Abstract
Background
Osteosarcomas (OSs) are predominantly generated by mesenchymal cells, which commonly develop in the distal and upper regions of the bones. OS has high mortality rates and treatment failures, mostly due to the presence of lung metastases.
Objectives
The present work aimed to scrutinize the anticancer potentials of aucubin against OS human osteosarcoma (MG-63) cells.
Materials and Methods
The proliferation of the control and aucubin-treated MG-63 cells was studied by an XTT assay. Various fluorescent staining assays were done to assess the endogenous reactive oxygen species (ROS) accumulation and apoptosis in the aucubin-treated MG-63 cells. The caspase-3, -8, and -9 activities were investigated using the assay kits.
Results
The XTT assay results confirmed that the aucubin treatment considerably reduced the MG-63 cells. The findings of the various fluorescent staining assays evidenced that the aucubin treatment remarkably promoted endogenous ROS accumulation and apoptosis in the OS cells. The caspase activities were also improved in the OS cells after the aucubin treatment.
Conclusion
The results of this work show that aucubin treatments inhibit cell proliferation and trigger apoptosis in MG-63 cells, suggesting its potential as a promising anticancer agent. These findings will facilitate the future development of aucubin as a talented anticancer agent to treat OS.
Introduction
Osteosarcoma (OS) is the predominant form of bone cancer in adolescents and is characterized by the appearance of malignant mesenchymal cells. The current global statistics for the year 2022 indicate that there were almost 20 million new cases of cancer and close to 10 million cancer deaths. Demographics-based predictions indicate that the annual number of new cases of cancer will reach 35 million by 2050, a 77% increase from the 2022 level (Bray et al., 2022). OS frequently develops on the epiphysis of long bones, with approximately two-thirds of cancers arising in the distal femur bones. The second most frequent location for OS is the proximal tibia, followed by the proximal humerus in third place. Typically, it impacts the elongated bones, specifically the distal femur (30%), proximal tibia (15%), and proximal humerus (15%) (Misaghi et al., 2018; Siddiqui et al., 2019). Roughly 10–15% of newly diagnosed OS patients have increased metastases, of which lung metastasis is the most common at 74%, followed by bone metastasis at roughly 9%, and combined bone and lung metastasis in about 8% of patients (Meltzer & Helman, 2021).
OS is characterized by the development of malignant osteoid, resulting from the abnormal growth of osteoblasts. This results in a decrease in bone marrow, high pressure, and metastasis within the bone, causing pain, swelling, and fractures. OS risk factors are elevated by various causes, including exposure to ionizing radiation, genetic disorders, and pre-existing bone diseases. Diagnosing the disease necessitates the use of radiography and magnetic resonance imaging (MRI) to examine the bone (Ferguson and Turner, 2018). As our understanding of the biology of OS has improved, the diverse nature and possible molecular abnormalities associated with OS have been partially documented. Nevertheless, when confronted with an inoperable disease, the options for treating OS patients who do not respond to or cannot tolerate chemotherapy drugs remain restricted (Arndt & Crist, 1999).
Apoptosis is a process of programmed cell suicide that plays a pivotal role in tumor development. It plays a key role in regulating tissue growth and controlling physiological processes (Mirkes, 2002). To trigger programmed cell suicide in tumor cells, specific proteins known as initiator and effector proteins must be activated. This activation can occur through either intrinsic signaling, which is triggered by internal cellular stress, or extrinsic signaling, which is initiated by external signals (Rowinsky, 2005). The proapoptotic caspase enzyme activation is crucial to the mechanisms of apoptosis. Caspases-9, which initiate apoptosis, and caspases-3, which execute the apoptosis, include regulators of cell death. Treatment failures in cancer therapy can have several negative consequences, including reduced chances of cancer survival, disease progression, and a poor prognosis. Additionally, there is a possibility that the tumor may develop resistance to subsequent chemotherapeutic therapies (Degterev et al., 2003). To ensure a higher chance of successful treatments, it is crucial to understand the fundamental principles of apoptosis.
The current treatment paradigm for OS involves a multimodal approach, typically combining surgery, chemotherapy, and occasionally radiation therapy. Surgical resection aims to remove the primary tumor while preserving limb function whenever possible (Wang, 2023). While chemotherapy has improved the overall survival rate to 60–75%, there has been no significant rise in survival rates over the past 20–30 years. In addition, chemotherapy frequently results in significant adverse effects such as ototoxicity, cardiotoxicity, and nephrotoxicity (Angulo et al., 2017). In addition, OS is distinguished by its resistance to therapy, as tumors only exhibit a response to high concentrations of chemotherapy and quickly develop resistance (Lilienthal & Herold, 2020). The progression of drug resistance is a significant barrier to making advancements in the therapy of OS (Hattinger et al., 2021). While there are various therapeutic approaches, the development of a noninvasive treatment with a potent drug with minimal or no adverse effects would be a major advancement in OS therapy (Fogh et al., 1977).
