Abstract
Background
Fucoidan is a phosphorylated polysaccharide extracted from seaweed that has a renal protective effect. However, whether Fucoidan can be used to prevent chronic kidney disease with mineral and bone disorders (CKD-MBD) is still a mystery and thus becomes the target of this research.
Materials and Methods
CKD-MBD mouse models were constructed, and the effects of Fucoidan and Klotho on CKD-MBD were determined through treatment of Fucoidan (100 mg/kg or 200 mg/kg) by gavage or tail vein injection of Klotho specific to small interfering RNA (siKlotho). The biochemical indicators related to renal function and bone metabolism in serum were detected by an automatic biochemical analyzer or enzyme-linked immunosorbent assay. Bone density was measured by X-ray. The effect of Fucoidan or siKlotho on kidney damage was tested by hematoxylin and eosin (H&E) staining or Elastica Masson-Goldner (EMG) staining. The expressions of Klotho, Runt-related transcription factor 2 (Runx2), and α-smooth muscle actin (α-SMA) in kidney tissue after Fucoidan treatment and/or siKlotho injection were quantified by quantitative reverse transcription-polymerase chain reaction (qRT-PCR), immunohistochemistry, or Western blot.
Results
Fucoidan treatment inhibited the levels of blood urea nitrogen (BUN), creatinine, alkaline phosphatase (ALP), phosphorus, intact parathyroid hormone (iPTH), and fibroblast growth factor 23 (FGF23), while promoting 1,25 (OH)2D3 levels in serum. Fucoidan also increased bone density in mice, alleviated kidney damage and fibrosis of kidney tissue, promoted the expression of Klotho and Runx2, and inhibited the expression of α-SMA in kidney tissue. However, the above-mentioned therapeutic effects of Fucoidan were all reversed by siKlotho.
Conclusion
Fucoidan prevents CKD-MBD by up-regulating Klotho levels.
Introduction
Chronic kidney disease-mineral and bone disorder (CKD-MBD) is an extension of the concept of renal bone disease or renal osteodystrophy. It describes a systemic metabolic disorder, which is characterized by abnormal levels of phosphorus, calcium, and other minerals in serum and tissue, parathyroid hormone (PTH), active Vitamin D, and fibroblast growth factor, as well as aberrant bone morphology such as bone mineralization (Ketteler et al., 2017; Yamada & Giachelli, 2017). The current treatment strategies for CKD-MBD include reducing the serum Pi level and the excessive secretion of PTH as much as possible and keeping the calcium concentration within the normal range (Bover et al., 2017; Isakova et al., 2017). However, these methods currently used in clinical practice have varying degrees of side effects, which are not ideal for the treatment of CKD-MBD, so novel treatment methods are necessarily required.
Seaweed is the most abundant species of marine plants, with phosphorylated polysaccharide as one of the main active ingredients (Brown et al., 2014). Fucoidan is a phosphorylated polysaccharide extracted from the marine natural product brown algae that possesses various functions in antioxidant, anti-tumor, anti-inflammation, and immunomodulation (Luthuli et al., 2019). Of note, Fucoidan has a certain therapeutic effect on kidney-related diseases. Chen et al. (2017) reported that oligo-Fucoidan can reduce the expression of α-smooth muscle actin (α-SMA) and inhibit tubulointerstitial fibrosis in CKD mice (Chen et al., 2017). Jia et al. (2016) found that low-molecular-weight Fucoidan can lessen blood creatinine, urea nitrogen, and urine albumin levels in CKD mice and plays a significant protective role in kidney damage and renal insufficiency (Jia et al., 2016). In addition, Katai et al. (2015) unveiled that wakame with Fucoidan as the main component can inhibit the level of Pi in the plasma of rats with a high-Pi diet (Katai et al., 2015). These evidences indicated the possibility of Fucoidan working as an effective drug for CKD-MBD. Klotho is a gene closely related to human aging (Xu & Sun, 2015), whose roles in maintaining kidney function and preventing kidney failure have attracted people’s attention (Buchanan et al., 2020). For instance, it was discovered that mice defective in the Klotho gene will develop a series of phenotypic changes similar to human aging, including growth retardation, arteriosclerosis, osteoporosis, and metabolic disorders (Kuro-o et al., 1997). Notably, most of the symptoms are also common in patients with chronic renal failure. Further study has unraveled that Klotho is highly expressed in kidney tissues (Dai & Yang, 2018). When chronic renal failure occurs, the Klotho level in patients will apparently be downregulated (Koh et al., 2001). Since Klotho has an intimate relationship with CKD, it is worth exploring the treatment of CKD from the perspective of the Klotho gene.
