Abstract
Background
Cyclophosphamide is one of the most extensively used chemotherapeutic drugs and is employed to treat several malignancies. Cyclophosphamide often comes with several unpleasant side effects. The primary negative consequence of cyclophosphamide in clinical chemotherapy is immunosuppression, which can lead to life-threatening complications.
Objectives
The present work was dedicated to exploring the therapeutic effects of Andrographis paniculata against cyclophosphamide-induced immunosuppression in mice.
Materials and Methods
The BALB/c mice were treated with 80 mg/kg of cyclophosphamide to achieve immunosuppression and treated with 250 and 500 mg/kg of A. paniculata extract. 10 mg/kg of levamisole was used as a standard drug. After the completion of the treatments, the body weights of the experimental animals were measured. The immune organs (spleen and thymus) were determined by standard methods. The levels of blood components were studied using an automated hematological analyzer. The macrophage phagocytic index, humoral response, and delayed immune response were studied by standard methods. The histopathological analysis was done on the heart, spleen, and thymus.
Results
The treatment with 250 and 500 mg/kg of A. paniculata extract substantially increased the body weight and elevated the immune organ index in the immunosuppressed mice. The changes in the levels of hematological parameters were successfully modulated by A. paniculata. The levels of macrophage phagocytic index and delayed and humoral immune responses were boosted by the A. paniculata extract treatment in the immunosuppressed mice. The outcomes of histopathological analysis exhibited that the A. paniculata extract attenuated the cyclophosphamide-induced damage to the heart, thymus, and spleen tissues.
Conclusion
The findings of the current work validate the immunomodulatory properties of A. paniculata extract in cyclophosphamide-induced immunosuppressed mice. Hence, it was clear that A. paniculata can be a talented immunomodulatory agent to treat immunological complications.
Introduction
Immunocompromised patients have increased dramatically in recent years for several reasons. Cancer is often regarded as one of the deadliest threats to human health and progress. Cancer treatment relies heavily on chemotherapy. One of the most common causes of death in cancer patients is secondary immunodeficiency brought on by chemotherapy treatments. Chemotherapy is effective against cancer, but it has serious side effects since it also harms healthy tissues. 1 Chemotherapy mainly focuses on killing the rapidly dividing tumor cells, thereby preventing cancer development. 2 Cyclophosphamide is one of the most extensively utilized chemotherapeutic agents and is employed to treat several malignancies. Apart from its therapeutic effects, cyclophosphamide frequently comes with unpleasant side effects. 3 The primary negative consequence of cyclophosphamide in clinical chemotherapy is immunosuppression. When chemotherapy compromises a patient’s immune system, it can lead to life-threatening complications such as secondary infections and immunodeficiency. 4
Cyclophosphamide is an alkylating chemical commonly utilized to treat several cancers, including ovarian, lung, and breast malignancies. Since immunosuppression is a general negative effect of cyclophosphamide, the drug is frequently employed to treat autoimmune diseases. 5 Furthermore, it has been utilized as an immunosuppressive drug for organ transplantation. 6 Side effects from cyclophosphamide are as varied as those from other chemotherapeutic medications and include a decrease in blood components. Extreme thrombocytopenia has been reported as a side effect. 7
Immune activities are vital to health maintenance since they are the host’s primary line of protection against infections. Aging, chronic illness, physical and emotional stress, and poor lifestyles all contribute to a decline in immunological function, which is a serious clinical issue worldwide. The immune system defends the body by rapidly responding to specific infections and germs via specialized receptors. Host-microbe interaction and subsequent disruption of homeostasis are the results of infections, external insults, or pathogenic microorganisms. Consequently, immunomodulation of the immune reaction may serve as a replacement for a wide range of diseases associated with immunodeficiency. 8
The maturation, maintenance, and function of the immune system are all vulnerable to a wide range of outside influences. For this reason, there has been a long-standing interest in modulation as a means of relieving disease by decreasing or increasing an organism’s immune reactivity to the invading antigen. 9 In addition to causing immunosuppression, presently existing immunomodulators have negative effects on human health. Thus, there is a growing interest around the world in finding better medicines and gauging their immunomodulatory potential. Immunomodulatory substances in natural products have been the subject of intense research and exploration in recent times. 10 There have been numerous research efforts aimed at discovering safe immunoenhancing drugs that can ameliorate myelosuppression and boost the immune response. Some plant extracts, for instance, have been studied for their ability to boost the immune response in Cy-immunosuppressed rats. 11
