Abstract
Background
Erigeron multiradiatus (Lindl.) Benth. has been used for years to treat various diseases in Traditional Tibetan Medicine (TTM). Our previous studies have shown that E. multiradiatus extract has important pharmacological effects, such as anti-myocardial ischemia–reperfusion (I/R) injury, anti-inflammatory, and anti-diabetic effects. To date, there have been no reports on using this extract to treat thrombosis.
Objectives
The aim of this study was to enrich the active components of E. multiradiatus and to further investigate its anti-thrombotic activity.
Materials and Methods
We used D101 macroporous adsorption resin to enrich the main active substances of E. multiradiatus and performed quantitative determination using ultra-high-performance liquid chromatography with a photodiode array detector (UHPLC-PDA). Furthermore, we investigated the anti-thrombotic effects of E. multiradiatus using three models: an arachidonic acid-induced platelet aggregation thrombotic zebrafish model, a phenylhydrazine-induced red blood cell (RBC)-injuring thrombotic zebrafish model, and a ponatinib-induced vascular endothelial cell (VEC)-injuring thrombotic zebrafish model.
Results
The yield of active component enrichment of E. multiradiatus was 4.95% using the D101 resin. We accurately determined eight compounds in the extract. Our results showed that these extracts had significant anti-thrombotic effects in the three different zebrafish models.
Conclusion
The enriched extract of E. multiradiatus could be used as a natural anti-thrombotic agent. Our results will provide a reference for reasonable clinical applications of E. multiradiatus.
Introduction
Thrombosis is the abnormal coagulation of blood due to the coagulation cascade during blood flow, which causes the body’s organs to become ischemic and infarcted, resulting in myocardial infarction, ischemia, and stroke (Bronze, 2018; Falk et al., 2013). Thrombosis, which plays an important role in cardiovascular disease due to a high pathogenicity and low cure rate, seriously threatens human health and life (Davì & Patrono, 2007; Halvorsen et al., 2014). Anti-thrombotic drugs are mainly used to treat anti-coagulation and anti-platelet aggregation, but there is no definitive treatment program for thrombotic diseases (Nakase & Suzuki, 2016; O’Connor et al., 2015). Two types of drugs are used in these cases. The first is anti-platelet aggregation drugs. One such drug is aspirin, which has a clear anti-platelet aggregation effect, but its side effects include bleeding, and patients can become resistant to aspirin (Li et al., 2014; Warkentin, 2012). Clopidogrel is the latest anti-platelet aggregation drug, but it lacks long-term data and is expensive (Bozzi et al., 2016). The second type of drug typically targets thrombin and combines warfarin and heparin (Warkentin, 2012). Heparin is the first-line choice for acute attacks of thrombotic diseases, but it also carries the risk of bleeding, so it is not suitable for long-term use (Esfandiar et al., 2012; Shi & Fu, 2021). Therefore, finding new drugs that can prevent and treat thrombosis while causing fewer adverse reactions is of great clinical significance.
Lately, traditional Tibetan medicine (TTM) has been gaining more and more attention worldwide for its distinct efficacy and minimal side effects (Ren et al., 2020; Wang, 2020). Erigeron multiradiatus (Lindl.) Benth. is a biennial or perennial herb that is mainly distributed across the Qinghai–Tibet plateau of China at altitudes ranging from 2600 to 4300 m (Editorial Committee of Chinese Flora, 1985). In TTM, E. multiradiatus has been used to treat a variety of diseases, including hyperpiesia, enteritis, diarrhea, and food poisoning, as well as fever and cough. Meanwhile, it is also used to promote blood circulation and remove blood stasis. Photochemical studies report the isolation and identification of many compounds in E. multiradiatus, such as flavonoids, phenolic acids, and sterols (Zhang et al., 2009; Zhang et al., 2013). Other studies have reported that crude extracts of E. multiradiatus have important pharmacological actions such as anti-inflammation, hepatoprotection, and anti-diabetes (Luo et al., 2008a, b; Luo et al., 2013). The compound breviscapine shows anti-myocardial ischemia–reperfusion (I/R)-injuring effect (Wang et al., 2009; Yang et al., 2021; Zhao et al., 2016). Until now, however, there have been no reports of using E. multiradiatus to treat thrombosis.
The zebrafish (Danio rerio), a small tropical freshwater fish of the family Cyprinidae, is widely applied in research into bioactive screening, toxicological evaluation, and new-drug discovery in traditional Chinese medicine (TCM) (MacRae & Peterson, 2015; Patton et al., 2021; Wu et al., 2022). Therefore, this fish is becoming a prevalent vertebrate model, attracting more and more attention due to its small size, short lifecycle, and high fertility rate, as well as the low investment in aquaculture required. Moreover, in comparison to mouse and other animal models, a zebrafish model has many advantages, such as high transparency, fewer test compounds, and high flux. Such a model is very suitable for thrombosis research (Williams & Poole, 2012), because zebrafish’s hemostatic and thrombotic processes are similar to those of humans, and it also shares most of the central homologous cytokines for platelet adhesion, activation, aggregation, and release reactions with humans (Lang et al., 2010; Zhu et al., 2016); these cytokines respond to anticoagulant and anti-platelet drugs commonly used in clinical treatment (Weyand & Shavit, 2014). Therefore, in this study, we used zebrafish as a model to study the anti-thrombotic activity of E. multiradiatus.
