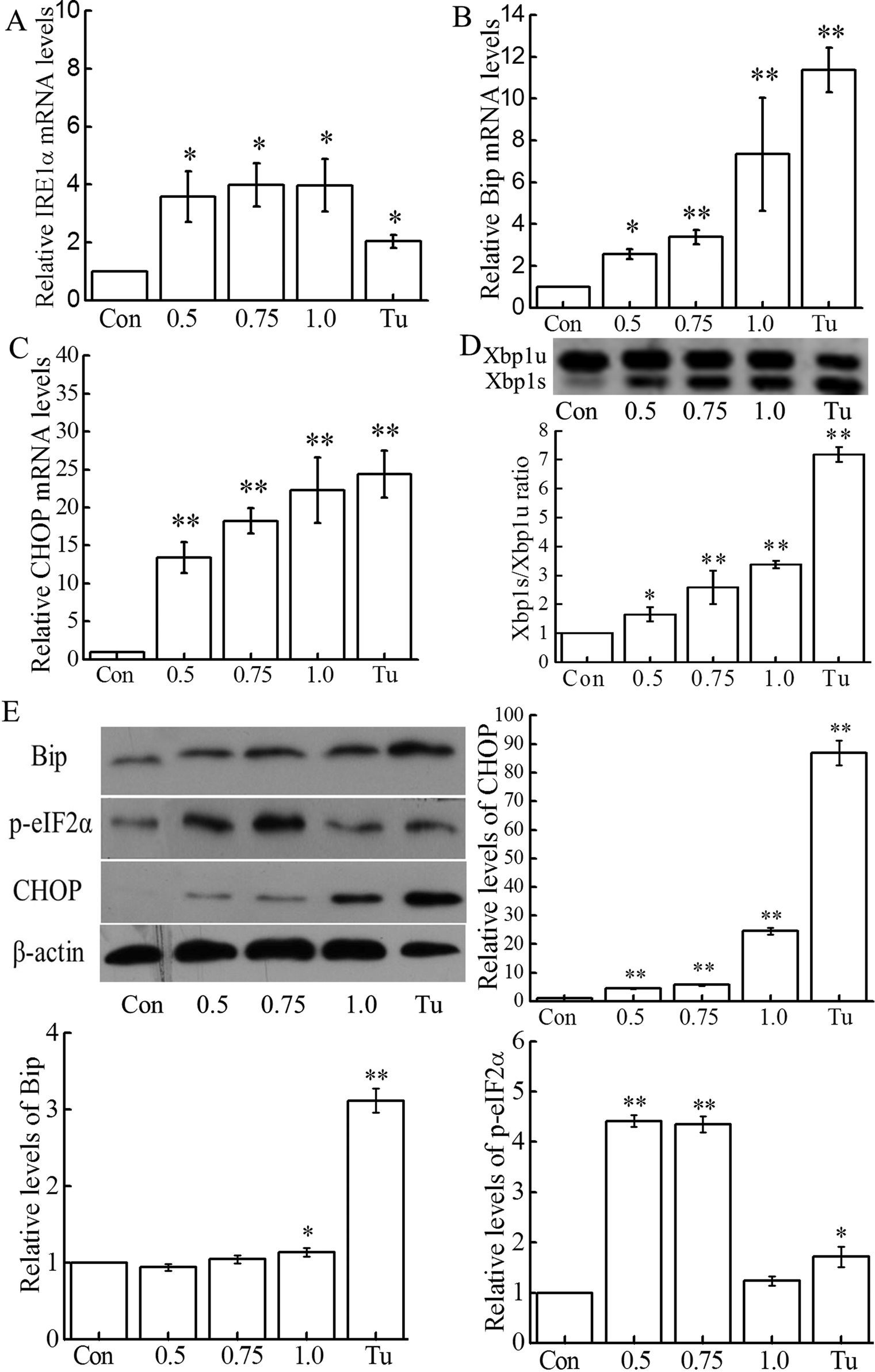
Effect of 2,4,6-TCP on endoplasmic reticulum (ER) stress. A, B, and C, After 12 hours of incubation with 2,4,6-TCP (0.5, 0.75, and 1.0 mmol/L), RNA was prepared and the mRNA expressions of inositol-requiring enzyme/endonuclease 1α (IRE1α), Bip, and CHOP were measured by quantitative polymerase chain reaction (qPCR) and normalized with β-actin and glyceraldehyde 3-phosphate dehydrogenase (GAPDH). D, The levels of Xbp1u and Xbp1s mRNA were measured using reverse transcription-polymerase chain reaction (RT-PCR) after exposure of mouse embryonic fibroblasts (MEFs) to 2,4,6-TCP for 12 hours. E, Equal amounts of total protein (about 30 μg) from 2,4,6-TCP treated for 12 hours and control cells were prepared for Western blotting analysis of Bip, CHOP, and p-eIF2α. β-Actin was used as a loading control. The blots are representative of 3 independent experiments, and their statistical results are presented in histograms. Cells treated with tunicamycin (Tu; 2.0 μg/mL) for 12 hours served as a positive control. All experiments were repeated at least 3 times. Data were presented as mean ± standard deviation (SD; n = 3). *
