Abstract
Background
Ginkgolide K (GK) has a protective effect on neurons and myelin sheath, and the function of astrocytes in phagocytizing myelin debris has attracted extensive attention in remyelination.
Objectives
This study focuses on the impact of GK on the phagocytosis of myelin debris by astrocytes and explores the possibility of treating demyelination.
Materials and Methods
Male C57BL/6 mice were used to establish a cuprizone (CPZ)-induced demyelination model. After being fed a normal or CPZ diet for 4 weeks, mice were intraperitoneally injected with PEG400 or GK for 14 consecutive days. GL261 and primary astrocytes were exposed to myelin debris. Immunohistochemistry/immunocytochemistry staining, western blot, RT-PCR, and other methods were used to detect the relevant indicators.
Results
Astrocytes engulfed myelin debris, leading to astrocyte reactivity with increased p-NF-kB/p65 and ATF6 expression and decreased nuclear factor erythroid 2-related factor 2 (Nrf2) expression, which was reversed by GK. Subsequently, astrocytes stimulated by myelin debris underwent self-apoptosis with increased expression of caspase-3 and BCL2-associated X (Bax), which was inhibited by GK. Simultaneously, GK efficiently promoted astrocytes to increase the production of neurotrophic ciliary neurotrophic factor (CNTF) and basic fibroblast growth factor (bFGF), speculating that increased CNTF/bFGF, decreased p-NF-kB/p65/ATF6, and up-regulated Nrf2 should participate in the protection of astrocyte apoptosis and build a beneficial microenvironment for myelin regeneration.
Conclusion
The results suggest that GK may have the potential to treat demyelination by promoting debris clearance and improving the brain microenvironment. However, further studies are needed to understand the physiological and pathological consequences of astrocytic phagocytosis and to investigate the possibility of using GK as a therapeutic application.
Introduction
Multiple sclerosis (MS) is a chronic inflammatory disease of the central nervous system (CNS) that is associated with prominent demyelination (Attfield et al., 2022). The current immunomodulatory treatment has been proven to be effective in treating the relapse or remission of MS but has failed to prevent disease progression (Lassmann et al., 2012; Piehl, 2021). Remyelination is a natural regeneration against demyelination, but the reasons for the failure or incompleteness of remyelination during MS have not been fully understood (Motavaf et al., 2017). Obviously, remyelinating therapy has become a new direction and challenge in treating MS (Plemel et al., 2017). However, the presence of myelin debris around the lesion site further exacerbated the remyelination. Therefore, removing myelin debris is crucial for effective remyelination after injury (Sen et al., 2022).
Traditionally, microglia are considered the major phagocytes. Recent studies have reported that astrocytes also have a phagocytic function and may play beneficial and detrimental roles during lesion development (Lee et al., 2021; Ponath et al., 2017). Astrocytes maintain the homeostasis of the brain through phagocytizing synapses, neuronal debris, axons, mitochondria, and pathological protein aggregates (Lee & Chung, 2021). Therefore, the phagocytosis of astrocytes may contribute to removing the cell debris or other wastes produced by brain tissue injury, further ameliorating neuroinflammation and neurological impairments after traumatic brain injury (Zhou et al., 2021), and improving the brain microenvironment after transient brain ischemia (Morizawa et al., 2017). Although accumulating evidence suggests that astrocytes participate in debris clearance in the brain, the reactivity of astrocytes to myelin debris during phagocytosis is still poorly understood.
Ginkgolide K (1,10-dihydroxy-3,14-didehydroginkgolide, GK) is a diterpene lactone extracted from the leaves of Ginkgo biloba. GK inhibited platelet activating factor (PAF)-induced platelet aggregation by antagonizing the PAF receptor, reversed the apoptosis induced by oxygen and glucose deprivation/reoxygenation (OGD/R) injury, and improved the neurobehavioral score and cerebral infarction volume after cerebral ischemia-reperfusion injury in a dose-dependent manner (Tao et al., 2017). Previous studies also revealed that GK exerted anti-inflammation, anti-oxidative stress, and neuroprotective effects in Parkinson’s disease (Miao et al., 2022) and experimental autoimmune encephalomyelitis (EAE). Based on the reactivity of astrocytes and the plasticity of GK targeting astrocytes, we tried to explore whether GK promotes astrocytes to phagocytize myelin debris, thus affecting the brain microenvironment and myelin regeneration (Yu et al., 2019).