Materials and Methods
Chemicals and Reagents
We purchased the reference compounds neochlorogenic acid (2101082), chlorogenic acid (21040904), scutellarin (21050603), and apigenin-7-O-glucronide (21033102) from Sichuan Weiqi Biological Technology Co., Ltd. (Chengdu, China). Cryptochlorogenic acid (20052601) and isochlorogenic acid B (20032602) were obtained from Chengdu Pufield Biotechnology Co., Ltd. (Chengdu, China), while isochlorogenic acids A (15100702) and C (15100816) were obtained from Chengdu Kangbang Company Limited (Chengdu, China). All eight chemicals had purities of 98%, as confirmed by high-performance liquid chromatography (HPLC). We used them for quality control (QC) purposes. Enteric-coated aspirin tablets (BJ54728) were obtained from Bayer Health Care AG (Chengdu, China) and Guanxinning tablets (1912804) from Chia Tai Whelk Bao Pharmaceutical Co., Ltd. (Chengdu, China). Other materials used in this study were dimethyl sulfoxide (DMSO; BCCD8942; Sigma-Aldrich [Merck, Kanton Bern, Switzerland]), arachidonic acid (C2123090; Shanghai Aladdin Biochemical Technology Co., Ltd., Shanghai, China), o-dianisidine (MKCC7501; Sigma-Aldrich [MilliporeSigma, Burlington, MA, USA]), phenylhydrazine (YH0170509; Shanghai Yihe Biotechnology Co., Ltd., Shanghai, China), and ponatinib (13771; MedChemExpress, Monmouth Junction, NJ, USA). HPLC-grade acetonitrile was obtained from Beijing Dikema Technology Co., Ltd. (Beijing, China). Formic acid was obtained from Tianjin Kemiou Chemical Reagent Co., Ltd. (Tianjin, China). We prepared ultra-pure water using a Milli-Q water purification system (MilliporeSigma).
The samples of E. multiradiatus collected during flowering from Ganzi, Sichuan Province. The authentication is according to the monograph Flora of China by Professor Hao Zhang (School of Pharmacy, Sichuan University, China). Herbarium voucher (No. EM190701) were prepared and deposited at the herbarium at the Institute of Qinghai-Tibetan Plateau, Southwest Minzu University. The herbs were dried in the sun.
Experimental Animals
Animal Care Ethics
All zebrafish experiments were conducted according to the guidelines of the Animal Ethics Committee of the Laboratory Animal Center of Southwest Minzu University (Chengdu, China). Feeding management met Association for Assessment and Accreditation of Laboratory Animal Care International (AAALAC) certification requirements (Cert. No. 001458).
Zebrafish Husbandry
We maintained an albino strain of melanin allele-mutant translucent zebrafish (Hunter Biotechnology, Inc., Zhejiang, China; License No. SYXK2012-0171) following standard protocols (Aleström et al., 2020). They were fed in 28°C water for 3 days. To prepare the water, we added 200 mg instant sea salt to 1 L reverse-osmosis water (conductivity, 450–550 S/cm; pH, 6.5–8.5; hardness, 50–100 mg/L CaCO3). We obtained embryos through natural spawning. Zebrafish larvae that were 3 days post fertilization (3 dpf) were used to determine maximum detectable concentrations (MDCs) of anti-platelet aggregation thrombosis, anti-red blood cell (anti-RBC)-injuring thrombosis, and anti-vascular endothelial cell (anti-VEC)-injuring thrombosis, as well as to evaluate the anti-thrombotic efficacy of the sample.
Sample Preparation
Sample Preparation of E. multiradiatus
The dried herb of E. multiradiatus was ground into powder. We then weighed 300.00 g of the powder, placed it in a round-bottomed flask, and added 2000 mL ethanol–water (70:30 v/v) solution. Reflux extraction was carried out in a water bath three times, 60 min per time. We then combined and centrifuged the filtrate at 4000 rpm for 20 min. The supernatant extract was transferred to a rotary evaporator and concentrated under vacuum to remove ethanol. Finally, we added deionized water to obtain sample solutions of 1500 mL.
The sample solutions were slowly added into glass chromatography columns (Shanghai Huake Experimental Devices and Materials Co., Ltd., Shanghai, China), 400-mm long with 30-mm inner diameter; the columns were then packed with D101 macroporous adsorption resin (Donghong Chemical Co., Ltd., Changzhou, China). We performed dynamic adsorption and desorption experiments by washing the resin columns with deionized water and and subsequently desorbing them using 75% ethanol at a flow rate of 2 BV/h. Subsequently, the eluent was collected, concentrated, and freeze dried to obtain 14.84 g crude extract. The yield of dry extract was 4.95%.
Standard Compound Solutions
Eight standard stock solutions (neochlorogenic acid; chlorogenic acid; cryptochlorogenic acid; scutellarin; apigenin-7-O-glucronide; and isochlorogenic acids A, B, and C) were individually prepared in methanol at a concentration of 1.0 mg/mL and then stored at 4°C. Next, we diluted these solutions with methanol to make a series of standard solutions with different concentrations. Enteric-coated aspirin tablets were dissolved with DMSO into solutions of 50.0 mg/mL, and Guanxinning tablets were dissolved in standard dilution water into solutions of 20.0 mg/mL. We stored both of these at 4°C.