Research interest in herbal plants and their products has increased rapidly in the modern era of pharmacy because of the low cost and widespread acceptance of plant bioactive chemicals in the management of several ailments with fewer adverse effects. Andrographis paniculata is a well-known herbaceous plant that belongs to the Acanthaceae family. 12 In Ayurveda, A. paniculata is utilized to treat a wide variety of ailments, including a sore throat, the flu, liver diseases, jaundice, dermatitis, intestinal worm infestation, and gonorrhea. 13 However, in Chinese medicine, the plant is used to cure diarrhea, laryngitis, gastrointestinal infections, and inflammation. 14 A. paniculata have several medicinal uses in modern pharmacology, including as an immune system stimulant, a treatment for pharyngotonsillitis and cardiac ischemia, and an agent with anticancer, antibacterial, anti-inflammatory, and anti-HIV properties.15, 16 However, the salutary properties of A. paniculata against immunosuppressive conditions have not been scientifically studied yet. Thus, the present study was dedicated to exploring the therapeutic properties of A. paniculata against cyclophosphamide-induced immunosuppression in mice.
Materials and Methods
Collection of Plant Material
The insect-bite-free fresh leaves of A. paniculata were collected from Bogor, West Java Province, Indonesia, in April 2023 with the help of local herbal plant experts located around the location.
Preparation of Plant Extract
One kg of fresh leaves of A. paniculata were rinsed with clean water, dried at room temperature, and then ground into powder. The leaf powder was then heat macerated with 2 L of distilled water at 60°C for 45 min, and then the suspension was collected and cooled to 37°C. The suspension was filtered using Whatman No. 1 filter paper, and the resultant extract was stored at 4°C until additional use.
Experimental Animals and Diets
The 6-week-old BALB/c mice weighing around 20 ± 4g were obtained from the institutional animal house and employed in the current study. All the mice were maintained at stable laboratory conditions with 24 ± 0.5°C temperature, 50%–60% air moisture, and 12-h/12-h day/night series. The mice were fed regular pellet food and permitted to access the water freely. All the mice were employed in the current work after a week of acclimation in the laboratory.
Experimental Procedures
The acclimated mice were distributed into six groups, with six in each (n = 6/group), as follows: I: control mice; II: cyclophosphamide-induced immunosuppressed mice; III: immunosuppressed + levamisole (standard drug)-treated mice; and IV and V: immunosuppressed + A. paniculata extract (250 and 500 mg/kg, respectively)-treated mice. The body weight of each mouse was quantified once every three days. The treatment with A. paniculata was done by oral gavage for 15 days for the mice in groups IV and V. Additionally, the mice in groups I and II were administered with the same quantity of distilled water, while the mice in group III received 10 mg/kg of levamisole through the oral route.
The immunosuppression in experimental mice from group II was achieved by administering 80 mg/kg of cyclophosphamide through the intraperitoneal route. Mice from group I were injected (i.p.) with the same quantity of saline solution. Following the last injection, the animals were subjected to 24 hr starvation and sacrificed. The immune organs like the spleen, thymus, and liver were taken out suddenly, rinsed with a precooled saline solution at 4°C, and blotted using filter paper. The tissues were then weighed precisely and utilized for the additional assays.
Quantification of the Immune Organ Index
Following the completion of the treatment (on the 16th day), the experimental mice were killed via cervical dislocation, and the organs were dissected. The dissected spleen and thymus organs were then rinsed with a saline solution and weighed precisely. The following formula was used to determine the organ index:
Organ index (mg/g) = Organ weight (mg)/body weight (g).
Hematological Analysis
The blood samples were gathered from the experimental mice and subjected to the analysis of hematological parameters. The levels of white blood cells (WBC), red blood cells (RBC), hemoglobin (Hb), polymorphonuclear cells, lymphocytes, monocytes, eosinophils, and mean corpuscular hemoglobin (MCH) were examined with the aid of an automated hematological analyzer (HEMAVET 950, Drew Scientific Group).
