Abstract
Background
Nasopharyngeal carcinoma (NPC), predominant in Southern China and Southeast Asia, is a malignant tumor that arises from the epithelial lining of the nasopharynx. Current NPC treatments result in unfavorable side effects. Natural compounds with anti-proliferative capabilities are gaining popularity as a way to mitigate the toxicity of radiation and synthetic antitumor drugs. Ponicidin, an ent-kaurane diterpenoid, has been demonstrated to possess several pharmacological activities, including antitumor, antibacterial, immunoregulatory, antiviral, and anti-inflammatory properties.
Materials and Methods
In the current investigation, the anti-carcinogenic activity of Ponicidin against nasopharyngeal cell line C666-1, has been investigated. The influence of Ponicidin on cell viability, apoptotic induction, mitochondrial membrane potential (MMP), reactive oxygen species (ROS) generation, and apoptotic markers has been examined. Doxorubicin was selected as the positive control for the experiments.
Results
The effect of Ponicidin on the viability of C666-1 cells revealed that its cytotoxic potential increased in a dose-dependent pattern and the optimum concentration chosen for further experiments was 7.5 µM. The Ponicidin-treated cells demonstrated a significant increase in the amount of TBARS, whereas it reduced the activity of superoxide dismutase (SOD) and glutathione (GSH) levels, indicating augmented oxidative stress and lower antioxidant activity in the C666-1 cells. The acridine orange/ethidium bromide (AO/EtBr) staining technique was used to assess morphological changes with respect to apoptosis, which confirmed its apoptotic nature. Furthermore, Ponicidin elevated the ROS generation, modified the mitochondrial membrane permeability, elevated the apoptotic marker levels (caspase-3, caspase-9, and Bax) and reduced the Bcl-2 expression in C666-1 cell lines.
Conclusion
The findings suggest that Ponicidin successfully inhibited cancer cell proliferation by following the mitochondrial apoptotic pathway and thus could be effectively utilized as a potent anti-cancer agent for the treatment of nasopharyngeal cancer.
Keywords
Introduction
The malignant tumor, nasopharyngeal carcinoma (NPC), develops from the upper portion of the pharynx. The disease is prevalent in specific racial and geographic regions of the world (Chen et al., 2019; Xiong et al., 2019). In a significant number of studies, multiple factors have been demonstrated to contribute to the occurrence of NPC, involving Epstein-Barr virus infection, genetic predisposition, drinking and smoking, and other environmental factors (Casanova et al., 2016; Tao & Chan, 2007). Contrary to other neck and head malignancies, non-differentiated squamous cell carcinomas—which make up the majority of NPC cases—have a high propensity to infiltrate nearby tissues and metastasize to local lymph nodes at an initial stage, which leads to poor prognosis in patients. Furthermore, local relapse and distant metastases are implicated in a significant proportion of NPC patients’ mortality (Fan et al., 2018; Luo et al., 2012). The abnormal activation of numerous proliferative pathways, such as Wnt, MAPKs, PI3-K, and EGFR, is considered to contribute to tumor progression (Gallardo et al., 2018).
The current treatment option for NPC is chemoradiation, however, this approach is said to function more efficiently for individuals with early-stage NPC than for those with late-stage NPC and regional tumor metastases (Tseng et al., 2019). In addition, the 5-year rate of survival of patients is just about 50%. The incorporation of a different therapeutic agent to cisplatin and 5-fluorouracil, the standard chemotherapy agents for the treatment of NPC, is one of the treatment approaches being researched for the disease (Daker et al., 2016). The need for novel therapeutic compounds is, however, largely justified by the multiple therapeutic setbacks and to evade toxicity related to radiation and synthetic anti-cancer drugs (Daker et al., 2013).
Patients seeking treatment are becoming interested in complementary and alternative medicine (CAM), as are healthcare professionals researching the efficacy of various treatments and management methods. Over 80% of cancer patients utilize CAM in addition to chemotherapeutic drugs during the first stages of their therapy, and up to 52% of patients with cancer choose one or more CAM therapies (Alsanad et al., 2014; Zeller et al., 2013). In China, the usage of traditional Chinese medicines (TCMs) for the treatment of tumors has a longstanding history. The volume of research supporting the ability of TCMs to treat cancer has grown in recent years, and their usage has gained wider acceptance around the globe. Celastrol, oridonin, and cinnamic acid have been demonstrated in prior research to exhibit anticancer activity through the activation of apoptosis and the suppression of NPC cell growth (Dai et al., 2019).
