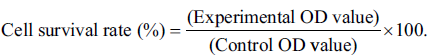Abstract
Background
Safflor yellow (SY) is a water-capacitive component of dried flowers, mainly from safflor in the safflower family, which contains a large amount of bruthin A, or Safflor yellow A (SYA) for short. Studies have reported the protective effects of SYA against oxidative stress injury in nerve cells.
Materials and Methods
In this study, H2O2-treated PC12 cells were used as experimental materials to explore how SYA plays a protective role against nerve cells after oxidative stress and to analyze the molecular mechanism of SYA. PC12 cells were treated with H2O2 and SYA solution. Cell proliferation is then detected with Cell Counting Kit-8 (CCK-8). The detection method is 5-(and 6)-chloromethyl-2′,7′-dichloro-dihydrofluorescein diacetate (CM-H2DCF-DA) staining reactive oxygen species (ROS). Levels of apoptosis-related proteins, including Bcl2-associated X protein (Bax), B-cell lymphoma (Bcl-2), caspase-3, and cleaved caspase-3, and oxidative stress regulators, namely, nuclear factor erythroid-2-related actor 2 (Nrf2) and heme oxygenase 1 (HO-1), were detected by western blotting.
Results
The CCK-8 assay results showed that the survival rate of H2O2-induced PC12 cells was significantly increased after SYA treatment. Fluorescence microscopy (CM-H2DCF-DA staining) indicated that SYA decreased H2O2-induced apoptosis in PC12 cells by reducing intracellular ROS. Western blotting analysis showed that SYA upregulated the expression of Nrf2, HO-1, and Bcl-2 and downregulated the expression of Bax, caspase 3, and cleaved caspase-3 in H2O2-induced PC12 cells.
Conclusion
SYA does protect the nerve cells from oxidative stress damage. Its mechanism of action is mainly related to whether the Nrf2/HO-1 pathway is activated.
Introduction
Ischemic cerebrovascular diseases account for 60%‒80% of cerebrovascular diseases and have relatively high morbidity, mortality, and disability rates worldwide. (Guidelines for Blood Pressure Management for Stroke Prevention and Treatment in China, 2017; Chinese Medical Association Neurology Branch & Chinese Medical Association Neurology Branch Cerebrovascular Disease Group, 2018) Oxidative stress injury eventually causes cell apoptosis and significantly impacts the occurrence and development of cerebral ischemic diseases (Yin et al., 2017). Regarding oxidative stress diseases, the reduction in the scavenging of reactive oxygen species (ROS) or its excessive production is generally believed to lead to its explosive release and induce the activation of related signaling pathways, damaging various tissues and organs (Circu & Aw, 2010; Xiong et al., 2016).
The nuclear factor erythroid-2-related factor 2/heme oxygenase-1 (Nrf2/HO-1) pathway is a multiorgan protective chain against different stressful environments. Nrf2 has a regulatory role in oxidative stress and the expression of encoded antioxidant proteins and inhibits oxidative stress. The endogenous antioxidant capacity of the brain can be increased by disrupting the Keap1-Nrf2 interaction or gene overexpression of Nrf2. The HO-1 gene is dependent on Nrf2, and its product is a powerful antioxidant. With the help of the Nrf2/HO-1 pathway, it can effectively exert anti-inflammatory and anti-oxidation effects, reduces the degree of damage to the mitochondria inside the cell, and automatically adjust the specific gravity of calcium elements to reduce problems such as apoptosis and iron porosis (Dong et al., 2017; Huang & Liu, 2017; Konrad et al., 2016) to prevent damage caused by the stress response. The above-mentioned are theoretical basis and practical basis for treating body organs under oxidative stress (Huang et al., 2015).
