Abstract
Background
Membranous nephropathy (MN), one of the primary pathogenic forms of adult nephrotic syndromes, frequently coexists with hypercoagulability and hyperviscosity. MN is prone to thrombosis, embolism, and other complications, leading to the accelerated occurrence of glomerulosclerosis and renal fibrosis. Therefore, it is particularly important to promote blood circulation and remove stasis through anticoagulant therapy. Salvianolate (SAL) is a Chinese patent anticoagulant commonly used in clinical practice to promote blood circulation and remove blood stasis. SAL plays an important role in alleviating urinary protein and renal pathological damage in MN patients.
Objectives
In the present study, we aimed to investigate the kidney-protective effect of SAL on MN in a rat model.
Materials and Methods
The rat model of MN was established by tail vein injection of cationic bovine serum albumin (C-BSA). After the treatment, urinary proteins, hypercoagulable state index (fibrinogen (Fib), D dimer (D-D)), hepatic and renal functions, renal pathology, and podocyte marker proteins were analyzed to explore the renal protective effect of SAL on MN rats and its underlying mechanism.
Results
In the modeled rats, we discovered a significant rise in urinary protein, a hypercoagulable state, and hypoproteinemia. Additionally, the expressions of Wilms’ tumor protein 1 (WT-1), podocalyxin (PCX), and vascular endothelial growth factor (VEGF) in renal tissues were significantly downregulated, indicating remarkable pathological damage to podocytes and renal tissues in MN rats. The expressions of the above-mentioned indices could be greatly reversed by SAL, which could also regulate the hypercoagulable state and suppress podocyte damage and renal pathological harm.
Conclusion
Our results suggested that the renal protective effect of SAL on C-BSA-induced MN was related to its ability to inhibit hypercoagulable states, upregulate the expressions of WT-1, PCX, and VEGF in the renal tissue, and repair podocyte injury.
Introduction
Membranous nephropathy (MN) is a primary glomerular disease that is accompanied by proteinuria, hematuria, edema, and hypertension as the main clinical manifestations. MN is one of the common pathological types of adult nephrotic syndromes, and its incidence is increasing year by year in China. It has been reported that the incidence of MN in China accounts for 30.8%–36.5% of primary glomerular diseases (Hu et al., 2020; Su et al., 2019), and the proportion can be as high as 43.9% in people aged more than 60 years (Nie et al., 2021). MN is often accompanied by the existence of hypercoagulability and hyperviscosity, which is prone to thrombosis, embolism, and other complications, promoting microcirculation disorders and accelerating the occurrence of glomerulosclerosis and renal fibrosis. Therefore, some experts have suggested that promoting blood circulation, removing stasis, and anticoagulation should be considered throughout the treatment of MN (Zou et al., 2019).
As a commonly used medicine for promoting blood circulation and removing blood stasis in clinical practice, salvianolate (SAL) has the effects of promoting blood circulation, nourishing blood, cooling blood, and channeling menstruation. In ancient China, there was even a theory that “one single Danshen works as well as Siwu”, which emphasizes the effect of Danshen in promoting blood circulation and removing blood stasis. SAL is the main active ingredient extracted from Danshen. Modern pharmacological studies have confirmed that SAL has the functions of anticoagulation, anti-platelet aggregation, correcting lipid metabolism disorders, anti-inflammation, improving microcirculation, and so on (Pang et al., 2020; Ren et al., 2019; Zheng et al., 2021), and it has been widely used in clinical treatment.
SAL has certain advantages in the treatment of MN. In the present study, we aimed to study the effect of SAL on urinary protein, hypercoagulable state index, hepatic and renal functions, and podocyte marker proteins in MN rats. Moreover, we also explored the renal protective effect of SAL on MN in rats. Collectively, our findings provided valuable insights into the therapeutic regimens of MN.