Plants of the Isodon and Rabdosia genera are the most extensively utilized folk medicines in China (Yang et al., 2012). An ent-kaurane diterpenoid obtained from Rabdosia and Isodon genus, Ponicidin, has been reported to possess several pharmacological activities, such as antibacterial, antiviral, immunoregulatory and anti-inflammatory properties (Islam et al., 2019). Several studies have suggested that Ponicidin exerts antitumor effects in hepatocellular cancer, lung cancer, and monocytic leukemia cells. This suggests that it could be employed as a potential cytotoxic drug (Liu et al., 2006; Liu et al., 2008; Zhang et al., 2007). However, there hasn’t been any significant research performed on the use of Ponicidin to treat NPC.
The objective of the current investigation is to examine the effectiveness of different dosages of Ponicidin on cell proliferation and apoptosis in vitro to ascertain the therapeutic potential of Ponicidin in nasopharyngeal cancer in C666-1 cancer cell lines. The influence of Ponicidin on the free radical scavenging analysis, cell viability by 3-(4,5-dimethylthiazoyl-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay, apoptotic induction by acridine orange/ethidium bromide (AO/EtBr), mitochondrial membrane potential (MMP) by Rh 123 staining, reactive oxygen species (ROS) generation by DCFH-DA staining, and apoptotic marker expression has been explored.
Materials and Methods
Chemicals
Chemicals such as Ponicidin and MTT were obtained from Sigma-Aldrich, USA. Assay kits were procured from Thermo Fisher Scientific, USA.
Free Radical Scavenging Analyses
Ferric-reducing Antioxidant Power Activity
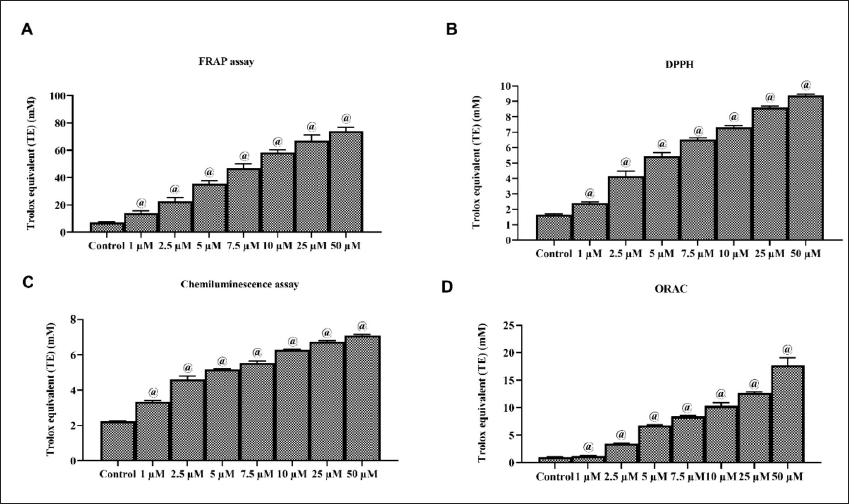
Ferric-reducing antioxidant power (FRAP) activity of Ponicidin was investigated using the procedure earlier described by Benzie and Strain (1996). Acetate buffer (300 mM), TPTZ solution (10 mM), and ferric chloride (20 mM) reagents were added together with the different concentrations of Ponicidin (1, 2.5, 5, 7.5, 10, 25, and 50 µM) and 1 mL of FRAP reagent. The reaction mixture (200 µL) was added into each well of the 96-well plate and incubated at 30°C for 10 min. After the incubation period, the final product was measured at 593 nm, and the FRAP activity was represented in terms of Trolox equivalent in milli Molar.
DPPH Radical Scavenging Activity
Ponicidin was tested for its ability to scavenge 2,2-diphenyl-1-picryl-hydrazyl-hydrate (DPPH) with the technique described by Brand-Williams et al. (1995). DPPH (150 mL) was suspended in 0.25 mM ethanol and then combined with Ponicidin at different concentrations (1, 2.5, 5, 7.5, 10, 25, and 50 µM) at 37°C for 30 min. After the incubation period, the absorbance of the final product was measured at 515 nm, and the DPPH activity was represented in terms of Trolox equivalent in milli Molar.