Aucubin, an iridoid glycoside specifically known as β-D-glucopyranoside, is found abundantly in the leaves of Aucuba japonica, Eucommia ulmoides, and Rehmannia glutinosa (Zeng et al., 2020). Several studies have already reported that aucubin has antioxidative, antidiabetic (Xue et al., 2009), gastroprotective (Yang et al., 2017), cardioprotective (Wu et al., 2018), antisepsis (Qiu et al., 2018), antipulmonary fibrosis (Zhou et al., 2017), neuroprotective (Chen et al., 2019), nephroprotective (Potočnjak et al., 2020), and hepatoprotective (Chang et al., 1983) activities. Nonetheless, there are no studies that highlight the anticancer effect of aucubin against OS. Consequently, the current study work is aimed to scrutinize the anticancer potentials of aucubin against OS human osteosarcoma (MG-63) cells.
Materials and Methods
Chemicals
The chemicals and reagents, such as aucubin, dimethyl sulfoxide (DMSO), and fetal bovine serum (FBS), were obtained from Sigma-Aldrich, USA. The biochemical markers were analyzed using kits obtained from Elabscience, USA.
Cell Culture
The MG-63 cells were procured from ATCC, USA, and cultivated in Dulbecco’s Modified Eagle Medium (DMEM) growth medium enriched with 10% FBS in a CO2 (5%)-controlled incubator. Cells were collected at 80% confluency for further fluorescence staining and biochemical analysis.
Cytotoxicity Assay
The XTT Cell Proliferation Assay Kit (Abcam, USA) was utilized to test the cytotoxicity of aucubin. In summary, MG-63 cells were grown in 96-well microtiter plates with growth media (100 µL) for 24 hours. Subsequently, they were exposed to various doses (3.13, 6.25, 12.5, 25, 50, 75, and 100 µM/mL). After a 24-hour incubation period, XTT/PMS solution (25 µL) was applied to each well for 2 hours, and cell growth was quantified at 450 nm (Czyżowska et al., 2022).
2′-7′-Dichlorodihydrofluorescein Diacetate (DCFH-DA) Staining
The influence of aucubin treatment on the accumulation of endogenous reactive oxygen species (ROS) generation cells was evaluated using DCFH-DA staining. The cells were cultured and exposed to aucubin at a dosage of 40 µM for 24 hours in a 24-well plate. Subsequently, the wells were supplemented with DCFH-DA dye (10 µL) and incubated for 10 minutes. The developed fluorescence was then investigated using a fluorescent microscope (Kim & Xue, 2020).
Dual Staining
The dual staining test was performed on the aucubin-treated MG-63 cells to scrutinize the apoptosis. The cells were exposed to 40 µM/mL of aucubin for another 24 hours. Subsequently, a solution consisting of acridine orange/ethidium bromide (AO/EtBr) (100 µg/mL) fluorescent stains was loaded into the wells and allowed to incubate for 5 minutes in a dark place. This was done to assess the occurrence of apoptosis in MG-63 cells using a fluorescent microscope (Liu et al., 2015).
4′,6-Diamidino-2-phenylindole (DAPI) Staining
The DAPI staining technique was employed to assess the apoptotic cells in the aucubin-exposed MG-63 cells. Cells were treated with 40 µM/mL of aucubin for 24 hours. Following the treatment, the cells were fixed in a paraformaldehyde (4%) solution and subsequently stained with DAPI (200 µg/mL) for 15 minutes. Subsequently, the impact of aucubin on apoptosis in MG-63 cells was investigated using a fluorescence microscope (Atale et al., 2014).
Analysis of Caspase Level
The cell lysates from MG-63 cells exposed to aucubin and control cells were prepared and used for the experiments. The caspase-3, -8, and -9 activities were scrutinized using assay kits following the methods indicated by the manufacturer (Elabscience, USA).
In Silico Analysis
In Silico Drug-likeness
The SwissADME web tool, which can be used for free at
Molecular Docking Analysis
The CB-Dock server, available at
Statistical Analysis
The statistical studies were done using GraphPad Prism, and the results are reported as a mean ± SD of three replicate assays. The significance threshold was determined by conducting the one-way analysis of variance (ANOVA) and Duncan’s multiple range test (DMRT) tests, with p < 0.05 being statistically significant.