Safflor is derived from the dried flowers of Carthamus tinctorius L. of the Compositae family and has a pungent taste and warm nature (National Pharmacopoeia Commission, 2015). Safflor extract contains yellow and red pigments, and safflor yellow (SY) includes hydroxy safflor yellow A (HSYA), safflor yellow A (SYA), safflor yellow B (SYB), and others (Duan et al., 2013). SY consists of a mixture of various water-soluble chalcone components, with water-soluble SYA as its effector (Le, 2015). From the history of cases, SY has long been used as a reagent of action in the treatment of cardiovascular diseases (Chen et al., 2017). Basic and clinical studies have demonstrated the efficacy and safety of SY (Yang et al., 2021). A meta-analysis of 10 randomized controlled trials, including 897 patients with ischemic stroke, showed that SY can improve the overall efficacy and safety of patients with ischemic stroke (Cui et al., 2011). Modern pharmacological studies have shown that its main effects include anti-inflammatory (Li et al., 2013), antioxidative stress, anti-apoptosis (Yang et al., 2019), thrombin inhibition (Zhu et al., 2005), and protection of the cardiovascular and nervous systems (Ablat et al., 2016; Zhang et al., 2015). Yan et al. (2020) have found that increased SY and HSYA levels significantly enhance cell expression activity in the liver and fat structure. The most influential are superoxide dismutase (SOD) and cellular mRNA activity. At the same time, it also promotes the expression of antioxidant enzymes and antioxidant factors in the oxidative stress system. Zhang and Zhang (2012) expressed their own views, experimentally proving that the effect of HSYA and SYB is to play an antioxidant effect by removing 1,1-diphenyl-2-pyrrolelidyl radicals and controlling the redox degradation of 2-deoxyribose in hydroxyl radicals. Thus, HSYA may inhibit apoptosis by activating the PI3K/AKT/GSK3B pathway (Chen et al., 2013). Bacchetti et al. (2020) have studied the regulatory effects of different concentrations of HSYA and SYA on antioxidants and demonstrated that SYA also has antioxidant effects.
Several investigations into the molecular mechanisms of HSYA have been conducted, but there are few studies on SYA, an equally important component of SY, so this study focused on the antioxidant effects of SYA on hypoxic-ischemic diseases. To our knowledge, only one latest article reported that SYA protects rat cardiomyocytes against anoxia/reoxygenation injury, possibly by inhibiting cellular oxidative stress and apoptosis (Duan et al., 2013). PC12 cells are a cell line derived from adult rat adrenal medullary pheochromocytoma with the properties of embryonic developmental neuro spinal cells, a commonly used neural cell line. The main research in this paper is to stimulate PC12 cells with H2O2 as an experimental model. To explore the protective effect of SYA produced inside cells on oxidative stress damage, the purpose is to explore the mechanism of SYA action.
Materials and Methods
Experimental Operation
SYA (purity ≥ 98%, as determined by HPLC) (Figure 1) was supplied by Shanghai Ronghe Pharmaceutical Technology Development Co., Ltd. (Shanghai, China). Edaravone was purchased from Jilin Boda Pharmaceutical Co. Ltd. (Jilin, China). The types of reagents used in cell culture are pheochromocytoma cell lines (PC12). Both DMEM and FBS were purchased from an American-type culture collection (ATCC). The Cell Counting Kit-8 (CCK-8) (C6005 500T) used was purchased from Cell and Molecular Biotechnology Co., Ltd. in Suzhou, China. Western blot reagents, including antibodies against Bax Bcl-2-associated X protein (Bax) (YT0455 20 µg), B-cell lymphoma (Bcl-2) (YM3041 50 µg), caspase-3 (YT0656 20 µg), and Nrf2 (YT3189 20 µg), were purchased from ImmunoWay Biotechnology Company (USA). Antibodies against HO-1 (27282-1-AP 50µL) were purchased from Proteintech (USA). Antibodies against cleaved caspase-3 (Asp175) (9661 20 µL) were purchased from Cell Signaling Technology (Danvers, USA). The fluorescent probe (E004-1-1 500T) belongs to the ROS fluorescent probe, has cell membrane permeability, can actively diffuse into the cell, and is purchased by the Institute of Biological Engineering in Nanjing, Jiangsu Province, China. ML385 (HY-100523, 5 mg) is a specific NRF2 inhibitor purchased from Jiangsu Kaiken Biotechnology Co., Ltd.
