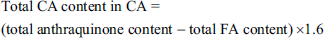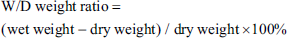Abstract
Background:
Leakage of the alveolar epithelial barrier can lead to pulmonary oedema, leading to organ dysfunction.
Objectives:
Combination anthraquinone (CA) and free anthraquinone (FA) were extracted from Rhubarb, and the prevention and therapeutic effects of Rhubarb anthraquinones (RA) on lipopolysaccharide (LPS)-induced lung injury were investigated.
Materials and Methods:
The CA and FA were extracted from Rhubarb by water solvent extraction and ethanol solvent extraction. The extracted RA content was determined using spectrophotometry and high-performance liquid chromatography (HPLC). A mouse model of inflammatory lung injury was established by LPS induction to study the mechanism of RA activity. Inflammatory factors were measured using an enzyme-linked immunosorbent assay. Changes in alveolar epithelial leakage were assessed using a permeability assay. Changes in lung injury were evaluated by histopathological and ultrastructural observations. Tight junction (TJ) markers and Ras homolog gene family member A (RhoA)/Rho-associated protein kinase 1 (ROCK1) pathway-related proteins were measured using immunohistochemistry and Western blotting. As an agonist of RhoA/Rock signalling, U46619 was used to further verify whether RA acts through this pathway.
Results:
RA could inhibit pulmonary inflammation by decreasing the level of inflammatory factors. The alveolar epithelial permeability was reduced, and the pathological injury of the lung tissue was alleviated after administration with RA. Furthermore, the expression of TJ proteins was up regulated and RhoA/ROCK1 signalling was inhibited in the presence of RA. The effects of RA on TJ proteins were partially reversed by U46619.
Conclusion:
RA effectively protects mice against inflammatory lung injury by enhancing alveolar epithelial TJ via RhoA/ROCK1 signalling.
Introduction
Acute lung injury (ALI) is a critical disease with high morbidity and fatality rates, placing a heavy burden on health systems. 1 Sepsis caused by inflammation has been shown to play an important role in the development of ALI. ALI mainly manifests as alveolar injury, uncontrolled inflammation and hypoxemia, which can progress to acute respiratory distress syndrome. It has been confirmed that alveolar epithelium hyper-permeability is one of the main mechanisms leading to the above syndromes. 2 Since there are currently no effective drugs to prevent or treat ALI, it is necessary to explore essential drugs from natural products.
Studies have shown that the alveolar epithelium acts as an abnormally dense barrier function. This barrier restricts the entry of some molecular substances into the airways. It regulates the diffusion barrier between cells. 3 Tight junction (TJ) proteins are a class of heteromeric protein complexes distributed between the lipid membranes of adjacent epithelial cells and are regulated by the alveolar epithelial TJ complex. 4 TJ proteins are occludin, claudins, some junctional adhesion molecules and zonula occludens (ZO). 5 Occludin combines with ZO-1 to form a crucial protein that forms the basis of the structure and is necessary to maintain the alveolar epithelial barrier integrity. 6 Claudin-5 is a major regulator of TJ permeability. 7 ZO-1 is the major scaffolding protein that stabilizes the TJ barrier through coupling to the prejunctional actin and myosin. 8 The Ras homolog gene family member A (RhoA)/Rho-associated protein kinase 1 (ROCK1) pathway plays an important role in mediating TJ dissociation, which can inhibit myosin II regulatory light chain (MLC) dissociation by activating the regulatory subunit 1 of MLC phosphatase target subunit 1 (MYPT1) phosphorylation, thereby disrupting TJ integrity upon actomyosin ring contraction. 9
Rhubarb, a Traditional Chinese medicine, has been proven to have anti-inflammatory properties.10, 11 One of the major active substances of Rhubarb is anthraquinone compounds, including a combination anthraquinone (CA) and free anthraquinone (FA). 12 Accumulating evidence indicated that Rhubarb anthraquinones (RA) compounds could prevent lung damage caused by inflammation. However, the potential mechanism remains unclear.13–15 In order to explore a therapeutic drug with fewer side effects and effective for the prevention and treatment of ALI, two main anthraquinone compounds were systematically extracted from Rhubarb, and the effects and mechanisms for relieving alveolar epithelial leakage of RA against inflammatory lung injury were comparatively investigated.