Quantitative Analysis Using Ultra-High-Performance Liquid Chromatography
We analyzed the samples using a Waters Ultra-High-Performance Liquid Chromatography (UHPLC) CLASS system (Waters Corp., Milford, MA, USA) equipped with a binary solvent delivery manager, auto-sampler manager, thermostatically controlled column compartment, and photodiode array detector (PDA). Empower 3 software (Waters) was used to control the instrument components and acquire, store, and analyze ultraviolet (UV) data. Separation was performed in an ACQUITY UHPLC High-Strength Silica (HSS) chromatography column (100 × 2.1 mm, 1.8 µm; Waters). We used formic acid in water (0.2%, v/v; A) and acetonitrile (B) for the mobile phase to carry out gradient elution. The UHPLC gradient elution procedure was optimized as follows: 10%–16% B (0–5 min), 16%–18% B (5–18 min), and 18%–24% B (18–26 min). The mobile-phase flow rate was set at 0.2 mL/min, the column temperature at 35°C, and the analytical volume of the reference sample was 1 µL. We then determined the content of the eight reference compounds in E. multiradiatus samples using the UHPLC-PDA system.
Evaluation of Anti-Thrombotic Efficacy
Effect of Anti-Platelet Aggregation Thrombus in Zebrafish
Maximum Detected Concentration in Zebrafish
We used 3-dpf zebrafish embryos to study the efficacy of anti-platelet aggregation thrombosis. Embryos were randomly placed in 6-well plates (Wuxi NEST Biotechnology Co. Ltd., Wuxi, China). Each well contained 30 fish embryos. We divided the larvae into five groups: normal control, model, positive, low-dose, and high-dose. E. multiradiatus extracts were diluted with distilled water to obtain a 2-mg/mL sample reserve solution. We dissolved arachidonic acid and aspirin in DMSO to prepare stock solutions, maintaining the final concentration at <0.5%. The normal control and model groups were treated with fish water, the positive group was treated with aspirin at a concentration of 50 µg/mL, and the two test groups were treated with E. multiradiatus solution at the indicated concentrations in Table 1. Zebrafish larvae of all five groups were incubated at 28°C for 3 h. Then, to each well of all groups except the normal group, we added arachidonic acid solution at a final concentration of 100 µM to induce thrombus formation. After treatment at 28°C for 90 min, the death rate of zebrafish larvae was observed and recorded, and the maximum tested concentration (MTC) of E. multiradiatus in each group was determined.
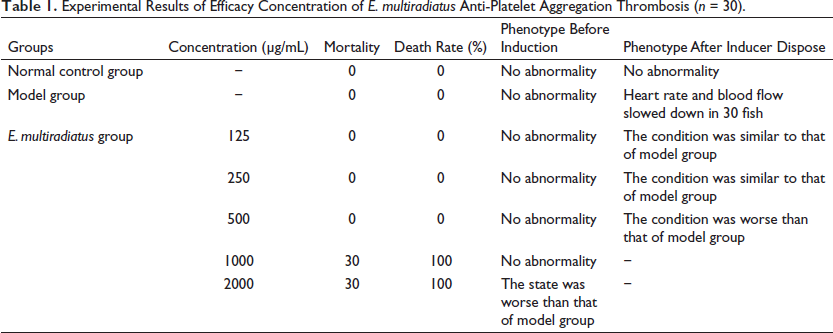
Experimental Results of Efficacy Concentration of E. multiradiatus Anti-Platelet Aggregation Thrombosis (n = 30).
Efficacy Evaluation of Anti-Platelet Aggregation Thrombosis
In this study, platelet aggregation thrombosis in zebrafish larvae was induced with arachidonic acid following a published method (Gao et al., 2019) with a slight modification. We randomly selected 3-dpf zebrafish embryos in the 6-well plates and treated 30 embryos per well with E. multiradiatus (50, 250 µg/mL) and aspirin (50.0 µg/mL) to a volume per well of 3 mL. At the same time, we established the normal control and model control groups. After treatment at 28°C for 3 h, all experimental groups except the normal control group were given arachidonic acid for 90 min at 28°C to establish a model of platelet aggregation thrombosis in zebrafish.
We prepared a staining solution by adding 50 mg o-dianisidine, 41 mg sodium acetate, and 1.1 mL 30% hydrogen peroxide solution into a volumetric flask to 50 mL. o-Dianisidine staining was used to detect expression of hemoglobin after establishment of the model. Next, we randomly selected 10 zebrafish larvae in each experimental group, placed them on glass slides, observed them under a dissecting microscope (SZX7; Olympus Corp. Tokyo, Japan), and obtained photographs using a charge-coupled device (CCD) camera (VertA1; Shanghai Tusen Vision Technology Co., Ltd., Shanghai, China).
Effect of Anti-RBC-injuring Thrombotic Activity in Zebrafish Larvae
Maximum Detected Concentration in Zebrafish
We used 4-dpf zebrafish embryos to study anti-RBC-injuring thrombosis. Embryos were randomly placed in 6-well plates; each well contained 30 fish larvae. We then similarly established the normal control, model, positive, low-dose, and high-dose groups. Concentration settings are shown in Table 2. The positive group was treated with Guanxinning tablets at a concentration of 2000 µg/mL. Zebrafish larvae of all groups were incubated at 28°C for 6 h. Then, to each well of all groups except the normal control group, we added phenylhydrazine solution to induce thrombus formation. After 18 h of exposure at 28°C, the death rate of zebrafish larvae was observed and recorded, and the MDC of E. multiradiatus in each model group was determined.
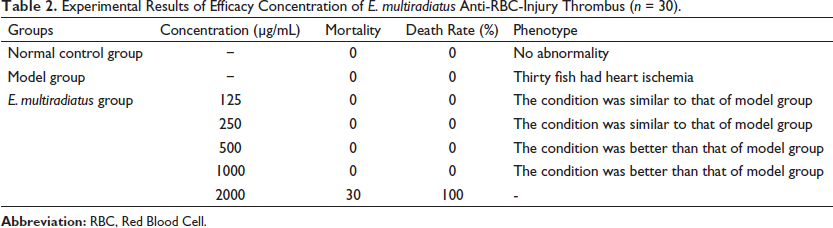
Experimental Results of Efficacy Concentration of E. multiradiatus Anti-RBC-Injury Thrombus (n = 30).