Fang et al. (2020) reported that Fucoidan can regulate the expression of Klotho in human proximal renal tubular epithelial cells so as to achieve a certain effect on improving renal cell senescence (Fang et al., 2020). However, it is unclear whether Fucoidan can improve CKD-MBD by regulating Klotho, which is worthy of further exploration.
Materials and Methods
Chemicals and Reagents
The following chemicals and reagents were used in this study: Fucoidan (F8315, ≥95%, Merck, German, dissolved in medium); 0.2% adenine (QC25-00240, Qcheng BIO, China); sodium pentobarbital (P-010, 1.0 mg/mL in methanol, Merck, German); phosphate buffered saline (PBS, 806552, Merck, German); xylene (A530011, ≥99%, Sangon, China) and ethanol (A500737, ≥99.7%, Sangon, China); citrate buffer (BL604A, Biosharp, China); and hydrogen peroxide solution (88597, 3%, Merck, German).
Animals
Eighty male C57BL/6 mice (SPF grade, 8 weeks old, 20–23 g) were purchased from Guangdong Medical Experimental Animal Center (
The mouse model of CKD-MBD was established according to the report of Lin et al. (2017) and Tani et al. (2020), and the selection of Fucoidan dose (100 and 200 mg/kg) was referred to in the previous literature (Wei et al., 2017; Yu et al., 2018). The experimental animals were distributed into 5 groups, namely, the control group, the model group, the model + 100 group, the model + 200 group, and the control + 200 group, with 10 mice in each group. The mice in the control group and the control + 200 group were fed with a standard chow containing 0.8% Pi until the 12th week, but those in the control + 200 group were additionally given 200 mg/kg Fucoidan by gavage daily from the 6th to the 12th week. The mice in the model group, model + 100 group, and model + 200 group were fed with chow containing 0.2% adenine and 0.8% Pi until the 6th week and with 0.2% adenine and 1.8% Pi from the 6th week to the 12th week. The mice in the model + 100 group and the model + 200 group also received 100 and 200 mg/kg of Fucoidan by gavage daily from 6th to 12th week, respectively.
To study the effect of Klotho-specific small interfering RNA (siKlotho, AM16708, ThermoFisher, USA) during Fucoidan treatment (200 mg/kg) of CKD-MBD mice, 30 mice were divided into the model + Fucoidan + negative control for siRNA (siNC, AM4611, ThermoFisher, USA) group, the model + Fucoidan + siKlotho group, and the model + siKlotho group. These three groups of mice received tail vein injections of siKlotho or siNC (10 nM in 200 µL of PBS) six times a week.
After the mice were euthanized by intraperitoneal injection of sodium pentobarbital (150 mg/kg) (Laferriere & Pang, 2020), their blood, kidney tissues, and femurs were collected for further research. The blood of mice was centrifuged to obtain serum. The contents of blood urea nitrogen (BUN), creatinine, alkaline phosphatase (ALP), and Pi in the serum were measured by an automatic analyzer (Cobas c111, Roche, Switzerland) according to the descriptions of previous studies (Andrukhova et al., 2018; Lin et al., 2017).
Enzyme-linked Immunosorbent Assay (ELISA)
The contents of intact parathyroid hormone (iPTH), fibroblast growth factor 23 (FGF23), and 1,25-dihydroxyvitamin D3 (1,25(OH)2D3, DHVD3) in the serum were measured by the Mouse iPTH ELISA Kit (E-EL-M0709, Elabscience, China), Mouse FGF23 ELISA Kit (E-EL-M2415C, Elabscience, China), and Mouse DHVD3 ELISA Kit (E-EL-0016C, Elabscience, China), respectively. In short, the operation step was to make the standard or sample (100 µL) react with the biotinylated antibody working solution (100 µL), horseradish peroxidase enzyme conjugate working solution (100 µL), substrate solution (90 µL), and stop solution (50 µL) in order. The absorbance at 450 nm was detected with a multiple detection reader (Fluoroskan Ascent FL, Thermo Fisher, USA).
