Abstract
Soft tissue mineralization was diagnosed in 19 captive 2-toed sloths (
Keywords
Two-toed sloths (
Soft tissue mineralization is generally caused by calcium and phosphorus imbalance—particularly calcium and phosphorus excess with extracellular deposition of amorphous calcium phosphate or hydroxyapatite crystal. Targeted tissues include those with natural acid secretion and an internal alkaline environment that allows for mineral deposition. 9 Mineralization is promoted in tissues with decreased mechanisms for inhibition of mineralization. 10 Susceptible sites include the gastric mucosa, renal cortex and pelvis, pulmonary interstitium and venules, and arteries throughout the body. 9,22 Causes of hypercalcemia and hyperphosphatemia include dietary calcium and phosphorus excess, dietary vitamin D excess, and primary or secondary elevations in parathyroid hormone or parathyroid hormone–related protein, as might occur with renal disease or as part of a paraneoplastic syndrome, respectively. 9,22 Soft tissue mineralization caused by these various etiologies is well described in domestic dogs, 19 –21 cats, 14 horses, 4 and pigs. 6 This study examined the gross and histologic lesions from 19 captive 2-toed sloths that had soft tissue mineralization at the time of death.
Materials and Methods
Nineteen captive 2-toed sloths of species
Results
Soft tissue mineralization of organs from 19 captive 2-toed sloths was detected by gross and histologic examination along with other visceral lesions (Supplemental Table 1). Clinical histories and results of perimortem blood biochemistry, hematocrit, urinalysis, and radiographs were provided for 8 sloths (Supplemental Table 2). Sloths ranged in age from 2 months to 41 years old, with a mean age of 12 years. Three sloths were aged as “adult” with exact age unknown. Soft tissue mineralization was equally severe in younger (<4 years;
In 6 of 19 sloths, mineralization was grossly evident, predominantly in the aorta and other great arteries. Affected arteries were palpably firm, brittle, and discolored white-grey. The vascular wall and endothelial surface were markedly thickened and irregular, and the lumen was partially occluded (Fig. 1). Histologically, arterial mineralization was identified in 11 of 14 sloths for which vascular tissue was submitted for histologic review. Mineralization varied from severe (9 sloths) to moderate (2 sloths) or absent (3 sloths). Vascular mineralization involved mostly arterioles and major arteries, with minor mineralization of veins in the lungs, gastric mucosa, and kidneys. The intima and internal elastic membrane were markedly hypertrophied with segmental mineralization. The tunica muscularis was extensively expanded by mineralization, with foci of cartilaginous and osseous metaplasia, fibroplasia, and smooth muscle hyperplasia (Fig. 2). Vascular lesions were consistent with medial arteriosclerosis (Mönckeberg sclerosis). Of these 11 sloths with arterial mineralization, 10 also had cardiac lesions characterized by overall cardiac enlargement (1 of 10 sloths), acute myocardial necrosis (4 of 10 sloths), and chronic myocardial fibrosis (5 of 10 sloths; Fig. 3). One sloth (No. 9) had atherosclerosis of great arteries with prominent subintimal plaques. Additionally, this sloth had extensive mineralization of the arterial tunica media with osseous and chondroid metaplasia, as was detected in others. The sloth depicted grossly (No. 1) had severe arterial mineralization and a confirmed dietary history of vitamin D, calcium, and phosphorus supplementation originally intended for birds cohoused with the affected sloth. Histologically, this sloth had marked parathyroid gland atrophy consistent with prolonged hypercalcemia, and the perimortem blood calcium level was significantly elevated at 12.7 mg/dl.