Materials and Methods
Animals and Drugs
Male C57BL/6 mice (10–12 weeks old) were purchased from Vital River Experimental Animal Technology Co. Ltd. (Beijing, China). The study was performed under the animal use protocol number: AWE202209006 and approved by the Ethics Committee of the Shanxi University of Chinese Medicine, Jinzhong, China. The experimental procedure was carried out in accordance with the guidelines of the International Council for Laboratory Animal Science. Mice were housed on a 12-h light/dark cycle with access to food and water ad libitum under clean conventional housing conditions. GK was purchased from Nanjing Dawdorf Biological Co.
Cuprizone-induced Demyelination Model
For the cuprizone (CPZ)-induced demyelination model, mice were fed with 0.2% (w/w) CPZ (Sigma-Aldrich, USA) in a chow diet ad libitum for 6 weeks. As an experimental design, mice were randomly divided into three groups (n = 8/per group) as follows: (1) control group = normal diet + solvent; (2) CPZ group = CPZ diet + solvent; and (3) CPZ + GK group = CPZ diet + GK. After being fed a regular or CPZ diet for 4 weeks, mice were intraperitoneally injected with PEG400 or GK (20 mg/kg, 200 µL/mouse) for 14 consecutive days. GK was dissolved with PEG400 ultrasound at a 10 mg/mL concentration for 5 min and then diluted with saline.
GL261 Cells
Mouse glioblastoma cells were cultured in complete medium (Dulbecco’s modified eagle medium (DMEM), 4500 mg/L glucose, 4 mM glutamine, and 1 mM sodium pyruvate) containing 1% streptomycin (10,000 µg/mL) and penicillin (10,000 units/mL) in a humidified incubator with 37°C and 5% CO2.
Primary Astrocytes
Primary astrocytes were obtained from postnatal (1 day of age) C57BL6 mice. Under sterile conditions, the cerebral cortex was dissected and placed in a complete medium (Thermo Science) containing 1% streptomycin-penicillin. Cells were dissociated into a single-cell suspension and then seeded in a 75-cm flask culture plate (Corning, USA) in a humidified incubator at 37°C and 5% CO2. When mixed glial cultures reached 90% confluence, microglia were repeatedly removed by constant shaking for 12 h at 180 rpm. The purity was close to 100% by glial fibrillary acidic protein (GFAP) immunocytochemistry staining.
Myelin Debris Coupled with carboxyfluorescein succinimide ester
Myelin debris was resuspended with sterile phosphate buffered saline (PBS) and labeled in a 50 M carboxyfluorescein succinimide ester (CFSE) solution for 30 min at room temperature (RT). After centrifugation at 14,000g for 10 min, the pellet was washed three times with PBS. CFSE-labeled myelin debris was resuspended with sterile PBS. Protein concentration was determined by wet weight (mg/mL, w/v).
Myelin Debris Coupled with pHrodo
Phrodo coupling of myelin debris was carried out according to the operation instructions (pHrodoTM Red, Invitrogen P36600). Briefly, myelin debris was mixed with sterile Phrodo and gently stirred at 30–40 rpm for 1–2 h RT. After washing with PBS, the tube was centrifuged at 20,000g for 1–2 min. After repeated washing, the pellet was resuspended with PBS and 5% dimethyl sulfoxide (DMSO). Protein concentration was determined by wet weight (mg/mL, w/v).
In vitro Phagocytosis of Myelin Debris by Astrocytes
GL261 and primary astrocytes (1×105/well) were plated in 12-well plates and respectively incubated with CFSE-conjugated myelin debris (5 mg/mL) or pHrodo-coupled myelin debris in the presence and absence of GK (50 µg/mL) for 24 h at 37°C with 5% CO2. Unphagocytosed myelin debris was washed out, and phagocytosis capacity was measured under fluorescence microscopy and by multifunctional Synergy H1 Hybrid Reader (BioTek, USA) using fluorescence 485 excited light.
