Abstract
Background
Cisplatin, the first-line drug for chemotherapy, often has limited treatment efficacy because of resistance and cancer recurrence mechanisms. Tetrandrine is a unique secondary metabolite of Stephania tetrandra. As a traditional Chinese medicine agent, tetrandrine has been reported to have antioxidant, anti-inflammatory, antitumor, and antiangiogenesis activities and has been shown to inhibit the proliferation and angiogenesis of colorectal, lung, and breast cancer cells; potential mechanisms underlying its activities include the promotion of tumor cell apoptosis, promotion of cell cycle arrest, and intensification of reactive oxygen species (ROS) production.
Objectives
The main treatments for oral cancer are chemotherapy, surgery, and radiotherapy; these treatments are often used in combination. Cancer cells easily develop cisplatin resistance; therefore, we investigated tetrandrine’s potential as a therapy for overcoming resistance to oral cancer drugs.
Materials and Methods
We used the cisplatin-resistant oral cancer CAR cell line (CAL27) as a research objected and applied inhibitor treatment to clarify the role of tetrandrine in cell death and mitochondrial dysfunction.
Results
Tetrandrine could effectively inhibit CAR cell proliferation and induce apoptosis, with a corresponding increase in ROS production in mitochondria. Moreover, tetrandrine increased caspase-9 and caspase-3 activity in CAR cells and induced apoptotic mRNA, caspase-3/-9, AIF, and Endo G overexpression. Our results indicate that tetrandrine induces apoptosis in CAR cells through a mitochondrial-dependent signaling pathway.
Introduction
In Taiwan, the incidence of oral cancer is increasing rapidly owing to increases in the number of people who smoke tobacco, consume alcohol, and chew betel nuts. Oral cancer sites include the upper and lower lips, buccal mucosa, tongue, upper and lower gums, retromolar triangle, the floor of the mouth, and hard palate.1–4 Currently, oral cancer treatment mainly involves surgical excision followed by chemotherapy and radiotherapy. Patients may experience side effects during treatment, such as pain, dysphagia, dry mouth, tissue fibrosis, and dysphagia. 5 Cisplatin and 5-fluorouracil (5-FU) are the main chemotherapy drugs employed for oral cancer treatment.6–8 Research has reported that the use of cetuximab (Erbitux) and other targeted drugs concomitantly with cisplatin + 5-FU treatment could improve the survival time of patients (cisplatin + 5-FU treatment alone: 4.4 months; combined treatment: 11 months).9, 10 Cetuximab is an epidermal growth factor receptor (EGFR) blocker. A substantial amount of EGFR is expressed on the membrane of oral cancer cells, and cetuximab blocks the EGFR transmission path, meaning that such cancer cells cannot grow and metastasize, thereby initiating cancer cell apoptosis and inhibiting cancer cell growth.11–13 Targeted drug therapy has been hampered by adverse reactions and drug resistance.14, 15 Compounds that inhibit other targets, such as AMP-activated protein kinase (AMPK) and AKT, and can inhibit the metastasis of oral cancer cells or induce autophagy or apoptosis are major goals in anticancer research and drug development.16–20
Cisplatin is a first-line chemotherapeutic for several cancers, but cisplatin resistance is a major cause of treatment failure and cancer recurrence. Cancer cells can become resistant to cisplatin through numerous mechanisms, such as a reduced ability of cancer cells to absorb cisplatin but an increased ability to excrete it, an increased metabolic capacity to detoxify cisplatin, an increased capacity to counteract cisplatin-induced oxidative stress and apoptosis, and an increased DNA repair capacity. 21 Many types of cancer cells are cisplatin-resistant, and thus, we hope to identify other treatment modalities to overcome this drug resistance.22–26
Tetrandrine isolated from Stephania tetrandra has antioxidant, antiaging, anti-cardiovascular, and antitumor properties. 27 The relationship between drug resistance and the autophagy mechanism of oral cancer is unexplored, and the roles of AMPK and AKT in the relationship between autophagy and drug resistance are unknown. Accordingly, we sought to identify targets that tetrandrine could act on, determine whether tetrandrine affects the apoptotic pathway of CAR cells, and reveal the transfer and message transmission paths of tetrandrine.
