Abstract
C4d is a byproduct of the activation of the classic and lectin complement pathways. Being routinely used as a marker for antibody-mediated rejection, the significance of C4d in native kidney disease is currently being widely studied. We evaluated glomerular and extraglomerular C4d staining in 82 biopsies of proliferative and nonproliferative glomerulonephritis diagnosed in our institution. The staining pattern of C4d was tabulated in various glomerular diseases. All biopsies of membranous nephropathy including membranous lupus nephritis (Class V) and immune complex-mediated membranoproliferative glomerulonephritis (MPGN) consistently showed C4d deposits along glomerular basement membrane mirroring the location of immunoglobulin and complement in these conditions. Conversely, other glomerular diseases like IgA nephropathy, postinfectious glomerulonephritis, focal segmental glomerulosclerosis, minimal change disease, and diabetic nephropathy showed variable mesangial and capillary wall C4d deposits. To summarize, the consistent pattern of C4d staining in membranous nephropathy (primary and secondary)and immune complex-mediated MPGN can be used as a valuable adjunct tool in establishing the diagnosis, especially when immunofluorescence findings are limited by inadequate sampling.C4d reactivity in other glomerular diseases are variable and may not aid as a diagnostic tool in renal biopsy evaluation.
Introduction
Assessment of complement abnormalities in glomerular diseases is emerging as a valuable tool for deriving insights into the underlying pathogenesis. Accessibility for C4d immunostaining as an adjunct in diagnosing antibody-mediated rejection in graft renal biopsies has aided its use in studying classical and lectin pathway complement activation in glomerulonephritis. C4d, a byproduct of C4 activation has the propensity to bind to the cell surface because of its thioester bond serving as a marker of complement activation. Detection of C4d deposition by either immunofluorescence (IF) or immunohistochemistry (IHC) techniques can be employed in demonstrating evident/masked antigen–antibody complex deposits in native kidney diseases.
C4d deposits in immune complex glomerulonephritis like membranous nephropathy (MN), IgA nephropathy (IgAN), immune complex-mediated membranoproliferative glomerulonephritis (MPGN), lupus nephritis (LN) and C3 glomerulopathy (C3G) have been studied and found to correlate with presence/absence of antigen–antibody complex deposition in glomerular capillaries to a variable extent.1-4 In addition, C4d IHC can be used as an indirect marker for the demonstration of immune complex deposition in these conditions when fresh tissue for IF is unavailable.
The pattern of variable C4d staining in glomerular diseases like MN, IgAN, focal segmental glomerulosclerosis (FSGS), minimal change disease (MCD), thin basement membrane disease, and thrombotic microangiopathy (TMA) has been elucidated in recent studies. Demonstration of C4d deposits in glomerular lesions in podocytopathies aided in understanding immune-mediated damage by complement activation in absence of evident antigen–antibody complex. Extraglomerular staining in peritubular capillaries (PTCs) and arterioles has been recorded especially in LN and hemolytic uremic syndrome (HUS).5-7 In combination with C1q, C4d stain can help in delineating various complement pathway activation and potentially assist in the prognostication of various renal diseases which can influence treatment outcomes. 8
With this knowledge of C4d staining patterns in various glomerular diseases, our group wanted to assess the utility of C4d as an ancillary technique in addition to routine light microscopy, IF studies, and ultrastructural evaluation in our tertiary care center.
Methods
Consecutive renal biopsies spanning from 2014 to 2020 received in our institution were reviewed for undertaking this descriptive study. Only biopsies with complete IF profiles, including IgG, IgA, IgM, C3, C1q, kappa, lambda, and electron microscopy findings were included in this study. The glomerular histology was classified as a proliferative and nonproliferative pattern of glomerular injury in correlation with IF findings.
C4d Immunohistochemistry:
Formalin-fixed paraffin-embedded tissue sections were stained using the mouse anti-human C4d monoclonal antibody (clone: C4D204; Abcam)by the immuno-peroxidase method using Ventana BenchMark system. 3-μm-thick sections were deparaffinized followed by heat-induced antigen retrieval in citrate buffer (pH 6) for 20 min. Primary antibody C4d was applied for 1 h at a dilution of 1:100 followed by DAB-based detection. Slides were counterstained with hematoxylin and mounted with DPX.