Materials and Methods
Extraction of RA
Rheum palmatum L. (R. acuminatum HOOK. F. and Thomson) was obtained from Li County, Gansu Province, and identified by a professor from the Pharmacy Department. The specimens were deposited in proprietary laboratories (Sample No. 20210016). Some appropriate improvements to the extraction details were made based on a previous study. 16 (1) CA: R. palmatum L. materials (2.4 kg) were crushed and extracted by reflux extraction with water thrice. The liquid medicine was then sieved, centrifuged and filtered. The filtrate was adsorbed by activated D101 macroporous resin (Donghong Chemical, China), followed by elution with pure water and 65% ethanol. The eluent was concentrated, and the pH was adjusted to 2.0, followed by filtration, concentration, decompression and drying. Finally, CA (7.7 g) was obtained. (2) FA: The precipitates produced during FA extraction were collected and extracted by reflux extraction with 8% hydrochloric acid−50% ethanol, followed by concentration. The concentrate was extracted with dichloromethane and concentrated. The concentrate was extracted with 5% sodium hydroxide. The pH of the extract was adjusted to 2.0, followed by elution and washing with water until the pH was neutral. After decompressing and drying, FA (17.5 g) was obtained.
Spectrophotometric Analysis
The emodin standard (Plant Biotech, Lanzhou, China) solution was diluted to yield different concentration solutions. The optical density (OD) value at 510 nm was measured by a UV-1900i ultraviolet–visible spectrophotometer (Shimadzu, Kyoto, Japan), and a calibration curve and regression equation were established. FA and CA samples were dissolved in methanol to determine the OD value via spectrophotometry at 510 nm. The total FA content in FA and CA was calculated using the regression equation. Based on the molecular mass ratio of emodin mono-oxygluconic acid and emodin of 1.6, the total CA content in CA was calculated. The total CA content in CA (calculated as emodin mono-oxyglucoside) was calculated based on a formula as follows:
High-Performance Liquid Chromatography (HPLC)
The experiments were detected using Dionex UltiMate 3000 HPLC (Thermo Fisher, MA, USA) with column oven, DAD detector, LPG-3400SD quaternary pump, a manual sampler with a sample loop and Shimadzu Intersil ODS C18. The parameters of the column were temperature 30°C, wavelength 254 nm, sensitivity 0.04 AUFS and injection volume 10 µL. Methanol was used as solvent A. About 0.1% phosphoric acid was used as solvent B. The flow rate of the mobile phase was 1.0 mL/min. The gradient elution program included 70%–80% A at 0 to 10 min and 80%–85% A at 10–30 min. The preparation of the aloe-emodin, rhein, emodin, chrysophanol and physcion standard (Plant Biotech) reference solution and test samples was the same as that of the emodin standard reference solution in spectrophotometric analysis. The peak area of the mixed standard solution was measured using HPLC. The regression equation was also established.
Ethics Approval
Healthy adult male and female BALB/c mice were obtained for this study. Mice weigh approximately 25–30 g. This project was approved by the Laboratory Animal Welfare Ethics Committee (certification No. D2021-262). International Guidelines were strictly followed throughout all handling procedures of laboratory animals. The mice were acclimatized in housing conditions under 24 ± 1°C, 45%–50% humidity. All mice were fed with standard food and water.