Measurement of Chemiluminescence
Ponicidin was tested for its ability to scavenge superoxide using a technique developed by Shimada et al. (1992). Using 10 mL of chemiluminescence (CL) solution, different concentrations (1, 2.5, 5, 7.5, 10, 25, and 50 µM) of Ponicidin, and 80 mL of xanthine oxidase, a reaction mixture was prepared. A control suspension was prepared with a HEPES buffer. In the next step, 200 mL of hypoxanthine substrate (0.72 mM) was added to the solution before it was applied to the luminometer. CL activity was measured for 10 min at 10 sec intervals in the luminometer. A total of three replicate tests were conducted and a relative luminescence value was determined. The scavenging capacity of Ponicidin was determined in terms of Trolox equivalent in milli Molar.
Estimation of oxygen radical antioxidant capacity
The oxygen radical antioxidant capacity (ORAC) level of Ponicidin was estimated by measuring its peroxyl radical scavenging capability with a kit by following the manufacturer’s instructions (Thermo Fisher, USA).
Collection of C666-1 and Vero Cell Lines
The C666-1 cancer cells and Vero cells were obtained from the ATCC, USA. After the cells were procured, they were grown in DMEM medium accompanied with 10% FBS in a humidified CO2 (5%) chamber at room temperature. The cells were trypsinized and used in subsequent studies after reaching 80% confluence.
MTT Assay
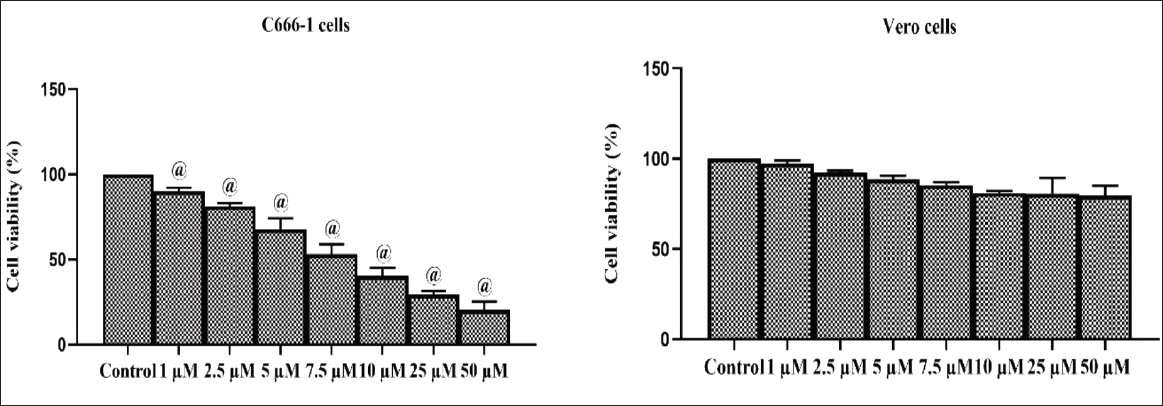
MTT assay was carried out to examine the viability of both normal Vero cells and C666-1 cells (Mosmann, 1983). The cells were grown onto 96-well plates with DMEM medium for 24 h at a density of 5 × 103 cells/well. Then, the C666-1 and Vero cells were maintained at room temperature for 24 h after each dose (5, 10, 15, 25, 50, 75, and 100 µM) of Ponicidin was administered. Afterwards, each well was incubated for 4 h following the addition of MTT (20 µL) and DMEM (100 µL). To dissolve the formazan crystals after the incubation period, 100 µL of DMSO was added. A microplate reader was used to measure absorbance at 570 nm for both control and treated cells.
