Abstract
Objectives:
Plumbagin has been displayed on growth inhibition in several cancer cells; however, the anticancer effects of plumbagin on cholangiocarcinoma (CCA) cell are still less information. This article will explore the potential anticancer effects and underlying mechanisms of plumbagin on CCA cells.
Materials and Methods:
Two types of CCA cell, including KKU-100 with slow growth and migration and KKU-M452 with rapid growth and migration, were treated with plumbagin and measured the growth inhibition and migratory suppression by sulforhodamine B, colony formation, acridine orange/ethidium bromide staining, wound healing, and Matrigel migration. Apoptosis induction, reactive oxygen species (ROS) formation, and mitochondrial function were analyzed by the flow cytometric method.
Results:
To compare the plumbagin effects on two types of CCA cells, the data indicated that plumbagin strongly reduced cell proliferation in a concentration- and time-dependent manner. At 72 h, IC50 values were 1.11 ± 0.12 µM for KKU-100 and 2.52 ± 0.40 µM for KKU-M452 cells, with inhibition of the cell cycle in the G0/G1 phase for KKU-100 and the G2/M phase for KKU-M452, respectively. Moreover, plumbagin decreased cell migration in both CCA cells in a dose-dependent manner. Furthermore, late apoptosis was significantly induced after treatment with plumbagin. Mechanistically, plumbagin increased ROS along with decreased mitochondrial function in CCA cells. Finally, the position of 5-hydroxy on the naphthoquinone ring of plumbagin was in a good position to form an H-bond with the amino group for all caspase 3, cytochrome C, and p21 binding sites, and these data indicated that plumbagin suppressed CCA cells via modulation of these proteins.
Conclusion:
Plumbagin strongly suppresses cell viability and stimulates apoptosis in CCA cells via induction of ROS and inhibition of mitochondrial function.
Introduction
Cholangiocarcinoma (CCA) is an aggressive tumor that originates from the epithelial bile duct cells. CCA is a main public health problem in the northeastern part of Thailand, and it is commonly found in men than women. 1 Risk factors of CCA are the consumption of raw fish that contains the Opisthorchis viverrini (liver fluke) infection, which causes inflammation in the epithelial bile duct along with fermented foods, which contain carcinogens (nitrosamines) in this region. Liver fluke stimulates chronic inflammation, leading to oxidative damage to the DNA of the infected biliary epithelium and malignant transformation. 2 The standard treatment is surgery followed by chemotherapeutic agents; however, the efficacy of treatments is still unsatisfactory, with reports of low cure rate and survival time. 2 Several studies indicated that chemotherapeutic drugs had a high toxicity and resistances in cancer patients. Therefore, the new alternative agents with high efficacy, less toxicity, and low resistance is required for CCA patients.
Plumbagin is an active compound that is extracted from the leaf, stem, bark, and root of the herbal medicinal plant Plumbago indica L. (Plumbaginaceae family). Plumbagin has been reported to have many biological actions, such as antiinflammatory, antibacterial, antimalarial, anticancer antiseptic, and antiprotozoa properties. 3 For cancer suppression, it has been documented that plumbagin decreases the proliferation of colon, lung, ovarian, breast, and prostate cancer, 4 along with inhibition of invasion, metastasis, and induction of apoptosis. 4 Nevertheless, the plumbagin effects of CCA are still less information and need to be investigated.
Plumbagin’s effects on cancer cell inhibition have been reported through several mechanisms. In osteosarcoma cells, plumbagin significantly reduced cell proliferation and induced apoptosis by reducing mitochondrial function and inducing reactive oxygen species (ROS) formation. 5 In esophageal squamous cell carcinoma, plumbagin activated cancer cells apoptosis by downregulating signal transducer and activator of transcription 3 (STAT3) along with upregulating tumor proteins p53 and p21 and further downregulating cyclin D1. 6 In cervical cancer cells, plumbagin suppressed the cell proliferation by strongly stopping the cell cycle at G2/M and S phase to G2/M phase with a reduction of protein-related cell death, cyclin A, B1, and E2. Furthermore, plumbagin induced apoptosis in cervical cancer cells through increasing protein-related apoptosis such as caspase 3 and 9. 7 In lung cancer cells, plumbagin significantly decreased the level of the epidermal growth factor receptor and its downstream signaling (Akt, NF-κB, Bcl-2, and survivin). 8 Therefore, the plumbagin effects on CCA are interesting to explore.