Immunohistochemistry and Immunocytochemistry Staining
Briefly, the slides of the brain (10 µm) and cultured astrocytes were blocked with 1% BSA at RT for 30 min. The immunostaining was performed with the following antibodies: anti-ABCA1 (Abcam, USA), anti-GFAP (Abcam), anti-Iba1 (Abcam), anti-DMBP (Abcam), anti-bFGF (Abcam), anti-CNTF (Abcam), anti-Nrf2 (Abcam), anti-NF-κB (Abcam), anti-Caspses3 (Cell Signaling Technology, USA), anti-Bax (Abcam), and anti-ATF-6 (Millipore, USA), followed by corresponding secondary antibodies at RT for 2 h. Results were visualized under fluorescent microscopy (BX60, Olympus Imaging America Inc., USA) in a blinded fashion. Quantification was performed by Image-Pro Plus 6.0 software.
Western Blot
The proteins isolated from the brain and astrocytes were separated by sodium dodecyl sulfate-polyacrylamide gel electrophoresis and transferred electrophoretically to a nitrocellulose membrane (Millipore). After blocking with 5% milk for 1 h at RT, the membranes were incubated with anti-ABCA-1, anti-CNTF, anti-bFGF, anti-NF-kB, anti-Nrf2, and anti-β-tubulin (Cell Signaling Technology) at 4°C for overnight, followed by horseradish peroxidase (HRP)-conjugated secondary antibodies (Abcam) at RT for 1 h. Immunoblots were developed with an enhanced chemiluminescence system (GE Healthcare Life Sciences) and measured using Quantity Software (Bio-Rad, Hercules, CA, USA). β-tubulin was used as an internal reference.
Quantitative Real-Time PCR
The total RNA of mice from the corpus callosum was isolated using a total RNA kit (Mei5bio, China). The reverse transcription of total RNA was achieved using a Prime Script TMRT reagent kit (Takara, Japan) to obtain cDNA. Quantitative PCR with SYBR Green Dye Gene Expression Assays was applied to determine gene expression using the ABI7500 system (Applied Biosystems, Carlsbad, CA, USA). The required primers were synthesized by Beijing Qingke Biological Engineering Technology Company (Beijing, China). The required primer sequences were as follows: TLR4 (forward: 5ʹ-AGCTTCTCCAATTTTTCAGAACTTC; reverse: 5ʹ-TGAGAGGTGGTGTAAGCCATGC), iNOS (forward: 5ʹ-GAGACAGG GAAGTCTGAAGCAC; reverse: 5ʹ-CCAGCAGTAG TTGCTCCTCTTC), and β-Actin (forward: 5ʹ-CCTCACTGTCCACCTTCC; reverse: 5′-GGGTGTAAAACGCAGCTC). The cycle threshold was determined for each sample as the initial increase in fluorescence above the background. β-actin was used as an internal control for normalization.
ELISA Assay
The concentrations of IL-1β, IL-6, and TNF-α were measured by sandwich ELISA kits (R&D System, USA) following the manufacturer’s instructions. Determinations were performed in duplicate, and the results were expressed as pg/mL.
Statistical Analysis
Data were presented as the mean ± standard error of the mean. The raw data were analyzed using a one-way analysis of variance with Tukey’s post hoc test using the GraphPad Prism 5.0 package (GraphPad Software, Inc., La Jolla, CA, USA). A p-value < 0.05 was considered to indicate a statistically significant difference.