Animal Experiments
Mice (n = 72) were randomized into the control, lipopolysaccharide (LPS), FA and CA groups. The FA, CA and LPS doses were selected based on previous studies.17, 18 Gavage needle (12#) was used for drug delivery to mice. The FA and CA groups were intragastrically administered with 114 mg/kg FA and CA in a 0.5% sodium carboxymethyl cellulose (CMC-Na) suspension, respectively. The control and LPS groups were administered CMC-Na with equal volume in the same way. LPS (15 mg/kg) was intraperitoneally injected to induce the sepsis model in the LPS, FA and CA groups 12 h later. The same amount of sodium chloride injection was administered to the control group. At 2 h after injection, the second gavage was administered to the four groups. 1% pentobarbital sodium was used to euthanize mice 24 h after injection. The blood and lung samples were taken for subsequent testing. In addition, five mice were randomly selected from the LPS, FA and CA groups 30 min before anaesthesia and injected with Evans blue (EB) dye (40 mg/kg) via tail vein. The same amount of phosphate-buffered saline was administered to the control group. Lung tissues were taken for permeability testing. Furthermore, other mice (n = 8) were randomly divided into the agonist and agonist + FA groups for pathway agonist experiments (n = 4 in each group). U46619 (Santa Cruz, Dallas, USA) was injected via a tail vein 30 min before the LPS challenge. The injected dose (0.375 mM, dissolved in 200 µL of saline) was based on a previous study. 19
Enzyme-Linked Immunosorbent Assay (ELISA)
The blood sample was obtained from mice eyeballs. After centrifugation, the upper serum sample was obtained and stored. The tumour necrosis factor-α (TNF-α), interleukin (IL)-1β, IL-10 and procalcitonin (PCT) levels in serum samples were detected using ELISA kits (Mibio, Shanghai, China).
Lung Wet/Dry (W/D) Weight Ratios
To a certain extent, the W/D weight ratio can reflect lung permeability and pulmonary oedema. The left lung tissues were removed, washed and weighed immediately. After drying (60°C, 48 h), the lung tissue was weighed. The numerical calculation was performed as follow:
EB Permeability Assay
EB (Aladdin, Shanghai, China) solution (40 mg/kg) was administered through the tail vein 30 min before euthanasia. The lungs were taken out and placed in formamide (Rhawn, Shanghai, China) solution (20 mL/kg). After incubation (37°C, 48 h) and centrifugation (2000× g, 5 min), the supernatant was obtained. The OD was detected at 620 nm using a spectrophotometer (YuanXi, Shanghai, China).
Hematoxylin and Eosin (H&E)
The samples were fixed in 4% paraformaldehyde for 72 h. After trimming, the samples were dehydrated, embedded, sliced into 3 µm and stained with H&E. The staining was examined by microscope (Nikon, Tokyo, Japan). The degree of tissue damage was assessed according to the criteria. 20
Immunohistochemical (IHC)
The paraffin sections were dewaxed in water and hydrated. Antigen retrieval was implemented in citrate buffer (pH 6.0). The sections were blocked and then incubated with a primary antibody at a 1:200 dilution overnight and a horseradish peroxidase-labelled streptavidin working solution (Maixin Biotech, Fuzhou, China) for 50 min. 3,3′-Diaminobenzidine (Zhongshan Golden Bridge, Beijing, China) and hematoxylin (Yike Biotech, Xian, China) were used for colour development and staining. The staining was examined by microscope. The degree of target protein staining was assessed using the multiplicative rapid scoring (QS) method. 20
Transmission Electron Microscopy (TEM) Observation
Mice were perfused using a 2.5% glutaraldehyde (Alfa Aesar, Shanghai, China) solution. Lung tissues were soaked in glutaraldehyde, cut into slices, fixed and then sent to the Electron Microscopy Center. The samples were subjected to wash, fix with osmium acid and dehydrated with a gradient of ethanol and acetone. After embedding in epoxy resin, the samples were cut into 50 nm ultrathin sections and stained with lead acetate-uranium. The sections were examined by TEM (Hitachi, Tokyo, Japan).
Western Blotting (WB)
The protein samples were loaded and subjected to sodium dodecyl sulfate-polyacrylamide gel electrophoresis (Kangwei Century) and transferred to a polyvinylidene fluoride membrane (Millipore, Burlington, MA, USA). The membrane was blocked and incubated with a primary antibody against β-actin (1:1000; Affinity, Changzhou, China), ZO-1 (1:1000; Abcam, Cambridge, UK), occludin (1:1000; Abcam), claudin-5 (1:1000; Abcam), RhoA (1:1000; Affinity), ROCK1 (1:1000; Affinity), MYPT1 (1:1000; Affinity), p-MYPT1 (1:1000; Bioss Biotech, Beijing, China), cyclooxygenase-2 (Cox-2) (1:1000; Affinity) and inducible nitric oxide synthase (iNOS) (1:1000; Affinity) at 4°C overnight; and incubated with the corresponding secondary antibodies for 1 h. The protein blots were visualized using an ECL chemiluminescence kit (Kangwei Century) by a Tanon automatic chemiluminescence gel imaging system (Tanon Techno, Shanghai, China).