In this study, we need to explore the anticancer properties of plumbagin against CCA cells by comparing two types of CCA cells, including KKU-100 with slow growth and migration and KKU-M452 with rapid growth and migration. Furthermore, we will examine the underlying mechanisms of plumbagin effects, including cell cycle arrest, antimigration, apoptosis induction, ROS formation, and mitochondrial dysfunction.
Materials and Methods
Cell and Cell Culture
Two types of CCA cells were used in this study including KKU100 with slow growth and migration and KKU-M452 with rapid growth and migration were kindly provided from the Faculty of Medicine at Khon Kaen University. Two cancer cells were cultured in Dulbecco’s Modified Eagle Medium (DMEM) with 10% fetal bovine serum (FBS) and 1% antibiotics. Every three days, the cells were replaced with the new complete media.
Sulforhodamine B (SRB) Method
The plumbagin effects on growth inhibition were examined by the SRB method. CCA cells were subcultured to 96-well culture plates (1×104 cells/well) for 24 h and exposed to varying concentrations of plumbagin (0−25 µM) for 24–72 h. CCA cells were then washed, fixed, stained with 0.4% SRB solution, and solubilized with 10 mM Tris base buffer after incubation periods. The optical densities were then read at a wavelength of 540 nm by a spectrophotometer.
Colony Formation Method
The plumbagin effects on colony forming ability inhibition were examined by colony formation methods. CCA cells were placed into 6-well culture plates (500 cells/well) for 24 h and exposed to varying concentrations of plumbagin (0–5 µM) for 24 h. Following the incubation periods, the cells were then washed and changed to the new complete medium every 2–3 days for 15 days. Finally, the colonies were then exposed to absolute methanol and stained with 0.25% crystal violet solution. The colonies were photographed with a Nikon camera and counted.
Cell Cycle Distribution Method
The plumbagin effects on cell cycle distribution were examined by the flow cytometric method. CCA cells were placed into 6-well culture plates (2.5×105 cells/well) for 24 h and then exposed to varying concentrations of plumbagin (0–5 µM) for 24 h. Following the incubation periods, the cells were fixed with 70% ethanol at −20°C for 24 h, cells were washed with PBS buffer, and treated with 500 µl of propidium iodide (PI) at room temperature for 30 min. Flow cytometry (BD Biosciences, San Jose, CA, USA) was used to examine cell cycle proportions using the BD Accuri C6 Plus software.
Acridine Oranged/Ethidium Bromide Staining Method
The plumbagin effects on apoptosis were examined by the acridine oranged/ethidium bromide (AO/EB) double staining method. CCA cells were cultured in 96-well culture plates (1×104 cells/well) for 24 h and exposed to varying concentrations of plumbagin (0–5 µM) for 24 h. Following the incubation periods, the CCA cells were washed with PBS buffer, added with AO/EB dye (1 µg/mL of each dye) for 15 min at room temperature. The images were observed and captured by a fluorescence inverted microscope (×20 magnification, CKX53, Olympus, USA).
Wound Healing Method
The plumbagin effects on migration were examined by the wound healing method. The CCA cells were placed into 24-well culture plates (2.5×105 cells/well) for 24 h, then the wound was made by scratching the cells with 0.2 mL pipette tips and exposed to varying concentrations of plumbagin (0–5 µM) for 24 h (KKU-M452) and 72 h (KKU-100). Following the incubation periods, the cell images were captured at 0, 24, and 72 h under phase-contrast microscopy (4× magnification, CKX53, Olympus, USA).