Materials and Methods
Materials
From Sigma-Aldrich (Merck KGaA, Darmstadt, Germany), we purchased tetrandrine, thiazolyl blue tetrazolium bromide (MTT), Dulbecco’s modified Eagle’s medium (DMEM), and an in situ Cell Death Detection Kit (fluorescein; Roche Diagnostics GmbH); unless indicated otherwise, other reagents and chemicals were purchased from the same supplier. Moreover, GeneTex (Hsinchu, Taiwan) was the source of all of the primary antibodies as well as the antimouse and antirabbit immunoglobulin G horseradish peroxidase–linked secondary antibodies employed in this study. Millipore (Merck KGaA, Darmstadt, Germany) supplied the Muse Caspase-3/-9 assay kits employed. In addition, from Molecular Probes (Thermo Fisher Scientific, Waltham, MA, USA), we procured 3,3’-dihexyloxacarbocyanine iodide (DiOC6(3)) and 2’,7’-dichlorodihydrofluorescein diacetate (H2DCFDA). Finally, HyClone (GE Healthcare Life Sciences; Logan, UT, USA) supplied L-glutamine, penicillin/streptomycin, trypsin-EDTA, and fetal bovine serum (FBS).
Cell Culture
From the American Type Culture Collection (Manassas, VA, USA), we obtained the CAR cell line (CAL27; cisplatin-resistant human oral cancer cells), which was established through the clonal selection of CAL27 by using 10 cycles of one passage treatment with 10–100 µM cisplatin followed by a recovery period of another passage.28, 29 The derived CAR cells were cultured in DMEM containing 100 U/mL penicillin, 100 µg/mL streptomycin, 10% FBS, 2 mM L-glutamine, and 100 µM cisplatin within a humidified atmosphere with 5% CO2 at 37°C; the medium employed for this culturing process was renewed at a frequency of 2–3 days. After cell staining using trypan blue, viable cells were counted, and the survival rate was calculated using a hemocytometer; the number of cells in the passage was controlled at 2–5 × 105 cells/mL. The cells were treated for 24 h with 0, 25, 50, 75, or 100 µM tetrandrine. Harvested cells were subsequently subjected to a cell viability test, a reactive oxygen species (ROS) production assay, a caspase-3/-9 assay, and a quantitative polymerase chain reaction (qPCR) analysis.
MTT Assay Execution for Viability Assessment
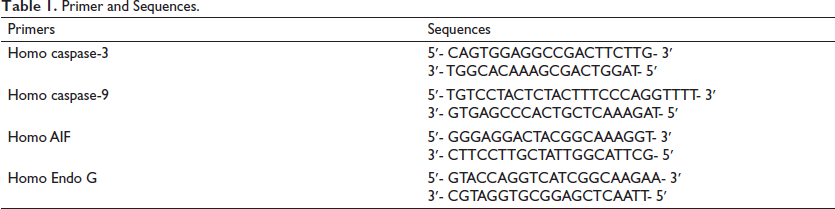
The CAR cells were plated in 96-well plates (2.5 × 104 cells per well) with and without exposure to tetrandrine at various concentrations (0, 25, 50, 75, and 100 µM) for 24 h to cause apoptosis; they were also pretreated with or without Z-VAD-FMK (10 mM) for 1 h, after which they were treated with MTT solution (0.5 mg/mL) for 2 h. Subsequently, we added dimethyl sulfoxide (100 µL) to the aforementioned wells for two reasons: culture medium replacement and formazan crystal dissolution. Optical density measurement was conducted at 570 nm through the use of a spectrophotometer.
Caspase-3/-9 Activity Assay
The CAR cells in six-well plates were exposed to tetrandrine at the following concentrations for 24 h: 0, 25, 50, 75, and 100 µM. After harvesting the cell lysates, we incubated the supernatants with the relevant reaction buffer by following the manufacturer’s protocols. Subsequently, a phase-contrast microscope was employed to visualize and photograph the cells.
Determination of Mitochondrial Electrical Potential and ROS Through Flow Cytometry
The CAR cells were exposed to tetrandrine at diverse concentrations (0, 25, 50, 75, and 100 µM) for 48 h. The cells were subsequently harvested, after which they were stained with 500 nM DiOC6(3) or H2DCF-DA for 30 min at 37°C and then examined through the use of flow cytometry, as previously described. 30
Apoptotic mRNA Level Analysis
The derived cells (1 × 106 cells/total) were exposed for 24 h with or without 50 and 75 µM tetrandrine. The assay was executed in accordance with the protocol for the use of the Qiagen RNeasy Mini Kit, as described previously.31, 32 RNA samples were processed with kit reagent for 30 min at 42°C in accordance with the instructions provided by the manufacturer (Applied Biosystems, Foster City, CA, USA). The following protocol was used in the subsequent qPCR: 2 min at 50°C, 10 min at 95°C, 40 15-s cycles at 95°C, and 1 min at 60°C with 1 µL of complementary reverse-transcribed DNA, 2X SYBR Green PCR Master Mix (Applied Biosystems), and forward and reverse primers (200 nM; Table 1). We conducted every assay in triplicate using an Applied Biosystems 7300 real-time PCR system, and we employed the 2–∆∆Ct method for the calculation of mRNA fold changes.