Results
GK Promotes Astrocytes to Phagocytize Myelin Debris
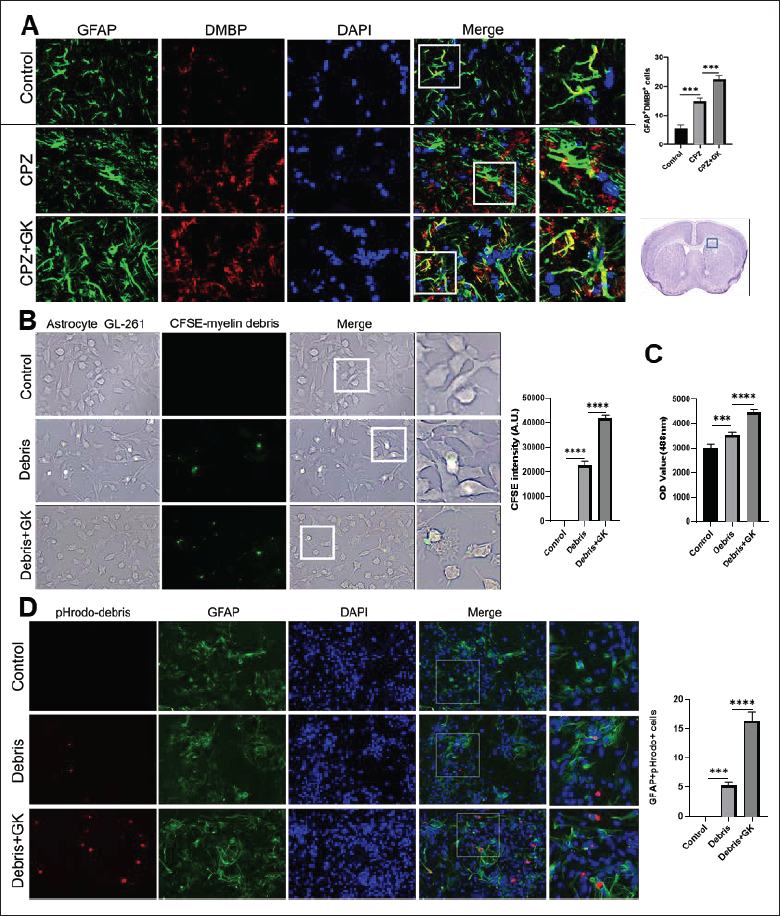
In recent years, the clinical significance of astrocytes phagocytizing myelin debris has attracted extensive attention. In the CPZ model, degraded myelin basic protein (DMBP) was co-localized with GFAP+ astrocytes, which were elevated by GK as compared to CPZ model mice (Figure 1A, p < 0.0001). Then, to further verify whether GK can promote the phagocytosis of myelin debris by astrocytes, we investigated the phagocytosis capacity of astrocytes exposed to myelin debris in vitro. The results showed that GK promoted GL261 astrocytes to phagocytize myelin debris coupled with CFSE, as compared with myelin debris alone (Figure 1B, p < 0.001). Using a fluorescence microplate reader, the fluorescence intensity in the myelin debris + GK group was significantly higher than that in the myelin debris group (Figure 1C, p < 0.0001). Furthermore, primary astrocytes were exposed to myelin debris labeled with pHrodo that could be red in a lysosomal acid environment. GFAP+ astrocytes co-localizating pHrodo were significantly elevated after GK addition (Figure 1D, p < 0.0001). Taken together, astrocytes can phagocytize myelin debris, while GK can further promote the phagocytic ability of myelin debris by astrocytes.
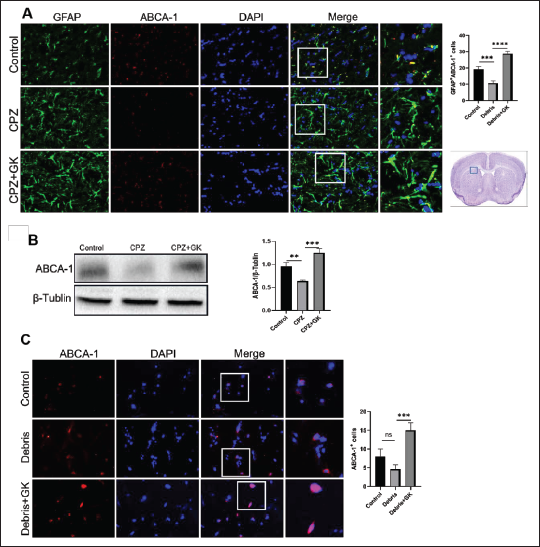
Previous investigations strongly support the hypothesis that ABCA1 is essential for astrocytic phagocytic capacity. Therefore, we detected the expression of ABCA1 on astrocytes after GK intervention in the CPZ mouse model. As shown in Figure 2A, GK significantly increased the numbers of GFAP+ astrocytes expressing ABCA1 by immunohistochemistry staining, as compared with CPZ model mice (p < 0.0001). The expression of ABCA1 protein in brain homogenate of mice treated with GK was also significantly higher than that of CPZ model mice by western blot (Figure 2B, p < 0.0001). To further test whether GK can induce ABCA1 expression on astrocytes exposed to myelin debris, we established an astrocyte model stimulated by myelin debris in vitro. The results showed that the expression of ABCA1 on primary astrocytes after GK treatment was higher than without GK (Figure 2C, p < 0.001). These results indicate that GK can promote astrocytes to upregulate ABCA1 expression, which should be related to the increased phagocytosis of myelin debris by astrocytes.