Bone Mineral Density (BMD) Measurement
After the animal was sacrificed, the femur was collected, and the surrounding soft tissues and ligament structures were removed. BMD was measured with the help of X-ray densitometers (Lunar-Prodigy, General Electric Company, USA).
Hematoxylin and Eosin (H&E) Staining and Elastica Masson-Goldner (EMG) Staining
The procedures of H&E staining and EMG staining were referred to in the previous study. H&E staining of kidney tissue was accomplished with the help of a H&E staining kit (SL7070, Coolaber, China). The kidney tissues were immersed in xylene and ethanol with gradient concentrations (100%-95%-80%-75%) after routine fixation, embedding, and sectioning. After washing with water, the tissue sections were soaked in hematoxylin staining solution for 10 min, differentiation solution for 30 s, tap water for 15 min, and eosin staining solution for 2 min. Following a rinse with tap water, the tissue sections were immersed in ethanol with gradient concentrations and xylene in sequence, and finally sealed with neutral resin (E675007, Sangon Biotech, China).
The EMG staining of tissue sections was performed by means of the EMG Staining Kit (1004850001, Merck, Germany). The operation steps were carried out strictly in accordance with the instructions in the manual. The results of H&E and EMG staining were observed under an optical microscope (BX53M, Olympus, Japan, 100×).
Immunohistochemistry
According to the previous study, the deparaffinized sections were placed in citrate buffer and then heated to 95°C for 15 min by microwave. The sections also needed to be soaked in a 3% hydrogen peroxide solution for 10 min to eliminate endogenous peroxidase activity. After being blocked by 5% bovine serum albumin (BSA, E661003, Sangon, China) for 30 min, the sections were incubated with the primary antibody Klotho (1:200, ab203576, Abcam, UK) at 4℃ overnight. The next day, the sections were incubated with the secondary antibody (1:1000, ab97051, Abcam, UK) at room temperature for 1 h. Subsequently, the sections underwent 3,3ʹ-diaminobenzidine (DAB) color development (DA1010, Solarbio, China), hematoxylin counterstaining, gradient alcohol dehydration, xylene permeabilization, and neutral resin mounting. The expression of Klotho in the kidney tissues of each group was observed under a microscope (BX53M, Olympus, Japan, magnification, 100× or 400×).
Western Blot
A Western blot was conducted as previously described (Alegria-Schaffer et al., 2009). In general, the protein was extracted from kidney tissue and quantified with the Protein Extraction Kit (BC3790, Solarbio, China) and the Bicinchoninic Acid (BCA) Protein Assay Kit (PC0020, Solarbio, China). The protein was separated by 6% sodium dodecyl-sulfate polyacrylamide gel electrophoresis (SDS-PAGE, P0686, Beyotime, China) and transferred to a polyvinylidene fluoride membrane (YA1701, Solarbio, China). After blocking by 5% BSA for 1 h, the membrane was reacted with following primary antibodies at 4°C overnight: Klotho (1:1000, 116 kDa, ab203576, Abcam, UK), Runt-related transcription factor 2 (Runx2, 1:1000, 57 kDa, ab236639, Abcam UK), α-SMA (1:1000, 42 kDa, #19245, cell signaling technology, USA), and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) (1:1000, 36 kDa, ab8245, Abcam, UK). GAPDH was used as an internal control. Then the membrane was cultivated with goat anti-rabbit (1:5000, ab6721, Abcam, UK) or goat anti-mouse (1:5000, ab6788, Abcam, UK) secondary antibodies for 2 h. The protein bands were visualized by iBright CL750 (Thermo Fisher, USA) with a West Pico ECL substrate (PE0020, Solarbio, China), the intensity of which was counted using ImageJ2x (Rawak Software, Germany).