Materials and Methods
Chemicals and Reagents Used
SAL for injection (Z20050248) was provided by Shanghai Green Valley Pharmaceutical Co. Benazepril hydrochloride tablets (X1902) were supplied by Shenzhen Salubris Pharmaceutical Co. Bovine serum albumin (BSA, A7906) was obtained from Beijing Solebo. Freund’s incomplete adjuvant (Bj-012-0392) was purchased from Sigma-Aldrich. Goat anti-rat IgG (20147-1) and FITC-labeled anti-goat IgG (111-095-003) were provided by Hebei Bohai Biological Co. Rabbit polyclonal antibody against rat Wilms’ tumor protein (WT-1) (BS-6983R), sheep polyclonal antibody against rabbit podocalyxin (PCX) (BS-5000R), and rabbit polyclonal antibody against rat vascular endothelial growth factor (VEGF) (BK-392) were supplied by Beijing Boaosen Biotechnology Co. TRNzol total RNA extraction reagent (Dp405-02) was obtained from Qiagen Biochemical Technology Co. PrimeScript™ RT Reagent Kit with gDNA Erase (RR047B) was purchased from TaKaRa. SYBR® Premix Ex Taq™ II (Tli RNaseH Plus) ROX Plus (RR82LR) was provided by TaKaRa. DL2,000 DNA marker (3427Q) was supplied by TaKaRa. Primers were synthesized using Invitrogen.
Experimental Animals
A total of 56 clean-grade healthy male SD rats weighing 180 ± 20 g were provided by the Experimental Animal Center of Hebei Medical University (certificate no. 1509116). The animals were bred in a standard laboratory environment: temperature (24 ± 2°C) and humidity (50 ± 5%). During the experiment, the rats were given free access to diet and water, and the padding was changed regularly. Animal experiments were approved by the Hebei University of Traditional Chinese Medicine Laboratory Animal Management and Ethics Committee (No. DWLL2019047) and were performed in accordance with national and institutional guidelines.
Experimental Design and Treatment Protocols
A total of 56 male SD rats were tested for negative urine protein. Nine rats were randomly selected for the control group, while the other rats were modeled. First, cationized BSA cationic bovine serum albumin (C-BSA) was prepared according to the modified Border method (Border et al., 1982). Freund’s incomplete adjuvant was mixed with 1 mg of C-BSA and 0.5 mL of PBS, followed by shaking and emulsification. Preimmunization was conducted by subcutaneously injecting prepared C-BSA in both armpits and groins of rats once every other day for three times. Finally, formal immunization was performed, and C-BSA was dissolved at a concentration of 16 mg/kg in 1 mL of PBS for tail vein injection three times a week, a total of 12 times. During modeling, one rat died, and the urine protein test of the other 46 rats was positive. Two rats were randomly selected for renal pathological examination to confirm the successful establishment of modeling.
The remaining 44 rats were randomly divided into the model group (n = 8), benazepril group (n = 9), SAL low-dose group (SAL-L, 16.7 mg/kg/day, n = 9), SAL medium-dose group (SAL-M, 33.3 mg/kg/day, n = 9), and SAL high-dose group (SAL-H, 66.7 mg/kg/day, n = 9) by the random number table method. Rats in the control group and model group were given distilled water (3 mL/mouse) by intragastric administration and normal saline (1.5 mL/mouse) by intraperitoneal injection daily. Rats in the benazepril group were given benazepril hydrochloride, which was dissolved in 3 mL of distilled water at a dose of 0.01 g/kg and injected intraperitoneally with normal saline (1.5 mL/mouse). Rats in each SAL group were given 3 mL of distilled water by intragastric administration, and the corresponding dose of SAL was dissolved in 1.5 mL of normal saline and given by intraperitoneal injection once a day for 4 weeks. The dosage of SAL in rats was converted from the dose of rats and humans (SAL-L 16.7 mg/kg, SAL-M 33.3 mg/kg, and SAL-H 66.7 mg/kg). The low dose of SAL was half of the medium dose, and the high dose of SAL was twice the medium dose. The diet, mental state, and reaction of the rats were observed every week during the experiment.
Urine Total Protein (24-UTP) and Biochemical Analyses
Urine protein was measured after 24-h before the end of the experiment, each rat was placed in a metabolic cage, fasting without water, and urine was collected for 24 h. A supernatant was recovered after the centrifugation, and the urine protein level was calculated using the biuret method (Guobing et al., 2001). For the biochemical test, 1% of sodium pentobarbital (150 mg/kg) was injected intraperitoneally for euthanasia. The blood sample (5 mL) was collected through the abdominal aorta and centrifuged at 3,000 rpm for 10 min. The upper serum was obtained by centrifugation and used for the detection of fibrinogen (Fib), D dimer (D-D), triglyceride (TG), total cholesterol (TC), serum total protein (TP), albumin (ALB), blood urea nitrogen (BUN), and serum creatinine (Scr).