Statistical Analysis
Data were statistically analyzed by GraphPad Prism version 8.0.1 (GraphPad Software, San Diego, CA, USA). Data were expressed as the mean ± standard deviation (SD). Differences between groups were compared using a one-way analysis of variance or rank-sum test. P <0.05 was considered statistically significant.
Results
FA and CA Contents
The regression equation of the emodin standard was Y = 0.0183X + 0.0939, R 2 = 0.9994. Results showed that emodin had a good linear relationship in a concentration range of 5.58–39.06 µg/mL. The total FA content in the FA sample was measured using a spectrophotometer with emodin as the standard, and the content was 96.58%. The total CA content in the CA sample was determined via regression equation, and the content (calculated as emodin monooxyglucoside) was 51.33%.
Contents of Different Ingredients in FA and CA
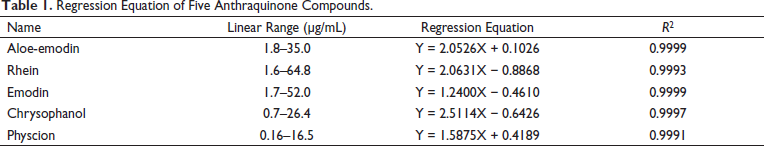
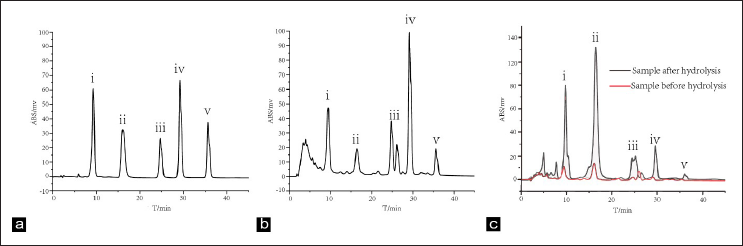
Table 1 shows the regression equation of five anthraquinones. Results showed that anthraquinones had a good linear relationship in the concentration range. HPLC results showed that the total FA content in the FA extract was 96.21%, the total FA content in the CA extract was 6.55%, and the total anthraquinone content was 35.53%. Figure 1 shows the HPLC chromatograms of the mixed reference substance, FA and CA from Rhubarb.
Regression Equation of Five Anthraquinone Compounds.
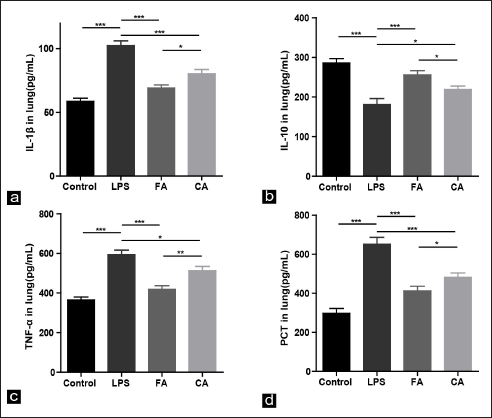
RA Suppressed LPS-induced Inflammation
Figure 2 shows the contents of four inflammation-related indexes in different groups. Compared to the control group, the TNF-α, IL-1β and PCT levels in the LPS group significantly increased, whereas the IL-10 level significantly decreased (all P <.001). After FA or CA intervention, the TNF-α, IL-1β and PCT contents significantly decreased, whereas the IL-10 content significantly increased. These data indicated that FA and CA could inhibit lung inflammation by decreasing the TNF-α, IL-1β and PCT contents but increasing the IL-10 content in the serum induced by LPS. RA might have a protective effect on inflammatory lung injury.