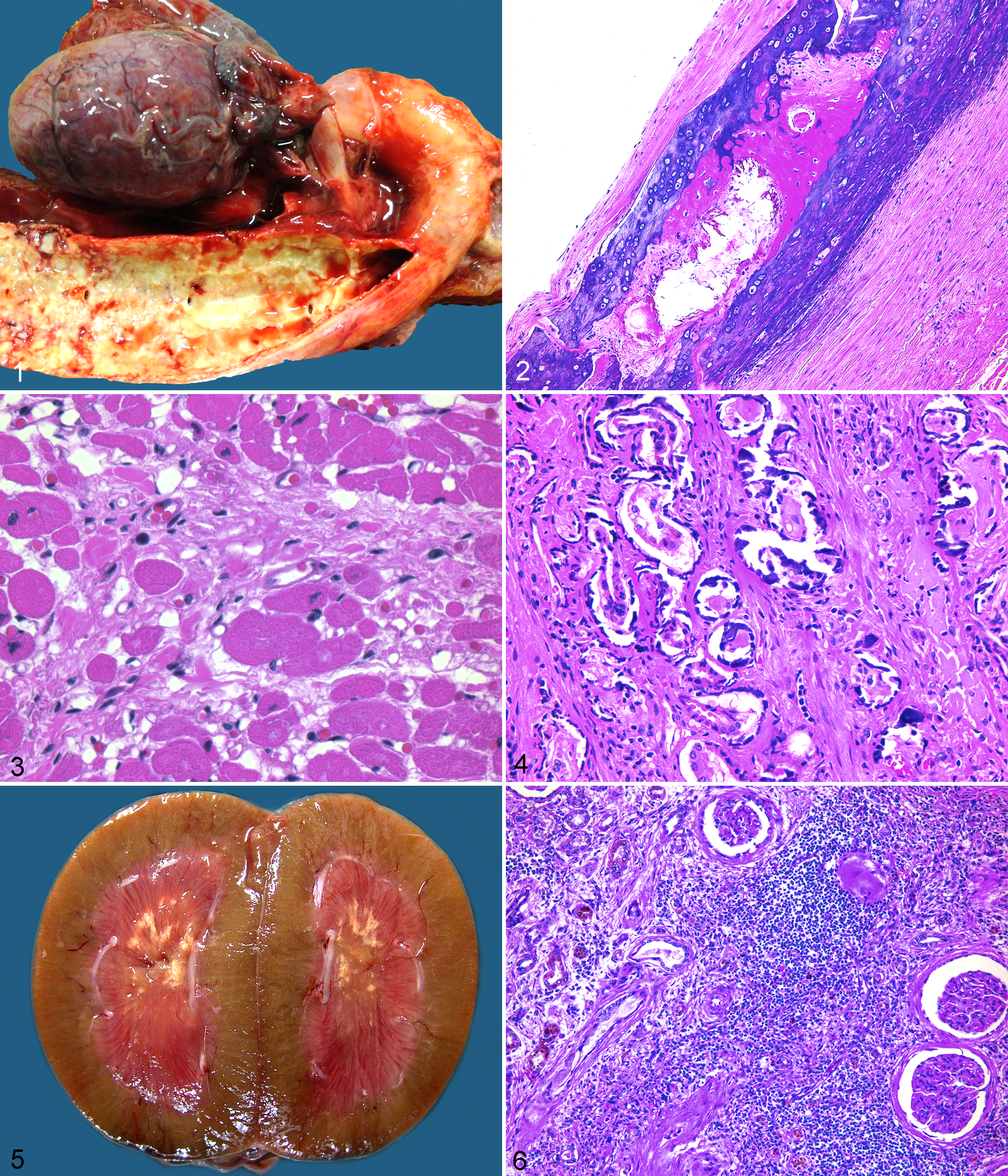
Soft tissue mineralization of the viscera was moderate to severe in all 19 sloths and mostly involved renal basement membranes (17 of 18 sloths for which kidneys were submitted for histologic review; 12 sloths were moderately to severely affected), gastric mucosa (17 of 17 sloths for which glandular stomach was submitted for histologic review; 15 sloths were severely affected), and lung (8 of 18 sloths with lungs submitted for histologic review; all moderately to severely affected). Two sloths had radiographic evidence and histologic confirmation of hyperostosis of the ribs.