Kidney Specimen Collection and Histopathological Study
After blood collection, kidney specimens were taken from sacrificed rats, the capsule was stripped, and the renal cortex of 10 mm × 2 mm × 2 mm was dissected and fixed in formalin-acetic acid-ethanol for 24 h. The specimens were then washed with water, subjected to ethanol gradient dehydration, and cut into 2-µm sections, followed by Masson’s staining and PASM staining. The changes in glomerular volume, morphology, basement membrane, and podocytes were recorded after observation under a light microscope at a magnification of 400×. Subsequently, a renal cortex of 1 mm × 1 mm × 1 mm was fixed with glutaraldehyde at 4°C, washed with PBS, fixed in 1% osmic acid, dehydrated in an ethanol gradient, and embedded in epoxy resin. The tissue was sectioned, followed by double staining of uranium acetate and lead citrate. The morphology of glomerular podocytes and basement membrane, as well as the deposition of immune complexes, were examined using transmission electron microscopy (TEM) at a magnification of 15,000×. The kidney samples of rats were prepared into 5-µm sections on ice, fixed with cold acetone, washed with PBS, sequentially incubated with FITC-labeled IgG primary antibody and secondary antibody, and sealed with glycerol phosphate buffer. IgG expression in the glomerular was observed under a fluorescence microscope at a magnification of 400×.
Immunohistochemistry
The kidney specimens were fixed in 10% neutral formaldehyde, dehydrated in an ethanol gradient, transparent by xylene, embedded by paraffin, sectioned, and stored overnight. Then the specimens were dewaxed and subjected to antigen retrieval, followed by incubation with primary and secondary antibodies. The expressions of WT-1, PCX, and VEGF at the protein level in the kidney tissues were examined under the microscope, and the result was expressed as dark brown, brown, light brown, or no color, where brown represents the positive results. Five fields were selected for each section, and the number of positive cells under the microscope (A) and staining intensity of positive cells (B) were scored at high magnification (400×). The results of immunohistochemical scoring were conducted by a semi-quantitative analysis, and the scoring standard was as follows: IHS = A × B, where A is the number of positive cells, 0 points for 0%–5%, 1 point for 6%–25%, 2 points for 26%–50%, 3 points for 51%–75%, and 4 points for 76%–100%; B is the staining intensity grade of positive cells, negative score 0, weak positive score 1, positive score 2, and strong positive score 3.
Gene Expression of WT-1, PCX, and VEGF in the Renal Tissues
Real-time PCR was used to detect the expressions of the above-mentioned genes. The total RNA was extracted from the fresh renal cortex by Trizol reagent, and then purified RNA was reverse-transcribed into cDNA. Primer sequences were as follows: WT-1 primer (length: 200 bp), forward 5′-CCCTACAGCAGTGACAATTTATAC-3′, reverse 5′-TACTGGGCACCACAGAGGAT-3′; PCX primer (length: 213 bp), forward 5′-CCTCCCACTGCACATTA-3′, reverse 5′-TCACTGTAGTCAAACACACGCTC-3′; VEGF primer (length: 133 bp), forward 5′-CAAAGCCAGCACATAGGAGAGAT-3′, reverse 5′-TTTTTGCAGGAACATTTACGTC-3′; GAPDH primer (length: 270 bp), forward 5′-TTCCTACCCCCAATGTATCCG-3′, reverse 5′-CCACCCTGTTGCTGTAGCATA-3′. PCR primers were synthesized using Invitrogen. GAPDH was selected as the housekeeping gene. The relative expressions of the target genes were calculated using the 2–∆∆Ct method.
Statistical Analysis
SPSS software 22.0 was used for data analyses. All data were expressed as mean ± standard deviation (SD). Normality tests and homogeneity tests of variance were performed for the measurement data. When normality and homogeneity of variance were evident, the t-test, one-way analysis of variance (ANOVA), and LSD technique were employed to compare two groups, multiple groups, and numerous comparisons between groups, respectively. When the distribution was not normal or the variance was uneven, the rank-sum test was used, and p < 0.05 was considered statistically significant.
Results
SAL and 24-UTP Measurements in MN Rats
Before the treatment, the 24-UTP of rats in each model group was significantly increased, indicating that the disease model was successfully established. There was no difference in 24-UTP of rats in each model group. After the treatment, the 24-UTP of rats in the model control group was further increased, while the 24-UTP of all treatment groups was decreased compared with the value before treatment. Compared with the model control group, the difference was significant (Figure 1). The results showed that both benazepril and SAL could reduce the level of urinary protein, and its effect in the SAL-M and SAL-H groups was slightly better compared with benazepril.