Efficacy Evaluation of Anti-RBC-injuring Thrombosis
To establish the drug protection group, 4-dpf zebrafish embryos were randomly selected and placed into 6-well plates. Thirty zebrafish per well were treated with E. multiradiatus (50, 1000 µg/mL) and Guanxinning (2000 µg/mL) to a volume per well of 3 mL. At the same time, we established the normal control and model groups. After treatment at 28°C for 6 h, all experimental groups except the normal control group were given phenylhydrazine for 18 h to establish a model of red blood cell (RBC)-injuring thrombus in zebrafish, following which o-dianisidine staining was used to detect expression of hemoglobin as described above. Similarly, we randomly selected 10 zebrafish from each experimental group, placed them on glass slides, observed them under the SZX7 dissecting microscope, and photographed them using the VertA1 CCD camera.
Effect of Anti-VEC-injuring Thrombotic Activity in Zebrafish Larvae
Maximum Detected Concentration in Zebrafish
We used 5-dpf zebrafish embryos to study anti-VEC-injuring thrombosis. The normal control, model, positive, low-dose, and high-dose groups were established as discussed earlier. Concentration settings are shown in Table 3. Each well contained 30 zebrafish. The positive group was treated with aspirin at a concentration of 50 µg/mL. To each well of all groups except the normal control group, we added ponatinib solution to induce thrombus formation. After 21 h of treatment at 28°C, the death rate of zebrafish larvae was observed and recorded, and the MDC of E. multiradiatus in each model group was determined.
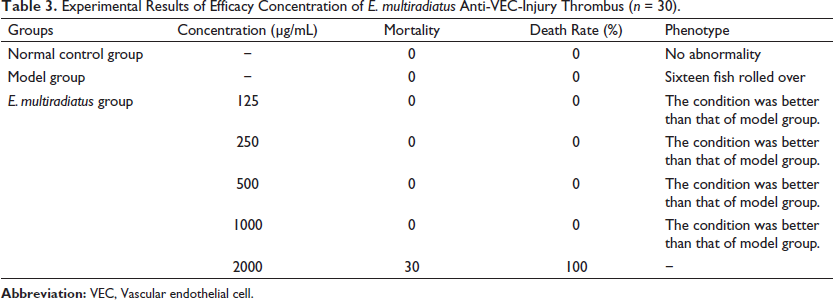
Experimental Results of Efficacy Concentration of E. multiradiatus Anti-VEC-Injury Thrombus (n = 30).
Efficacy Evaluation of Anti-VEC-injuring Thrombosis
To establish the drug protection group, 5-dpf zebrafish embryos were, respectively, randomly selected in 6-well plates. Thirty zebrafish per well were treated with E. multiradiatus (50, 250 µg/mL) and aspirin (50.0 µg/mL) to a volume per well of 3 mL. At the same time, we established the normal control and model groups. Except for the normal control group, all experimental groups were given ponatinib for 21 h to establish a model of vascular endothelial cell (VEC)-injuring thrombosis in zebrafish. Then, we used o-dianisidine staining to detect expression of hemoglobin, and we observed and photographed 10 randomly selected zebrafish as described previously.
Statistical Analysis
We used NIS-Elements D advanced image processing software v3.20 (Nikon, Tokyo, Japan) to collect data and analyze the heart staining intensity of zebrafish. Anti-platelet aggregation thrombosis, anti-RBC-injuring thrombosis, and anti-VEC-injuring thrombosis were evaluated by statistical significance. All data are presented as the mean ± standard error of the mean (SEM). Differences between two groups were analyzed using a two-tailed Student’s t-test. We used one-way analysis of variance (ANOVA) to compare multiple groups. p < 0.05 was considered statistically significant.
Results
Optimization of Extraction and Separation Conditions
Sample Preparation Optimization
We investigated the effects of extraction method, extraction solvent, and extraction time on extraction rate. Two commonly used extraction methods were studied: heating reflux and ultrasonically assisted extraction. Optimal extraction conditions were compared and optimized, and the material transfer rate of E. multiradiatus was determined using UHPLC. We used neochlorogenic acid, chlorogenic acid, cryptochlorogenic acid, apigenin-7-O-glucronide, scutellarin, and isochlorogenic acids A, B, and C as chemical markers to evaluate E. multiradiatus quality. Extraction solvent (100% ethanol with 100% or 70% methanol), the number of times extraction was performed (once or twice), and durations of extraction (15, 30, and 45 min) were selected as the sample extraction factors. The results showed that ultrasonic extraction was the more suitable extraction method because it produced relatively larger peak areas for the separated components. Then, 70% methanol was used as the extract solvent, and the sample was extracted twice for 30 min each. Finally, the accurately weighed powder (approximately 0.1 g) was suspended in 20 mL of 70% methanol (v/v) in a 50-mL volumetric flask and sonicated (Kun Shan Ultrasonic Instruments Co., Ltd., Kun Shan, China) for 30 min at room temperature. The extraction was repeated once. Then, the final volume was replenished to 50 mL with 70% methanol (v/v). The solvent was centrifuged at 4000 rpm for 5 min, and the supernatant was stored at 4°C as the test solution. Before injection and analysis, we filtered the test solution through a 0.22-µm polytetrafluoroethylene (PTFE) syringe filter and injected it into the UHPLC system. Three replicate UHPLC analyses were performed per sample.