Results
Effect of Aucubin Treatment on the MG-63 Cell Viability
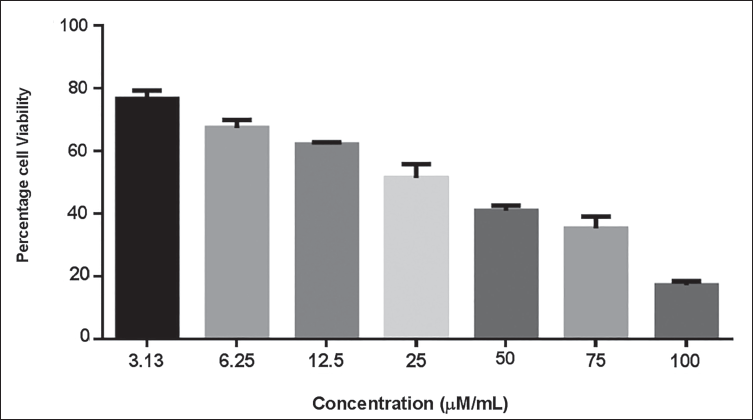
Figure 1 presents the results of the XTT cytotoxicity assay, illustrating the influence of aucubin on the proliferation of MG-63 cells. Aucubin treatment considerably reduced the MG-63 cell viability at a dosage range of 3.13, 6.25, 12.5, 25, 50, 75, and 100 µM/mL. The increasing dosage of aucubin had a minimal inhibitory effect on the development of MG-63 cells (Figure 1). The IC50 concentration of aucubin was noted at 40 µM/mL in MG-63 cells, and this same dosage was chosen for subsequent studies.
Effect of Aucubin Treatment on the Viability of Human Osteosarcoma (MG-63) Cells. Aucubin Treatment Significantly Reduced the Viability of MG-63 Cells at a Dose Range of 3.13–100 µM/mL. The IC50 Concentration of Aucubin was Noted at 40 µM/mL for MG-63 Cells. The Data are Displayed as the Mean ± SD of Three Measurements. Using GraphPad Prism Software, One-way Analysis of Variance (ANOVA) and Duncan’s Multiple Range Test (DMRT) Assays are Used to Statistically Analyze the Results.
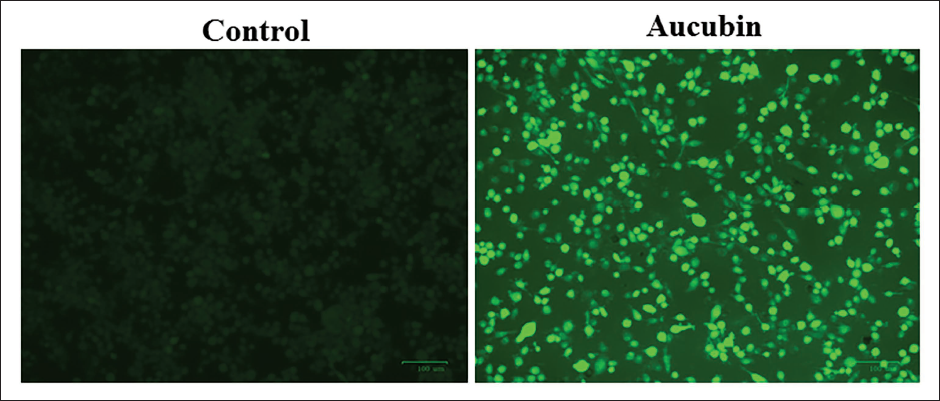
Effect of Aucubin on ROS Generation in MG-63 Cells
Figure 2 exhibits the results of the influence of aucubin treatment on the generation of endogenous ROS in OS cells. The treatment of OS cells with 40 µM/mL of aucubin led to a considerable elevation in green fluorescence compared to the control. The higher green fluorescence is a result of the heightened production of endogenous ROS in MG-63 cells treated with aucubin (Figure 2).
Effect of Aucubin on the Reactive Oxygen Species (ROS) Accumulation in the Human Osteosarcoma (MG-63) Cells. The Endogenous ROS Production Levels in the Control and Aucubin-treated MG-63 Cells were Assessed Using 2′-7′-Dichlorodihydrofluorescein Diacetate (DCFH-DA) Staining. The Treatment of MG-63 Cells with 40 µM/mL of Aucubin Resulted in a Considerable Increase in Green Fluorescence Compared to the Control, Which Indicates the Heightened Accumulation of Endogenous ROS in Aucubin-treated MG-63 Cells.