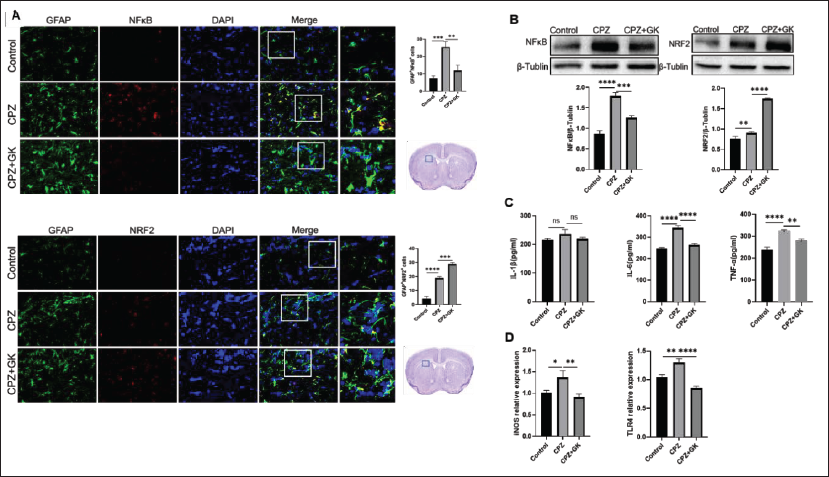
GK Regulates the Rebalance of Inflammatory and Oxidative Responses by Astrocytes Exposed to Myelin Debris
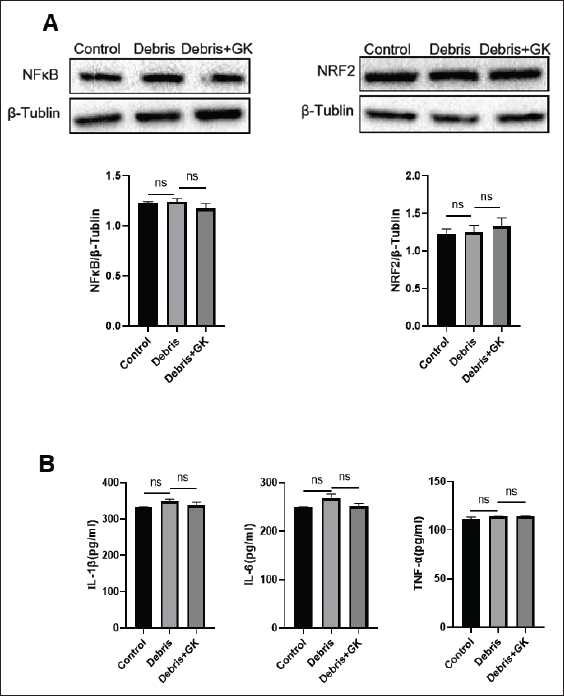
In the CPZ model, the expression of p-NF-kB increased while the expression of nuclear factor erythroid 2-related factor 2 (Nrf2) decreased in astrocytes as compared with control mice (Figure 3A; p < 0.001 and p < 0.01, respectively). Similar results also appeared in brain homogenate by western blot (Figure 3B; p < 0.01 and p < 0.0001, respectively). IL-6 and TNF-α protein as well as iNOS and TLR4 mRNA in brain homogenate of CPZ model mice were also higher than those of control mice (Figure 3C and D; p < 0.05, p < 0.01, and p < 0.0001, respectively). On the contrary, GK decreased astrocytes expressing p-NF-kB and increased astrocytes expressing Nrf2 (Figure 3A; p < 0.01 and p < 0.001, respectively), which was consistent with that from brain homogenate by Western blot (Figure 3B; p < 0.001 and p < 0.0001, respectively). Similarly, GK inhibited the production of IL-6 and TNF-α (Figure 3C; p < 0.0001 and p < 0.01, respectively) and the expression of iNOS and TLR4 mRNA (Figure 3D; p < 0.01 and p < 0.0001, respectively). However, inconsistent with expectation, when primary astrocytes were exposed to myelin debris in vitro, myelin debris did not affect the expression of p-NF-kB and Nrf2 and the production of IL-1β, IL-6, and TNF-α (Figure 4A and B). These results suggest that the phagocytosis of myelin debris is not enough to affect astrocytes’ inflammatory and oxidative responses in vitro, and the inflammatory and oxidative responses in the brain of CPZ model mice may be more related to the microglial responses or astrocyte-microglia interaction.