Mobile-phase Optimizing
After performing the above analysis, we comprehensively compared the samples’ chromatograms under different elution conditions. The results showed that degree of separation and peak shape were good when elution conditions for E. multiradiatus samples were as follows: formic acid in water (0.2%, v/v; A) and acetonitrile (B) were used as the elution solvent. The UHPLC gradient elution procedure was optimized as follows: 10%–16% B (0–5 min), 16%–18% B (5–18 min), and 18%–24% B (18–26 min). Therefore, we selected these conditions for the analysis of the samples. Chromatographic results are shown in Figure 1.
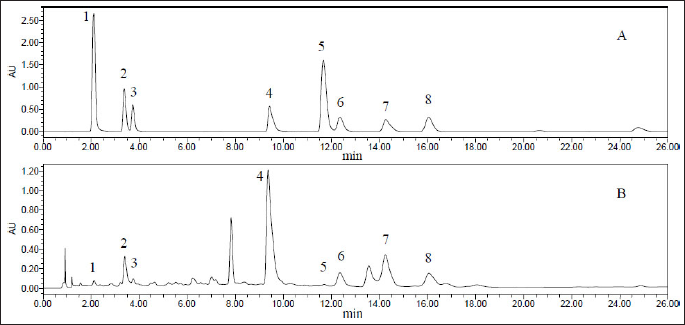
Validation of the UHPLC-PDA Method
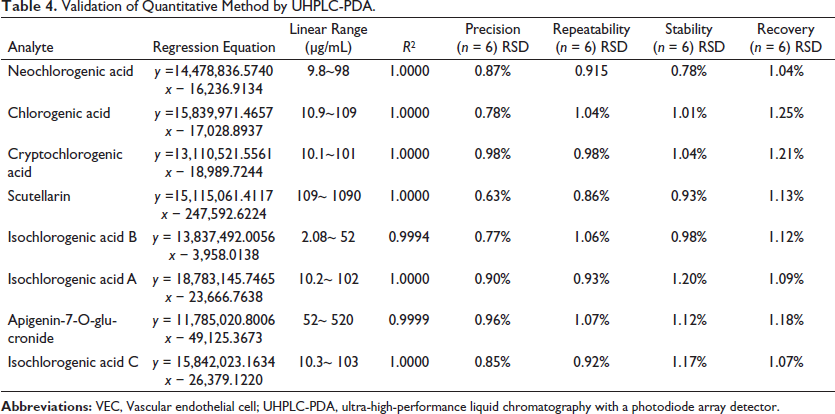
To achieve better resolution in the analyte data, we optimized chromatographic conditions, including composition and flow rate of the mobile phase, column temperature, and column type. Chromatographic conditions were also optimized for better resolution and higher detection of each component; these conditions included mobile-phase composition and flow rate, column temperature, column type, and detection wavelength. A validation study of the analytical method was carried out to determine the specificity, sensitivity, linearity, accuracy, precision, and applicability of the method. At the same time, we quantitatively detected the eight analytes in the medicinal materials under optimal conditions. Validation results using the quantitative method with UHPLC-PDA are shown in Table 4.
Validation of Quantitative Method by UHPLC-PDA.
Linearity
We evaluated linearity at five non-zero concentrations and the resultant regression curves. The calibration regression equation, correlation coefficient, and linear ranges for each analyte are presented in Table 4.
Precision
Instrument precision was determined by analyzing six replicates of same standard solution on the same day. The combined relative standard deviation percentage (%RSD) for all eight analytes ranged from 0.63% to 0.98%, indicating good instrument precision.
Repeatability
To evaluate repeatability, we prepared six test solutions in parallel for analysis according to the preparation method described above. The data in Table 4 show extraordinary reproducibility of the samples. The %RSD of the repeatable experimental analysis was only 0.86%–1.07%, showing that the method had good repeatability.
Stability of Method Extracts
We evaluated sample stability via repeat analysis of the same samples at 0, 2, 4, 8, 12, and 24 h after preparation. The stability of the extract during UHPLC analysis of the sample was tested; the results are shown in Table 4. The results ranged from 0.78% to 1.20%, indicating that the samples were stable within 24 h at room temperature.
Method Accuracy
To test the accuracy of the established method, we performed a recovery test. A known content of the standard substance fo each component (equivalent to half the content of eight compounds in the test sample) was spiked into six sample solutions (0.05 g). The recoveries of the eight compounds are summarized in Table 4. The recoveries were 97.39%–98.45%, and the % RSDs were 1.04%–1.25%. The levels of recovery and associated variations were acceptable.