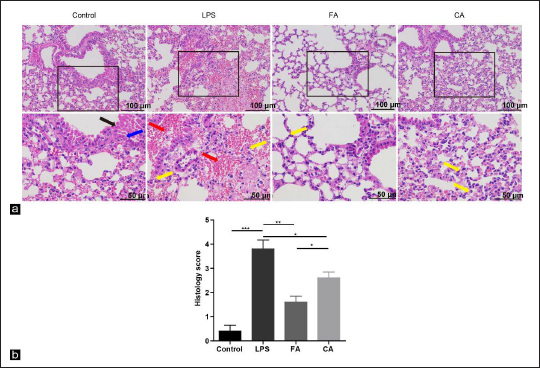
RA Ameliorated LPS-induced Lung Pathological Damage
To explore the morphological efficacies of RA on lung injury in mice, H&E staining was performed to examine the pathological differences in the lungs (Figure 3a). Normal structures of lung tissues were observed in the control group by optical microscopy. Pathological changes were found in the LPS group, including different degrees of thickened alveolar septum, haemorrhage and neutrophil infiltration. FA or CA administration significantly reduced the severity of lung pathological damage. Compared to the LPS group, the pathology scores indicated that FA and CA treatment significantly ameliorated LPS-induced lung injury in mice (Figure 3b).
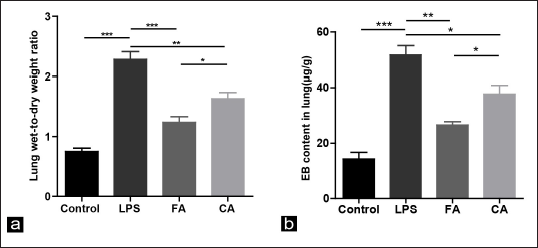
RA Attenuated LPS-induced Lung Injury via Reducing Alveolar Epithelial Permeability
To identify the roles of RA on lung leakage, the extent of alveolar epithelial permeability was determined by the lung W/D weight ratio and EB permeability assay (Figure 4a and b). Compared to the control group, the lung W/D weight ratio and EB leakage substantially increased in the LPS group (all P <.001). In contrast, FA or CA treatment significantly reduced the lung W/D weight ratio and suppressed EB leakage in the presence of LPS. These results suggested that LPS induced alveolar epithelial hyperpermeability, resulting in lung injury and pulmonary oedema, whereas FA and CA protected against lung injury via ameliorated alveolar epithelial permeability.
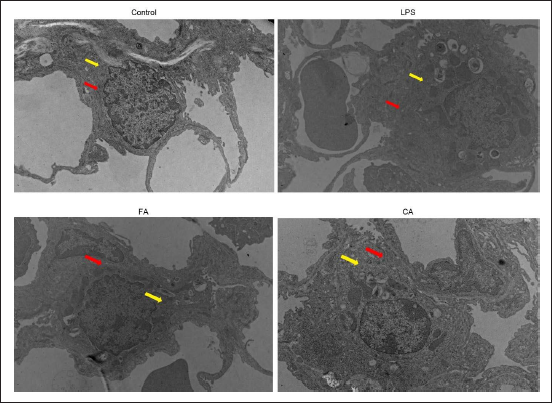
RA Improved the TJ Barrier Ultrastructure in Inflammatory Lung Injury
In Figure 5, electron microscopy showed the ultrastructural contrast of lungs in each group. The structure of alveolar epithelial cells and TJ gaps were normal in the control group. However, alveolar epithelial irregular thickening, widening of TJ gaps between epithelial cells, and mitochondrial vacuolation in epithelial cells were checked in the LPS group. After FA intervention, the TJs of epithelial cells were narrow, mitochondrial vacuolation was normal and the swelling and thickening of epithelial cells were reduced. The changes in epithelial cells and TJs in the CA group were between the FA and LPS groups. These results suggested that RA ameliorates LPS-induced ultrastructural changes in the TJ barrier of alveolar epithelial cells in mice.