Assessment of Oxidative Stress Markers
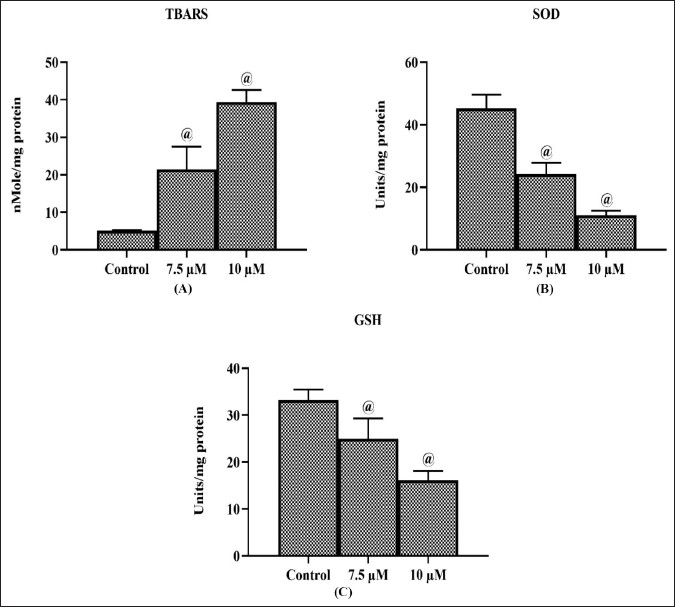
The levels of oxidative stress markers such as TBARS, superoxide dismutase (SOD), and glutathione (GSH) were estimated in the untreated and Ponicidin (7.5 and 10 µM) administered C666-1 cells by assay kits and following the instructions provided by the supplier.
Estimation of ROS Generation
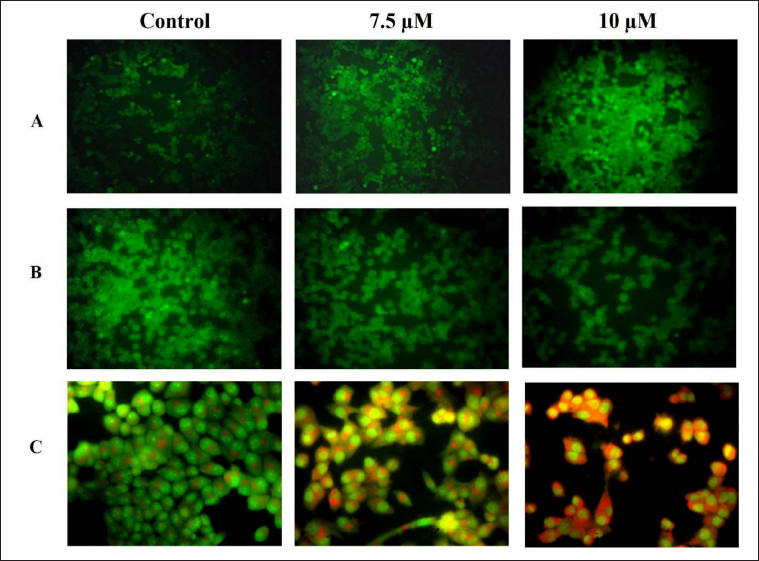
A DCFDA staining technique was employed to investigate the effect of Ponicidin on ROS generation within C666-1 cells. Cells were grown in 24-well plates for 24 h and then treated with 7.5 µM of Ponicidin and 2 µg of DOX for another 24 h. By staining with 10 µL of DCF-DA, ROS generation levels in the Ponicidin-treated C666-1 cells were determined. A fluorescent microscope was used to determine the level of ROS production based on the intensity of the fluorescent light.
Estimation of MMP
The Rh-123 staining assay was employed to determine the MMP level. For the investigation, C666-1 cells were loaded into the 24-well plate containing DMEM medium and treated for 24 h at 37°C with 7.5 µM of Ponicidin and 2 µg of DOX. Rh-123 dye (10 µg/mL) was added to the wells by mixing and the mixture was allowed to incubate for 30 min. Finally, fluorescent microscopy was utilized to assess the level of MMP.
AO/EtBr Dual Staining Approach
AO/EtBr staining technique was employed to assess the level of apoptosis of untreated and Ponicidin-treated C666-1 cells. Cells were loaded on a 24-well plate at a density of 5 × 105 cells per well in DMEM medium and treated with 7.5 µM of Ponicidin and 2 µg of DOX and incubated at 37°C for 24 h. Then, AO/EtBr stain (100 µg/mL) at a ratio of 1:1 was added to each well by mixing for 5 min at room temperature. Finally, a fluorescent microscope was utilized to detect apoptosis by measuring the fluorescence intensity.