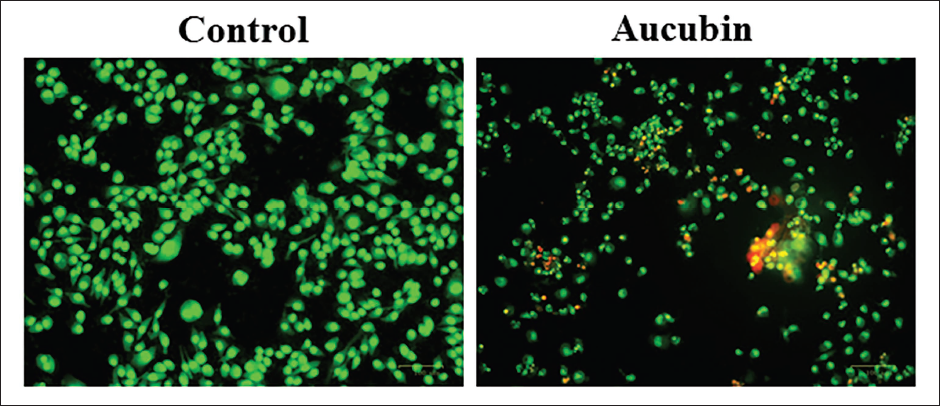
Effect of Aucubin Treatment on the Apoptosis in MG-63 Cells
The dual staining technique was employed to measure the apoptosis in both the control and aucubin-exposed MG-63 cells (Figure 3). After being treated with 40 µM/mL of aucubin, the MG-63 cells exhibited increased levels of yellow and orange fluorescence, providing evidence of the presence of apoptotic cell deaths in both early and late apoptotic stages, respectively.
Effect of Aucubin Treatment on the Apoptosis in Human Osteosarcoma (MG-63) Cells. The Dual Staining Technique was Employed to Measure the Apoptosis in Both the Control and Aucubin-exposed MG-63 cells. 40 µM/mL of Aucubin Treatment to the MG-63 Cells Exhibited Increased Levels of Yellow and Orange Fluorescence, Which Proves the Presence of Apoptotic Cell Deaths in Both Early and Late Apoptotic Stages, Respectively.
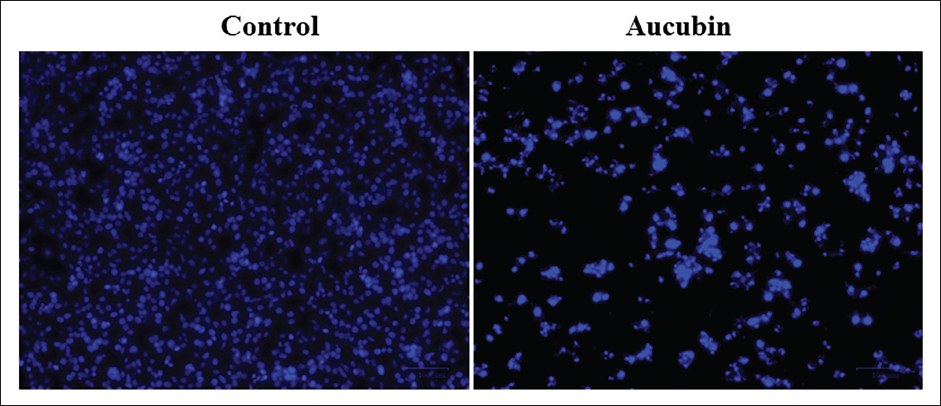
Treatment with Aucubin Induces Apoptotic Cell Death in MG-63 Cells
Figure 4 shows the findings of a DAPI staining assay conducted on OS cells exposed to aucubin. After exposure to 40 µM/mL of aucubin, the MG-63 cells exhibited various signs of apoptosis, such as chromatin condensation, membrane damage, the formation of apoptotic bodies, and a reduction in cell count.
Effect of Aucubin Treatment on the Apoptosis in the Human Osteosarcoma (MG-63) Cells. The 4′,6-Diamidino-2-phenylindole (DAPI) Fluorescent Staining Assay was Conducted on Control and Aucubin-treated MG-63 Cells to Study Apoptotic Cell Death. After Exposure to 40 µM/mL of Aucubin, the MG-63 Cells Exhibited Various Signs of Apoptosis, Such As Chromatin Condensation, Nuclear Damage, Membrane Rupture, the Formation of Apoptotic Bodies, and a Reduction in Cell Count.