Quantitative Reverse Transcription-Polymerase Chain Reaction (qRT-PCR)
The RNA from tissues or cells was extracted by the Total RNA Extraction Kit (R1200, Solarbio, China). The relative expression of Klotho was determined using the FastKing One-Step Reverse Transcription-Fluorescence Quantitative Kit (FP314, Tiangen, China). GAPDH was employed as the reference gene as needed. The data were shown on a real-time PCR detection system (CFX96, Bio-rad, USA). The primers were listed as follows: Klotho forward: 5ʹ-GATGGCAGAGAAATCAACACAGT-3ʹ and reverse: 5ʹ-ACTACGTTCAAGTGGACACTACT-3ʹ; GAPDH forward: 5ʹ-CTCTCTGCTCCTCCTGTTCGACAG-3ʹ and reverse: 5ʹ-GTGTAATCATATTGGAACATGTAG-3ʹ. The relative expression was standardized using the 2–∇∇Ct method (Livak & Schmittgen, 2001).
Statistical Analysis
Data were analyzed using the Graph Prism v8.0 (Graphpad Software, California, USA) and represented as mean ± standard deviation. Differences among multiple groups were analyzed by one-way analysis of variance. p < 0.05 was accepted to be statistically significant.
Results
Effect of Fucoidan Treatment on Serum Biochemical Indexes in CKD-MBD Mice
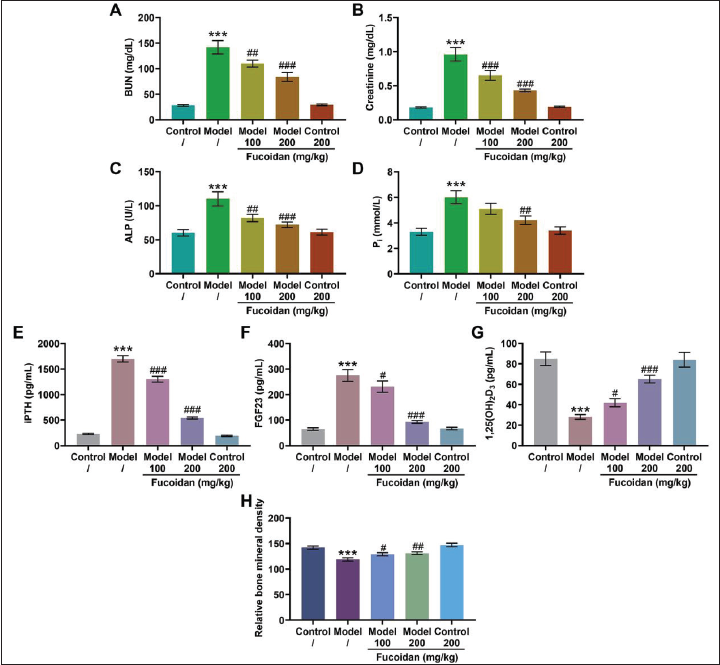
We found that compared with the control group, the serum levels of BUN (Figure 1A, p < 0.001), creatinine (Figure 1B, p < 0.001), ALP (Figure 1C, p < 0.001), and Pi (Figure 1D, p < 0.001) in the model group were significantly increased. In addition, the serum of the model group mice exhibited higher levels of iPTH and FGF23 compared with that of the control group mice (Figure 1E and F, p < 0.001). Interestingly, Fucoidan treatment markedly reduced the above-mentioned serum biochemical indicators in the model group in a concentration-dependent manner but had no significant effect on the serum biochemical indicators of mice in the control group (Figures 1A–F, p < 0.05). Not only that, Fucoidan treatment abrogated the decrease in serum 1,25(OH)2D3 content and BMD caused by modeling (Figure 1G and H, p < 0.05).
Figures 2A and B shows the histopathological changes of mouse kidney tissues, in which the white arrow indicated lumen expansion, the black arrow signified the shedding of renal tubular epithelial cells, and the yellow arrows depicted degenerative changes in the wall of arterioles. As delineated in Figure 2A, compared with that of the control group, the kidney surface of the model group mice was irregular, and kidney atrophy occurred. In addition, the kidney tissue of the model group mice also showed the shedding of renal tubular epithelial cells, the invasion of inflammatory cells, and the expansion of the tubular cavity. Strikingly, Fucoidan alleviated the renal tubular damage in model mice in a concentration-dependent manner. Figure 2B describes the results of EMG staining. We could notice that the kidney tissue of the model mice had a large number of fibrotic structures accompanied by luminal expansion, but the glomerular structure remained normal, and no obvious degenerative changes were observed in the arteriolar walls. However, Fucoidan treatment could alleviate kidney fibrosis in the model mice.