Primer and Sequences.
Statistical Analysis
All experiments were performed in triplicate, and relevant data are presented as mean ± standard deviation. All statistical analyses involved a one-way analysis of variance, and p values < 0.05 were considered as statistical significance.
Results
Tetrandrine Lowers CAR Cell Viability
The survival rates (according to MTT detection) of CAR cells treated with tetrandrine were significantly higher (p < 0.05) than those of untreated cells, highlighting the high growth-inhibitory activity of tetrandrine.
Effects of Tetrandrine on Mitochondrial Electrical Potential and ROS in CAR Cells
We used H2-DCFDA to detect increased ROS in drug-resistant pancreatic cancer (CAR) cells following tetrandrine treatment; the results indicated that tetrandrine induced a dose-dependent increase in ROS.
Effect of Tetrandrine on Caspase-Dependent Apoptosis in CAR Cells
An analysis of the influence of tetrandrine on the caspase activity of the cells of interest indicated that tetrandrine activated caspase-3/-9.
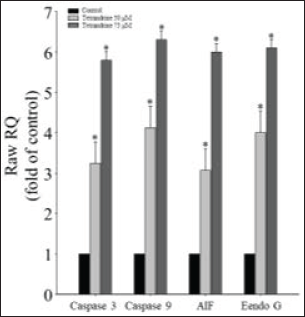
Effects of Tetrandrine on Apoptotic mRNA Expression in CAR Cells
Discussion
In line with the gradual improvement in extraction and separation technology, the active ingredients of traditional Chinese medicine have gradually begun to be used as effective antitumor and chemotherapy resistance–reversing drugs,33, 34 and these natural products afford clear reversal effects in cancer cells. 35 For example, when Ganoderma lucidum polysaccharide combined with cisplatin was used to treat ovarian cancer drug–resistant cells (SKOV 3/DDP), GST protein expression was downregulated, lessening cisplatin resistance. 36 Astragalus polysaccharides have favorable antitumor effects, reduce the toxicity and side effects of chemotherapy, improve immunity, and can increase the sensitivity of HeLa cells to cisplatin by regulating autophagy; they can also reverse A549/DDP cells’ resistance to cisplatin and exhibit sensitizing effects. 37
The pathways of apoptosis, otherwise known as programmed cell death, are either intrinsic or extrinsic. Intrinsic pathways depend on mitochondria-controlled signaling, and extrinsic pathways are dependent on death receptors that are expressed on the cell membrane. 38 The main molecules implicated in the pathway that involves mitochondrial-dependent intrinsic apoptosis are cytochrome c, Bax, cleaved caspase-3, Apaf-1, and cleaved caspase-9. 38 Disrupting the apoptotic process (a physiological process) engenders the uncontrolled growth of cells, consequently increasing the malignancy risk. For this reason, this process has been harnessed as an anticancer treatment; DNA damage with chemotherapy, with or without radiotherapy, is applied to cause the apoptosis of cancer cells. 39 Cisplatin’s anticancer action involves DNA lesion creation, a response to DNA damage, and mitochondrial apoptosis. 40 Our cell viability assessment results indicate that tetrandrine caused apoptosis, with the reduction in CAR cell viability being significantly dose-dependent (Figure 1). The expression of multi-drug-resistance-related proteins (MRPs) often causes tumors to develop drug resistance and lose their sensitivity to drugs; the actions of these MRPs are a key cause of chemotherapy failure. Some natural compounds widely found in plants inhibit tumor growth and reverse drug resistance mediated by MRPs. 41 Accordingly, natural compounds may be helpful for treating patients with cisplatin-resistant oral cavity squamous cell carcinoma. 28
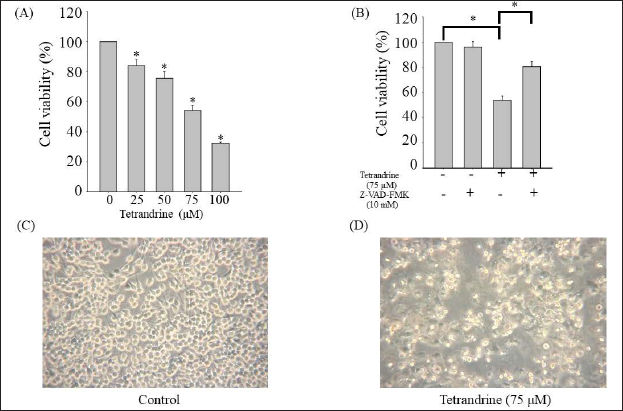