Matrigel Migration Method
The plumbagin effects on migration were examined by the Matrigel migration method. CCA cells were placed in the upper chamber of an Inserted-well (2×104 cells/well) with serum-free medium and varying concentrations of plumbagin (0–5 µM) for 24 h. In the lower chambers, complete DMEM medium was added. Following the incubation periods, the migrated cells to the lower chamber surface were fixed with absolute methanol for 30 min at −20°C and then stained with 0.25% crystal violet. The percentage of migrated cells was photographed under a phase‑contrast microscope (20× magnification, CKX53, Olympus, USA) and further counted in three different fields.
Apoptosis Method
The plumbagin effects on apoptosis were examined by the flow cytometric method. CCA cells were placed into 6-well culture plates (2.5×105 cells/well) for 24 h and exposed to varying concentrations of plumbagin (0–5 µM) for 24 h. Following the incubation periods, the cultured cells were then washed with cold-PBS buffer for three times, and 100 µL of 1× binding assay buffer was added. The suspension cells were then added with 5 µL of Annexin V-FITC and 1.5 µL of PI solution at room temperature for 15 min in the dark. The fluorescent signal of apoptosis was measured by flow cytometry (BD Biosciences, San Jose, CA, USA) using BD Accuri C6 Plus software.
ROS Formation Method
The plumbagin effects on ROS formation were examined by the flow cytometric method. CCA cells were placed into 6-well culture plates (2.5×105 cells/well) for 24 h and exposed to varying concentrations of plumbagin (0–5 µM) for 24 h. Following the incubation periods, the suspension cells were treated with 25 µM DCF-DA fluorescent probe at 37°C for 30 min. Subsequently, ROS production was measured by flow cytometry (BD Biosciences, San Jose, CA, USA) using BD Accuri C6 Plus software.
Mitochondrial Function Method
The plumbagin effects on mitochondrial function were examined by the flow cytometric method. CCA cells were placed into 6-well culture plates (2.5×105 cells/well) for 24 h and exposed to varying concentrations of plumbagin (0–5 µM) for 24 h. Following the incubation periods, the cells were stained with the JC-1 fluorescent probe and the fluorescent signal was measured by flow cytometry (BD Biosciences, San Jose, CA, USA) using BD Accuri C6 Plus software as aforementioned at 527 and 590 nm, respectively.
Computational Study
Plumbagin was docked with the AutoDock4.2 program 9 from The Scripps Research Institute, USA, into caspase 8 (PDB ID: 3kjn), cytochrome C (PDB ID: 5ty3), and p21-activated kinase-1 (PAK1) (PDB ID: 6b16). First of all, the ligands and macromolecules were separated and then redocked to validate the target binding sites. For ligands, hydrogen and Gasteiger charges were added to all atoms, but polar hydrogen and Kollman charges were added to macromolecules. The grid maps were calculated with AutoGrid. Larmarckian genetic algorithm was a method for conformational searching for the docking of flexible ligands into rigid protein binding sites. All run parameters were kept at their default settings except the number of genetic algorithm runs, which was 100. The docked conformations were clustered using a tolerance of 2 Å RMSD.
Statistical Analysis Method
All results are represented as the mean±SE. and the difference was tested using the Student’s t test between the control and plumbagin treatment groups. The significant difference was set as *p < 0.05.
Results
Plumbagin Suppresses Cells Viability and Colony Formation
SRB method was used to analyze the viability of CCA cells after following the treatment with plumbagin (0–25 µM) for 24–72 h. Two types of CCA cells, namely, KKU-100 (slow growth and migration) and KKU-M452 (rapid growth and migration), were used in this study to compare the plumbagin effects on these two cells. This data indicated that plumbagin strongly reduced cell proliferation in a concentration- and time-dependent manner with very low IC50 values (Figure 1A & 1C). IC50 values were 2.52 ± 0.39, 1.42 ± 0.10, and 1.10 ± 0.12 µM for 24, 48, and 72 h in KKU-100 cells and 5.97 ± 0.73, 4.04 ± 0.33, and 2.52 ± 0.40 µM in KKU-100 cells, respectively. Overall, plumbagin suppressed cell viability both in CCA cells with the same effects and in KKU-100 cells, which were more sensitive to plumbagin than KKU-M452 cells.