Measurement of Phagocytic Index
The macrophage phagocytic level was measured by the neutral red test. Following the completion of the treatments, the mice were injected (i.p.) with 1 ml of CRBCs (20%) to immunize them. The mice were then sacrificed after 30 min. The cells were collected and grown for 48 h, and then 0.075% neutral red (100 µl) was mixed into each well and maintained for 2 h. Then the cells were rinsed thrice with saline solution, and then a 100 µl solution (50% ethanol and 1% acetic acid, 1:1) was mixed into the wells overnight at 37°C and then absorbance was taken at 540 nm.
Determination of Humoral and Delayed-type Hypersensitivity (DTH) Response
To measure the hypersensitivity response, the experimental mice were injected with 200 µg of methylated BSA (mBSA) at three locations in the abdomen, along with 200 µl of a 1:1 emulsion of CFA and PBS. After eight consecutive immunizations, the experimental mice were injected with 20 µl of 5 mg/ml mBSA in PBS into one rear footpad, while the other rear footpad was administered only with 20 µl of PBS. The swelling of the footpad was determined at 18, 42, and 66 h after the treatment with the aid of a Series 7 spring-loaded caliper. The degree of the DTH responses was measured from variances in the thickness of footpads between the treatment groups.
Histopathological Analysis
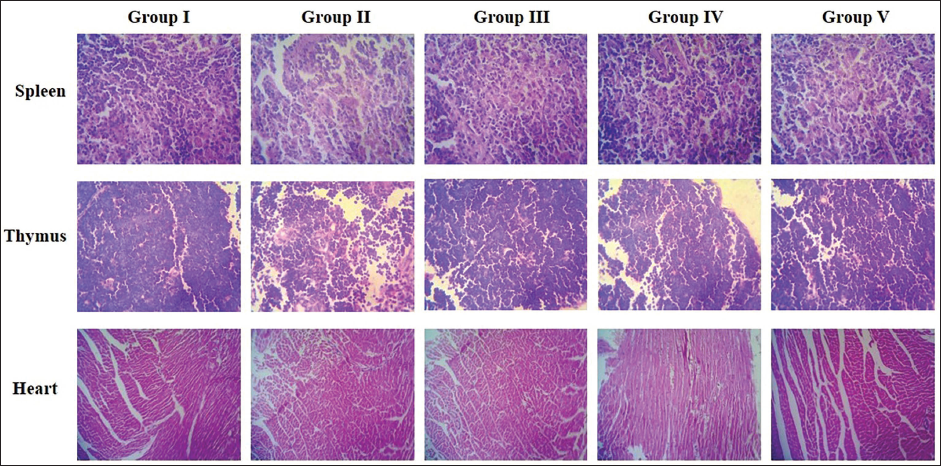
The spleen, heart, and thymus tissues of the experimental mice were dissected after sacrificing under anesthesia. The collected tissues were then rinsed with a saline solution, dehydrated, and fixed with 10% formalin. The tissues were then dehydrated using graded ethanol and finally paraffinized. Then tissues were cut at 5 µm size, stained with hematoxylin and eosin, and assessed using an optical microscope at ×40 magnification to detect the histological changes.
Statistical Analysis
The results obtained from each assay were statistically investigated using GraphPad Prism version 6. The values are revealed as mean ± SD of the triplicate measurements and studied by one-way ANOVA and Tukey’s post hoc test. p < .05 was fixed as statistically significant.
Results
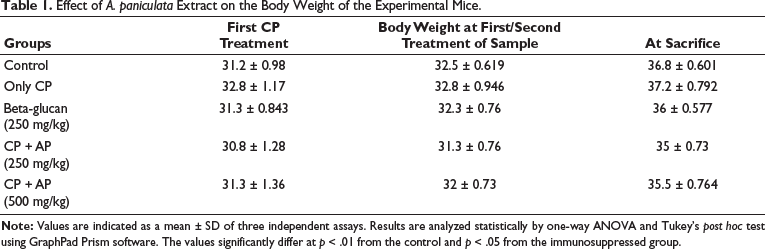
Effect of A. paniculata Extract on the Body Weight of the Experimental Mice
Table 1 reveals the influence of A. paniculata extract treatment on the body weight of the experimental mice. A remarkable reduction in body weight was observed in the mice after the challenge with 80 mg/kg of cyclophosphamide, which confirms the onset of immunosuppression. Interestingly, treatment with 250 and 500 mg/kg of A. paniculata extract substantially improved the body weight of the immunosuppressed mice. The standard rug levamisole treatment also increased the body weight of the immunosuppressed mice, which evidences the therapeutic activity of the A. paniculata extract.