GK Prevents Apoptosis by Astrocytes Exposed to Myelin Debris
We further observed the outcome of astrocytes phagocytizing myelin debris in vitro. As shown in Figure 5A, after primary astrocytes were exposed to myelin debris, cell death increased by lactate dehydrogenase (LDH) assay (p < 0.0001), while GK effectively inhibited cell death (p < 0.001). Further observation showed that GL261 astrocytes exposed to myelin debris could cause nuclear pyknosis under a microscope bright field. At the same time, GK significantly prevented nuclear pyknosis (Figure 5B). Similarly, primary astrocytes exposed to myelin debris showed uneven nuclear staining and small fragments in nuclei. GK can prevent this phenomenon under a fluorescence microscope (Figure 5C). These results reveal that astrocytes exposed to myelin debris should lead to self-apoptosis, which can be inhibited by GK.
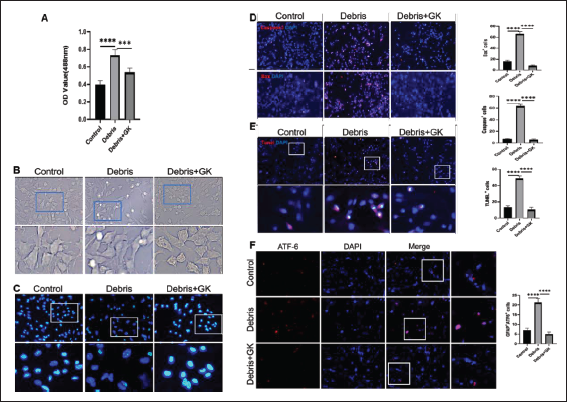
Next, we detected the apoptosis-related molecules and cell apoptosis. As expected, the exposure to myelin debris induced astrocytes to upregulate caspase-3 and Bax and increase TUNEL+ apoptotic cells (Figure 5D and E; p < 0.0001). GK inhibited the expression of caspase-3 and Bax and the apoptosis of astrocytes (Figure 5D and E; p < 0.0001). Subsequently, based on a literature study, we detected apoptosis-related molecules in ER stress. The results showed that the expression of ATF6 increased in astrocytes exposed to myelin debris, which was also inhibited by GK (Figure 5F, p < 0.0001), suggesting that the signal pathway in the endoplasmic reticulum may be involved in the apoptosis of astrocytes exposed to myelin debris.
GK Promotes Neurotrophic Factors by Astrocytes Exposed to Myelin Debris
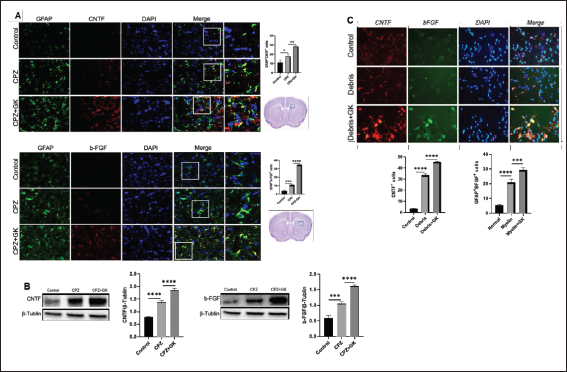
Finally, we tried to understand how GK protects astrocytes from apoptosis induced by exposure to myelin debris. As anticipated, GK significantly increased GFAP+ astrocytes expressing CNTF and bFGF in the corpus callosum, as compared with CPZ model mice (Figure 6A; p < 0.0001 and p < 0.01, respectively). The expression of CNTF and bFGF protein in brain homogenate of mice treated with GK was also significantly higher than that of CPZ model mice (Figure 6B, both p < 0.0001), suggesting that the myelin debris produced by demyelination in CPZ mice could stimulate astrocyte to generate CNTF and bFGF. GK also further promoted the production of CNTF and bFGF in vitro cultured astrocytes exposed to myelin debris (Figure 6C; p < 0.0001 and p < 0.001, respectively).