Estimation of Oxidative Stress and Apoptotic Biomarker Levels
Commercial ELISA assay kits (Thermo Fisher, USA) were utilized to estimate the oxidative stress markers (TBARS, SOD, and GSH) and apoptotic markers (Bax and Bcl-2 protein levels as well as the activities of caspase-3 and caspase-9 proteins) in control and Ponicidin-treated C666-1 cells at two different concentrations (7.5 and 10 µM).
Statistical Analysis
GraphPad Prism software was used to evaluate the results obtained. The one-way ANOVA and Tukey’s post hoc test were used to thoroughly examine the results. The results were calculated as the mean ± SD of 3 different tests, and a p < 0.05 was established as a significant level.
Results
Impact of Ponicidin on the Free Radical Scavenging Potential
Different free radical scavenging experiments such as DPPH, FRAP, ORAC, and CL assays were used to examine the in vitro antioxidant capacity of Ponicidin, and the findings are illustrated in Figure 1. The treatment with different Ponicidin concentrations significantly eliminated the free radicals. Ponicidin considerably (p < 0.05) reduced the number of free radicals, including superoxide, peroxyl, and DPPH radicals. It also exhibited dose-dependent ferric-reducing capabilities with increasing concentration, demonstrating its in vitro free radical scavenging capabilities. This result supports Ponicidin’s ability to effectively scavenge free radicals.
Impact of Ponicidin on the Cytotoxicity of C666-1 and Vero Cell Lines
The cytotoxic potential of Ponicidin toward both healthy Vero cells and the NPC C666-1 cells was evaluated, and the findings are presented in Figure 2. Our results demonstrated that the Ponicidin treatment considerably (p < 0.05) reduced the growth of C666-1 cells dose-dependently. Ponicidin, on the other hand, did not affect the viability of Vero cells at the same doses. At the highest concentration of Ponicidin, the viability of Vero cells was slightly reduced. This finding demonstrates Ponicidin’s cytotoxicity toward C666-1 cells. Ponicidin’s IC50 for the C666-1 cells was determined to be 7.5 µM, therefore this concentration was used for further experiments.
Impact of Ponicidin on the SOD and GSH Level and TBARS Level in the C666-1 Cells
In the NPC C666-1 cells, the influence of the Ponicidin administration on the TBARS, SOD, and GSH levels was assessed, and the findings are depicted in Figure 3. The Ponicidin-treated cells (7.5 and 10 µM) showed a significant (p < 0.05) enhancement in the amount of TBARS (Figure 3A). Ponicidin administration decreased the activity of SOD (Figure 3B) and GSH levels significantly (Figure 3C). These results indicated that administering Ponicidin to C666-1 cells significantly reduced the antioxidant levels and increased oxidative stress.
Impact of Ponicidin on the ROS Generation, MMP State, and Apoptosis in C666-1 Cells
The impact of the Ponicidin treatment on the amount of intracellular ROS formation in the C666-1 cells is demonstrated in Figure 4A. C666-1 cells exposed to 7.5 µM of Ponicidin exhibited enhanced green fluorescence than the untreated cells. The enhanced intracellular generation of ROS in the treated cells was evidenced by an increased green fluorescence. Additionally, the standard drug DOX exhibited higher green fluorescence, confirming the increased ROS generation (Figure 4A). The MMP state of untreated and treated cells is shown in Figure 5B. In the control cells, which can be distinguished by their bright green fluorescence, an elevated MMP level was observed. However, 7.5 µM of the Ponicidin supplied to C666-1 cells resulted in the depletion of MMP as indicated by the reduced green fluorescence (Figure 4B). Rh-123 stained cell number was reduced as a result of the DOX treatment, which supports the diminished MMP state. The effect of the Ponicidin administration on apoptotic cell death in the C666-1 cells is shown in Figure 4C. The intensity of the yellow and orange fluorescence in the Ponicidin-treated cells is more prevalent than in the control, indicating that there were increased early and late apoptotic cell deaths. The cells treated with the DOX drug also displayed stronger yellow and orange fluorescence, which indicates an increase in apoptotic cell death (Figure 4C).