Cell Culture and Cytotoxic Assay
PC12 cells are a type of cell line derived from adult rat adrenal medulla pheochromocytoma, a commonly used neural cell line. PC12 cells need to be grown in a DMEM-rich environment with 10% FBS and 1% penicillin. Before the experiment begins, cells need to be conceived in a humidity incubator at 37°C with a CO2 content of 5%. The different concentrations of SYA or H2O2 were chosen to treat PC12 cells. The concentration of SYA ranged from 10 to 500 mg/L; the concentration of H2O2 ranged from 200 to 800 µM. PC12 cells were incubated in a 96-well plate at 37°C and 5% CO2 for 24 h, removed from the old culture medium, added into different concentrations of SYA or H2O2, and incubated for 4 h. Cell proliferation and toxicity were detected by CCK-8. Test the absorbance of light at 450 nm with the microplate reader module in the Molecular Device Spectroscopy Max M5E tool.
Analysis of Cell Viability
Six different sets of experiments are performed under each set of conditions, divided into blank control group and experimental group (H2O2), SYA (10 mg/L) + H2O2, SYA (50 mg/L) + H2O2, SYA (100 mg/L) + H2O2, and edaravone (10 µM) + H2O2. Place PC12 cells in a cell culture incubator with a box size of 100 µL/well, then incubate at 37°C at 5% CO2 concentration for 24 h, followed by placing the cells in H2O2 solution at a concentration of 500 µM for 4 h. This study was conducted in six independent experiments. Cell viability is measured using the CCK-8 test method.
Analysis of Intracellular ROS Levels
Transfer the cells to a 6-well plate environment for 24 h. Three different concentrations of SYA are then set to be added to the Petri dish followed by gestation for 24 h, 10, 50, and 100 mg/L, respectively. The final step is to add H2O2 culture at a concentration of 500 µM for 4 h. PC12 cells (3×106) were harvested, washed using phosphate buffered saline, and stained with 5-(and 6)-chloromethyl-2ʹ,7ʹ-dichlorodihydrofluorescein diacetate (CM-H2DCF-DA) (10 µM), and incubated for 30 min. A fluorescence microscope (Leica DM2500) with absorbance at 488 nm (excitation) and 530 nm (emission) was used to continuously collect images to observe changes in DCF fluorescence in cells.
Western Blot Analysis
The PC12 cells were divided into four groups: control, model (H2O2), SYA (10 mg/L) + H2O2, and SYA (10 mg/L) + H2O2 + ML385 (5 µM). The proteins are separated by the influence of 10% sodium dodecyl sulfate-polyacrylamide gel electrophoresis. The isolated protein is then transferred to the polyvinylidene fluoride membrane. Treat with a 5% concentration of skim milk for 2 h followed by incubation overnight with biological antibodies to Nrf2, HO-1, Bax, Bcl-2, caspase-3, lysis caspase-3, or β-actin. The next day washes three times with ST and incubated with the secondary antibody for 2 h at normal room temperature (37°C). Bands were visualized using the electrochemiluminescence method. Images were acquired using a two-color laser imaging system (LICOR Odyssey). Protein bands were analyzed using ImageJ software and compared with the gray value of β-actin to analyze the protein expression level.
Statistical Analysis
SPSS software (version 25.0) was used to manage and analyze all experimental data. There are two more reference criteria for the results, including the mean and the standard deviation. Data variance is meant to reflect differences between cells. The LSD t-test is used for comparisons between different processing groups. Statistically the probability <0.05 is considered a reasonable range.