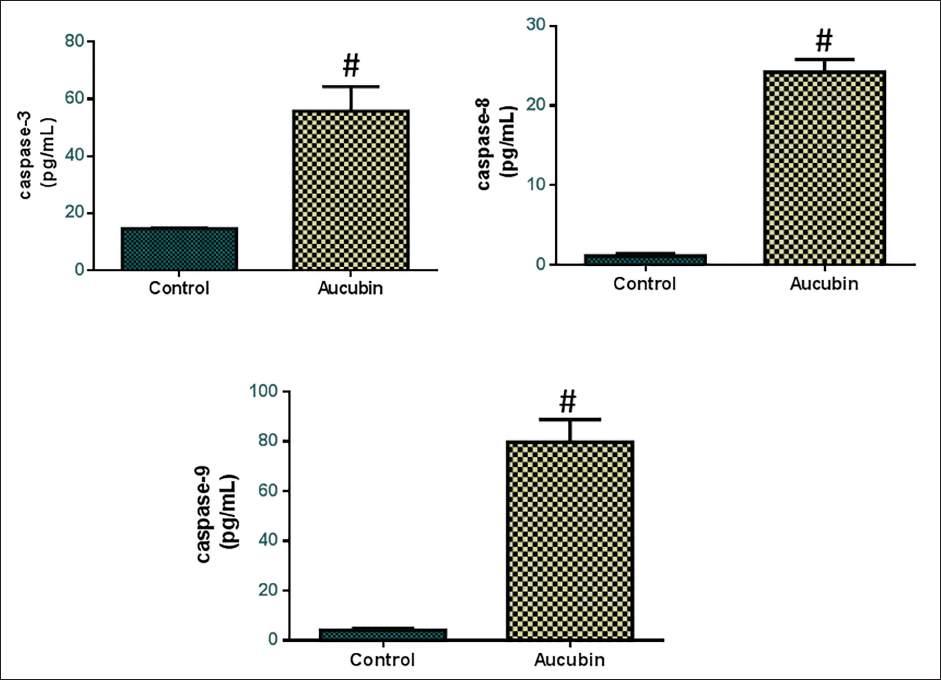
Effect of Aucubin Treatment on the Caspase Expressions in the MG-63 Cells
The apoptotic markers and caspase enzyme activities of the aucubin-treated OS cells were studied using kits (Figure 5). In the control cells, the caspase-3, -8, and -9 enzyme activities were decreased. Nevertheless, the treatment of MG-63 cells with 40 µM/mL aucubin led to a substantial elevation in caspase-3, -8, and -9 activities in comparison to the control. Therefore, it was apparent that aucubin induces apoptosis in the OS cells by promoting caspase activities.
In silico Analysis of Aucubin
Pharmacokinetic Properties and Predicted Values of Aucubin.
Target Prediction
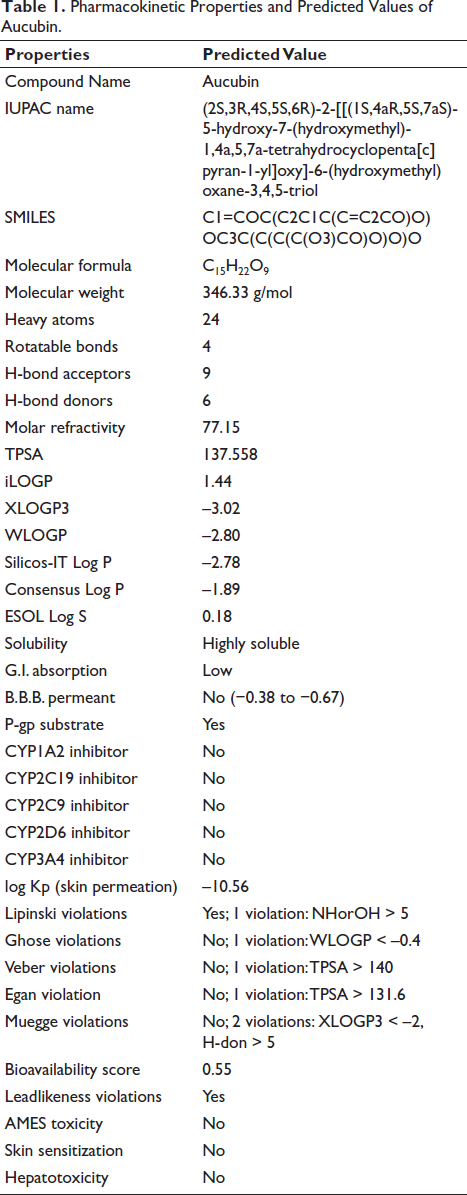
Currently, well-established bio-/chemo-informatics strategies can provide viable support in predicting the most likely small compound targets. The SwissTarget web tool is utilized to make predictions regarding the most likely protein targets of small compounds. The present study was limited to the top 15 human targets. The compound was calculated to have a 53.3% likelihood of binding to lyase receptors and a 20% likelihood of binding to protease enzymes Figure 6. Based on the results of the molecular docking tests, it was determined that the molecule has a 20% likelihood of binding to enzymes.
Top Targets of Aucubin as Predicted Using SwissTarget.