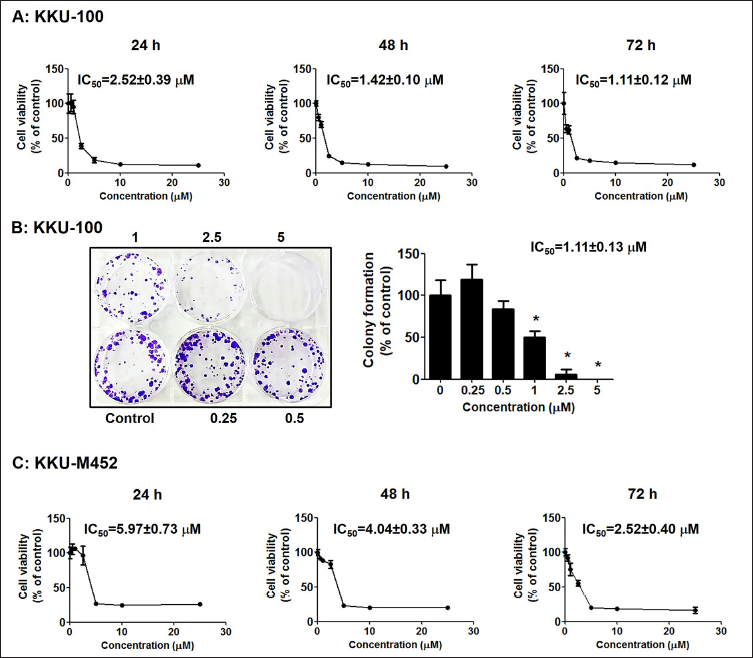
To confirm the cell viability of SRB results and a colony-forming assay was further performed. In this method, KKU-100 cells were represented as the CCA model because KKU-M452 cells did not perform colony formation. The data revealed that plumbagin considerably decreased the colony formation rate of KKU-100 cells (Figure 1B), with an IC50 value of 1.11 ± 0.13 µM.
Plumbagin Arrests Cell Cycle Distribution and Reduces Cells Number
Flow cytometric analysis was used to analyze the cell cycle distribution of CCA cells following treatment with plumbagin (0–5 µM) for 24 h. The G0/G1 phase cell cycle arrest was indicated in KKU-100 cells when the cells were exposed to plumbagin for 24 h. Furthermore, the induction of G2/M phase arrest was detected in KKU-M452 cells when compared with the control group (Figure 2A and B). Slowly growing CCA cells had suppressed cell cycles in the G0/G1 phase; however, rapidly growing cells had suppressed cell cycles in the G2/M phase.