Results
SYA Was Non-Cytotoxic to PC12 Cells
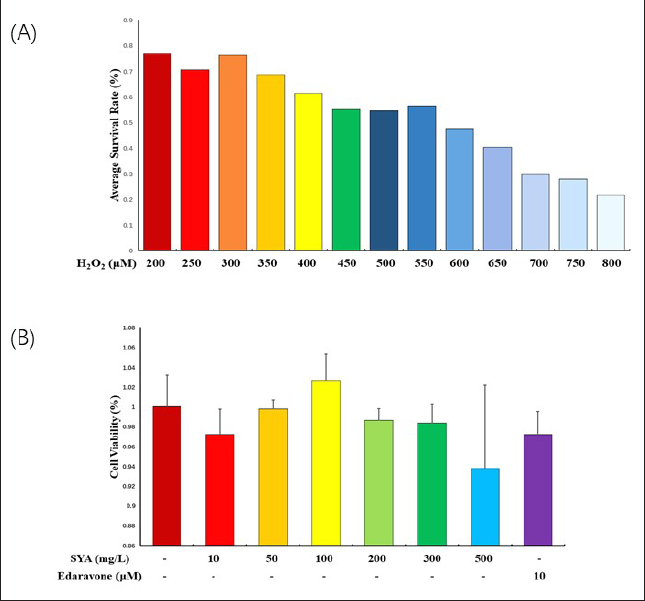
We observed the cell proliferation effects of H2O2 and SYA on PC12 cells by performing the CCK-8 assay. As shown in Figure 2A, H2O2 at concentrations of 200‒800 µM significantly inhibited the proliferation of PC12 cells at 450 µM (p < 0.001), with an IC50 of 500 µM. SYA (10‒500 mg/L) showed no significant cytotoxicity to PC12 cells, even at a higher concentration of 500 mg/L (Figure 2B). Based on the aforementioned observations, 500 µM H2O2, and 10, 50, and 100 mg/L SYA were selected for subsequent experiments. The CCK-8 assay experiment is to still test the relative proliferation rate of PC12 cells, and select the experiment with a concentration of 10 µM as the positive control group.
SYA Increased H2O2-treated PC12 Cell Viability
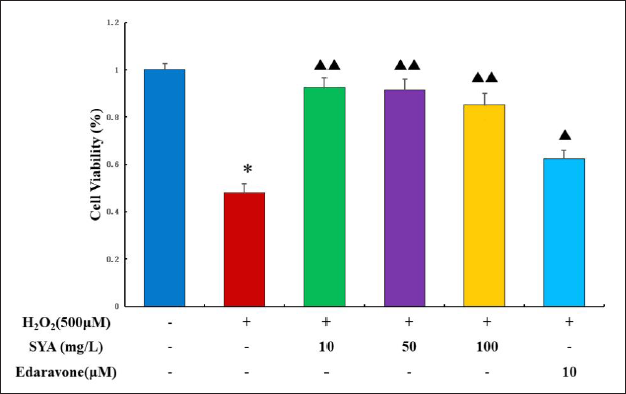
In addition, we also assessed the protective strength of SYA content against H2O2-induced oxidative stress damage in PC12 cells. As shown in Figure 3, the CCK-8 assay results found that the SYA-treated experimental group had a higher survival rate than the H2O2-induced PC12 cells compared to the model group (p < 0.001).
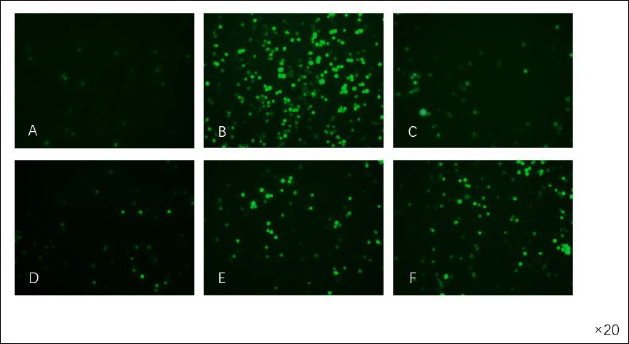
SYA Reduced H2O2-induced Intracellular ROS Production
Compared with those in the model group, intracellular ROS levels were significantly reduced in the SYA-treated group. After CM-H2DCF-DA staining, a fluorescence microscope was used to continuously collect images to observe changes in DCF fluorescence in the cells. The experimental results show that the sensitivity of PC12 cells treated with H2O2 to green fluorescence is increased, but treatment with SYA weakened the green fluorescence (Figure 4). From this we know that SYA can reduce H2O2-induced production of intracellular ROS, thus exerting an antioxidative stress effect.