Glomerular C4d staining intensity was scored semiquantitatively on a scale of 0 to 3 + as 0 (negative), 1+, 2+, and 3 + . The variable staining pattern among glomeruli was interpreted as segmental/global and focal/diffuse similar to glomerular lesions. The location of C4d staining is mapped out based on location in the capillary wall, mesangium, tubular basement membrane, and PTCs. The character of the capillary wall C4d stain was labeled as either a continuous or irregular granular pattern. The evaluation was performed by 2 renal pathologists and a consensus was reached.
Results
The results of glomerular C4d staining in 82 biopsies of proliferative and nonproliferative glomerular diseases are tabulated in Tables 1 and 2. One biopsy of graft recurrent glomerular disease included was FSGS, NOS.
Nonproliferative Glomerular Diseases

Proliferative Glomerular Diseases
Among nonproliferative glomerulopathy, all 29 biopsies of primary (phospholipase A2 receptor [PLA2R]+, n = 24) (Figure 1A) and secondary MN (PLA2R, n = 5) (Figure 1B) showed consistently diffuse continuous granular C4d deposits in capillary wall. Of the primary MN biopsies, under electron microscopy 4 biopsies showed stage I, 6 biopsies showed stage II, 10 biopsies showed stage III and 4 biopsies showed stage IV deposits.
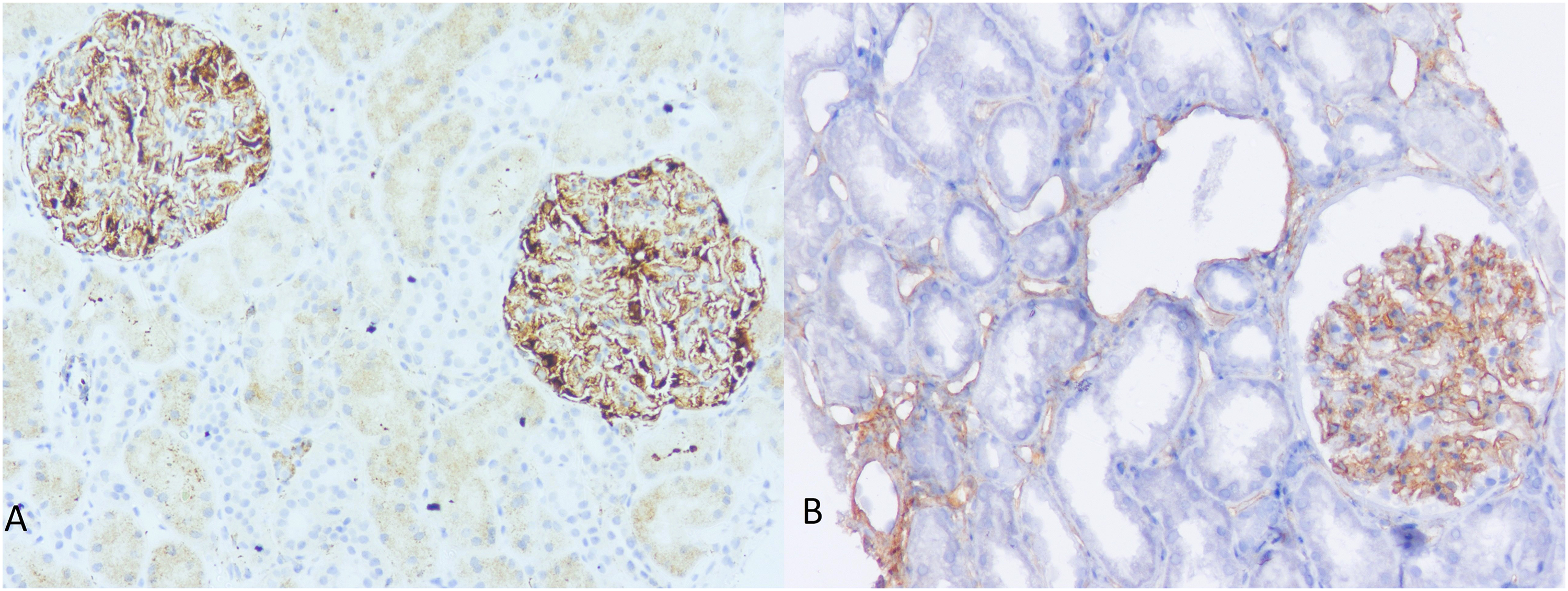
C4d staining pattern in primary and secondary membranous nephropathy. (A) Antiphospholipase A2 receptor (PLA2R) antibody-positive membranous nephropathy with diffuse and global continuous C4d deposits along the capillary wall (200×). (B) Membranous lupus nephritis with continuous C4d deposits along GBM and extraglomerular uptake in peritubular capillaries (200×).