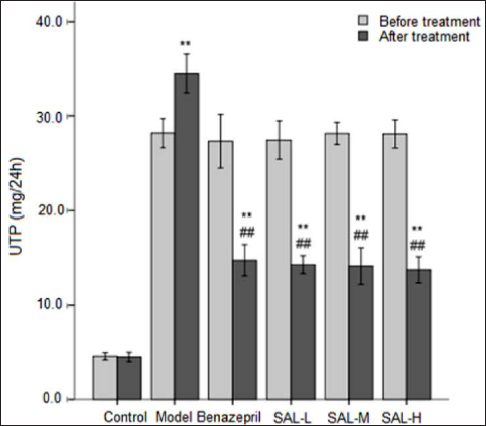
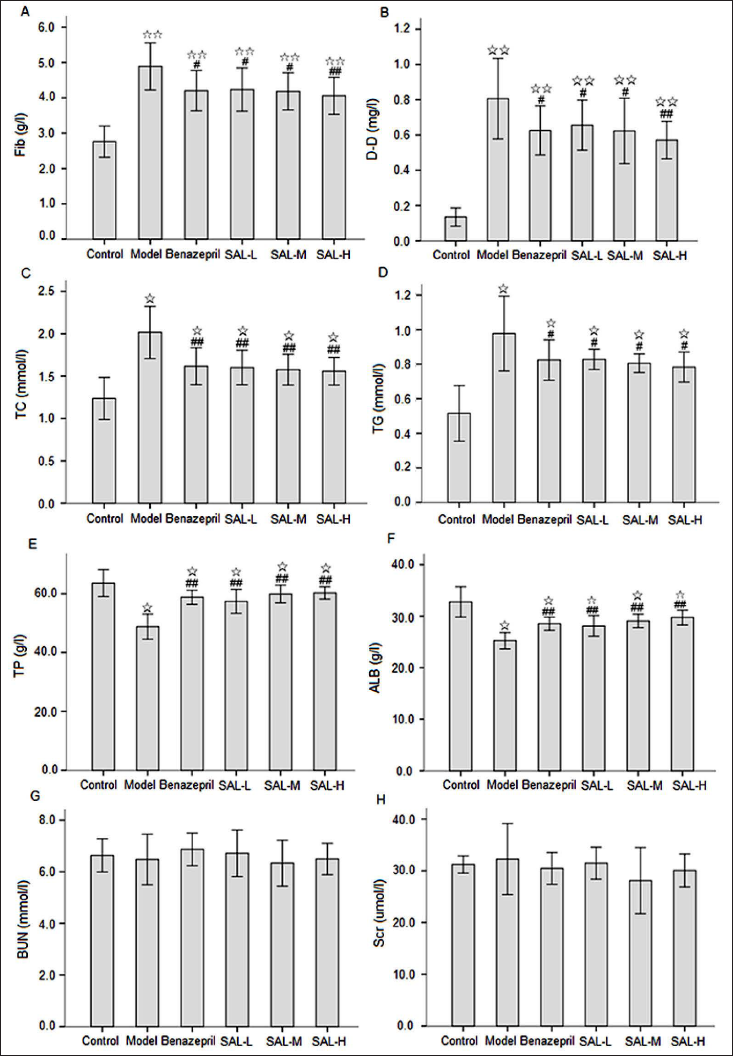
SAL and Biochemical Measurements
Hypercoagulable state, blood lipid, and serum protein levels in MN rats revealed the effectiveness of SAL treatment (Figure 2). Figure 2 shows that compared with the control group, the contents of Fib, D-D, TC, and TG in the model group were increased, while the contents of TP and ALB were decreased. Compared with the model control group, the contents of Fib, D-D, TC, and TG in all treatment groups were significantly decreased to different degrees. The contents of TP and ALB were significantly increased. There were no differences in the contents of Fib, D-D, TC, TG, TP, and ALB between the benazepril group and SAL treatment groups. Moreover, there was no difference among different SAL treatment groups. In addition, there was no significant difference in the contents of BUN and Scr among all groups. The results showed that there were a hypercoagulable state, hyperlipemia, and hypoproteinemia in MN rats, which was consistent with the urinary protein level. SAL and benazepril could inhibit the hypercoagulable state, decrease the blood lipid, and increase the serum protein level, and the effect of SAL in the SAL-M and SAL-H groups was more significant.
SAL and Renal Histopathology, Immunopathology
To observe the effect of SAL on renal pathological damage in MN rats, we observed the glomerulus by light microscopy, TEM, and immunofluorescence (Figure 3).