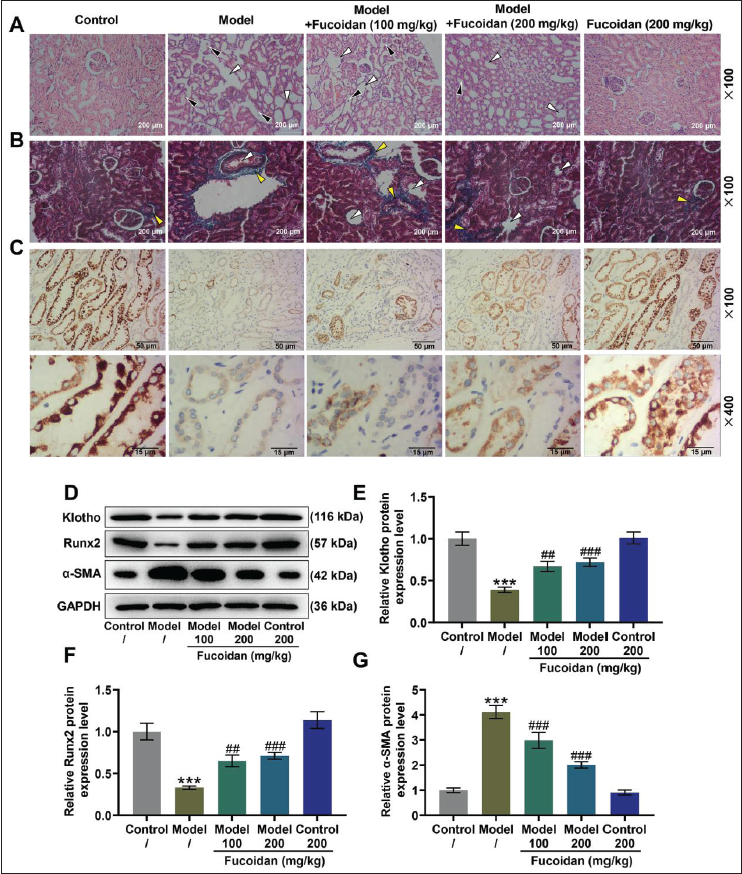
Through immunohistochemistry, the expression of Klotho in the mice of the model group fed with high Pi was significantly lower than that of the control group (Figure 2C). In addition, the quantification of Klotho expression in kidney tissue by Western blot also reached the same conclusion (Figure 2D and E, p < 0.001). At the same time, downregulation of Runx2 and upregulation of α-SMA were found in the kidney tissue of the model group (Figure 2D and F–G, p < 0.001). Nevertheless, Fucoidan dose-dependently reversed the decrease in Klotho and Runx2 expressions and the increase in α-SMA expression caused by modeling (Figures 2C–G, p < 0.01). Since 200 mg/kg Fucoidan had a more obvious protective effect on the kidneys of CKD-MBD mice, this concentration was selected for subsequent experiments.