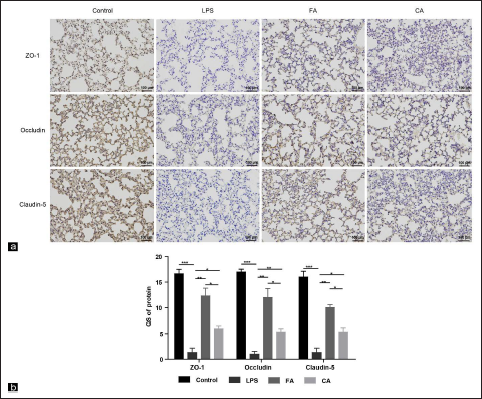
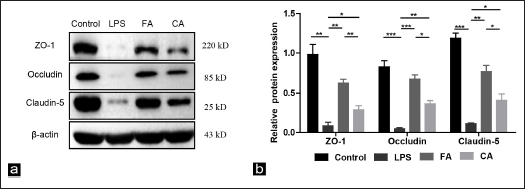
RA Increased TJ Expression in Lung Tissues of Mice in Inflammatory Lung Injury
TJs are a major factor in maintaining a normal alveolar epithelial barrier system. Occludin, claudin-5 and ZO-1 are key parts of TJs in epithelial cells. We used the above three indicators as TJs to detect their expression. TJ expression in lung tissues was detected by IHC, and IHC staining showed that TJ staining was fully expressed along the alveolar epithelium (Figure 6a). Staining quantification showed that Occludin, claudin-5 and ZO-1 levels significantly decreased in the LPS group. In contrast, Occludin, claudin-5 and ZO-1 expression was significantly upregulated in mice pretreated with FA or CA (Figure 6b). As detected by WB, Occludin, claudin-5 and ZO-1 expression were significantly downregulated in LPS group, which was remedied by RA. In addition, the remedial effect of FA was more effective than CA (Figure 7a and b). The above results suggested that RA could upregulate Occludin, claudin-5 and ZO-1 expression. These results were consistent with the IHC.
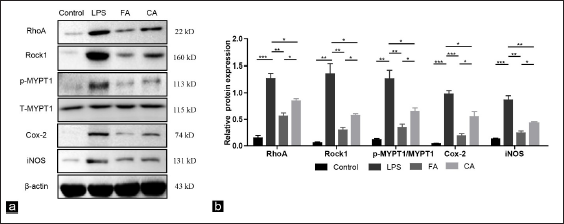
RA Inhibited the RhoA/ROCK1 Pathway and Inflammation-related Protein in LPS-induced Lung Injury
To demonstrate whether RA could mediate the RhoA/ROCK1 pathway in lung tissues of mice induced by LPS, WB was used to analyze pathway protein expression. Analysis of the bands showed that the expression levels of RhoA, ROCK1 and downstream p-MYPT1/MYPT1 in the LPS group were significantly increased than those in the control group (P <0.001, P <0.01 and P <0.01). Compared to the LPS group, FA treatment downregulated RhoA, ROCK1 and p-MYPT1/MYPT1 expression, and the inhibitory effects of FA were due to CA. Furthermore, the changes in protein expression of inflammation-associated proteins Cox-2 and iNOS were also consistent with the above results (Figure 8a and b). These data suggest that RA inhibits inflammatory lung injury and the RhoA/ROCK1 signalling pathway.
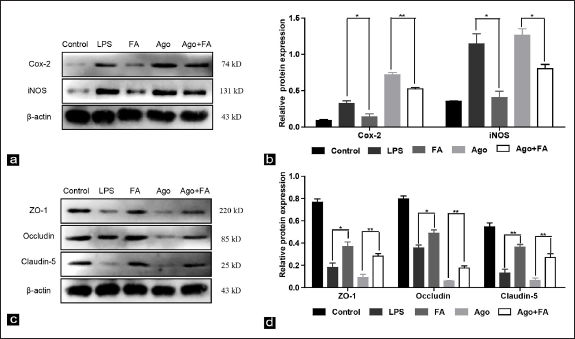
RA Regulated TJs Through the RhoA/ROCK1 Pathway in LPS-induced Lung Injury
U46619 is an agonist of RhoA. 21 To further investigate whether RA could regulate TJs through the RhoA/ROCK1 pathway in lung tissues of mice induced by LPS, U46619 was used to activate RhoA and validated with the more potent FA. WB results indicated that compared with the LPS group, Cox-2 and iNOS protein expression levels were lower in the FA group (Figure 9a and b). Conversely, the ZO-1, occludin and claudin-5 protein expression levels were higher in the FA group than in the LPS group, whereas the protective effects of FA on TJs were partially abolished by the intervention of U46619 (Figure 9c and d). All the above results further verified that RA regulates TJ expression by inhibiting the RhoA/ROCK1 signalling pathway.