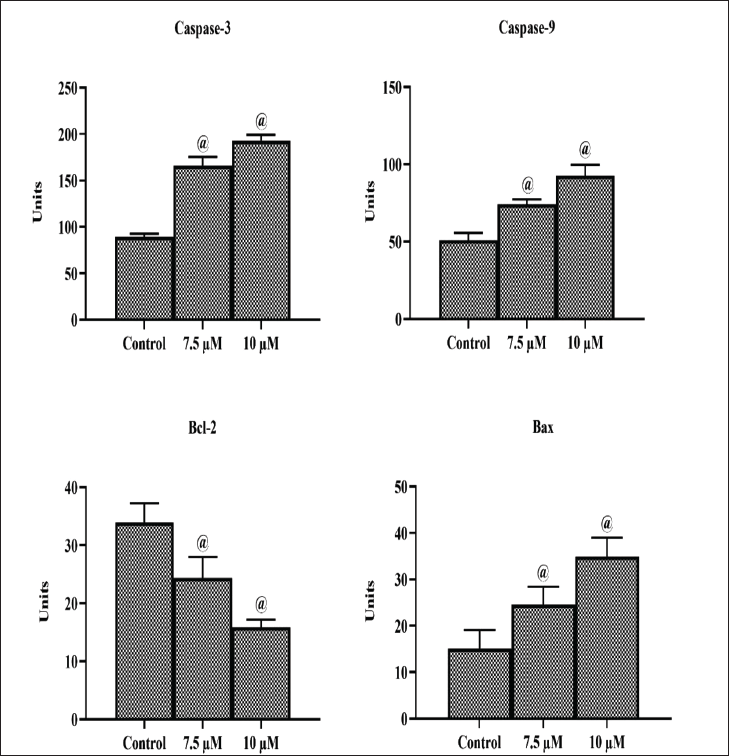
Effect of Ponicidin on the Caspase-3, Caspase-9, Bax, and Bcl-2 Levels in the C666-1 Cells
To examine whether activation/repression of apoptotic markers including caspase-3, caspase-9, Bcl-2, and Bax are involved in Ponicidin-induced apoptosis on C666-1 cells, the estimation of these parameters was carried out by ELISA technique. The findings of the investigation into the impact of Ponicidin treatment on caspase-3, caspase-9, and Bax/Bcl-2 levels are shown in Figure 5. Caspase-3, caspase-9, and Bax levels were considerably (p < 0.05) elevated in C666-1 cells after receiving 7.5 and 10 µM of Ponicidin. In the C666-1 cells, the Ponicidin treatment also resulted in a decline in Bcl-2 levels, confirming its anti-carcinogenic potential (Figure 5).
Discussion
NPC is a type of malignant tumor that arises from the nasopharynx’s epithelial cell linings (Ferlay et al., 2015). Patients with locoregionally late stages of NPC typically receive challenging treatments that are frequently toxic with myriad side effects. In an attempt to discover naturally occurring substances with substantial apoptosis-inducing activity and low toxicity, this study was focused on assessing the cytotoxic potential of a terpenoid Ponicidin on nasopharyngeal cancer cell lines.
Naturally occurring substances with animal and plant origins are gaining popularity in the research of anticancer treatments because they have been shown to interfere with the carcinogenic process at a specific stage (Xiong et al., 2019). A wide variety of plant-derived compounds including Ponicidin have been researched for their capability to inhibit cancer cell proliferation of different types of malignancies (Kim et al., 2015).
Plant-derived terpenoids act as effective anti-cancer molecules by limiting cancer cell growth and promoting programmed cell death. In the past, kaurene diterpenes that were isolated from a variety of medicinal plants were frequently utilized to treat cancer and inflammation (Xia et al., 2019). Bio-organic scientists are especially interested in the small bioactive compound, ent-kaurane diterpenoid, due to its extensive anti-tumor properties (Liu et al., 2012). Ponicidin is one such ent-kaurane diterpenoid that has been evaluated for its anticancer potential against nasopharyngeal cancer cells in this investigation.