Discussion
We previously found that GK treatment effectively alleviated behavioral abnormalities and improved demyelination in CPZ mice (Li et al., 2019). In this study, we further focused on the role of GK in targeting astrocytes to phagocytize myelin debris. The results showed that GK promoted astrocytes to phagocytize myelin debris, inhibited inflammatory signals, and induced antioxidant molecules. Astrocytes that have phagocytized myelin debris exhibited nuclear pyknosis, up-regulated the expression of caspase-3 and Bax, and increased TUNEL+ cell apoptosis, suggesting that astrocytes phagocytized myelin debris may lead to self-apoptosis. Meanwhile, GK promoted the formation of CNTF and bFGF, which should be related to anti-apoptosis and remyelination.
Efficient clearance of myelin debris by phagocytosis is critical to eliminate inhibitory signals interfering with oligodendrocyte progenitor cells (OPCs) activation, recruitment to the site of demyelination, and/or differentiation into myelinating mature oligodendrocytes (Franklin & Ffrench-Constant, 2017; Lampron et al., 2015). Traditionally, microglia are considered the primary phagocytes. Recent studies have reported that astrocytes also have phagocytic functions (Konishi et al., 2020; Lee & Chung, 2021; Lee et al., 2021; Ponath et al., 2017; Sanchez-Mico et al., 2021; Tao et al., 2017), providing a new idea for the therapy of demyelination. ABCA1 participates in astrocytic phagocytosis to remove damaged debris (Chen et al., 2021). Consistent with this observation, our results also revealed that GK promoted astrocytes to phagocytize myelin debris, possibly through the up-regulation of ABCA1 expression. How GK induces ABCA1 to promote astrocytic phagocytosis of myelin debris remains unclear. Limited information demonstrated that SIRT1 activator E1231 protects from experimental atherosclerosis and lowers plasma cholesterol and triglycerides by enhancing ABCA1 expression (Feng et al., 2018). Loss of SIRT1 in vivo reduces expression of a variety of liver X receptor targets involved in lipid metabolism, including ABCA1 (Li et al., 2007). Our latest results showed that GK induced the expression of SIRT1 in H2O2-induced PC12 neurons and reduced cellular senescence (unpublished data). These results suggest that GK induced AST upregulates the expression of ABCA1 and promotes the phagocytosis of myelin debris, which might be related to SIRT1. The detailed cellular and molecular mechanisms remain to be further clarified.
Besides, ABCA1 is known to have anti-inflammatory and anti-oxidative effects in macrophages (Tang et al., 2009). Interestingly, overexpression of ABCA1 decreased TNF-α and IL-6 production in endothelial cells (Stamatikos et al., 2019), suggesting that ABCA1 could form a negative feedback regulatory axis with inflammatory responses. Therefore, the mechanism by which ABCA1 participates in phagocytosis and anti-inflammation still needs to be further explored. The last puzzling question is why astrocytes that phagocytize myelin debris will undergo self-apoptosis while GK further promotes phagocytosis but reduces apoptosis. Consistent with our results, telocytes enhanced M1 macrophage differentiation and phagocytosis while inhibiting apoptosis (Huang et al., 2021). When a particle is recognized by phagocytic receptors, various signaling pathways are activated to initiate phagocytosis. Generally speaking, cell responses associated with phagocytosis can be controlled by parallel signaling pathways. There are several studies to clarify the signal cascade of astrocyte apoptosis, including cytosolic Ca2+ elevation, oxidative stress, NF-κB activation, mitochondrial dysfunction, ER stress, and protease activation. Therefore, we speculate that myelin debris stimulates astrocytes to enter a reactive state, including activation, inflammatory response, and oxidative stress, which may promote cell apoptosis, while GK may reduce apoptosis by inhibiting inflammatory and oxidative stress.