In order to confirm 2,4,6-TCP-induced ER stress, we analyzed the protein expressions of Bip and CHOP, as well as the level of p-eIF2α by Western in MEFs. Indeed, 2,4,6-TCP exhibited a dose dependent, stimulating effect on the expression of the CHOP proteins. However, the Bip expression was upregulated only by 1.0 mmol/L 2,4,6-TCP (
2,4,6-Trichlorophenol-Induced Apoptosis
Mouse embryonic fibroblasts were exposed to increasing concentrations of 2,4,6-TCP ranging from 0.5 to 1.0 mmol/L, and apoptosis was assayed after 24 hours by flow cytometry with the AV/PI staining. As shown in Figure 4A and B, apoptosis was increased when cells were treated by 0.75 and 1.0 mmol/L 2,4,6-TCP (
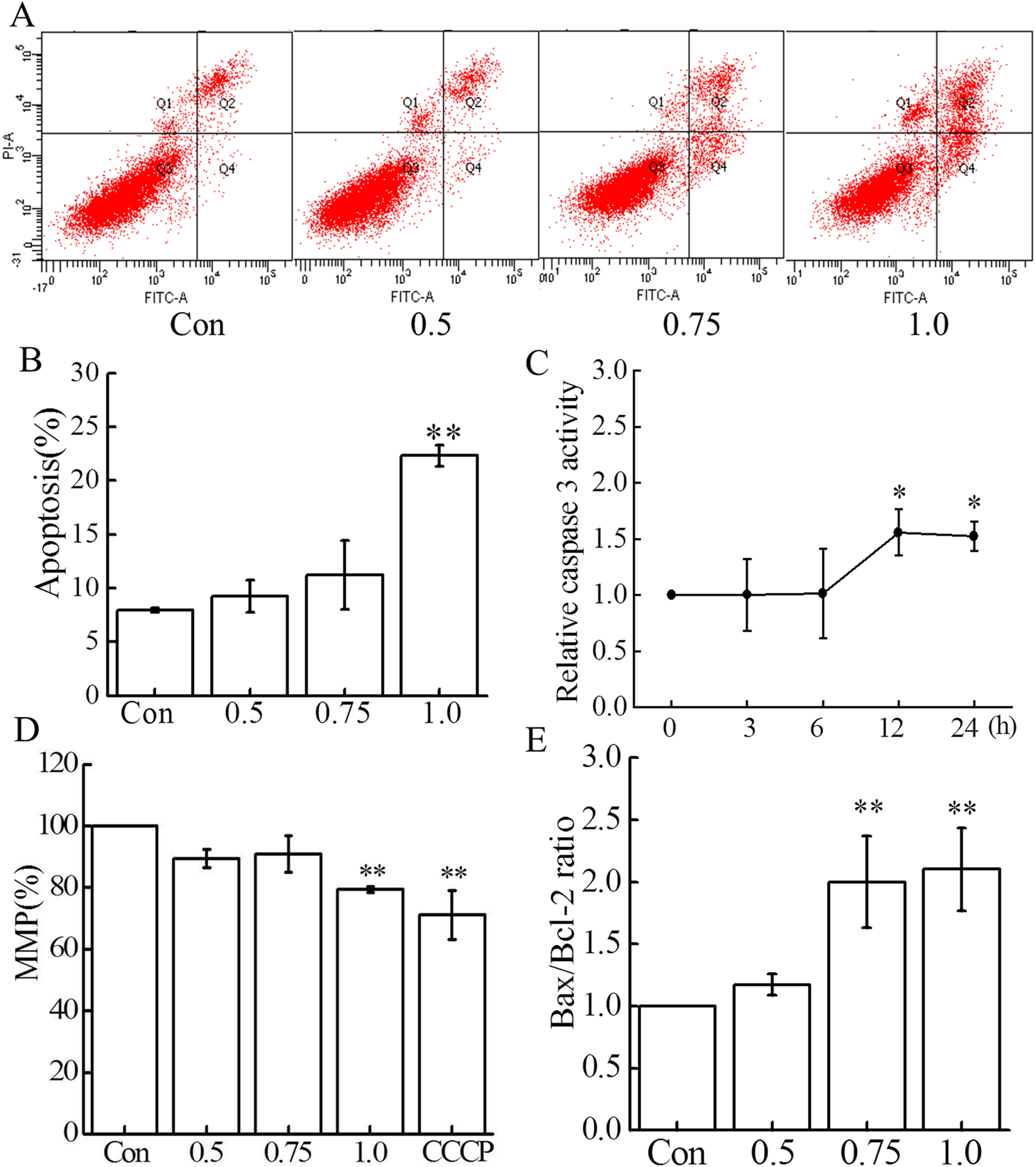
Effect of 2,4,6-TCP on apoptosis. A and B, The cells were exposed to 2,4,6-TCP (0.5, 0.75, and 1.0 mmol/L) for 24 hours, and apoptosis was detected with annexin V (AV)/propidium iodide (PI) staining by flow cytometry. Data presented in panel (A) are a representative of 3 independent experiments, and their statistical results are presented in panel (B). C, Effect of 2,4,6-TCP on caspase 3 activity. The cells were exposed to 2,4,6-TCP (1.0 mmol/L) for different time points, caspase 3 activity