Effect of A. paniculata Extract on the Body Weight of the Experimental Mice.
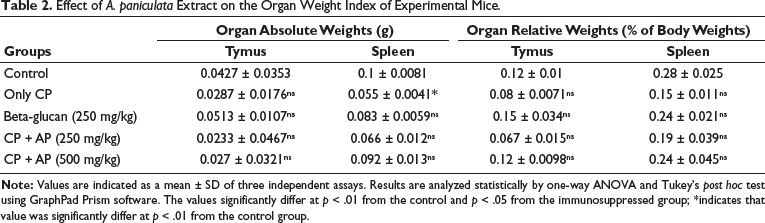
Effect of A. paniculata Extract on the Organ Weight Index of Experimental Mice
The impact of A. paniculata extract on the changes in the weights of the organs, such as the spleen and thymus of the experimental mice, was examined, and the outcomes are illustrated in Table 2. The immunosuppression caused by the cyclophosphamide challenge resulted in a remarkable reduction of organ weights, including the spleen and thymus, when compared to the control. However, the treatment with 250 and 500 mg/kg of A. paniculata extract remarkably boosted the weight of the spleen and thymus in the immunosuppressed mice. The activity of the A. paniculata extract was supported by the results of the standard drug levamisole treatment, which also increased the organ weight index in the immunosuppressed mice.
Effect of A. paniculata Extract on the Organ Weight Index of Experimental Mice.
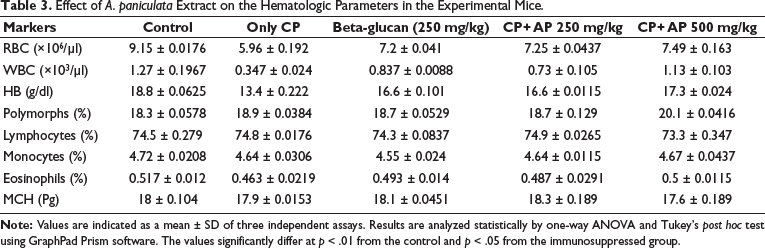
Effect of A. paniculata Extract on the Hematological Parameters in the Experimental Mice
The changes in the levels of hematological parameters such as WBC, RBC, Hb, lymphocytes, neutrophils, monocytes, eosinophils, and MHC was determined, and the findings were revealed in Table 3. The immunosuppressed mice demonstrated a significant reduction in the levels of WBC, RBC, Hb, and eosinophils, but no major alterations were noted in the status of lymphocytes, neutrophils, monocytes, or MHC. Interestingly, treatment with 250 and 500 mg/kg A. paniculata extract considerably regulated these changes and set back the normal levels of hematological parameters in the immunosuppressed mice. The therapeutic effects of A. paniculata extract were supported by the results of standard drug treatment with levamisole.
Effect of A. paniculata Extract on the Hematologic Parameters in the Experimental Mice.
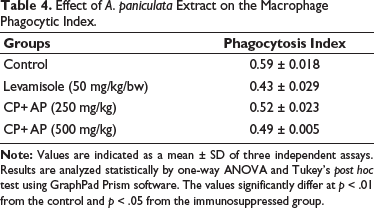
Effect of A. paniculata Extract on the Macrophage Phagocytic Index
The level of macrophage phagocytic index in both control and experimental mice was studied, and the results were demonstrated in Table 4. The cyclophosphamide-induced immunosuppressed mice demonstrated a considerable elevation in the phagocytic index. Fascinatingly, treatment with A. paniculata extract (250 and 500 mg/kg) remarkably suppressed the levels of phagocytic index in the immunosuppressed mice. The levamisole treatment also reduced the phagocytic index, which supports the activity of A. paniculata extract.