Molecular Docking Analysis of Aucubin
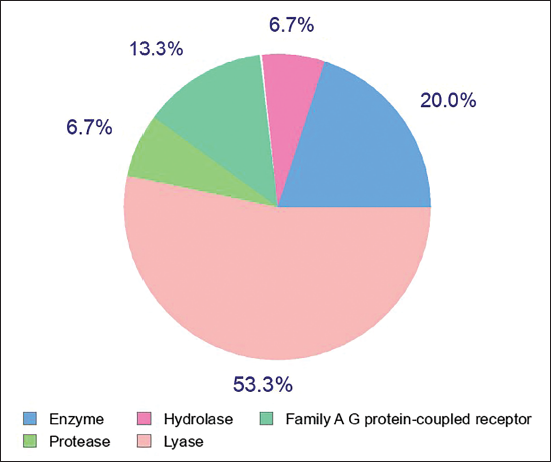
Docking analysis of the molecule aucubin revealed a good docking percentage within the active site of the Crystal Structure of CSF1R (PDB ID: 6T2W) with −7.28 kcal/mol, respectively, revealed better docking affinity, and the molecule demonstrated binding affinity within the binding locations of the co-crystallized ligand (Figure 7). The compound formed hydrogen bonds with GLN547, VAL548, ARG549, and ARG816 and residual hydrophobic interaction with PHE593, ALA629, and LEU799. In silico docking outcomes demonstrated that compound aucubin has the least docked score (−7.2 kcal/mol), respectively (Table 2), suggesting that the determined compound is promising against the Crystal Structure of CSF1R (PDB ID: 6T2W).
2D and 3D Molecular Interaction View of the Aucubin Against the Crystal Structure of CSF1R (PDB ID: 6T2W).
Molecular Docking Scores and Residual Amino Acid Interactions of Aucubin Against Crystal Structure of CSF1R (PDB ID: 6T2W).

Discussion
OS is the predominant type of bone cancer and originates from undifferentiated mesenchymal cells that produce bone tissue. Approximately 80% of OS have morphological abnormalities in osteoblastic cells, resulting in the production of defective osteoid structures. This leads to a lack of differentiation at the histological level. OS is predominantly generated by mesenchymal cells, which commonly develop in the distal and upper regions of the bones, leading to a notable limitation of mobility (Adamopoulos et al., 2017). OS has high mortality rates and treatment failures, mostly due to the presence of lung metastases. OS cells typically exhibit a marked tendency (de Azevedo et al., 2020). Currently, surgical resection is the main method used to treat patients with OS. Interestingly, the bioactive components of natural substances also have a pivotal role in the therapy of OS (Cascini & Chiodoni, 2021). Hence, the present work focuses on understanding the anticancer activity of aucubin against OS cells.
The cytotoxicity assay is a valuable tool in toxicology as it provides insights into cell proliferation, death, and metabolic processes in response to the test compounds (Bharti et al., 2016). This work employed the XTT assay to examine the cytotoxic properties of aucubin at different concentrations in terms of their capacity to impede cell proliferation in MG-63 cells. The XTT assay is a process that involves the enzymatic reduction of tetrazolium salts by the mitochondrial dehydrogenase enzyme, which is found in live cells. The cells are cultured with XTT, which is transformed by live cells into a colored formazan product (Pierce et al., 2008). The cells’ metabolic activity directly indicates the intensity of the color produced by the formazan product. The XTT assay measures the quantity of formazan product directly in the medium (Huyck et al., 2012). The current findings exhibited that the aucubin treatment considerably reduced the MG-63 cell viability.
ROS are substances that are produced as a result of the metabolic processes that occur in mitochondria. Cancer cells possess elevated amounts of baseline ROS, which makes the cells more susceptible to extreme oxidative stress. Thus, an abundance of ROS is likewise lethal to OS cells. Optimal levels of ROS are necessary for normal cellular processes, but an overaccumulation of ROS can trigger apoptosis and block the cell cycle in cancer cells (Büssing et al., 1999). Oxidative stress, caused by excessive generation of ROS, leads to the destruction of large molecules, initiates many signaling pathways, and finally leads to cell death by apoptosis. ROS plays a crucial role in initiating apoptosis in cancer cells. ROS accumulation, which plays a direct role in regulating mitochondrial functions, has been involved in cell death through many pathways (Panieri & Santoro, 2016). As a result, certain ROS-inducing anticancer drugs’ ability to induce cell death is largely dependent on the excessive production of ROS (Reczek & Chandel, 2018). It has been shown that many chemotherapeutic drugs trigger programmed cell death (apoptosis) and self-degradation of cells (autophagy) in tumor cells by producing ROS (Tang et al., 2019). The findings of this work highlighted that the aucubin treatment effectively promoted the production of endogenous ROS in the OS cells, which is evidenced by the considerably increased green fluorescence compared to the control. These outcomes proved that the aucubin treatment can induce oxidative stress-regulated apoptosis in OS cells by promoting ROS generation.