Our results demonstrate that tetrandrine reduced mitochondrial membrane potential (∇Ψm), which induced substantial ROS production (Figure 2). The manifestation of mitochondrial outer membrane permeabilization (MOMP) varies substantially; moreover, following mitochondrial permeabilization, considerable caspase activation takes place, often culminating in programmed cell death in a short space of time (usually a matter of minutes).42, 43 Cytochrome c is the most crucial mitochondrial intermembrane protein released following MOMP. After cytochrome c reaches the cytoplasm, it binds transiently to Apaf-1, a crucial caspase linker molecule. This culminates in major conformational alterations to Apaf-1; it oligomerizes into a heptamer wheel and exposes the caspase activation and recruitment domain (CARD). 44 Our results indicate that induced overexpression of caspase-3/-9, which are apoptotic proteins, induced mitochondrial dysfunction (Figure 3). The Apaf-1 CARD domain binds to the CARD domain of the initiator caspase procaspase-9 to form apoptotic bodies. In apoptotic bodies, caspase-9 dimerization culminates in its activation; this action leads to the cleaving and activation of executioner caspase-3 and caspase-7, resulting in rapid cell death. The presence of cytochrome c is vital for the mitochondria-dependent activation of caspases. 45
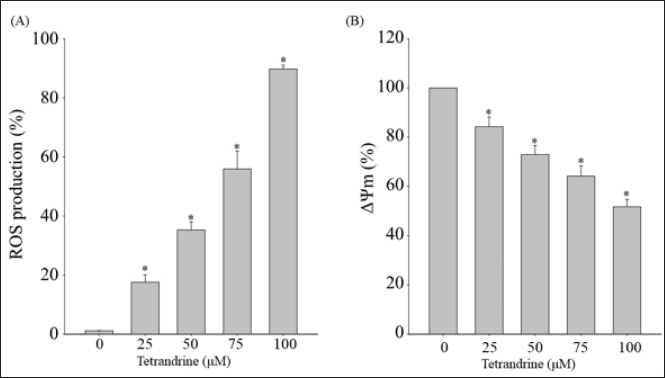
We revealed that tetrandrine causes cell apoptosis. CAR cells treated with tetrandrine had significantly elevated levels of caspase-3 and caspase-9. Increased caspase-3/-9 activity results in mitochondria being dependent on the apoptosis pathway and on caspase-3 and caspase-9 for morphological alterations; the subsequent increase in ROS production is suggestive of mitochondrial dysfunction, which results in inadequacies in energy reserves and intracellular signaling pathway activation. We found that tetrandrine can induce CAR cell apoptosis through mitochondria-dependent signaling pathways, which, along with tetrandrine, increase ROS production to reduce mitochondrial membrane potential. Tetrandrine stimulated the CAR cell-related upregulation of caspase-3, caspase-9, caspase-3/-9, AIF, and EndoG mRNA expression, indicating that tetrandrine induces apoptosis (Figure 4). We detected that tetrandrine affected the apoptosis of CAR cells, and we evaluated this effect. Our study findings indicate that tetrandrine has potential use for enhancing gemcitabine-induced apoptosis in CAR cells. In mitochondrial-dependent cell death, the changes in caspase-3 and caspase-9 are the key points that indicate the reaction of autophagy or apoptosis. In addition, the situation of ROS raising could trigger a common event upstream of the inflammasome leading to apoptosis or autophagy.45, 46
Conclusion
We revealed that tetrandrine resulted in a significant dose-dependent reduction in CAR cell viability. Increased caspase-3/-9 activity results in mitochondria being dependent on caspase-3, caspase-9, and apoptosis pathways for morphological alterations; the subsequent increase in ROS production is suggestive of mitochondrial dysfunction, which results in inadequacies in energy reserves and the activation of intracellular signaling pathways.
Summary
This study demonstrated that tetrandrine induced the apoptosis of CAR cells through increasing ROS production in mitochondria. Moreover, colorimetric assays revealed that tetrandrine increased caspase-9 and caspase-3 activity in CAR cells, indicating that CAR cell apoptosis was induced via a mitochondrial-dependent signaling pathway. According to the malfunction of mitochondria activity, the Endo G and AIF intervene in apoptosis in the nucleus.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This study was supported by the Department of Medicine, Kaohsiung Armed Forces General Hospital. Funding for this study was provided under the Plan 106-05.
Statement of Informed Consent and Ethical Approval
Approval from the ethics committee is not required due to the use of cell lines.