was analyzed with caspase 3 Activity Assay Kit. D, Cells were treated with 2,4,6-TCP for 24 hours, and then MMP was examined with 5,5,6,6-tetrachloro-1,1,3,3-tetraethylbenzimidazolylcarbocyanine iodide (JC-1) staining by flow cytometry. Cells treated for 30 minutes with carbonyl cyanide m-chlorophenyl hydrazone (CCCP) were used (50 μmol/L) as a positive control for MMP decrease. E, After 24-hour incubation of mouse embryonic fibroblasts (MEFs) with 2,4,6-TCP (0.5, 0.75, and 1.0 mmol/L), RNA was prepared and the messenger RNA (mRNA) expressions of Bax, Bcl-2 were measured by quantitative polymerase chain reaction (qPCR). All experiments were repeated at least 3 times. Data were presented as mean ± standard deviation (SD; n = 3). *
The mitochondrial apoptosis pathway is involved in many aspects of programmed cell death. To further uncover the involvement of mitochondria in 2,4,6-TCP-induced apoptosis, we examined MMP and the Bax/Bcl-2 mRNA ratio, both known parameters of mitochondrial distress. As shown in Figure 4D and E, 2,4,6-TCP produced a dose-dependent decrease for MMP, and a concentration-dependent increase for the Bax/Bcl-2 ratio after 24 hours exposure to 2,4,6-TCP.