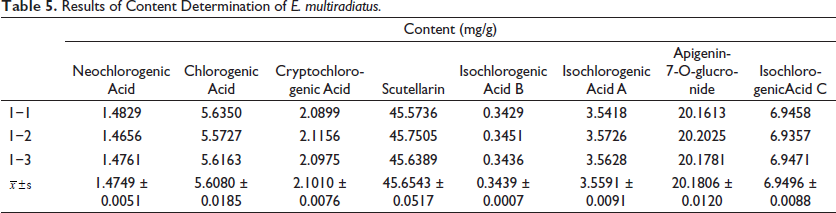
Quantitative Analysis of E. multiradiatus Samples
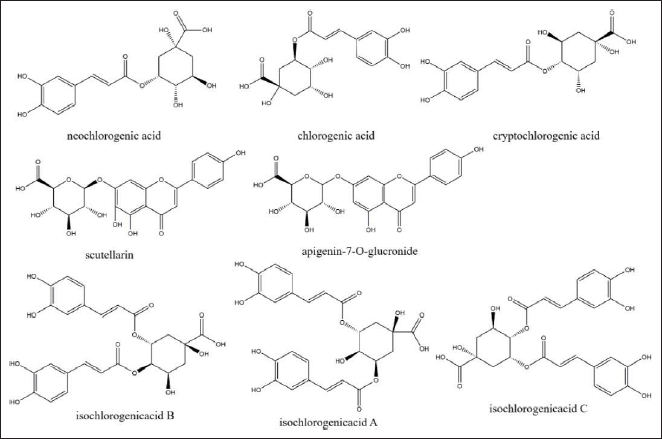
We pretreated 0.1-g crude extract of E. multiradiatus to perform UHPLC analysis. By subsequent comparison with the reference substance, a total of eight components were determined, containing: neochlorogenic acid, chlorogenic acid, cryptochlorogenic acid, apigenin-7-O-glucronide, scutellarin, and isochlorogenic acids A, B, and C (Figure 1). Their molecular structures are shown in Figure 2. The contents of each component in the sample were calculated according to the regression equation shown in Table 5.
Molecular Structures of Eight Compounds.
Results of Content Determination of E. multiradiatus.
Anti-Platelet Aggregation Thrombosis
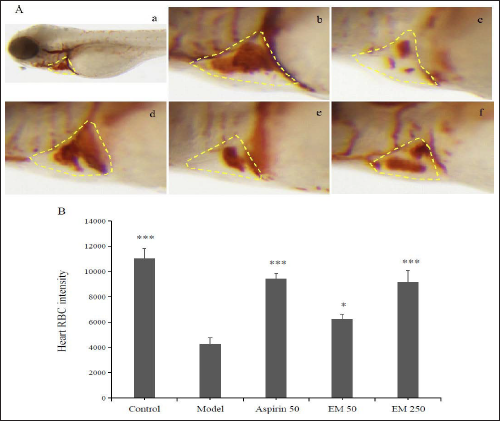
Under experimental conditions, we employed a zebrafish model of arachidonic acid-induced thrombosis to investigate the role of E. multiradiatus in thrombosis. In addition, this study was the first to investigate the toxic effects of E. multiradiatus on the survival and development of zebrafish embryos. Significantly, the heart rate and cardiac blood flow of 3-dpf zebrafish embryos slowed down after incubation with arachidonic acid for 90 min. After 3 h of pre-treatment in E. multiradiatus at a volume ranging from 50.0 µg/ml to 250 µg/ml, embryonic development was not affected. However, the proportion of dead or deformed embryos increased significantly when the dosage exceeded 500 µg/mL; when it was >1000 µg/mL, all embryos died. Therefore, the drug dose must be controlled within a safe range. We limited the concentration of E. multiradiatus to 250 µg/mL to evaluate the efficacy of platelet aggregation thrombosis. When the MTC of E. multiradiatus in the zebrafish was 250 µg/mL, no significant difference was obtained in efficacy in comparison to the normal control and model groups in Table 1.
In the positive control group, 3-dpf zebrafish embryos were given different doses of E. multiradiatus (50 or 250 µg/ml) or aspirin for 3 h and then incubated in embryonic medium containing arachidonic acid for 90 min. Figure 3 shows that E. multiradiatus significantly increased cardiac blood flow in arachidonic acid-induced zebrafish, and the effect became more and more obvious as dosage increased. When the MTC of E. multiradiatus reached 250 µg/mL, the effect of anti-platelet aggregation thrombosis was basically similar to that of aspirin (50 µg/mL).
Anti-RBC-injuring Thrombus
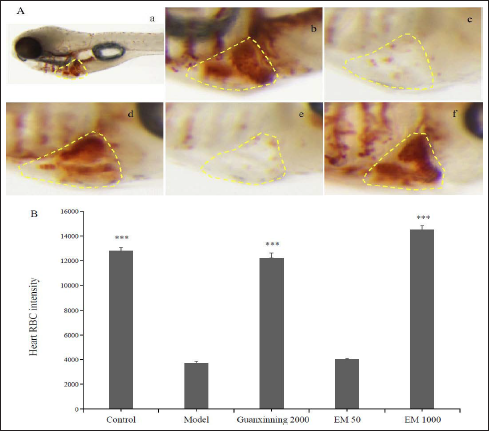
Similarly, we used phenylhydrazine to establish a model of anti-RBC-injuring thrombus zebrafish to study the role of E. multiradiatus in antithrombosis. We observed cardiac ischemia in 4-dpf zebrafish embryos incubated in phenylhydrazine for 18 h. Symptoms were alleviated by the administration of E. multiradiatus. When the administered dosage was <1000 µg/mL, heart ischemia obviously recovered; when dosage exceeded 2000 µg/mL, all the zebrafish died. Therefore, the drug dose must be controlled within a safe range. The concentration of E. multiradiatus was limited to 1000 µg/mL (Table 2).
Next, we administered E. multiradiatus (50, 1000 µg/ml) or Guanxinning to 4-dpf zebrafish embryos for 6 h as positive controls, and then incubated them in phenylhydrazine-supplemented embryonic medium for 18 h. As shown in Figure 4, we found that E. multiradiatus significantly improved cardiac ischemia in phenylhydrazine-induced zebrafish, and with the increase in dosage, the effect became more obvious. When the concentration of E. multiradiatus reached 1000 µg/mL, the effect intensity was higher than that of Guanxinning (2000 µg/mL).