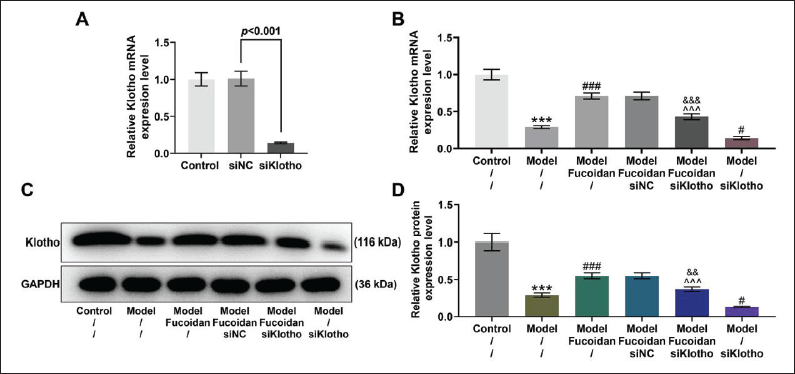
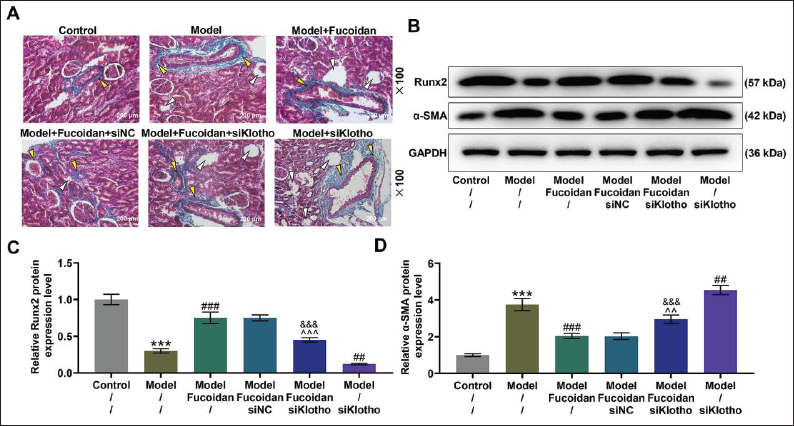
Figure 3A shows that the transfection of siKlotho can effectively suppress the expression of Klotho in mouse kidney tissue (p < 0.001). In addition, siKlotho abrogated Fucoidan treatment-induced up-regulation of Klotho in the kidney tissue of mice in the model group (Figures 3B–D, p < 0.01). Notably, when Klotho was silenced, the fibrosis of the kidney tissue in the model group became more serious, and the renal protective effect of Fucoidan was also attenuated (Figure 4A). Similarly, the expression of Runx2 was decreased, while the expression of α-SMA was increased in the Model + siKlotho group compared with those in the model group (Figures 4B–D, p < 0.01); however, these Fucoidan treatment-induced changes of Runx2 and α-SMA expressions were reversed by siKlotho (p < 0.01).
Discussion
CKD-MBD is a systemic disorder of mineral and bone metabolism caused by CKD. CKD is accompanied by two main complications, namely, secondary hyperparathyroidism (sHPT) and vascular calcification (Elias et al., 2018). Disorders of these organs can lead to abnormal bone formation and bone resorption, and abnormal bone metabolism can cause bone deformation and bone pain and increase the risk of fractures (Nickolas et al., 2010). These symptoms are attributed to the reduction of renal phosphate excretion in CKD patients, and renal insufficiency results in low expression of α-1-hydroxylase, which reduces the content of calcitriol. In order to maintain the steady state of serum calcium and Pi, the expressions of PTH and FGF-23 will be increased accordingly (Mejía et al., 2011). A high level of PTH will bring about increased bone loss, and a high level of FGF23 will reduce hyperphosphatemia at the cost of inhibiting 1,25(OH)2D3, leading to sHPT (Stubbs et al., 2007). BUN and creatinine are two indicators used clinically to evaluate renal function (Lyman, 1986), the up-regulation of which is consequent on abnormal kidney function. ALP is a sign of bone formation and mineralization. Beddhu et al. (2009) found that the increase in serum ALP levels is highly related to the mortality of CKD patients (Beddhu et al., 2009).
Based on the above findings, we studied the changes in the above-mentioned biochemical indicators and BMD after Fucoidan treatment. It came out that the serum levels of BUN, creatinine, ALP, Pi, iPTH, and FGF23 in the model group were remarkably higher, but the 1,25(OH)2D3 and BMD levels were lower when compared with those in the control group. These indicated that the kidney function of the mice had been severely damaged after modeling, accompanied by bone abnormalities and metabolic disorders such as aberrant calcium and Pi levels. However, Fucoidan treatment can significantly improve the above-mentioned symptoms. Not only that, Fucoidan had a certain alleviating effect on renal tubular epithelial cell shedding and renal tissue fibrosis in CKD mice, which is similar to the conclusions of Wang et al. (2012). They synthesized some Fucoidan derivatives, including over sulfated, phosphorylated, aminated, acetylated, and benzoylated Fucoidan, and low-molecular-weight Fucoidan derivatives, to compare their antioxidant and anti-coagulant activities. Subsequently, they found that Fucoidan derivatives have a protective effect on the kidneys of CKD rats and that benzoylated Fucoidan derivatives (150 mg/kg/day) and benzoylated low-molecular-weight Fucoidan derivatives (150 mg/kg/day) possess stronger antioxidant activity than native Fucoidan (150 mg/kg/day). In addition, Chen et al. (2021) reported that multiple drug combinations, including oligo-Fucoidan, fucoxanthin, and L-carnitine have an inhibitory effect on renal fibrosis in CKD mice and can reduce the level of creatine in the serum of CKD mice with doses of 10 (oligo-Fucoidan), 300 (fucoxanthin), and 50 (L-carnitine) mg/kg/day (Chen et al., 2021).