Renal mineralization was detected in 17 of 18 sloths for which kidneys were submitted for histologic review and was centered on Bowman capsules and the basement membrane of proximal and distal renal tubules, with occasional diffuse mineralization of tubules. Interstitial mineralization was common and extensive, and foci frequently incorporated veins and arterioles (Fig. 4). Of 17 affected sloths, 3 had birefringent, fan-shaped crystals within cortical and medullary renal tubules, consistent with calcium oxalate crystals. Each of these sloths had grossly visible renal mineralization, nephro-, and/or uroliths diagnosed radiographically antemortem and at necropsy. The composition of crystals and liths was not determined or reported on any case (Fig. 5). Of the 17 sloths, 11 had moderate to severe degenerative renal lesions characterized by fibrosis of the interstitium, tubular basement membranes, Bowman capsules, and glomerular mesangium. Fifteen of the sloths had lymphoplasmacytic interstitial nephritis, which ranged from mild (4 sloths) to moderate (5 sloths) to severe (6 sloths; Fig. 6). Moderate to severe renal disease was most prevalent in sloths >5 years old, and degenerative renal lesions were often associated with equally advanced soft tissue and renal mineralization.
Of 17 sloths with stomach submitted for histologic evaluation, all had moderate to severe gastric mineralization of the glandular mucosa. Mineralization in 1 case (sloth No. 19) was grossly evident and characterized by marked irregular mucosal thickening and black-grey mucosal discoloration with erosions and ulcers (Fig. 7). Histologically, mineralization in all cases was centered on a wide band in the mid–lamina propria and extending throughout the mucosa in severe cases. Mineralization effaced blood vessels and gastric glands and was associated with mild lymphoplasmacytic inflammation, abundant mucosal edema, and erosion or ulceration (Fig. 8).
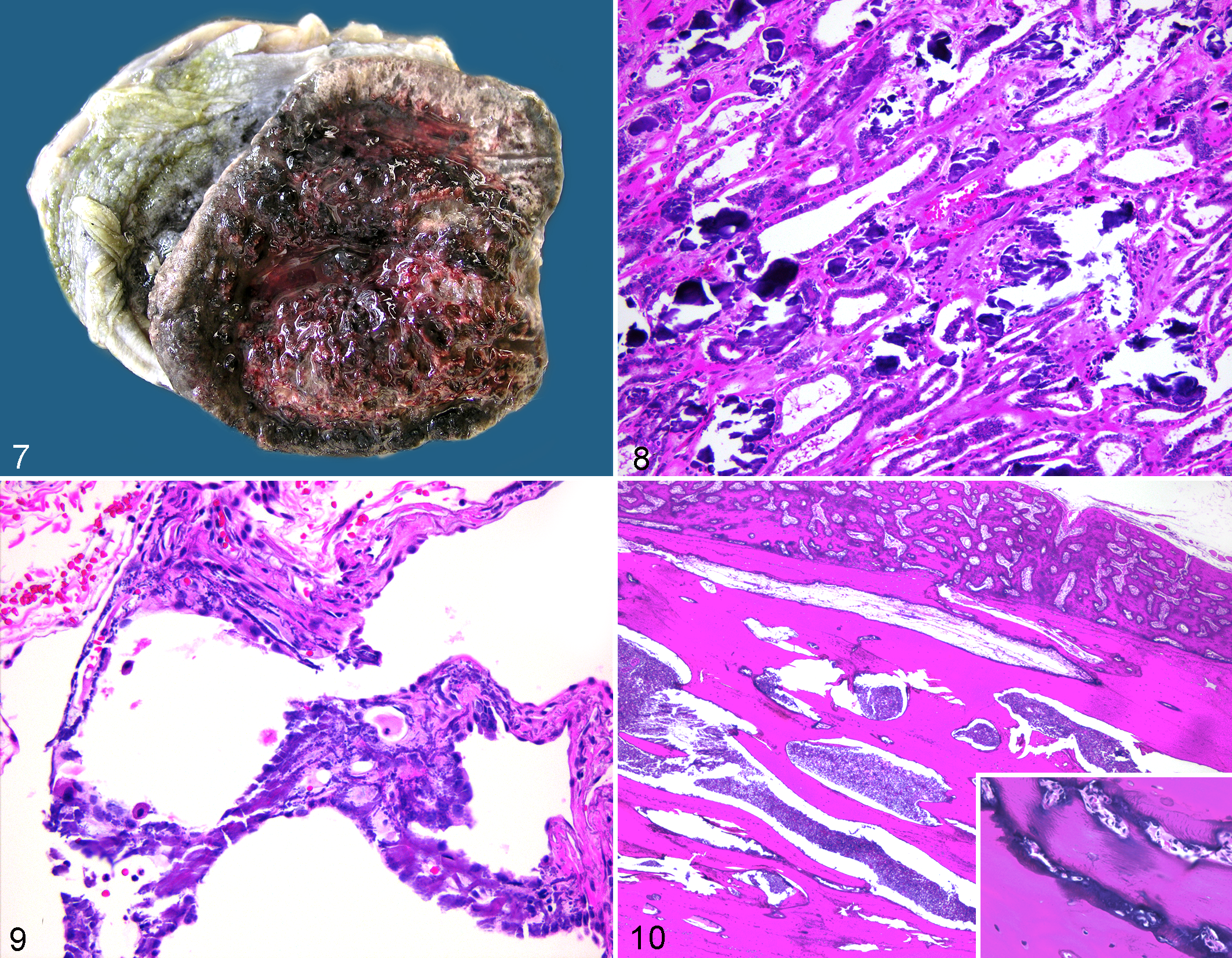
Pulmonary mineralization was detected in 8 of 18 sloths with lung submitted for histologic evaluation and varied from moderate (4 sloths) to severe (4 sloths) with mineralization of arteriolar and venular walls and the interstitium. In severe cases, mineralization effaced the alveolar architecture (Fig. 9).