Effect of A. paniculata Extract on the Macrophage Phagocytic Index.
Effect of A. paniculata Extract on the Humoral and Delayed Hypersensitivity Response in Experimental Mice
Table 5 demonstrates the influence of A. paniculata extract treatment on the humoral and delayed hypersensitivity reactions in the experimental mice. The immunosuppressed mice demonstrated a considerable diminution in the immune response. Nonetheless, the treatment with A. paniculata extract (250 and 500 mg/kg) remarkably boosted both the humoral and delayed hypersensitivity responses in the immunosuppressed mice, which confirms the immune-boosting effects of the A. paniculata extract. The treatment with levamisole also boosted the immune response in the experimental mice.
Effect of A. paniculata Extract on the Humoral and Delayed Hypersensitivity Response in Experimental Mice.

Effect of A. paniculata Extract on the Heart, Thymus, and Spleen Histopathology of the Experimental Mice
The effect of A. paniculata extract on the histopathology of the heart, spleen, and thymus was analyzed, and the findings were revealed in Figure 1. The control mice demonstrated intact and tightly arranged cardiomyocytes, thymocytes, and splenocytes in the heart, thymus, and spleen, respectively. However, the cyclophosphamide-induced immunosuppressed mice revealed a considerable loss in the cardiomyocytes, thymocytes, and splenocytes, respectively. Furthermore, damaged and necrotic cells were also observed on tissue histopathological images. Captivatingly, the A. paniculata extract (250 and 500 mg/kg) effectively inhibited cell damage and cell loss and revealed normal cell appearances in the heart, spleen, and thymus tissues with intact nuclei, which was found to be similar to the control. The standard drug treatment with levamisole also attenuated the histological changes in the immunosuppressed mice’s heart, spleen, and thymus. These outcomes proved that the A. paniculata extract ameliorated the histological damage in the immunosuppressed mice.
Discussion
Cyclophosphamide or its metabolite-induced immunosuppression may suppress both cell-mediated immunity and humoral immunity. Different cell populations within the immune system are involved in host somatic activities like aging, mental state, inflammation, and infection. 17 Following infection or inflammation, the immune system guards against both foreign stimuli and internal changes. 18 Therefore, it is crucial for patients undergoing chemotherapy treatment to understand how to restore their immune function while in an immunosuppressed state. An impaired immune response to antigens, such as those found on tumor cell surfaces, is defined as immunosuppression. Many factors, including long-term infections, toxic chemical buildup, and excessive radiation exposure, have been linked to this condition. 19 Immunological system regulators are compounds that can either stimulate or reduce the immunological response. Phytochemicals, the natural compounds of herbal origin identified, are well-known natural immunomodulators. 20 Therefore, bioactive compounds and extracts of the plants can be employed to regulate an immunocompromised immune response. In this work, the immunomodulatory properties of the A. paniculata extract were studied in immunosuppressed mice.
Cyclophosphamide-induced myelosuppression is a major problem for chemotherapy-treated cancer patients. Immune mechanisms rely heavily on WBC, which ingest pathogens and produce antibodies. 21 The evaluation of immunocompromised ailments is aided by alterations in the WBC count. However, WBCs have a short lifespan and require a steady supply of stem cells from the bone marrow to differentiate. 22 All components of blood, including leukocytes, platelets, and RBCs, are affected by hematopoietic depression. 23 There is substantial evidence that cyclophosphamide reduces blood components. 24 The bone marrow, the source of WBC, is the organ most negatively impacted by immunosuppressive therapy, as measured by WBC and platelet counts. It is crucial to regulate an equilibrium between RBC and WBC numbers for optimal immunological function. 25 Cyclophosphamide remarkably diminished the peripheral RBCs, WBCs, Hb, and platelets, which is supported by the earlier work. 26 Similar to the findings of several other studies,27, 28 we found that the numbers of erythroblasts, MCH, lymphocytes, monocytes, and neutrophils were also significantly decreased after cyclophosphamide administration in mice. We found that A. paniculata extract protected against cyclophosphamide-induced myelosuppression by increasing peripheral RBC, WBC, Hb, eosinophils, monocytes, neutrophils, lymphocytes, and MHCs.