Of 5 MCD biopsies, none of them showed any C4d deposits along the capillary wall. Three of them showed nonspecific podocyte uptake and one of them with mesangial C4d deposits (Figure 2A). Three biopsies of diabetic nephropathy were included, all of them showing the absence to trace mesangial C4d deposits.
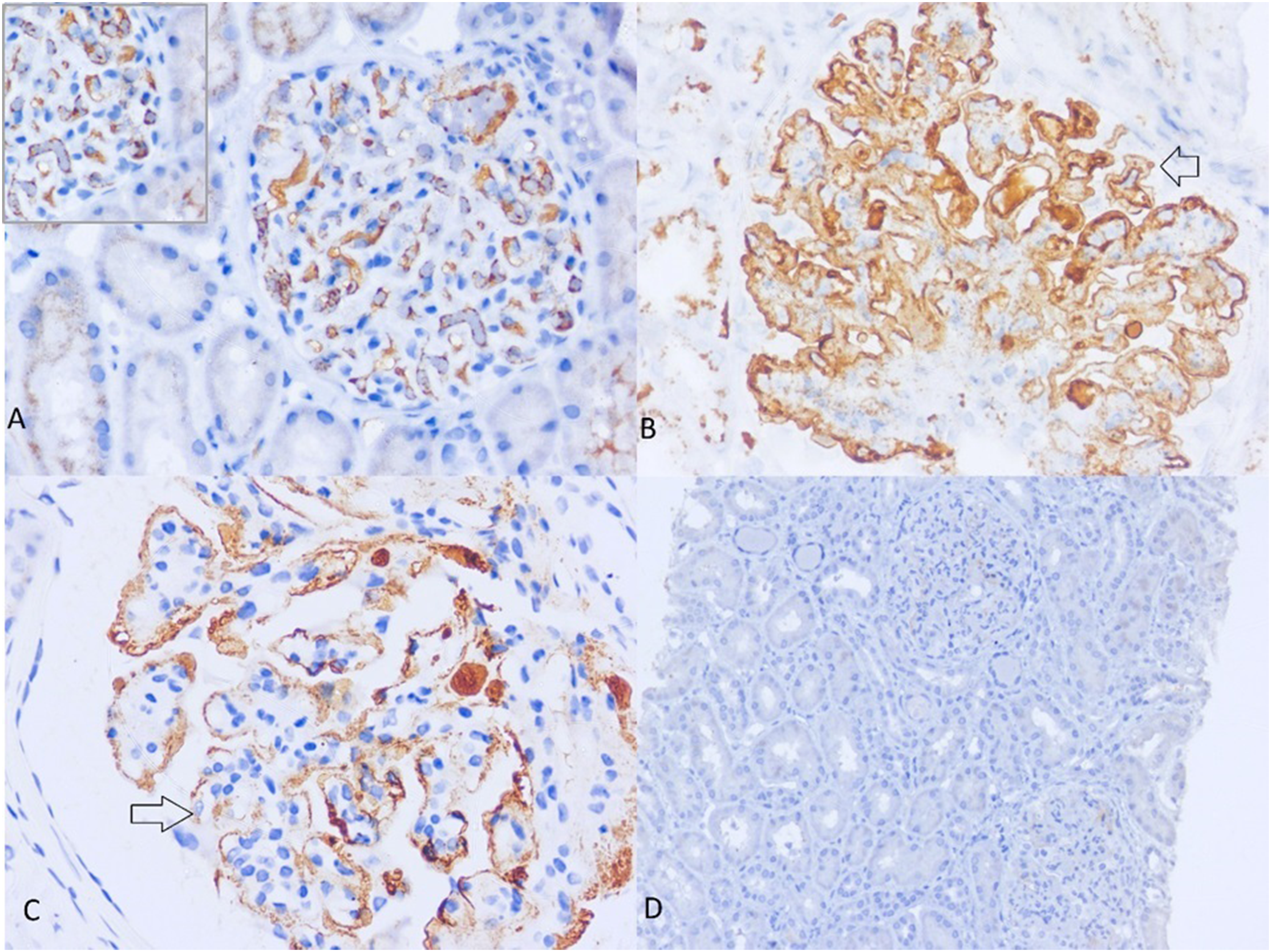
C4d staining pattern in glomerular diseases. (A) A biopsy of minimal change disease with distinct mesangial cell and podocyte uptake of C4d (400×). There are no C4d deposits in the capillary wall. (B) Immune complex-mediated membranoproliferative glomerulonephritis (MPGN) with continuous granular C4d deposits in the subendothelial and subepithelial location of the capillary wall (400×)similar to the “tram track” like appearance seen in silver methenamine stain. (C) and (D) Variable C4d reactivity in C3 glomerulopathy. (C) In one biopsy, which displayed a membranoproliferative pattern of injury with endocapillary hypercellularity in light microscopy, there is an irregular deposition of C4d (arrow) in the capillary wall (600×). (D) Another biopsy with mesangial proliferation was negative for C4d (100×).
One patient of glomerular TMA showed focal segmental capillary wall c4d deposits, consistent with the evidence of widespread complement activation along renal vasculature in HUS. 7
Among proliferative glomerular diseases, all 4 biopsies of immune complex-mediated MPGN (IgG and C3) showed diffuse continuous capillary wall granular deposits of C4d in subendothelial and subepithelial locations distinctively highlighting the “tram track” like appearance (Figure 2B). C3G (n = 3) biopsies showed variable C4d deposits, 2 of them with absent glomerular deposits (Figure 2D). Of them, 1 showed mesangioproliferative and the other showed membranoproliferative injury. One patient showed irregular deposits along GBM which displayed a membranoproliferative pattern of injury with endocapillary hypercellularity (Figure 2C).