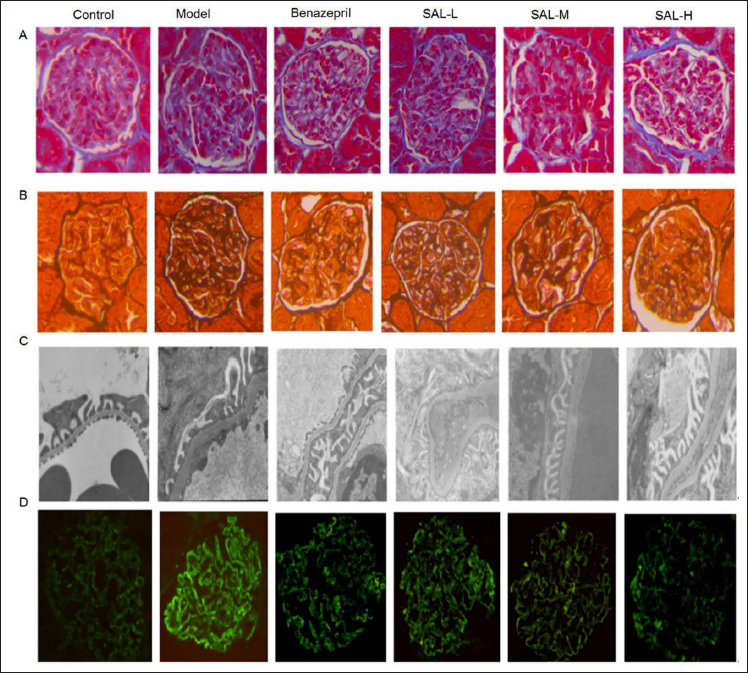
Light microscopy showed that there was no significant change in glomerular morphology in the control group. In the model group, the glomerular volume was increased irregularly, some capillary loops were twisted and expanded, the immune complex of the glomerular subepithelium was deposited, and the basement membrane was formed with a diffuse and uneven thickening “spike”. Compared with the model group, the glomerular volume and capillary loop dilation were not obviously changed in the benazepril group and SAL treatment groups, the glomerular subepithelial immune complex showed scattered granular deposition, the basement membrane was slightly thickened with a few “spikes”, and the degree of pathological damage was lighter compared with the model group to varying degrees.
According to the TEM findings, the basement membrane was not thickened, the podocyte foot processes were arranged in a regular and orderly manner, and the glomerular podocyte structure of the control group was normal. In the model group, there was an extensive deposition of electron-dense material under the epithelium. Moreover, a large number of fusions or disappearance of podocyte foot processes, the formation of “spike”, irregular thickening, distortion of the basement membrane, and irregular stratification were also observed. Compared with the model group, the benazepril group and SAL treatment groups showed a small amount of subepithelial electron density deposition, partial fusion of podocyte foot processes, and less thickening of the basement membrane to varying degrees.
The results of IgG immunofluorescence showed that there was no deposition of immune complex in the glomerulus of control rats, and the fluorescence intensity was negative. In the model group, glomerular IgG was diffusely distributed along with glomerular capillary loops and part of mesangial areas, and the fluorescence intensity was strongly positive. Compared with the model group, IgG in the benazepril group and SAL treatment groups was distributed in capillary loops and mesangial region in fine granular form, and immunofluorescence deposition of IgG was weakly positive in the benazepril group and SAL treatment groups, while it was lighter compared with the model group to varying degrees.
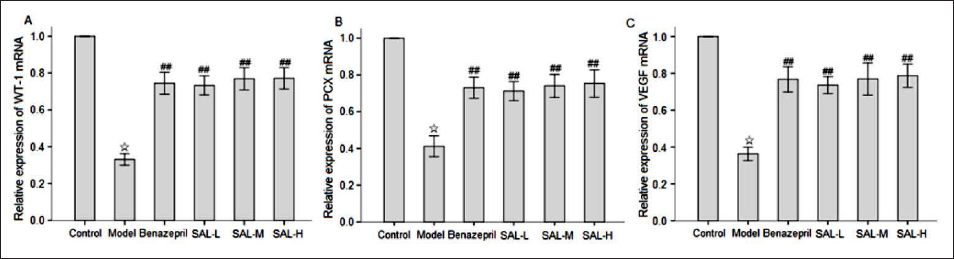
SAL Treatment and Gene Expressions of WT-1, PCX, and VEGF in the Renal Tissues of MN Rats
Both WT-1 and PCX are specific markers expressed in mature glomerular podocytes, which play a vital role in maintaining the morphology, structure, and filtration barrier of podocytes. VEGF can promote the growth, differentiation, and proliferation of glomerular podocytes, and it plays a crucial role in maintaining the structure of glomerular capillaries and the integrity of the filtration barrier. In normal renal tissue, VEGF is mainly expressed in podocytes, and only a small amount is expressed in distal renal tubular epithelial cells and collecting duct (CD) epithelial cells. To observe the effects of SAL on marker proteins such as WT-1, PCX, and VEGF in the renal tissue of MN rats, immunohistochemistry and real-time PCR were used to detect the above indicators.