Discussion
Chlorophenols, including 2,4,6-TCP, have been defined as priority pollutants with high health risks for both animals and humans. Therefore, the use of CPs has been strictly limited in many countries. 4 However, CPs are still affecting the quality of air, water, and soil 3,4 and, consequently can be taken up by plants and ingested by animals and humans. In the present study, we found that exposure to 2,4,6-TCP led to oxidative stress, ER stress and apoptosis in MEFs.
Previous study has shown that some CPs, such as 4-chlorophenol, 2,4-dichlorophenol, 2,3,4-tirchlorophenol, and pentachlorophenol inhibited cell growth and decreased cell viability. 32 Whether 2,4,6-TCP has similar effects need to be further investigated. Here, we showed that MEFs exposure to 2,4,6-TCP changed their morphology together with reducing their viability. Our observation, similar to those from previously published studies with Chinese hamster 7 and Vero cell lines, 35,37 suggests the broad cytotoxic effect of 2,4,6-TCP on mammalian cells. Our next goal was, subsequently, to identify the cellular factors involved in 2,4,6-TCP-induced cytotoxicity.
The role of ROS in CPs-induced cytotoxicity has been demonstrated by using either various cell culture systems or animal models. While pentachlorophenol was shown to increase ROS production in
The ER stress is considered as another important toxic effect of phenol analogs.
27,44,45
Studies have suggested that nonylphenol induced ER stress in both rat testicular Sertoli cells
27
and the neuronally differentiated PC12 cells.
46
To test whether 2,4,6-TCP could induce ER stress
Among ER stress-induced proteins, CHOP is known to be involved in ER stress-induced cell death.
50,51
Induction of CHOP by 2,4,6-TCP led us to ask whether 2,4,6-TCP could induce apoptosis. Previous studies have demonstrated that caspase 3 activation was involved in 2,4,5-trichlorophenol and pentachlorophenol-induced apoptosis in human lymphocytes
33
and mouse hepatocytes,
52
respectively. These findings revealed that caspase 3 is a critical player in CPs-induced apoptosis. Additionally, many other CPs were shown to induce cell death via apoptosis.
31,33,34
In the present study, the increases in both apoptosis and activated caspase 3 (a key executor of apoptosis) in cells exposed to 2,4,6-TCP demonstrated the responsibility of this compound in cytotoxicity leading to cell death. Furthermore, we showed that 2,4,6-TCP disturbed the mitochondrial function by decreasing MMP while increasing the Bax/Bcl-2 ratio. These results suggest an involvement of mitochondria in 2,4,6-TCP-induced apoptosis. Similar observations were made with primary cultured
In conclusion, the present study showed the roles of 2,4,6-TCP in oxidative stress, ER stress, and apoptosis, which altogether are contributing to cytotoxic effects in MEFs. These findings will contribute to further understand the potential health risk of the widely use of 2,4,6-TCP for humans.
Footnotes
Acknowledgments
We thank Dr Christophe E. Redon (Laboratory of Molecular Pharmacology, National Cancer Institute, National Institutes of Health, USA) for the critical reading of the manuscript.
Author Contributions
Xiaoning Zhang contributed to conception and design, acquisition, analysis, and interpretation; drafted the manuscript; critically revised the manuscript; gave final approval; and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Xiaona Zhang contributed to acquisition and analysis, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Zhidan Niu contributed to acquisition, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Yongmei Qi contributed to design, critically revised the manuscript, gave final approval, and agrees to be accountable for all aspects of work ensuring itegrity and accuracy. Dejun Huang and Yingmei Zhang contributed to conception and interpretation, critically revised the manuscript, gave final approval, and agree to be accountable for all aspects of work ensuring itegrity and accuracy.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the National Natural Science Foundation of China (20907019), the Program for New Century Excellent Talents in University (NCET-10-0464), the Fundamental Research Funds for the Central Universities (Lzujbky-2013-241), and the Specialized Research Fund for the Doctoral Program of Higher Education (20110211110032).