Two sloths had hyperostosis noted radiographically on the ribs 1 to 2 years prior to death. The ribs were grossly irregular with cortical thickening. Histologically, there was subperiosteal proliferation of woven and lamellar bone. Woven bone had extensive scalloping and remodeling with osteoblast proliferation. Medullary trabecular bone was markedly thickened and lamellar with narrowed trabecular spaces such that cortical and trabecular bone was difficult to differentiate histologically (Fig. 10). Bone lacunae were surrounded by irregular and excessive globular deposition of basophilic matrix (Fig. 10, inset).
One geriatric sloth (No. 18) had moderate arterial and renal mineralization with transitional cell carcinoma of the urinary bladder. Parathyroid glands were histologically normal, and there was mild renal interstitial fibrosis and interstitial nephritis. With no advanced renal disease or abnormal dietary history, soft tissue mineralization in this case was thought to be due to paraneoplastic hypercalcemia related to transitional cell carcinoma.
Perimortem blood chemistry, complete blood cell count, and urinalysis were available on 8 of the 19 sloths with soft tissue mineralization and are summarized with terminal clinical signs (Supplemental Table 2). Of these 8 sloths, 2 sloths had clinicopathologic renal failure based on marked elevation of serum urea, creatinine, and/or calcium and phosphorus, 7 which correlated with severe histologic renal disease. Prior to death, 4 of the 8 sloths had marked elevation of calcium, and 3 had elevated phosphorus. Interestingly, all 8 sloths had urine-specific gravities that appeared isosthenuric with or without azotemia, although reference ranges for urine-specific gravity are not available on this species. Polyuria and polydipsia were noted clinically in only 1 sloth (No. 3), and this sloth had normal serum urea with mild histologic interstitial nephritis but severe renal and visceral mineralization. Other sloths with soft tissue mineralization presented with emaciation (3 sloths) or anorexia and weakness (6 sloths).
Discussion
This study retrospectively examined 19 captive 2-toed sloths with lesions of soft tissue mineralization and characterized the gross and histologic lesions. Soft tissue mineralization in 2-toed sloths occurred in visceral locations typically described in other species 9 with marked mineralization of the gastric mucosa, kidneys, major arteries, and lungs. The cause of soft tissue mineralization is speculated.
