Abstract
Objective
Secondary hyperparathyroidism (SHPT) is a major complication in patients with chronic kidney disease (CKD). SHPT is related to chronic kidney disease-mineral bone disorder, leading to increased morbidity and mortality. Etelcalcetide is intravenously administered at the end of hemodialysis (HD). Etelcalcetide differs from the oral calcimimetic cinacalcet because it reduces gastrointestinal adverse events, thereby improving therapeutic effects. Etelcalcetide has only been approved by the U.S. Food and Drug Administration for several months. Therefore, there have only been a few reports regarding treatment of SHPT using etelcalcetide. This study aimed to evaluate the efficacy of etelcalcetide in patients on HD with SHPT.
Methods
Nine patients on HD (four men and five women, aged 58 ± 10 years) were enrolled in this study. All of the patients received etelcalcetide (5–10 mg, three times a week after HD). The observation period was 4.4 ± 1.0 months.
Results
All of the patients showed a significant reduction in serum parathyroid hormone levels during the observation period (−59% ± 20%). No significant adverse effects were observed.
Conclusions
Although this study had an uncontrolled small group and a short observation period, our results suggest that etelcalcetide could be a promising agent for SHPT treatment.
Keywords
Introduction
Chronic kidney disease (CKD) induces several complications.1–3 Secondary hyperparathyroidism (SHPT) is one of the most important complications of CKD, leading to vascular calcification and cardiovascular diseases.4,5 The main mechanism underlying SHPT involves an increase in parathyroid hormone (PTH) levels, which may exacerbate an imbalance in calcium and phosphorus metabolism.6,7 Furthermore, abnormalities in these parameters are associated with hospitalization. 8 The estimated healthcare costs are more than three times higher for patients with CKD and SHPT than for those without SHPT. Therefore, treatment of SHPT may reduce the economic burden of SHPT in patients with CKD.9–11 The updated Kidney Disease Improving Global Outcomes guidelines suggest that conventional treatment of SHPT in patients with CKD is limited to administration of the vitamin D analog, calcitriol, or dietary phosphate binders to reset the levels of serum calcium, phosphorus, and PTH. 12 However, administration of a vitamin D analog increases serum calcium and phosphorus levels. 13 Therefore, effective therapies for SHPT that can control PTH levels without inducing hypercalcemia are needed.
Etelcalcetide, a novel calcimimetic agent, is a synthetic peptide agonist of the calcium-sensing receptor and differs from cinacalcet because it shows mild gastrointestinal symptoms, nausea, and vomiting. Furthermore, etelcalcetide may be administered via a hemodialysis (HD) venous line, thereby decreasing the oral medication load. Additionally, etelcalcetide and its metabolite are not absorbed in the fiber of polysulfone dialyzers. 14
There have been few reports on the use of etelcalcetide for SHPT because etelcalcetide has only been recently approved by the U.S. Food and Drug Administration.15–17 In this study, we investigated the efficacy and safety of etelcalcetide in Japanese patients on HD.
Methods
Patients and study protocol
Nine HD patients with CKD were enrolled in the study. They underwent outpatient HD three times a week at the Department of Nephrology, Kindai University Nara Hospital, Kindai University Faculty of Medicine (Nara, Japan) or Seiwadai Clinic (Nara, Japan). All of these patients were Japanese and older than 20 years. Patients on precipitated calcium carbonate or phosphate binders and active vitamin D analogs, and those with serum intact PTH (iPTH) levels >180 pg/mL and serum calcium levels ≥8.4 mg/dL were eligible for this study. The major exclusion criteria were a history of parathyroidectomy or parathyroid intervention before the study, congestive heart failure, acute coronary syndrome, poorly-controlled hypertension, and diabetes mellitus. Eligible patients received a bolus injection of etelcalcetide (5–10 mg) three times a week for 2 to 5 months (Table 1). The safety or tolerability of etelcalcetide was evaluated using vital signs, laboratory findings, 12-lead electrocardiograms (ECGs), and adverse events, such as gastrointestinal side effects, nausea, and vomiting. The dose was increased by 5 mg if iPTH levels were not decreased intermediately during the observation period and no adverse events were reported. Administration of etelcalcetide was stopped when serum calcium levels were less than 8.4 mg/dL or symptomatic hypocalcemia was recognized. Blood samples were collected at the start of HD and at the beginning of the week. Serum levels of iPTH, calcium, phosphorus, and alkaline phosphatase were measured before and at the end of the observation period of the study.