Antioxidants such as DPPH is a free radicals at a normal temperature that can receive hydrogen or electron to form a stable diamagnetic molecule. The DPPH radical scavenging analysis is regarded as a reliable method for efficiently assessing antioxidant capabilities (Das et al., 2016; Xu et al., 2013). The scavenging effect of Ponicidin on DPPH radical was observed in a dose-dependent fashion. Additionally, the Fe3+ to Fe2+ reducing the capacity of Ponicidin was assessed with the help of FRAP assay, which evaluates the reducing potential of antioxidants (Fe, 2015). FRAP assay also revealed the reducing nature of Ponicidin in a concentration-based manner, confirming its remarkable antioxidant potential. Moreover, its antioxidant activity was further confirmed by ORAC and CL assays. The findings of this investigation are in concordance with a previous report which proved the anti-oxidant potential of Ponicidin by DPPH and ABTS assays (Duan et al., 2019).
The MTT cytotoxic assay is crucial for determining cytotoxicity while evaluating anticancer drugs since it attempts to measure the number of viable cells in the culture (Gordon et al., 2018). It was discovered that Ponicidin declined the viability of C666-1 cells in a concentration-dependent trend. A similar cell viability reduction in hepatoma, human leukemia, and colorectal cell lines was observed upon Ponicidin treatment (Zhao et al., 2011).
Oxidative stress in cancer cells is brought about by an increase in ROS, which limits the synthesis of antioxidants (Yao et al., 2021). SOD enables the catalysis of superoxide anions to oxygen and hydrogen peroxide conversion while providing protection against lipid peroxidation. Lipid peroxidation is triggered by the development of ROS, which frequently results in the production of excessive MDA, which interferes with normal cell activity and results in cancer (Kruk & Aboul-Enein, 2017). The Ponicidin-treated cells demonstrated a significant increase in the amount of TBARS, whereas it reduced the activity of SOD and GSH levels, indicating augmented oxidative stress and lower antioxidant activity in the C666-1 cells.
ROS are crucial in triggering tumor cell death (Yang et al., 2016). The ROS-sensitive fluorometric probe DCFDA was utilized to estimate the amount of ROS produced within the cells in order to determine whether ROS are involved in the Ponicidin-regulated cell death. An elevated intracellular generation of ROS in the Ponicidin-treated cells was shown by an increased green fluorescence. A similar augmented ROS generation was induced by Ponicidin in gastric carcinoma cells (Zhang et al., 2019). To further understand the mechanism of cell death triggered by Ponicidin, the AO/EtBr staining technique was used to assess morphological changes with respect to apoptosis. The living cells displayed green fluorescence due to acridine orange penetration into the cellular membranes, but the apoptotic cells were displayed as orange entities due to nuclear shrinkage, which confirms the stimulation of the apoptotic process. A comparable morphological change was observed by the treatment of the natural compound brassinin in C666-1 cells (Yin et al., 2022).
The majority of the ROS produced inside a cell arise from the mitochondria. As they regulate ATP synthesis and the MMP controls the release of apoptogenic proteins into the cytosol during apoptotic signal transduction, mitochondria play a crucial role in the induction of apoptosis (Singh et al., 2019). One of the mitochondria-related characteristics of tumour cells is an elevated MMP. Research has shown that oxidative stress, DNA damage, and mitochondrial membrane depolarization may all induce cells to undergo apoptosis (Mani et al., 2020). Depolarization of the MMP was clearly observed in the Ponicidin-treated C666-1 cells, which is in concordance with a study that reported a similar reduction of MMP by Ponicidin in lung cancer cells (Liu et al., 2006).
Apoptosis, a physiologically biological process, helps to maintain cellular homeostasis. However, apoptotic pathways have a tendency to become dysregulated, leading to a number of pathological characteristics that aid in carcinogenesis (Koff et al., 2015). The antitumor cell inhibitory mechanisms of numerous cytotoxic drugs, thus, involve the stimulation of malignant cell apoptosis, which is crucial in the treatment of cancer (Kang et al., 2014). Caspases are a group of cysteine proteases that contribute to apoptotic cell death. The two key pathways involved in the processes of apoptosis are the intrinsic pathway and the extrinsic pathway, which each entail two significant caspase cascades. Cleaved caspase-9 is an active form of caspase-9, which is an initiator caspase that can trigger caspase-3 in the apoptotic signaling cascade. When a cell undergoes apoptosis, caspase-3 is cleaved to produce the active caspase-3 enzyme, which breaks down several cellular proteins and causes morphological alterations and DNA fragmentation (McIlwain et al., 2015).