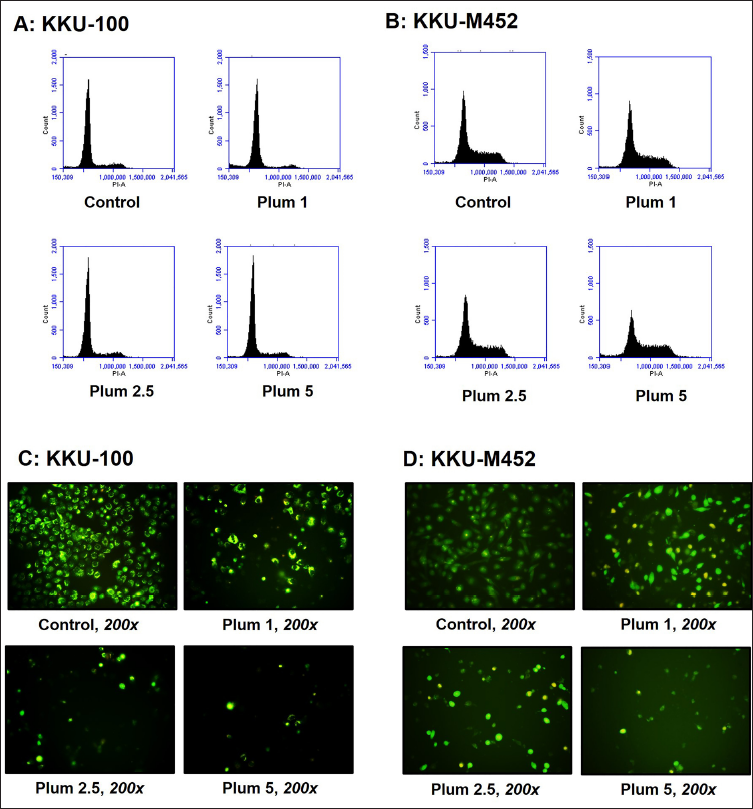
To confirm the cell cycle arrest results and the plumbagin effects on cell number and morphology were performed by AO/EB staining method. After cells were treated with plumbagin, the number of CCA cells decreased in a significant and dose-dependent manner in two types of CCA cells. Morphological changes were detected with membrane blebbing and cell shrinkage, which are features of apoptosis. When the CCA cells were incubated with 2.5 µM plumbagin, almost all the KKU-100 and KKU-M452 cells shrank, and no cells with normal morphological features were detected (Figure 2C and D). Two types of CCA cells were demonstrated to undergo apoptosis after treatment with plumbagin and showed the same effects.
Plumbagin Decreases Cell Migration
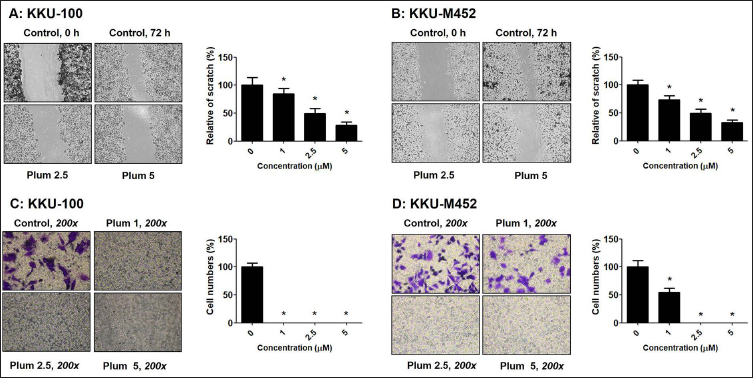
The wound healing and Matrigel migration method were used to analyze the migration of CCA cells after following treatment with plumbagin (0–5 µM). The results revealed that plumbagin could significantly suppress KKU-100 and KKU-M452 cells migration in a dose-dependent manner (Figure 3A and B). Furthermore, a similar result was observed in the Matrigel migration method. Plumbagin treatment in KKU-100 and KKU-M452 cells led to a reduced percentage of migrated cells (Figure 3C and D) in a dose-dependent manner as measured by the wound healing assay. At a high dose of plumbagin (2.5 µM), plumbagin significantly suppressed migrated cells, and no cells were detected at the high dose. The migration results demonstrated that plumbagin suppressed cell migration in slowly migrating cells, KKU-100 cells, than in rapidly migrating cells, KKU-M452 cells.
Plumbagin Activates Cell Apoptosis, Induces ROS Formation, and Decreases Mitochondrial Function
Flow cytometric analysis was used to analyze CCA cells’ apoptosis following treatment with plumbagin (0–5 µM) for 24 h. Plumbagin strongly stimulated late apoptosis in two CCA cells in a dose-dependent manner, especially in KKU-100 cells (Figure 4A-B).