Clinical characteristics.

M: male; F: female.
These patients were treated as per the protocol and a retrospective waiver of consent was obtained from the Clinical Study Ethics Review Board of Kindai University Nara Hospital (approval number: 17-9).
Ethical approval
All of the procedures performed in this study involving human participants were in accordance with the ethical standards of Kindai University Nara Hospital Research Committee, as well as the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Data analysis
Statistical significance for differences was determined by the interquartile range and Mann–Whitney U test. All analyses were performed using StatView (SAS Institute, Cary, NC, USA). Statistical significance was defined as P < 0.05.
Results
Four male and five female patients were enrolled in the study. The mean (SD) age of the patients at onset of SHPT was 58 ± 10 years, while the observation period was 4.4 ± 1.0 months. The mean etelcalcetide dose at the end of the study was 6.1 ± 2.2 mg. The initial doses of vitamin D and phosphate binder were continued during the observation period, except when calcium levels reached below 8.4 mg/dL. Serum iPTH levels were significantly decreased during therapy compared with baseline levels (626 ± 326 versus 258 ± 207 pg/mL, P = 0.01, Figure 1). However, no changes were observed in the levels of serum calcium, phosphorus, and alkaline phosphatase during the observation period (calcium, 9.2 ± 0.8 and 9.1 ± 1.1 mg/dL; phosphorus, 5.6 ± 1.2 and 5.0 ± 1.1 mg/dL; and alkaline phosphatase, 257 ± 122 and 244 ± 120 IU/L before and during therapy, respectively) (Figure 2a–c). Except for one patient, the dosage of calcium carbonate or vitamin D analogs was increased when etelcalcetide treatment was started (Table 1). No adverse events, such as nausea, vomiting, hypotension, headache, muscle spasms, anemia, or abnormal 12-lead ECGs, were reported during the observation period.