Figure 4 shows that large amounts of WT-1, PCX, and VEGF proteins were expressed in the glomerular podocytes of the control group, which were distributed in a continuous linear pattern along the capillary loop. A small amount of VEGF was expressed in the distal renal tubular epithelial cells. Small amounts of WT-1, PCX, and VEGF proteins were found in the glomerular podocytes of the model group, the staining intensity was weak, and the difference was statistically significant compared with the control group (p < 0.01). The expressions of WT-1, PCX, and VEGF in the benazepril group and SAL treatment groups were scattered along the glomerular capillary loop, and the staining intensity was significantly increased compared with the model group (p < 0.05). Although the expressions of WT-1, PCX, and VEGF in the benazepril group and SAL treatment groups were different, there was no statistical significance (p > 0.05).
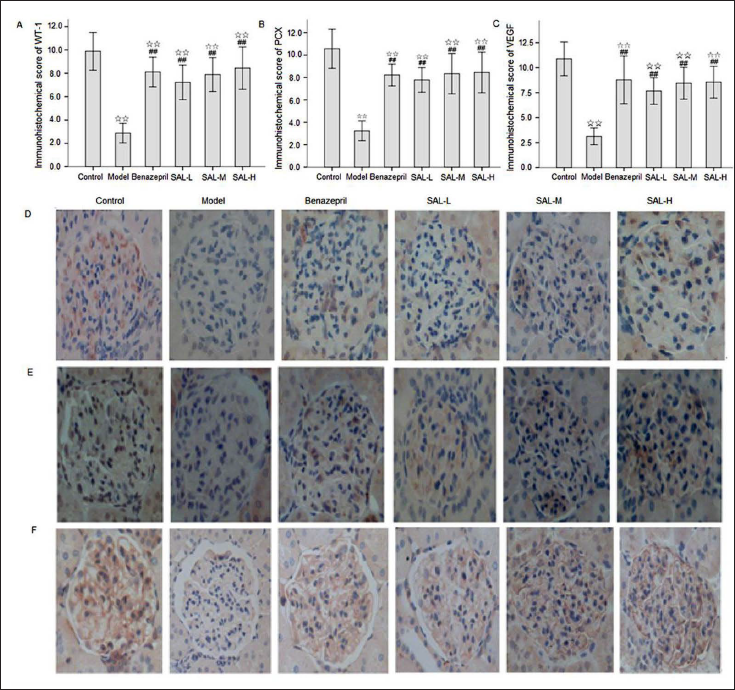
Figure 5 shows that compared with the control group, the expressions of WT-1, PCX, and VEGF at the mRNA level in the model group were significantly decreased (p < 0.05). Compared with the model group, the expressions of WT-1, PCX, and VEGF at the mRNA level were significantly increased in the benazepril group and SAL treatment groups (p < 0.01), while there was no statistically significant difference in the expressions of WT-1, PCX, and VEGF at the mRNA level between the benazepril group and SAL treatment groups (p > 0.05).
Discussion
MN is a common pathological type of chronic glomerulonephritis. The main pathogenesis is that the antigen-antibody immune complex activates complement, leading to the formation of the membrane attack complex C5b9 to induce podocyte injury, filtration barrier damage, and proteinuria. Therefore, the urinary protein levels reflect the degree of cell damage, which can be used as an important index to evaluate the treatment effect of MN. With the increase of urinary protein, the macromolecular protein in the blood is gradually decreased, forming hypoproteinemia. Meanwhile, water permeates the periphery, resulting in increased blood viscosity. This puts the blood in a state of high coagulation, which makes it easy to form thrombosis and emboli. The increase of compensatory synthetic protein and lipoprotein in the liver and the occurrence of hyperlipidemia, in combination with the application of hormones and diuretics in clinical treatment, can further aggravate the hypercoagulable state. The prevalence of MN has been rising annually in China, and it is frequently associated with hypercoagulability and hyperviscosity. Additionally, thrombosis, embolism, and other consequences are common in MN. Therefore, the creation of a workable treatment plan for MN is urgently required.