Discussion
Inflammation is a systemic response. LPS administration can induce a strong systemic inflammatory response, leading to multiple organ dysfunction including lung, intestinal and other organs. 22 ALI is a life-threatening clinical syndrome in which alveolar epithelial damage, interstitial pulmonary oedema and neutrophils are the main pathological changes. 23 Effective drug therapy is urgently needed in the clinic to prevent and treat ALI. LPS is an endotoxin that can induce the release of various inflammatory factors, such as IL-1β and TNF-α. LPS can induce pulmonary inflammation to maximize 24 h after administration and is widely used to establish the ALI model.24, 25 Released pro-inflammatory cytokines can be detected in serum after LPS administration. Inflammatory mediators can change the structure and function of the alveolar epithelial barrier, and the damaged barrier loses the ability to clear alveolar fluid, resulting in pulmonary oedema. A stable alveolar epithelial barrier plays an important role in the inflammatory response and oedema clearance, which may serve as a potential treatment strategy. 26
Rhubarb has a variety of pharmacological activities such as anti-tumour, regulating bacteria, anti-inflammatory and anti-fibrosis. It is currently recognized that the main components that play a role are anthraquinones. 27 Rhubarb has been used clinically in China for thousands of years and is usually administered orally. Zhao et al. 28 found that analytes were widely distributed in most tissues after the gavage of RA extract in rats. Therefore, as part of a systemic multi-organ inflammatory injury study, intragastric administration of RA was chosen for this study. In our study, FA and CA were extracted from Rhubarb, which met the quality-control requirements, and a systematic comparative study was conducted on the protective effects of RA on the alveolar epithelial barrier. We found that RA attenuated LPS-induced lung injury in mice by repressing the inflammatory response.
TJs between adjacent cells, which serve to seal the paracellular space and selectively prevent the passage of molecules, are essential to protect the function and integrity of the epithelial barrier. 29 The structure and dysfunction of the alveolar epithelial barrier are causes of pulmonary oedema, and their permeability is mainly regulated by TJs. TJs consist of transmembrane proteins, such as occludins, claudins and cytoplasmic plaque (ZO-1, ZO-2 and ZO-3). 30 Claudins, the most widely expressed transmembrane proteins among TJs, are regarded as primarily sealing, ion-selective paracellular channel-forming and water-permeable paracellular barriers. Claudin-5 plays a major role in sealing. 31 Occludin comprises four transmembrane domains, two extracellular loops and one intracellular loop, which exerts adhesion between epithelial cells. 32 As bridges between TJs and cytoskeletal proteins, ZO proteins connect claudins and occludin with the cytoskeleton for protein complex aggregation to TJ domains. ZO-1 regulates TJ assembly, which is critical for TJ chain and membrane formation. 33 In this study, LPS can damage the alveolar epithelial barrier structure and increase permeability. In contrast, RA can significantly improve this barrier dysregulation and alleviate lung injury. In addition, as three indicators of TJs and alveolar epithelial barrier integrity, their expression was downregulated after LPS stimulation and upregulated after RA treatment.
Inflammatory infiltration is one of the important factors in ALI. TNF-α and IL-1β are the keys to initiating lung injury in the early stage of inflammation. TNF-α mainly recruits leukocytes into lung tissues, and IL-1β can induce macrophages and monocytes to promote lung injury.26, 34 IL-10, is a key protective cytokine produced by immune cells that protect against lung injury.35, 36 PCT, a sensitive biomarker, is often used to detect lung inflammation in humans. 37 Results indicated that RA could significantly inhibit TNF-α, IL-1β and PCT expression in the serum of mice with LPS-induced lung injury and increase IL-10 expression, thereby exerting an inhibitory effect on inflammation.