Box plot showing serum iPTH levels. Etelcalcetide significantly decreased serum iPTH levels at the end of the observation period compared with baseline levels. iPTH: intact parathyroid hormone.
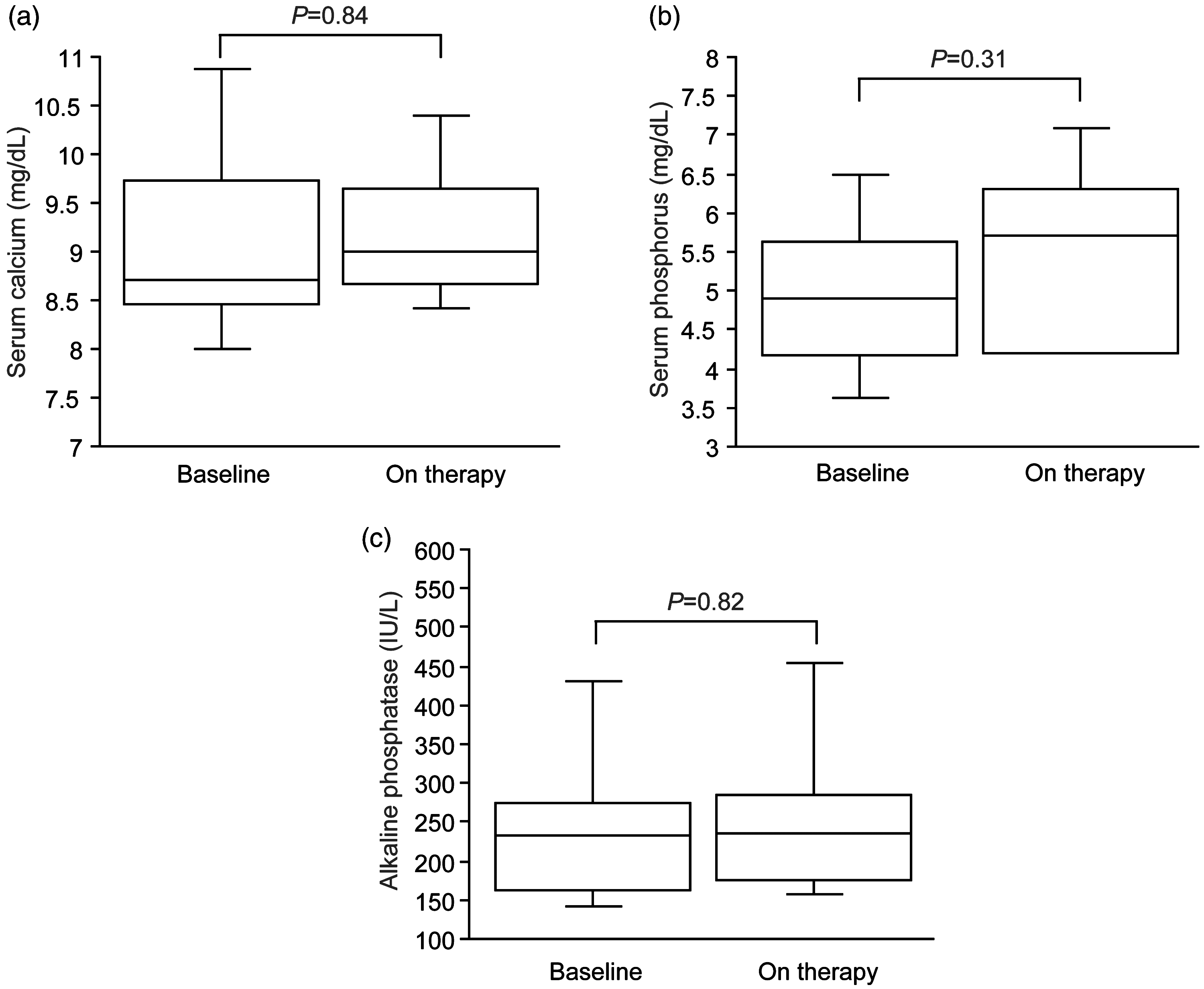
Box plots showing levels of serum calcium, phosphorus, and alkaline phosphatase. No changes in the levels of serum calcium, phosphorus, and alkaline phosphatase were observed during the observation period. (a) serum calcium, (b) serum phosphorus, and (c) serum alkaline phosphatase levels.
Discussion
In this study, we evaluated the efficacy and safety of etelcalcetide in Japanese patients on HD with SHPT. The iPTH-lowering effect in our study was strong, but different from that previously reported.15,18,19 No significant differences in other bone metabolic markers were observed. There were no adverse events during the observation period.
Previous reports showed reduced serum calcium levels in patients on etelcalcetide compared with those on cinacalcet. 15 However, no significant decrease in serum calcium levels was observed in our study. This observation may be attributed to several reasons. First, the doses of carbonate or active vitamin D analogs were freely increased in our protocol when serum calcium levels were decreased below 8.4 mg/dL. Second, calcium concentrations of the dialysate in our institute are relatively high (2.75 mEq/L).
The level of iPTH was decreased in our study by almost 60%, which is similar to that previously reported.15,18,19 Serum levels of intact fibroblast growth factor 23 (FGF23), bone alkaline phosphatase, and tartrate-resistant acid phosphatase-5b are suppressed after treatment with etelcalcetide.15,18,19 Therefore, etelcalcetide may modulate the balance between osteoblasts and osteoclasts, thereby improving bone density and quality. Our study clearly showed that administration of etelcalcetide improved levels of the bone metabolic marker iPTH. Therefore, further investigations are required to study the effect of etelcalcetide on FGF23, bone alkaline phosphatase, and tartrate-resistant acid phosphatase-5b.