Dietary vitamin D excess was considered likely in 3 of the 19 sloths in this study. In 1 case, chronic unintentional supplementation of vitamin D, calcium, and phosphorus was documented. This sloth had severe soft tissue mineralization with marked parathyroid gland atrophy consistent with prolonged hypercalcemia. In 2 other sloths, vitamin D toxicosis was diagnosed by pathognomonic bone lesions, which included hyperostosis of subperiosteal and trabecular bone and excess deposition of glycosaminoglycan matrix. 22
Calcium and phosphorus imbalance due to secondary renal hyperparathyroidism was suspected in 13 of the 18 sloths for which kidney was submitted for histologic evaluation. Renal disease is a common cause of morbidity and mortality in captive sloths 12,13,18 and was prevalent in this study group, in which all 18 sloths showed mild to severe renal inflammation or degeneration. Degenerative renal lesions were most significant in sloths ≥5 years old. Additionally, antemortem renal failure was demonstrated clinicopathologically in 2 sloths, correlating with postmortem end-stage renal lesions. Renal disease causes metastatic and dystrophic mineralization by several mechanisms; importantly, reduced renal clearance results in serum hyperphosphatemia and renal secondary hyperparathyroidism and hypocalcemia or, ultimately, hypercalcemia. Calcium and phosphorus imbalance can culminate in extracellular deposition of amorphous calcium phosphate or hydroxyapatite crystals in key soft tissues, including the great arteries. 11 Renal mineralization was common (17 of 18 sloths for which kidney was submitted for histopathology) with Bowman capsules, tubular basement membranes, and perivascular and mural mineralization of interstitial blood vessels associated with mild to severe mononuclear nephritis. It is unclear if inflammatory renal lesions were reactive following soft tissue mineralization of the kidney or if interstitial nephritis was primary and ultimately led to renal insufficiency and secondary soft tissue mineralization. In sloths >5 years old, degenerative renal disease can be an important cause of morbidity and mortality 12,13,17 with secondary soft tissue mineralization likely. Interestingly, 5 of 18 sloths had moderate to severe visceral and/or vascular mineralization with histologically mild renal lesions. Additionally, of 8 sloths with soft tissue mineralization for which perimortem blood work was available, only 2 showed clear clinicopathologic renal insufficiency, suggesting that in at least some cases, the soft tissue mineralization may not be caused by primary renal disease.
Paraneoplastic hypercalcemia associated with transitional cell carcinoma of the urinary bladder may have contributed to soft tissue mineralization in 1 geriatric sloth that had mild renal inflammation with moderate mineralization of arteries and lungs. Transitional cell carcinoma is known to produce parathyroid hormone–related protein and has been shown to induce hypercalcemia in humans and other species. 15 Unfortunately, serum to confirm this suspicion was not available.
Gastric mucosal mineralization is well described in many mammalian species and is particularly associated with serum hypercalcemia and hyperphosphatemia in association with uremia. 17 Gastric glands secrete substantial amounts of hydrochloric acid, in contrast to an internal alkaline environment, allowing for extracellular deposition of amorphous calcium phosphate and hydroxyapatite crystals. 9 Mineralization causes local mucosal edema, reactive fibroplasia, and generally mild lymphoplasmacytic inflammation with mucosal erosion or ulceration, 17 as was seen in these sloths. Loss of gastric glandular function may have been clinically important in these sloths, exacerbating anorexia and emaciation, as was seen in 6 of 8 sloths for which clinical perimortem data were obtained. Gastric mineralization was detected radiographically and histologically in all 17 sloths for which glandular stomach was submitted for histopathology. Thirteen of these sloths also had histologically moderate to severe renal disease and/or uremia, which likely exacerbated gastric mineralization. In 3 sloths, gastric mineralization was marked and accompanied by only mild renal disease, and a dietary cause of soft tissue mineralization was suspected.