What mechanism was implied in the protective effects of RA on LPS-induced inflammatory lung damage? To address this question, the effects of RA on the RhoA/ROCK1 pathway were investigated. Previous works of literature have revealed that the RhoA/Rock signalling produces a marked effect on apoptosis, immune regulation and regulating the body’s coagulation function.38–40 Moreover, the RhoA/Rock signalling provides certain role in the inflammatory response. The RhoA/Rock pathway can activate nuclear factor-κB and release inflammatory factors, thereby causing the accumulation of many inflammatory factors. 41
Rho is a GTP-binding protein in Ras homologous genes, of which RhoA is the most representative. RhoA can catalyse GTP hydrolysis after binding to GTP. 42 RhoA protein is expressed in all human tissues. It is mainly located in the plasma membrane, cytoplasm and other cell parts. RhoA plays role in cell growth and cytoskeleton regulation. 43 Rock, including ROCK1 and Rock2, is a downstream factor of RhoA. ROCK1 mostly exists in nonneural tissues, while Rock2 is in the central nervous system. 44 Rock causes the release of inflammatory mediators. 45 Hypoxia or inflammation can activate the Rho/Rock pathway, and the activated Rho combines with Rock, causing the upregulation of intracellular Ca2+ concentrations and phosphorylation of MYPT-1, a downstream effector protein of Rock. The activity of MLC phosphatase is inhibited, and MLC phosphorylation further enhances myosin contractility, remodels the cytoskeleton and causes abnormal expression and distribution of intercellular TJs, ultimately resulting in abnormalities in cell permeability and barrier function. 46 Yin et al. 47 found that Connexin 40 promotes lung barrier leakage through interaction with ROCK1 and phosphorylation of MYPT1 in LPS-induced ALI mice. This suggests that there is a mutual connection between RhoA/ROCK1 signalling and TJ. Our study also demonstrated that RA may regulate TJ expression via inhibiting the RhoA/ROCK1 signalling. As an agonist, U46619 can cause activation of the RhoA/Rock signalling. 48 Cox-2 and iNOS are two integral factors involved in the regulation of inflammation. Another previous study found that the expression of Cox-2, iNOS and TJ proteins was reduced after U46619 intervention. U46619 can attenuate the inhibitory effect of dexmedetomidine on colon cell barrier damage. 49 Similar to these findings, our results indicated that RA downregulated RhoA, ROCK1 and p-MYPT1 expression but upregulated TJs expression, whereas U46619 showed the opposite effect, suggesting that the protective effects of RA on LPS-induced lung injury were associated with inhibition of the RhoA/ROCK1 signalling pathway.
This study also has some limitations. First, RA is a complex crude extract. It remains unclear which monomer or several monomers work together to exert an anti-inflammatory effect, and this is what the next research needs to explore. Second, U46619 is a nonspecific agonist of RhoA. Future studies should focus on applying small interfering RNA or short hairpin RNA at the cellular level to further clarify whether the anti-inflammatory effects of RA are related to RhoA/ROCK1 downregulation. Third, which are the immune target cells of RA after inflammatory lung injury in the body? This is currently in progress in the next research.
Conclusion
In summary, RA may enhance TJ expression by inhibiting the RhoA/ROCK1 pathway, improving alveolar epithelial permeability and alleviating lung inflammation, thereby ameliorating lung injury in mice. This will provide theoretical support for the clinical application of RA and a new idea for the treatment of ALI.
Footnotes
Abbreviations
Summary
Proposed model for the roles of RhoA/ROCK1 pathway regulated by RA in the protection of epithelial barrier dysfunction. In LPS-induced ALI, RA pretreatment inhibits the RhoA/ROCK1 pathway, leading to a decrease of TNF-α, IL-1β and an increase of IL-10, which contributes to inhibiting TJ opening and ameliorating epithelial barrier dysfunction.
Acknowledgment
The authors thank the Cuiying Medical Center of Lanzhou University Second Hospital for providing the necessary support for this study.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This work was supported by the Doctoral students Training Research Fund of Lanzhou University Second Hospital (No.YJS-BD-32), the Key Projects of Traditional Chinese Medicine Research of Gansu Province (No. GZKZ-2020-7), the Science and Technology Research Projects of Chongqing Education Commission (No. KJQN201902706 and No. KJQN201802704) and the Gansu Provincial Natural Science Young Foundation (No. 21JR1RA161).
Statement of Informed Consent and Ethical Approval
The experimental animal welfare ethics committee of the Lanzhou University Second Hospital approved this animal experiment.