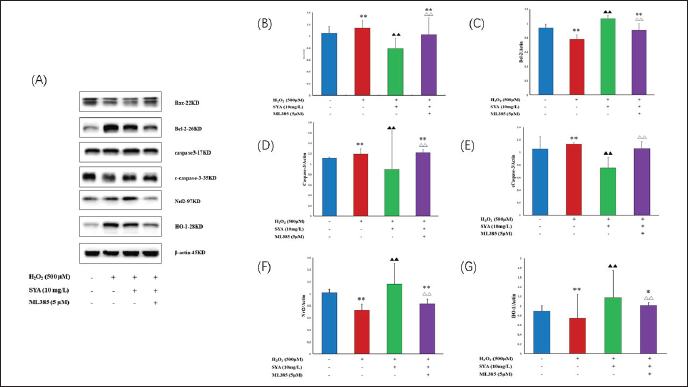
SYA Inhibited H2O2-induced Apoptosis and Upregulated the Expression of Nrf2 and HO-1
Western blotting detects protein expression in cells. According to the experimental results, the protein content of Bcl-2 in PC12 cells induced by H2O2 decreased, and the protein content of Bax and caspase-3 increased, and cleaved caspase-3 expression (p < 0.001). By contrast, after SYA (10 mg/L) treatment, Figures 5A–E shows that the protein content of Bcl-2 is significantly increased compared to the original. In contrast, the protein content of Bax, caspase-3, and lysed caspase-3 showed a sharp downward trend. Western blot results also showed that SYA (10 mg/L) upregulated Nrf2 and HO-1 expression (p < 0.001). Simultaneously, we added ML385, an Nrf2 antagonist, and found that the previously increased expression of Nrf2 and HO-1 was suppressed. (p < 0.001). These results indicate that SYA protects PC12 cells against H2O2-induced apoptosis and exerts an antioxidant effect by activating the Nrf2/HO-1 pathway (Figures 5A, F, and G).
Discussion
The herb Safflor is widely used for the relief of hypertension, coronary heart disease, and ischemic cerebrovascular diseases (Qu et al., 2016). As stated in the “Introduction” section, the protective effects of SY in ischemic stroke have been investigated previously, and anti-thrombosis (Zeng et al., 2002), anti-coagulation (Sun et al., 2010), anti-oxidation (Sun et al., 2012), anti-inflammation (Ye & Gao, 2008), and anti-calcium dysregulation (Xu et al., 2019) effects have been observed. Multiple studies have demonstrated that oxidative stress plays a key role in nerve cell injury after an ischemic stroke (Fan et al., 2011; Sun et al., 2012; Tian et al., 2004; Wei et al., 2005).
In the first part of this study, we explained that PC12 cells induced by H2O2 were used to simulate some of the characteristics of oxidative stress damage in human nerve cells. The results of the cell viability assay indicated that this model was successful. The survival ratio of PC12 cells induced by H2O2 can be significantly enhanced after the cells are treated with SYA. Different macromolecules react with endogenous ROS in cells and cause oxidative stress. We found that H2O2 treatment increased intracellular ROS levels, whereas SYA treatment decreased these levels. The above experimental conclusions all show that SYA does protect H2O2-induced PC12 cells from oxidative stress damage. Theoretically, it is an effective way to exert antioxidant effects by increasing the ability to scavenge ROS or reducing the production of ROS after adding antioxidants. Notably, we found that low concentrations of SYA (10 mg/L) caused a stronger reduction in intracellular ROS production than high concentrations of SYA (100 mg/L). The reason for this phenomenon may be the anti/pro-oxidative functions of antioxidants. Under physiological conditions, the addition of antioxidants excessively scavenges ROS, leading to a redox imbalance, which can also induce cell signaling disorders and trigger the initiation of a self-compensating pro-oxidative system in the body (Chen et al., 2012).