SAL is the main active ingredient extracted from the Traditional Chinese medicine Danshen. Modern pharmacological studies have confirmed that SAL has the effects of anticoagulation, anti-platelet aggregation, and improvement of microcirculation, and it has been widely used in the clinical treatment of chronic kidney diseases. It has been found that salvianolic acid can increase the activities of total superoxide dismutase (T-SOD), glutathione peroxidase (GPx), and catalase (CAT) and lower the levels of malondialdehyde (MDA), reactive oxygen species (ROS), and NADPH oxidase 4 (NoX-4) in 5/6 nephrectomized rats. It can enhance the activation of the Akt/GSK-3β/Nrf2 signaling pathway, inhibit NF-κB signaling pathway transduction, and achieve renal protection by reducing anti-inflammation, anti-oxidation, and kidney damage (Zhang et al., 2019). In a meta-analysis consisting of 12 randomized controlled trials, SAL is found to protect kidney function, inhibit inflammation, and fight oxidation by affecting the levels of Scr, BUN, 24-h UTP, hypersensitive C-reactive protein, and interleukin-6, thus improving the clinical efficacy of patients with diabetic nephropathy (Shen et al., 2020). Another study on salvianolic acid in the rat model of minimal change disease has shown that salvianolic acid in combination with low-dose prednisone can reduce urinary proteins, improve blood biochemical indicators and renal function indexes, activate Nrf2/HO-1 and PPARγ/Angptl4 signal transduction, and ameliorate podocyte injury, alleviating renal pathological damage in rats with minimal change disease (Wang et al., 2019). Studies have shown that salvianolic acid can improve renal dysfunction and reduce the expression of transdifferentiation-related proteins, including fibronectin (FN), α smooth muscle actin (α-SMA), and transforming growth factor β (TGF-β), in rats with renal fibrosis. In addition, SIRT1-mediated autophagy can also be activated to weaken epithelial-mesenchymal transdifferentiation in renal fibrosis and delay the process of renal fibrosis (He et al., 2020). However, the application of SAL in MN is still rare.
As a zinc finger-like transcription factor, WT-1 plays a vital role in the development and maturation of the kidney. It is mainly expressed in the nucleus of mature glomerular podocytes but not in the mesangial region, tubule region, or interstitial region, and it is one of the podocyte-specific molecular proteins (Chen et al., 2020; Husain et al., 2018). Studies have found that, compared with normal rats, the expression of WT-1 in glomerular podocytes in nephropathic rats is decreased, which is positively correlated with the decrease of podocyte number and podocyte apoptosis, showing important reference significance for predicting the degree of glomerular injury and glomerular sclerosis (Schumacher et al., 2011). PCX is a negatively charged hiatus membrane protein in the apical membrane area of glomerular podocytes, which plays a fundamental role in maintaining glomerular filtration through the membrane charge barrier, protecting glomerular structure stability, and alleviating glomerular injury (Lin et al., 2015). Studies have found that the urinary protein level in nephropathic patients is correlated with the number of podocytes and the amount of podocyte marker protein PCX, and the higher the 24-h UTP in patients, the more podocytes are shed in urine, and the higher the content of PCX in urine (Sanchez-Niño et al., 2019; Wang et al., 2010). VEGF can promote the growth, differentiation, and proliferation of glomerular podocytes, and it plays a crucial role in maintaining the structure of glomerular capillaries and the integrity of the filtration barrier. In the normal kidney, VEGF is mainly expressed in podocytes, and a small amount is expressed in distal renal tubule epithelial cells and CD epithelial cells. In glomerular diseases, VEGF mainly promotes the recovery of glomerular diseases by stimulating angiogenesis, vascular reconstruction, and vascular repair (Tanaka et al., 2015; Xu et al., 2021; Yang et al., 2021).