Our previous studies showed that CKD is characterized by an increase in oxidative stress and inflammation and protein kinase C β-dependent glomerular endothelial dysfunction. 20 Interestingly, cinacalcet treatment decreases oxidative stress and improves endothelial function. 21 Further prospective studies are needed, but treatment with calcimimetic peptides, including etelcalcetide, may exert possible beneficial effects on cardiovascular diseases in patients on HD.22–25
The conventional therapy of SHPT mainly includes vitamin D analogs, phosphate binders, or calcimimetics. Vitamin D analogs may not only decrease PTH in SHPT, but also affect absorption of calcium and phosphate from the intestine, leading to hypercalcemia, hyperphosphatemia, and elevated serum FGF23 levels. These conditions induce vascular and ectopic calcification. The oral calcimimetic drug cinacalcet is widely used in treating SHPT. A previous meta-analysis showed that cinacalcet could achieve the efficacy endpoints in control of SHPT. 26 Increases in FGF23 levels may be associated with adverse effects, including coronary artery calcification, left ventricle hypertrophy, and mortality.27–32 The Evaluation of Cinacalcet Hydrochloride Therapy to Lower Cardiovascular Events (EVOLVE) study clearly showed that administration of cinacalcet decreased FGF23 levels, leading to a significant reduction in heart failure, sudden death, and cardiovascular mortality. 33 Etelcalcetide, a novel, second generation calcimimetic, appears to be superior to cinacalcet. A greater than 50% reduction from baseline in mean serum PTH levels during the efficacy assessment phase was shown to be 52% in etelcalcetide and 40% in cinacalcet. 34 Moreover, a previous study showed a 30% decrease in FGF23 levels in the etelcalcetide treatment group, and this was a more distinct reduction than cinacalcet. 33 Gastrointestinal symptoms, nausea, and vomiting are major adverse events of oral SHPT treatment, resulting in poor adherence to treatment. A recent report showed that gastrointestinal side effects in the cinacalcet group were higher than those in the etelcalcetide treated group (46.7% vs. 37.8%, respectively). However, etelcalcetide leads to more frequent adverse effects of asymptomatic and symptomatic hypocalcemia that can be more distinct at the beginning of administration. 34 Therefore, administration of intravenous etelcalcetide may improve treatment efficacy and reduce the medication burden.
The unit costs of etelcalcetide from several European countries can be 15% or 30% higher than cinacalcet. 35 However, according to an analysis performed in the United Kingdom, the incremental cost effectiveness ratio for etelcalcetide is GBP 14,778 per-quality adjusted life year, which is lower than the National Institute for Health and Care Excellence threshold (GBP 20,000–30,000). 36
In summary, our study suggests that etelcalcetide is useful for improving the prognosis of patients with SHPT. However, a large number of case–control studies will be needed in the future to be able to evaluate this treatment.
Footnotes
Acknowledgements
Parts of this manuscript were presented at the 11th international congress of ISHD. We wish to thank Kazumasa Tanaka, Ryoichi Hirakawa, Yasuhiro Akai, and Yasuhiro Horii for consultation on the work-up of the patients and this study.
Declaration of conflicting interest
The authors declare that there is no conflict of interest.
Funding
A.M. has received research grants from Otsuka Pharmaceutical, Teijin Pharma, Torii, Taisho Toyama Pharmaceutical, and Sanofi. A.M. has also received speaker honorarium from Ono Pharmaceutical, Kyowa Hakko Kirin, Takeda Pharmaceutical, Otsuka Pharmaceutical, MSD, Novartis Pharma, Kissei Pharmaceutical, Boehringer Ingelheim, Chugai Pharmceutical, Kowa, and Mylan. This work was supported by JSPS KAKENHI Grant Number 17K09720.