To determine if the cytotoxicity of aucubin to the MG-63 cells was due to apoptosis, dual staining was conducted. Apoptosis-related alterations in the cell membrane can be identified with AO/EtBr staining under a fluorescence microscope. The purpose of this staining method is to differentiate between healthy and apoptotic cells (Ribble et al., 2005). Dual staining is a widely used technique for identifying and measuring apoptosis, both in terms of quality and quantity. The principle relies on the capacity of AO to infiltrate the undamaged membranes of normal cells, leading to the emission of green fluorescence. On the other hand, EB can enter cells undergoing apoptosis that have damaged membranes, causing them to emit orange-red fluorescence (Gherghi et al., 2003). The findings of this work demonstrated an increase in apoptotic and necrotic cells following aucubin treatment.
Apoptosis is a mechanism of programmed cell suicide that serves to eliminate cancerous or defective cells and regulate overall cell growth using various molecular pathways. Apoptosis plays a key role in impeding the advancement of tumors by removing cells that have undergone genetic alterations or are in a premalignant state. Blocking apoptosis in mutant cells is frequently seen as a fundamental component of a successful anticancer treatment, as it can prevent their transformation into malignant cells. When apoptosis fails, it can lead to unregulated cell proliferation and, eventually, the development of tumors. Furthermore, cancer cells can avoid programmed cell death, multiply quickly, and display resistance to therapies used to treat cancer (Rathore et al., 2017). To achieve successful cancer therapy, it is necessary to specifically target and eliminate OS cancer cells. Inducing cell apoptosis and sensitizing resistant tumor cells are crucial in OS therapy (Bouralexis et al., 2003). In this work, the findings of the DAPI analysis evidenced that the aucubin treatment effectively promoted apoptosis in the OS cells.
The caspase is a protease family enzyme that plays a key role in regulating apoptosis. The caspases are categorized into three distinct groups as per their function: executioner (caspase-3), initiator (caspase-9), and inflammatory caspases (caspase-5) (Ghavami et al., 2009). Caspase-3 plays a key role in cancer treatment because it is responsible for breaking down various regulatory proteins that inhibit cell function and are implicated in programmed cell death. Furthermore, caspase-3 activation not only triggers apoptosis but also has a role in cleaving various substrates, hence increasing the susceptibility of tumor cells to undergo apoptosis (Boice & Bouchier-Hayes, 2020). Caspase-9 is the main controller of apoptosis that occurs within the mitochondria and is overexpressed during the onset of apoptosis (Yuan & Akey, 2013). Apoptosis is triggered by the decrease in mitochondrial membrane potential, causing the release of cytochrome c and the caspase-3 activation. Caspase-3, functioning as an intracellular protease, has an essential role in the initial phases of apoptosis. Inducing caspase-3 activity triggers the breakdown of PARP, leading to the excessive utilization of ATP and NAD+ by the catalytic receptor mechanism, finally culminating in cell death (Garrido et al., 2006). In this work, the findings suggested that the aucubin treatment significantly elevated the caspase-3, -8, and -9 activities in the OS cells. The results confirmed that aucubin can trigger apoptosis in OS cells.
Pharmacokinetic and toxicological profile ADME/Tox refers to the processes of drug absorption, distribution, metabolism, toxicity, and excretion. The in silico ADME/Tox profile is a valuable tool for forecasting the pharmacological characteristics of potential sample drugs, particularly during the preclinical phases. To enhance ADME/Tox predictions, in silico techniques have been implemented. These models have been explicitly utilized to enhance drug optimization and prevent late-stage failures, which are significant as they result in substantial wastage of time and money (Durán-Iturbide et al., 2020).