The intrinsic apoptotic process is regulated by proteins of the Bcl-2 family. Several apoptotic signals cause Bax, a Bcl-2 family member that promotes apoptosis, to be activated. Bax translocation from the cytosol to the mitochondria is thought to trigger apoptosis by lowering the potential of the mitochondrial membrane, releasing cytochrome c, and activating caspases. The anti-apoptotic protein, Bcl-2, is substantially elevated in many malignancies as compared to healthy normal cells (Radha & Raghavan, 2017). In particular, Bcl-2/Bax controls the release of cytochrome c into the cytosol from mitochondria, which causes caspase activation and finally leads to apoptosis (Zhao et al., 2015). Tumor cells can develop resistance to apoptosis through the initiation of enhanced Bcl-2 levels or the suppression or reduction of Bax protein levels (Dabrowska et al., 2016).
In this study, Ponicidin treatment led to increased expression of Bax proteins and downregulated Bcl-2 expression, which resulted in a lowering of the Bcl-2/Bax ratio, indicating that Ponicidin-induced apoptotic cell death. Moreover, it is possible that Ponicidin exerts anti-cancer activity through a caspase-dependent mechanism given the activation of caspase-3/9 that was observed in the NPC cells in our investigation.
Ponicidin has been proven in several studies to be able to cause apoptosis in a variety of cancer cell types, and a number of mechanisms behind Ponicidin-mediated apoptotic cell death have also been established. In the colorectal cell lines, Ponicidin triggered apoptotic cell death by downregulating the MEK and Akt signalling pathways and upregulating the expression of the proteins caspase-3, Bax, and p38 (Dabrowska et al., 2016). Liu et al. (2015), suggested that the anti-tumor activity of Ponicidin is accomplished by cell death through caspase-3 activation and inhibition of the PI3K/Akt signaling pathway in gastric cells. By increasing the production of ROS, Bcl-2 expression, decreasing the Bax and caspase-3 expression, and possibly through a signaling pathway controlled by JAK2 and STAT3, Ponicidin was found to induce apoptotic cell death in gastric carcinoma cells (Zhang et al., 2019). In this study, we demonstrated that high ROS-induced oxidative stress and dysregulation of apoptosis-associated genes promote apoptosis in C666-1 cells.
Conclusion
This study provides the evidence of in vitro anti-cancer potential of Ponicidin against the NPC C666-1 cells. Our findings suggest that Ponicidin substantially inhibited the cell viability of C666-1 cells. It also increased ROS production and reduced the MMP level of C666-1 cells. Ponicidin-induced apoptosis in the C666-1 cells by increasing the expression of caspases and Bax and decreasing the Bcl-2 expression. Overall, our findings support the in vitro anticancer property of Ponicidin against NPC, and more research into this framework might contribute to the development of a powerful anticancer medicine to effectively treat NPC.
Footnotes
Summary
NPC, predominant in southern China and southeast Asia, is a malignant tumor that arises from the epithelial lining of the nasopharynx. Ponicidin, an ent-kaurane diterpenoid, has been demonstrated to possess several pharmacological activities, including antitumor, antibacterial, immunoregulatory, antiviral, and anti-inflammatory properties.
Elevated ROS concentration causes mitochondrial outer membrane pores to be ruptured, lowering the MMP and releasing apoptotic caspases, leading to cell death via the mitochondrial pathway.
Acknowledgment
This work was supported by Changshu Hospital affiliated with Nanjing University of Chinese Medicine, Suzhou Jiangsu, 215500, China.
Abbreviations
NPC: nasopharyngeal carcinoma; TCMs: traditional Chinese medicines; MTT: 3-(4,5-dimethylthiazoyl-2-yl)-2,5-diphenyltetrazolium bromide; FRAP: ferric reducing antioxidant power; DPPH: 2,2-diphenyl-1-picryl-hydrazyl-hydrate; CL: chemiluminescence; ORAC: oxygen radical antioxidant capacity; GSH: glutathione; ROS: reactive oxygen species; MMP: mitochondrial membrane potential.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