Other researchers have found that CPA treatment results in a significant loss of body weight, suggesting direct toxicity, as well as a loss of lymphatic organ weight associated with myelosuppressive depletion of peripheral lymphoid cells. 29 In the current study, our results also indicated that the A. paniculata extract effectively increased the body weight of the cyclophosphamide-induced immunosuppressed mice.
Immune organs (such as the spleen and thymus) are essential players in the body’s immune function. 30 The immune system is crucial to human health because it controls and regulates the autoimmune response, keeping it in check. 31 Immune system dysfunction has been linked to a wide range of disorders, including autoimmunity, inflammation, and cancer. 32 Zhu et al. 33 found a close relationship between the spleen and thymus in terms of their roles in both cellular and humoral immunity. Immunological cells can rest and be stimulated by antigens in the spleen, leading to an immunological response. 34 The thymus has a role in controlling peripheral T-cell maturation and is the site of T-lymphocyte differentiation and maturation. 35 Lymphocyte proliferation is reflected in an enlarged spleen and thymus, but organ atrophy may be responsible for a diminished immune response. 36 Therefore, the spleen and thymus indexes can be employed as early indications to probe the immunopharmacological pathways in animal models as a result of dietary supplementation. Immune function and prognosis can be determined from spleen index values. 37 The thymus and spleen are responsible for producing lymphocytes, and larger quantities of these cells signify a healthier immune system. Consequently, if these two organs begin to atrophy, it could lead to a compromised immune system. Mouse models of cyclophosphamide-induced immunosuppression showed diminished spleen and thymus indices in our investigation. This research showed that thymus and spleen atrophy and weight loss in immunosuppressed mice could be attenuated by administering A. paniculata extract.
The immune response relies heavily on macrophages. 38 Macrophages control both the innate and adaptive immune responses, ensuring that the body is adequately protected from harmful microorganisms. It is common knowledge that macrophages are exceptionally secretory cells. In response to stimulation, macrophages secrete a wide range of cytokines that act as signaling molecules for the immune response. Macrophages initiate phagocytosis in response to stimulation and secrete several effector chemicals, including NO, to defend the host against infection and injury. 39 In the current study, a decreased phagocytic index was observed in the immunosuppressed mice. However, the A. paniculata extract boosted the macrophage phagocytic index in the cyclophosphamide-induced immunosuppressed mice.
The effects of cyclophosphamide on the spleen, heart, and thymus were examined through histological examinations. In an earlier study, it was highlighted that cyclophosphamide causes damage to the body’s organs and tissues. 40 The current study found that cyclophosphamide-induced damage to the spleen, thymus, and heart, leading to the death of splenocytes in the spleen, thymocytes in the thymus, and cardiocytes in the heart, respectively. 41 In addition, the treatment with A. paniculata extract effectively prevents the damage caused by cyclophosphamide on the spleen, thymus, and heart tissues. Moraes et al. 42 found that rat tissue damage caused by cyclophosphamide was mitigated by a combination of propolis and Caesalpinia pyramidalis extracts, which validates the current study’s findings.
Conclusion
In summary, this work validates the immunomodulatory properties of A. paniculata extract in cyclophosphamide-induced immunosuppressed mice. The treatment with A. paniculata extract remarkably increased body weight and thymus and spleen organ indexes. Furthermore, the levels of hematological indices were modulated by the A. paniculata extract. The A. paniculata extract treatment increased phagocytic index and humoral and delayed immune responses, and attenuated histopathological changes in the heart, spleen, and thymus in the cyclophosphamide-induced immunosuppressed mice. Hence, it was clear that A. paniculata can be a talented immunomodulatory agent. However, additional studies are still recommended in the future to gain a clear understanding of the salutary properties of A. paniculata.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Approval and Informed Consent
The protocols for the animal experiments were verified and approved by the Institutional Animal Ethics Committee of KMCH College of Pharmacy, Coimbatore (Approval No. KMCRET/ReRc/I.P/62/2022, dated 23/07/2022).
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