Anti-VEC-injuring Thrombus
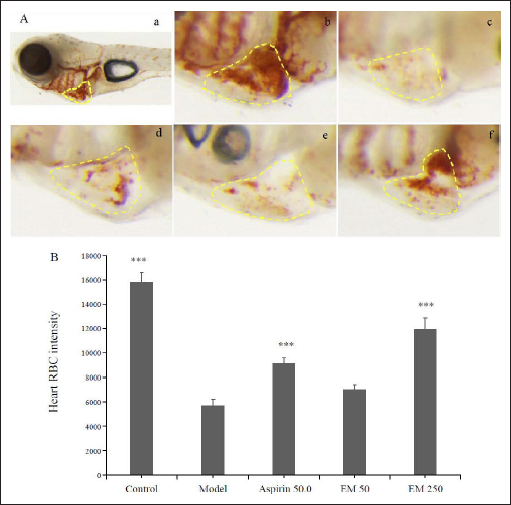
We used ponatinib to establish a model of anti-VEC-injuring thrombosis in zebrafish in order to study the anti-thrombotic effect of E. multiradiatus. We observed that after 5-dpf zebrafish embryos were incubated in ponatinib for 21 h, and 16 of the 30 embryos rolled over. When the concentration of E. multiradiatus was <1000 µg/mL, the rollover rate was improved, but when it was >2000 µg/mL, all zebrafish died. Therefore, the drug dose must be controlled within a safe range, and we limited the concentration of E. multiradiatus to 1000 µg/mL in Table 3.
Next, we administered different doses of E. multiradiatus (50, 250 µg/ml) or aspirin as positive controls, followed by incubation in ponatinib-supplemented embryonic medium for 21 h. We discovered that E. multiradiatus was effective in the treatment of VEC-injuring thrombosis and displayed a dose-dependent effect in Figure 5. When the concentration of E. multiradiatus reached 250 µg/mL, the effect intensity was significantly higher than that of aspirin (50 µg/mL).
Discussion
Formation of thrombi is considered as the main cause of cardiovascular and cerebrovascular diseases (Alkarithi et al., 2021). Thrombosis is a complex and progressive process involving many factors: VEC damage, changes in blood flow status, and increased blood coagulation (Iwanaga et al., 2021; Jamiolkowski et al., 2016; Neubauer & Zieger, 2022). Any one of these three factors can cause thrombus, but due to the complexity of the coagulation process in the blood, all three thrombosis formations are often interacting at the same time. Therefore, anticoagulation therapy is the main means of treating and preventing thrombotic diseases. Anticoagulant and anti-platelet drugs play similar roles in clinical medicine (Ni et al., 2021). The former mainly include warfarin, rivaroxaban, apixaban, and edoxaban, which are typically used to treat atrial fibrillation. Anti-Platelet drugs include aspirin, ticlopidine, clopidogrel, and cilostazol. Aspirin is a clinically important anti-platelet and anti-thrombin drug (Lu et al., 2015; Tan et al., 2013). However, these drugs can cause serious adverse reactions along with a risk of hemorrhagic complications (Fu et al., 2021).
In comparison to western drugs, traditional herbal medicines have significant anti-thrombotic effects with the advantages of low toxicity and low side effects, which can significantly reduce the risks of thrombosis treatment (Lv et al., 2015). Traditional herbal medicines have thousands of years of practical experience behind their usage in the treatment of thrombotic diseases. For example, Guanxinning tablets are traditionally used to treat coronary artery disease (CAD). In accordance with the Chinese medicine theory, the main active ingredients in this recipe are Salvia miltiorrhiza Bge. and Ligusticum chuanxiong Hort., which dilate blood vessels, promote blood circulation, and resolve blood stasis (Wang et al., 2016). Therefore, we used Guanxinning tablets as a positive drug in our zebrafish model.
In a previous study, we found that a single bolus injection of E. multiradiatus extract reduced symptoms of acute myocardial I/R injury in rats. Our results also showed that the mechanism of E. multiradiatus was related to inhibition of I/R-induced inflammatory response, inhibiting activation of the nuclear factor κ-light-chain-enhancer of activated B cells (NF-κB), and c-Jun N-terminal kinase (JNK) signaling pathways after reperfusion (Xu et al., 2015). In the present study, we obtained different extracts of E. multiradiatus using a new purity method and accurately determined eight constituents thereof using UHPLC-PCA. Two important flavonoids, scutellarin, and apigenin-7-O-glucronide, could play important roles in the anti-thrombotic effect. It is reported that scutellarin is reported to have evident anti-thrombotic effect and that the underlying mechanisms might be intimately related to the effect on anti-platelet aggregation. Both scutellarin and apigenin-7-O-glucronide are main bioactive constituents of E. breviscapus. Previous research has shown that E. breviscapus flavones can significantly inhibit the formation of thrombi induced by adenosine diphosphate (ADP), arachidonic acid, and platelet-activating factor (Shen et al., 2000). Meanwhile, scutellarin can dose-dependently decrease the ADP-induced platelet aggregation rate in rats, thereby improving hemorheological parameters and preventing thrombosis and platelet aggregation. In the study, percentages of scutellarin and apigenin-7-O-glucronide were about 4.5% and 2.0%, respectively, in the E. multiradiatus extract, which contributed to this anti-thrombotic effect (Song et al., 2011).