Of 4 biopsies of PIGN with a diffuse proliferative pattern of glomerular injury, none of them showed any evidence of C4d deposits, despite evident immune complex deposits in IF. All 3 biopsies recovered subsequently in 6 to 8 weeks with normal C3 and C4 levels. Eight biopsies of IgAN included showed variable mesangial and segmental capillary wall granular C4d stain.
Twenty 4 biopsies of LN were included in the study, of which 4 biopsies were class III LN, 10 biopsies were class IV LN and 4 biopsies were class V LN. Six biopsies had a combination of classes (Class III/IV + V LN). Segmental/focal to global/diffuse irregular granular GBM and mesangial C4d deposits correlated with the location of immune complex reactivity in Class III/ IV LN. Biopsies with Class V and class III/IV + V lesions had global and diffuse continuous granular GBM deposits similar to MN biopsies. Eight of them showed extraglomerular C4d staining patterns in PTC (Figure 1B) and tubular basement membrane.
One patient with proliferative glomerulonephritis with monoclonal IgG deposits showed diffuse strong irregular C4d deposits along GBM.
A subset of 6 biopsies included in the study had insufficient glomerular sampling for IF studies. Of them, 1 patient showed capillary wall C4d stain assisted in labeling as MN given the diffuse continuous granular pattern as described earlier, in combination with “spike” and “crater” lesions in silver methenamine stain. A patient of crescentic glomerulonephritis with focal, continuous capillary wall C4d deposits (suggests immune complex etiology) and another patient of Diabetic nephropathy with diffuse continuous capillary wall C4d deposits raised the possibility of underlying immune complex disease (MN in the latter patient). IF on paraffin sections performed in the above 3 biopsies confirmed the presence of immune complex deposits. The remaining 3 biopsies had focal staining with variable intensity in the mesangium and the capillary wall.