Of 14 sloths for which great vessels were submitted for histology, 11 had moderate to severe arterial mineralization with osteochondrogenic metaplasia. Arterial mineralization can be metastatic or dystrophic and is categorized pathologically into intimal and medial mineralization. 3 While intimal mineralization is generally due to dystrophic causes, such as atherosclerosis, medial mineralization is generally metastatic, involving initial vascular smooth muscle stress and dysfunction, followed by myocyte apoptosis of various causes, including serum hypervitaminosis D, serum mineral excess, uremia, metabolic and inflammatory disorders, and chronic hypertension. 2,16 Damaged vascular smooth muscle myocytes release microvesicles, which contain significant amounts of calcium, promoting extracellular mineralization. 15 Chronic vascular stress or significant vascular apoptosis can lead to extensive mineralization of the extracellular matrix. 3,5,8,11 Vascular smooth muscle has phenotypic plasticity that promotes osteochondrogenic metaplasia. 3,5,8,11 Osteochondrogenic metaplasia is generally irreversible; it is driven by vitamin D, calcium, and phosphorus; and it is amplified in cases of vitamin and mineral excess and uremia. 3,5,8 Vascular and visceral mineralization is controlled by molecules that act as inhibitors of calcification and specifically include matrix Gla protein, fetuin A, osteopontin, and osteoprotegerin. 3,8,10 Chronic hypercalcemia and hyperphosphatemia can promote loss of mineralization inhibitors, and chronic vascular damage can overwhelm inhibitory mechanisms. 3,8
Arterial mineralization is a dangerous lesion that causes systemic hypertension and promotes vascular smooth muscle damage, which further induces arterial mineralization. Arterial mineralization commonly results in left-sided cardiac hypertrophy, cardiac ischemia, and cardiovascular insufficiency and mortality in people. 3,8 Interestingly, 10 sloths with significant arterial mineralization also had cardiac lesions, which included gross cardiac hypertrophy, chronic myocardial fibrosis, and acute myocardial necrosis, which may be consistent with vascular compromise and multiphasic myocardial ischemia as well as chronic hypertension, although hypertension was not examined clinically in any of the sloths in this study. Importantly, the prevalence of degenerative cardiac lesions in adult sloths without arterial mineralization has not been previously reported; therefore, a correlation of arterial mineralization and cardiac disease in these study sloths is speculative and extrapolated from disease progression that has been well established in people with arterial mineralization.
Pulmonary interstitial and vascular mineralization was moderate to severe in 8 of 18 sloths for which lung was submitted for histologic evaluation. The lung is a common site of soft tissue mineralization in most mammals due to the interface of acid and alkaline microenvironments within the pulmonary parenchyma and blood vessels, enhancing extracellular deposition of amorphous calcium phosphate and hydroxyapatite crystal. 1,9 Chronic and advanced pulmonary mineralization reduces pulmonary elasticity and can predispose animals to secondary pulmonary infection and exercise intolerance. 1 Interestingly, 2 of 8 sloths with moderate to severe pulmonary mineralization also had inflammatory pulmonary disease consisting of bronchopneumonia and pulmonary abscessation, which may have been due in part to abnormal respiratory clearance function caused by advanced pulmonary mineralization.
In conclusion, soft tissue mineralization is a common finding in captive 2-toed sloths and can be associated with dietary imbalance of calcium, phosphorus, and vitamin D, as well as underlying renal disease and degenerative cardiac disease. Excess minerals and vitamins may be inadvertently consumed by sloths in multispecies exhibits—particularly exhibits cohousing exotic birds and New World primates, which often require diets containing higher levels of vitamin D and calcium. The pathologic findings in this and other reports 12 suggest that sloths are sensitive to dietary mineral and vitamin excess. Hypocalcemia, hypercalcemia, and hyperphosphatemia in captive sloths can be associated with renal insufficiency, and renal disease does appear to be common in sloths in this study as well as previous reports. 12,13,18 Routine evaluation of 2-toed sloths should include physical examination, serial radiographs, blood chemistry panel and complete blood cell count, urinalysis, and vitamin D and parathyroid hormone levels for the early detection of soft tissue mineralization, renal insufficiency, and calcium:phosphorus imbalance or vitamin D excess. Postmortem examination of sloths should include gross and histologic evaluation of all major viscera and especially representative specimens of major arteries, heart, lungs, parathyroid glands, kidneys, bone, and glandular mucosa of the stomach. Complete dietary and medical history along with comprehensive necropsy of deceased sloths can aid greatly in the understanding of soft tissue mineralization and important degenerative diseases of 2-toed sloths and may help to promote the longevity and overall health of this unique species in captivity.
Footnotes
Acknowledgements
We thank Roy Brown at Histology Consulting Service and Colorado Diagnostic Medicine Center for expert slide preparation. We are indebted to the following institutions for submission of sloth tissues and historical data: Denver Zoological Foundation, San Antonio Zoo, Mesker Park Zoo, Buffalo Zoo, Lion County Safari, Nashville Zoo, St Francis Hospital, Santa Barbara Zoo, Toledo Zoo, Gulf Coast Avian and Exotics, Potawatomi Zoo, Ellen Trout Zoo, El Paso Zoo, and the Mantella Collection.
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