In recent years, the main animal models of MN have included the PMN model, the C-BSA rat model, the DPP Ⅳ rat model, anti-renal tubular antigen mouse model, and the aminopeptidase A mouse model. The C-BSA rat model of glomerular subepithelial immune complex deposition, diffuse heterogeneous basement membrane thickening, and “pin process” generation is the rat model that most closely resembles the pathological manifestations of human MN, so we chose this model. Benazepril, as a commonly used drug in the treatment of MN, has the effect of reducing urinary protein, so it is often used as a control drug in many clinical or experimental studies. In the present study, the renal tissue of rats in the model group showed obvious pathological damage, such as diffuse and uneven thickening of the glomerular basement membrane, deposition of subepithelial immune complex accompanied by “spike” formation, diffuse and strip deposition of IgG in basement membrane area, an increase of blood Fib, D-D, TC, and TG, and a decrease of TP and ALB. The expressions of WT-1, PCX, and VEGF in renal tissues were decreased, which was consistent with the quantitative proteinuria and pathological changes in renal tissues, suggesting that the deficiency of WT-1, PCX, and VEGF was involved in the podocellular injury process of MN. These lesions were reduced with benazepril treatment. Compared with the model group, SAL treatment groups showed alleviation of kidney tissue injury in MN model rats to a certain extent; the contents of Fib, D-D, TC, and TG were decreased; and the contents of TP and ALB were increased to varying degrees. Moreover, SAL treatment groups also exhibited increased levels of WT-1, PCX, and VEGF in the kidney tissue of MN rats to varying degrees. These results suggested that the protective effect of SAL on the kidney was related to improved hypercoagulability, alleviated podocyte injury, maintenance of the normal podocyte structure, and the integrity of the glomerular filtration barrier. The effect of SAL showed a dose-dependent manner. However, when the dose was increased to a certain level, the effect remained unchanged. In addition, there was no significant difference in BUN and Scr between different groups, which may be related to the short course of disease and mild pathological damage.
Conclusion
Our results provided important experimental evidence, supporting that MN could be treated by reducing proteinuria, increasing serum protein, and alleviating podocyte and renal pathological damage. These effects could inhibit hyperco-agulability; upregulate the expressions of WT-1, PCX, and VEGF in the renal tissue; repair podocyte injury; and maintain the integrity of the glomerular filtration barrier. Moreover, these results indicated that SAL was a promising drug for the treatment of MN. However, further clinical trials are still required to confirm these findings.
Footnotes
Summary
Our results provided important experimental evidence, supporting that MN could be treated by reducing proteinuria, increasing serum protein, and alleviating podocyte and renal pathological damages. These effects could inhibit hypercoagulability, upregulate the expressions of WT-1, PCX, and VEGF in the renal tissue, repair podocyte injury, and maintain the integrity of the glomerular filtration barrier. Moreover, these results indicated that SAL was a promising drug for the treatment of MN. However, further clinical trials are still required to confirm these findings.
Abbreviations
MN: Membranous nephropathy; SAL: Salvianolate; PCX: Podocalyxin; VEGF: Vascular endothelial growth factor; C-BSA: Cationized BSA; Fib: Fibrinogen; D-D: D dimer; TG: Triglyceride; TC: Total cholesterol; TP: Total protein; ALB: Albumin; BUN: Blood urea nitrogen; Scr: Serum creatinine; TEM: Transmission electron microscopy; CD: Collecting duct; T-SOD: Total superoxide dismutase; GPx: Glutathione peroxidase; CAT: Catalase; MDA: Malondialdehyde; ROS: Reactive oxygen species; FN: Fibronectin; α-SMA: α smooth muscle actin; TGF-β: Transforming growth factor β; WT-1, Wilms’ tumor protein 1; NoX-4: NADPH oxidase 4.
Authors’ Contributions
Wenjun Chen and Jinchuan Tan participated in the research design. Wenjun Chen, Suzhi Chen, Fenngwen Yang, Huiming Yan, and Huijie Zhou performed the experiments, analyzed the data, and were the major contributors in writing the manuscript. Fengwen Yang provided technical guidance and participated in data acquisition and analysis. Wenjun Chen and Jinchuan Tan confirmed the authenticity of all the raw data. All the authors read and approved the final version of the manuscript.
Availability of Data and Materials
The data generated in the present study may be requested from the corresponding author.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was funded by grants from the Hebei Natural Science Foundation Project (Nos. H2019423037 and H2020423049) and the Scientific Research Project of the Hebei Administration of Tradition Chinese Medicine (Nos. 2020337 and 2020065).
Statement of Informed Consent and Ethical Approval
The animal-related protocols complied with the guidelines of the Animal Ethics Committee of Hebei Medical University (No. DWLL2019047). All efforts were made to minimize the suffering of all the animals.