Researchers have investigated the molecular mechanisms by which HSYA protects neural cells from oxidative stress, including the inactivation of the p38MAPK pathway and AMPK-mediated inhibition of nuclear factor-kappa B (Du et al., 2019; Zhao et al., 2020). Hu et al. (Hu et al., 2016) confirmed that HSYA exerts anti-apoptotic effects through the PI3K/Akt/Nrf2/HO-1 pathway to alleviate myocardial ischemia‒reperfusion injury. By contrast, regarding the mechanism of antioxidative stress in ischemic diseases, Huang and Liu (Huang & Liu, 2017) found that the Nrf2/HO-1 pathway was closely related to the inflammation and oxidative stress of resveratrol in rats with myocardial ischemia‒reperfusion injury. It downregulated the levels of the inflammatory factor myeloperoxidase (MPO) and upregulated antioxidant levels, namely, SOD and glutathione peroxidase (GPx). According to our literature review, further research on the evidence regarding the antioxidative stress of SYA in ischemic cerebrovascular disease via the Nrf2/HO-1 pathway is necessary.
In addition, we explored the mechanism of action of SYA in its ability to resist oxidative stress responses. We found that the transcription factor Nrf2 is the main regulator of the entire redox process, regulating the degree to which antioxidants and phase II detoxification enzymes are expressed in antioxidant defences. HO-1, a phase II detoxification enzyme, protects multiple organs against oxidative stress. In addition to its antioxidant effect, Nrf2 can improve apoptosis and inflammatory pathways. The complete Nrf2/HO-1 pathway exerts multiple effects, such as anti-oxidation, anti-inflammatory effects, reduction of mitochondrial damage, and regulation of Ca2+ influx and cell death, affecting the outcome of the disease.
The results of this study show that after the H2O2 treatment, the protein expression of Bax, caspase-3, and lysis caspase-3 is increased, while the protein expression of Bcl-2 is reduced. After SYA treatment in the experimental group, the expression protein content of Bax, caspase-3, and cleavage aspartase-3 in H2O2-induced PC12 cells decreased, and the protein content of Bcl-2 increased, effectively reducing the phenomenon of apoptosis in cells after H2O2 action. And we also found that other levels of H2O2-induced PC12 cells that had been treated with SYA also changed. The protein content associated with the Nrf2/HO-1 pathway was significantly increased. After the pathway inhibitor ML385 was added to the culture solution, SYA underwent the opposite transformation of the protective effect of oxidative stress on PC12 cells, which inhibited the Nrf2/HO-1 pathway. These findings provided evidence that allowed us to speculate that the antioxidative stress effect of SYA is partially dependent on the activation of the Nrf2/HO-1 pathway.
In studies of diseases in the treatment of the nervous system, most of the literature on the function and use of Nrf2/HO-1 has focused on neurodegenerative diseases (Cui et al., 2018; Huang et al., 2017), but few studies have focused on acute ischemic stroke. This study shows for the first time that SYA activates an antioxidative stress mechanism by upregulating the Nrf2/HO-1 pathway, protecting nerve cells from damage. This study demonstrated that the Nrf2/HO-1 pathway is a potential therapeutic target for future ischemic stroke.
Moreover, previous research (Huang et al., 2017) has reported that the Nrf2 pathway protects neurons from the loss of mitochondrial membrane potential (MMP) in neurodegenerative diseases, reducing ROS production and protecting cells from apoptosis. Similarly, this study confirmed that apoptosis can be inhibited through the Nrf2/HO-1 pathway. As discussed, our research suggests that SYA can be further investigated, starting with the reversal of MMP reduction in ischemic stroke. By contrast, SYA may be a therapeutic agent for neurodegenerative diseases.