Discussion
As proposed by Sethi et al 1 in 2015, C4d glomerular staining is a marker of classical/lectin pathway activation associated with immune complex glomerulonephritis. The classical pathway of complement is activated by antigen–antibody complex followed by cleavage of C4 domains into C4a and C4b. C4b is then cleaved in the presence of C4b binding proteins into C4c and C4d. Thus, the deposition of C4d is regarded as an indirect sign of complement activation.
C4d deposition in PTCs is used as a diagnostic marker of antibody-mediated rejection. Owing to the ease of performing IHC, C4d is increasingly being used as an ancillary technique in demonstrating evident and masked immune complex deposits in native glomerular diseases. However, it can be used in resource-scarce centers with a lack of IF to delineate the primary etiology of glomerular disease. We observed the location, pattern, intensity, and character of C4d deposition in glomerular diseases with proliferative and nonproliferative glomerular morphology diagnosed in our center.
C4d positivity in MN is well-documented in various studies due to complement activation and C5b-9 deposition. C4d deposits were localized in GBM with a granular appearance similar to our study. We observed that the GBM staining pattern in all our biopsies, including those with early changes (stage I) under electron microscopy were continuously granular, unlike the pattern in proliferative glomerulonephritis. The secondary MN biopsies did not show any mesangial deposits of C4d although there was immune complex deposition in IF.
C4d staining in glomerular diseases like MCD, FSGS, and diabetic nephropathy was variable. Hence, cautious interpretation of C4d staining in mesangium is recommended in nonproliferative glomerular diseases. In addition, C4d deposition can precede the development of FSGS. 9 Although more studies are needed to document them to speculate on the pathogenesis, they are likely to be due to nonspecific entrapment.
All our biopsies of immune complex-mediated MPGN showed a continuous granular pattern of c4d deposits along GBM in subendothelial and subepithelial locations while C3G showed variable C4d deposits. C3G studies by Sethi et al and Bashir et al 10 showed a majority of them being C4d negative, and C4d reactivity in our C3G biopsies is in concordance with the possibility of precipitation of alternate pathway abnormalities by infections and lectin pathway activation. Thus, C4d deposition in MPGN does not exclude the possibility of C3G. 11 In addition, C4d was negative in all our PIGN biopsies which implies that it may not help differentiate early PIGN versus C3G as well, especially in children. 12 These findings are similar to other C4d studies on PIGN. 13
C4d deposition in nonsclerosed glomeruli and kidney disease progression in IgAN were evaluated extensively in recent literature.14-16 Mesangial and arteriolar deposits of C4d have been described in IgAN.14,15 None of our biopsies showed any arteriolar deposits. IgAN mesangial C4d deposits are considered one of the earliest prognostic variables for patients with idiopathic IgAN and normal kidney function. This deposit pattern is associated with increased and persistent disease activity and worse kidney outcomes. 14 However, the significance of segmental GBM positivity in our biopsies is unknown.
C4d deposits in LN biopsies paralleled the location of antigen–antibody as first demonstrated by Kim and Jeong et al. However, the extent of C4d reactivity did not correlate with disease activity. The pattern of C4d deposits was continuous granular in class V LN in contrast with irregular granular deposits in class III/IV LN. This is in alignment with our observation that when endocapillary hypercellularity is associated with immune complex-mediated glomerular diseases, the pattern of c4d deposition was irregular. Two biopsies showed the presence of C4d deposits in PTCs similar to observations by Li et al. 17 Although our study sample size is small, we propose this pattern of continuous granular reactivity in class V LN may be used to exclude its possibility, especially when light microscopic findings are inconclusive for early membranous LN.
To conclude, very few studies assess C4d staining patterns and character critically in various proliferative and nonproliferative glomerular diseases. The specific and consistent continuous granular GBM staining pattern of C4d in our biopsies of MN (including membranous LN) and immune complex-mediated MPGN can be used as a valuable adjunct tool in establishing the diagnosis, especially when IF findings are limited by inadequate sampling. In contrast to the early promise of C4d stain in the evaluation of C3G and PIGN, C4d reactivity in these glomerular diseases are variable and may not aid as a diagnostic tool in renal biopsy evaluation.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