Absorption was forecasted based on the features of water solubility, lipophilicity, and intestinal absorption percentage. The water solubility was determined using the Silicos IT LogSw descriptor of SwissADME and the results are shown in Table 1. LogSw data for our compound is projected to range from –2.80 to –3.02. In the SwissADME LogSw scale, the molecule with a value of 0.18 is deliberated to be highly soluble. Lipophilicity was evaluated by calculating the logarithm of the n-octanol/water partition coefficient, which was projected using the Consensus LogPo/w descriptor of SwissADME. LogPo/w is intricately connected to transport mechanisms, such as membrane permeability, and the dispersion of substances to various tissues and organs (van de Waterbeemd, 2008). An effective strategy to achieve good oral bioavailability, which involves both permeability and solubility, is to maintain a modest logP value within the range of 0–3 (Zerroug et al., 2019). The logPo/w value for our compounds was projected to be –2.80. The anticipated BBB permeability values of our compounds are predicted to be within a range of –0.38 to –0.67. The estimation of metabolism was conducted using SwissADME, which assessed the inhibition of key cytochromes (CYP) belonging to the P450 superfamily. These cytochromes include CYP1A2, CYP2C19, CYP2C9, CYP2D6, and CYP3A4. The molecule was anticipated to inhibit CYP1A2, while it was expected to have little to no inhibitory effect on CYP2C19, CYP2C9, and CYP3A4. Contrarily, the compounds were projected to have no inhibitory effect on CYP2D6. Excretion mostly occurs through a mix of hepatic and renal clearance, and it is influenced by bioavailability. It plays a crucial role in establishing the dosage required to achieve steady-state concentrations (Małkiewicz et al., 2019). The hepatotoxicity and AMES toxicity of the sample drugs were anticipated using pkCSM-pharmacokinetics. The hepatotoxicity description indicated that the chemical does not possess hepatotoxicity. Drug-likeness descriptors, determined using the Lipinski and Veber criteria, were computed using SwissADME. Lipinski’s rule of five asserts that optimal absorption or permeation occurs when the molecular weight (MW) is less than 500 Da, the number of hydrogen bond donors (HBDs) is less than 5, the LogP value is less than 5, and the number of hydrogen bond acceptors (HBAs) is less than 10. Veber et al. discovered two other significant characteristics: the number of rotatable bonds (NBR) being less than 10 and the polar surface area (PSA) being less than 140 Å2 (Małkiewicz et al., 2019). Examinations of the compound revealed a single breach of these regulations, implying that it would exhibit orderly absorption or penetration.
Currently, well-established bio-/chemo-informatics methods can provide support in predicting the targets of small compounds. Ligand-based target prediction has demonstrated exceptional accuracy and rapid identification of accurate protein targets for compounds in the field of drug discovery (He et al., 2019). The SwissTarget web tool is utilized to make predictions regarding the most likely protein targets of small compounds. The present work was limited to the top 15 human targets. The compound was calculated to have a 53.3% affinity for lyase receptors and a 20% affinity for proteases. According to the findings of the docking studies, the molecule was estimated to have a 20% likelihood of binding to enzymes.
A docking study of the molecule aucubin revealed a better docking score within the active site of the crystal structure of CSF1R (PDB ID: 6T2W) with −7.28 kcal/mol, respectively. This demonstrated better docking affinity, and the molecule showed binding affinity within the binding locations of the co-crystallized ligand. The compound formed hydrogen bonds with GLN547, VAL548, ARG549, and ARG816 and residual hydrophobic interaction with PHE593, ALA629, and LEU799. Docking findings exhibited that the compound aucubin has the least docked score (−7.2 kcal/mol), respectively (Table 2), suggesting that the determined compound is promising against the crystal structure of CSF1R (PDB ID: 6T2W).
Conclusion
The present work shows that aucubin treatments inhibit cell proliferation and trigger apoptosis in different staining, like AO/EtBr, ROS, and DAPI, in MG-63 cells. MG-63 cells increased their endogenous ROS production, decreased their viability, and underwent apoptotic cell death via increasing their caspase activities in response to the aucubin treatment. Hence, aucubin possesses the capacity to serve as a hopeful therapeutic option for OS treatment. Furthermore, additional studies are needed to identify the specific molecular mechanisms underlying the anticancer activity of aucubin. This knowledge will facilitate the future use of aucubin to treat OS.
Summary
OS frequently develops on the epiphysis of long bones, with approximately two-thirds of cancers arising in the distal femur bones. As our understanding of the biology of OS has improved, the diverse nature and possible molecular abnormalities associated with OS have been partially documented. Hence, we aimed to scrutinize the anticancer potentials of aucubin against OS MG-63 cells. The aucubin-treated MG-63 cells were studied by an XTT assay. Staining assays were done to assess the endogenous AO/EtBr, DAPI, and ROS accumulation and apoptosis in the aucubin-treated MG-63 cells. The caspase-3, -8, and -9 activities were investigated using the assay kits. The results of this work show that aucubin treatments inhibit cell proliferation and trigger apoptosis in MG-63 cells, suggesting its potential as a promising anticancer agent.
Footnotes
Abbreviations
DMSO: Dimethyl sulfoxide; FBS: Fetal bovine serum; AO/EtBr: Acridine orange/ethidium bromide; ROS: Reactive oxygen species; CYP: Primary cytochromes; MG-63: Human osteosarcoma; PSA: Polar surface area; MW: Molecular weight; HBSs: Hydrogen bond donors; DAPI: 4′,6-Diamidino-2-phenylindole.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