We also determined six derivatives of caffeoylquinic acid in E. multiradiatus extract. Caffeoylquinic acid and its derivatives are widely found in certain medicinal and edible plants. The anticoagulant mechanism of chlorogenic acid mainly inhibits the activity of certain coagulation factors (Choi & Kim, 2017), measured activated partial thromboplastin time (APTT), prothrombin time (PT), and thrombin time (TT) in rat platelets. They found that chlorogenic acid can prolong APTT, PT, and TT values and significantly inhibit thrombin activity. This analysis suggested that chlorogenic acid might indirectly change the common coagulation pathway, induce reduction of thrombin activity, or inhibit related coagulation factors to exert anti-thrombotic effect.
In TTM, E. multiradiatus is often used in clinical treatment to improve blood flow, promote blood circulation, and eliminate blood stasis (Zhang et al., 2009). Our results can support the clinical application of folk medicine. In the present study, we established three zebrafish models: platelet aggregation thrombosis induced by arachidonic acid, VEC-injuring thrombosis induced by ponatinib, and RBC-injuring thrombosis induced by phenylhydrazine. Intensity of cardiac RBC staining is negatively correlated with tail vein thrombus length, indicating that such intensity can be used to assess thrombosis (Wang et al., 2016).
Thrombus decreased significantly in all three models after treatment with positive drugs (aspirin and Guanxinning tablets). We also confirmed that arachidonic acid, ponatinib, and phenylhydrazine could induce thrombus formation in zebrafish. E. multiradiatus extract had an obvious curative effect in all three zebrafish thrombus models in a dose-dependent manner. This indicated that E. multiradiatus had an anti-thrombotic effect and that its mechanism of action might be related to protecting blood vessel endothelium, restoring RBCs, and inhibiting platelet aggregation. The efficacy of traditional medicine is usually the result of multi-component and multi-target interactions (Xia & Chen, 2012). It is still uncertain whether the anti-thrombotic effect of E. multiradiatus is regulated by multiple components and targets or caused by sequential pathological events. For example, previous studies showed that caffeoylquinic acid might activate adenylate cyclase to increase the concentration of cyclic adenosine monophosphate (cAMP) and inhibit platelet aggregation, and it also showed that higher doses of caffeoylquinic acid can depolymerize platelet clots, prolong bleeding time, reduce the procoagulant effect of platelets, and prevent thrombosis (Fu et al., 2021; Guo et al., 2021). Although our results showed the anti-thrombotic effect of E. multiradiatus in three different models, further studies are necessary to clarify the molecular mechanism underlying this activity.
Conclusion
In the present study, two flavonoids and six caffeoylquinic acid-type compounds were accurately determined using UHPLC-PDA. These compounds could be the main active constituents in E. multiradiatus extract. These results demonstrated that this extract had a significant anti-thrombotic effect in three different zebrafish models. The enriched E. multiradiatus extract could be used as a natural anti-thrombotic agent. Our results will provide a reference for reasonable clinical applications of E. multiradiatus.
Summary
In TTM, E. multiradiatus has been used to treat a variety of diseases. Other studies have reported that crude extracts of E. multiradiatus have important pharmacological actions such as anti-inflammation, hepatoprotection, and anti-diabetes. The compound breviscapine shows anti-myocardial I/R-injuring effect. Until now, however, there have been no reports of using such extracts to treat thrombosis. We used D101 macroporous adsorption resin to enrich the main active substances of E. multiradiatus and performed quantitative determination using ultra-high-performance liquid chromatography with a photodiode array detector (UHPLC-PDA). Furthermore, we investigated the anti-thrombotic effects of E. multiradiatus in an arachidonic acid-induced platelet aggregation thrombotic zebrafish model, a phenylhydrazine-induced RBC-injuring thrombotic zebrafish model, and a ponatinib-induced VEC-injuring thrombotic zebrafish model. We accurately determined eight compounds in the extract. Our results showed that these extracts had significant anti-thrombotic effects in the three different zebrafish models.
Footnotes
Abbreviations
TTM: Traditional Tibetan medicine; TCM: Traditional Chinese medicine; HPLC: high-performance liquid chromatography; QC: Quality control; DMSO: Dimethyl sulfoxide; MDCs: Maximum detectable concentrations; VEC: Vascular endothelial cell; UHPLC: Ultra-high-performance liquid chromatography; PDA: Photodiode array detector; UV: Ultraviolet; HSS: High-Strength Silica; CCD: Charge-coupled device; SEM: Standard error of the mean; ANOVA: One-way analysis of variance; PTFE: Polytetrafluoroethylene; %RSD: Relative standard deviation percentage; MTC: Maximum tested concentration; CAD: coronary artery disease; JNK: c-Jun N-terminal kinase; ADP: Adenosine diphosphate; APTT: Activated partial thromboplastin time; PT: Prothrombin time; TT: Thrombin time; cAMP: Cyclic adenosine monophosphate.
Acknowledgments
The authors are immensely thankful to the Southwest Minzu University and Tibetan Plateau Ethnic Medicinal Resources Protection and Utilization Key Laboratory of National Ethnic Affairs Commission of the People’s Republic of China and Sichuan Qiang Yi Medical Resources Protection and Utilization Technology Engineering Laboratory for offering all the necessary facilities and infrastructure for uninterrupted conduct of this work.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This project was funded by grants from the National Natural Science Foundation of China [NO. 31870314], the National Key Research and development Program of China [NO. 2019YFC1712503], and Southwest Minzu University Basic Scientific Research Business Special Fund project for Central Universities [NO. 2021HQZZ01].
Ethical Approval and Informed Consent
Compliance with Ethical Standards: All applicable international, national, and/or institutional guidelines for the care and use of experimental animals were followed. This study involved no human participants or samples obtained from patients.