A large amount of animal data and clinical observations provided sufficient evidence that Klotho, a protein produced by the kidney that has anti-aging and phosphaturic properties, is not only an early biomarker for CKD (Sanchez-Niño et al., 2020) but also an intermediary for the development of CKD and CKD complications, including vascular calcification and aging (Hu et al., 2011; Lim et al., 2019). Artificially supplementing Klotho is beneficial to inhibit the development of kidney fibrosis and CKD (Neyra & Hu, 2016). In our study, the decrease in Klotho levels in CKD mice can be reversed by Fucoidan treatment. Moreover, Fucoidan treatment also promoted the expression of Runx2 but suppressed the expression of α-SMA in CKD mice. Runx2 is a key regulator that modulates the differentiation and proliferation of osteoblasts and promotes bone formation (Komori, 2020). A study has demonstrated that TSA treatment can restore the level of Runx2 in adenine mice, which is vital in restoring CKD-caused bone loss (Lin et al., 2017). The up-regulation of α-SMA usually means an aggravation in the degree of renal fibrosis (Li et al., 2019; Sun et al., 2016). On this basis, it can be concluded that Fucoidan treatment can relieve bone loss and kidney fibrosis in CKD mice.
By silencing Klotho, we explored whether the mechanism of Fucoidan in the treatment of CKD-MBD is related to the upregulation of Klotho. The results verified that after silencing of the Klotho, the regulatory effects of Fucoidan on renal fibrosis and expressions of Runx2 and α-SMA were reversed, indicating that Fucoidan protects kidneys by regulating Klotho’s expression. This conclusion is similar to the results of Fang et al. (2020) that Fucoidan prevents kidney aging by reducing the expression of Klotho (Fang et al., 2020). Moreover, in contrast to Fang et al.’s (2020) study, our novelty lies in the first proposal that Fucoidan ameliorated mineral and bone disorders in CKD by upregulating Klotho expression.
Conclusion
In a word, our research fully proves the mechanism by which Fucoidan improves CKD-MBD by upregulating the expression of Klotho, providing a new method and theoretical basis for the prevention and treatment of CKD-MBD patients. Nevertheless, in vitro experiments are additionally needed to validate our results in the future. Besides, to establish its clinical applications, more clinical trials are demanded to confirm the safety, other potential effects, and feasibility of Fucoidan in alleviating kidney and bone damage in patients with CKD-MBD.
Footnotes
Summary
Fucoidan attenuated kidney and bone damage in CKD-MBD mice by upregulating Klotho levels.
Acknowledgments
This work was supported by the 2019 Science and Technology Project of the Changzhou Municipal Health Commission [ZD201925].
Abbreviations
CKD-MBD: chronic kidney disease-mineral and bone disorder; iPTH: intact parathyroid hormone; α-SMA: α-smooth muscle actin; BUN: blood urea nitrogen; ALP: alkaline phosphatase; FGF23: fibroblast growth factor 23; H&E: hematoxylin and eosin; EMG: Elastica Masson-Goldner; DAB: 3,3ʹ-diaminobenzidine; BCA: bicinchoninic acid; GAPDH: glyceraldehyde-3-phosphate dehydrogenase; qRT-PCR: quantitative reverse transcription polymerase chain reaction; sHPT: secondary hyperparathyroidism; Runx2: Runt-related transcription factor 2; PTH: parathyroid hormone; BMD: bone mineral density; BSA: bovine serum albumin.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The mice were reared in the Nanfang Hospital Central Animal Laboratory and had free access to food and water. Our research was approved by the Nanfang Hospital Animal Ethics Committee with the approval number S202008005.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the 2019 Science and Technology Project of the Changzhou Municipal Health Commission [ZD201925].
Informed Consent
Not applicable.