Conclusion
In conclusion, this study demonstrated that SYA had no obvious cytotoxicity in normal PC12 cells, and encouragingly it showed significant protection against H2O2-treated PC12 cells. We also provided evidence that SYA reduced H2O2-induced apoptosis in PC12 cells by way of reducing intracellular ROS, and its mechanism was related to the upregulation of the Nrf2/HO-1 pathway. Thus, our results provide experimental evidence that SYA can be used as an antioxidant treatment for nerve cell oxidative stress injury.
Footnotes
Summary
SY is derived from the dried flowers of C. tinctorius L. of the Compositae family and is its main water-soluble component. SYA is the main active ingredient of SY and is commonly used in the clinical treatment of ischemic cardiovascular and cerebrovascular diseases. Studies have reported the protective effects of SYA against oxidative stress injury in nerve cells. In this study, PC12 cells treated with H2O2 were used as a model to explore the protective effect of SYA on oxidative stress injury and explore its possible molecular mechanism. PC12 cells were treated with H2O2 or SYA. Cell proliferation was determined using a CCK-8 assay. ROS were stained with CM-H2DCF-DA. Levels of apoptosis-related proteins, including Bax, Bcl-2, caspase-3, and cleaved caspase-3, and oxidative stress regulators, Nrf2 and HO-1, were detected by western blotting. The CCK-8 assay results showed that the survival rate of H2O2-induced PC12 cells was significantly increased after SYA treatment. Fluorescence microscopy (CM-H2DCF-DA staining) indicated that SYA decreased H2O2-induced apoptosis in PC12 cells by reducing intracellular ROS. Western blotting analysis showed that SYA upregulated the expression of Nrf2, HO-1, and Bcl-2 and downregulated the expression of Bax, caspase 3, and cleaved caspase-3 in H2O2-induced PC12 cells. SYA can protect nerve cells from oxidative stress injury, and its mechanism was related to the activation of the Nrf2/HO-1 pathway.
Acknowledgments
Thanks for the support of the Shanghai Traditional Chinese Medicine Diagnosis and Treatment Model Innovation Pilot Construction Project (ZY (2018-2020)-FWTX-6013), the Three-Year Action Plan for “Strengthening Traditional Chinese Medicine” in Hongkou District, Shanghai (HGY-YSZK-2018-10), Shanghai Sailing Program (2021YF1447800), and the Scientific Research Project of Traditional Chinese Medicine of Shanghai Hongkou District Health Committee (HKQ-ZYY-2020-15). We appreciated Dr. Zhang Yian and Mrs. Ni Wenwen for their practical suggestions on the manuscript. We would like to thank Editage (
Abbreviations
Bax: Bcl2-associated X protein; Bcl2: B-cell lymphoma 2; CCK-8: Cell Counting Kit-8; CM-H2DCF-DA: 5-(and 6)-chloromethyl-2ʹ,7ʹ-dichloro-dihydrofluorescein diacetate; GPx: glutathione peroxidase; HO-1: heme oxygenase 1; HSYA: hydroxy safflor yellow A; MMP: mitochondrial membrane potential; MPO: myeloperoxidase; Nrf2: nuclear factor erythroid-2-related actor 2; ROS: reactive oxygen species; SOD: superoxide dismutase; SY: safflor yellow; SYA: safflor yellow A; SYB: safflor yellow B; PC12: pheochromocytoma cell lines.
Authors’ Contributions
Dr. Wang and Dr. Chen designed the study. Dr. Chen and Dr. Gu drafted the main manuscript text. Dr. Chen, Dr. You, and Dr. Xie collected the data. Dr. Chen and Dr. Wang contributed to the bioinformatics analysis. Dr. Lu prepared Figures 1–![]() and the graphical abstract. All authors reviewed the manuscript.
and the graphical abstract. All authors reviewed the manuscript.
Data Availability Statement
The data used to support the findings of this study are available from the corresponding author upon request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
This article does not contain any studies with human participants or animals performed by any of the authors.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.