Microglia are classically considered phagocytes. However, astrocytes have been highlighted as phagocytes in the last few years. Microglia and astrocytes participate in developmental myelination by engulfing excessive, unneeded myelin. They are also involved in the clearance of degenerated myelin debris for accelerating remyelination or engulfing healthy myelin sheaths for inhibiting remyelination (Xu et al., 2023). Astrocytes are capable of phagocytosis and can provide a compensatory mechanism if microglia are unable to execute phagocytosis (Konishi et al., 2020). It was reported that astrocytes phagocytosed adult hippocampal synapses and eliminated unnecessary excitatory synaptic connections for circuit homeostasis, thereby supporting cognitive function (Lee et al., 2021). However, LCN2-expressing astrocytes acquire a phagocytic phenotype and are able to uptake myelin, leading to progressive demyelination in Lcn2-/- mice (Wan et al., 2022). These studies raised the following questions: (1) what is the essential difference between astrocyte and microglia phagocytosis; (2) how microglia and astrocytes cooperate to complete phagocytic function; and (3) whether different signal molecules and pathways closely regulate astrocytic phagocytosis, demyelination, and remyelination. Based on these limited results, we speculate that GK protection against myelin debris-mediated apoptosis in astrocytes should be related to the regulation between ATF6, NF-kB, and Nrf2 and the increase of CNTF and bFGF production. Of course, how GK affects these molecules and thus affects cell apoptosis still needs careful stripping and verification.
Conclusion
GK promoted astrocytes to phagocytize myelin debris, accompanied by the upregulation of ABCA1, inhibition of NF-kB, and increase of Nrf2, CNTF, and bFGF, ultimately reducing the apoptosis of astrocytes stimulated with myelin debris. Future research is anticipated to generate novel strategies for treating CNS demyelination.
Footnotes
Summary
When myelin debris was exposed to astrocytes, GK intervention promoted astrocytes to engulf myelin debris, possibly through the upregulation of ABCA1 expression, accompanied by the inhibition of inflammatory molecules and the formation of neurotrophic factors, leading to the improvement of the microenvironment for myelin sheath protection or regeneration.
Abbreviations
MS: Multiple sclerosis; CNS: central nervous system; GK: Ginkgolide K; EAE: experimental autoimmune encephalomyelitis; CPZ: cuprizone; CFSE: carboxyfluorescein succinimide ester; RT: room temperature; DMEM: Dulbecco’s modified eagle medium; PBS: phosphate buffered saline; DMSO: dimethyl sulfoxide; BCA: bicinchoninic acid; ABCA1: ATP binding cassette transporter A1; GFAP: glial fibrillary acidic protein; Iba1: ionized calcium binding adapter molecule 1; DMBP: degraded myelin basic protein; CNTF: ciliary neurotrophic factor; bFGF: basic fibroblast growth factor; Nrf2: nuclear factor erythroid 2-related factor 2; NF-kB: noncanonical nuclear factor-kappa B; Bax: BCL2-associated X; ATF6: activating transcription factor 6; HRP: horseradish peroxidase; PCR: polymerase chain reaction; ELISA: enzyme-linked immunosorbent assay; LDH: lactate dehydrogenase; IL-1β: interleukin 1β; IL-6: interleukin 6; TNF-α: tumor necrosis factor-α; ROS: reactive oxidative species.
Authors’ Contributions
BX and CM designed the experiments and checked all experimental data. XL, KL, and ZD involved in the establishment of CPZ model and intervention of GK. QW performed Western blot. XL, KL, and LS performed astrocyte culture and treatment, ELISA, RT-PCR, immunocytochemistry, and immunohistochemistry. JY participated in the data analysis. DM, BX, and CM proofread and finalized the article.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
All experimental protocols were approved by the Ethics Committee of Shanxi University of Chinese Medicine (AWE202209006, Approval date: October 7, 2022).
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This work was supported by the grants from the National Natural Science Foundation of China (81473577 to C-GM, 81903596 to QW), the Young Scientists Cultivation Project of Shanxi University of Chinese Medicine (2021PY-QN-09 to L-JS), the Shanxi Applied Basic Research Project (201901D211538 to L-JS), the Leading Team of Medical Science and Technology, Shanxi Province (2020TD05 to C-GM), and the basic research project of the cultivation plan of the scientific and technological innovation ability of Shanxi University of Chinese Medicine (2020PY-JC-02 to L-JS).