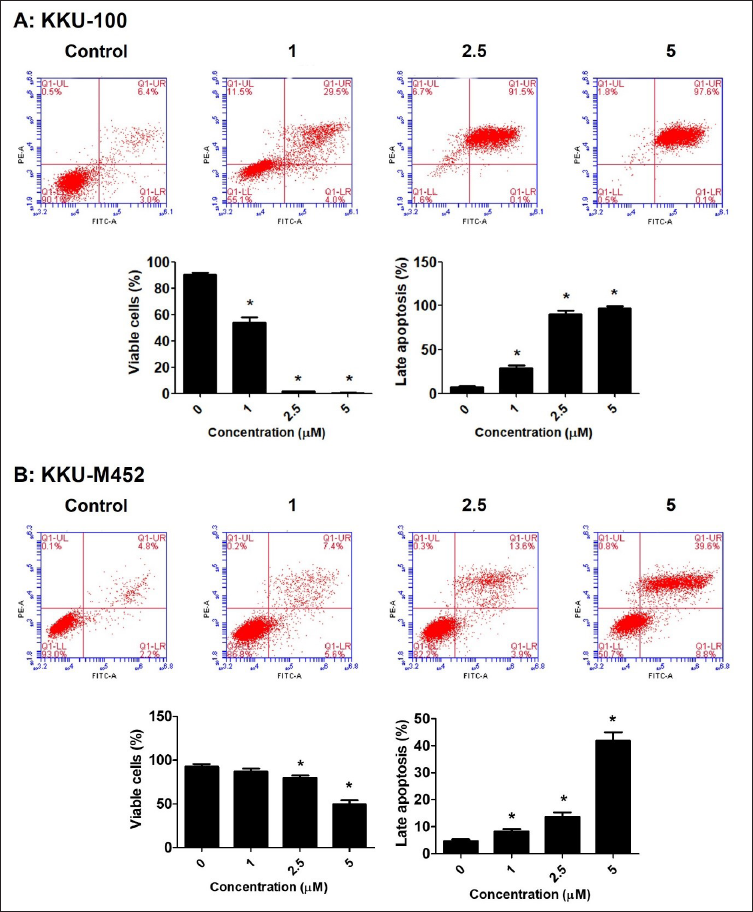
The data indicated that viable cells were observed in a very low percentage with the plumbagin-treated group at high doses (5 µM). A higher dose of plumbagin strongly induced late apoptosis in approximately 97% of KKU-100 cells and 39% of KKU-M452 cells, which is consistent with late apoptosis. This study demonstrated that the plumbagin-treated group stimulates late apoptosis in both CCA cells.
The mechanism of apoptotic induction was explored by ROS formation and mitochondrial function. After incubation with plumbagin for 24 h, the flow cytometric analysis confirmed that plumbagin strongly increased the formation of ROS generation in a dose-dependent manner (Figure 5A and B), especially in KKU-100 cells than KKU-M452 cells. Furthermore, mitochondrial function method was indicated that plumbagin induced the loss of mitochondrial membrane potential (MMP) or mitochondrial dysfunction in both CCA cells, indicating that plumbagin strongly increased the percentage of depolarized cells, JC-1 monomers induction (Figure 5C-D) along with decreased JC-1 aggregates. The data indicated that plubagin-induced cell apoptosis was accompanied by ROS production and mitochondrial dysfunction.
Computational Study
The binding site affinity was performed by molecular docking with AutoDock version 4.2. The X-ray crystal structures from the PDB ID, namely, 3kjn, 5ty3, and 6b16, were selected as templates for caspase 8, cytochrome C, and P21 (RAC1) activated kinase 1 (PAK1), respectively. The crystal ligands were redocked to validate the template binding site, and the results show that the docked orientations are like the conformations in the X-ray crystal structure, with RMSD of less than 2. Then, these templates were used for docking with plumbagin.
After 100 docking runs between plumbagin with caspase 8, cytochrome C, and PAK1, the docked conformations of similar orientation (RMSD < 2 Å) were clustered and shown in Table 1. The binding energy of plumbagin was −6.03 kcal/mol with the member of 99 for caspase 8, −7.20 kcal/mol with the member of 83 for cytochrome C, and −5.49 with the member of 100 for PAK1. The more negative values of binding energy and percentage of members in a cluster correspond to the greater predicted binding affinities. Plumbagin forms two hydrogen bonds with His317 and Cys360 for caspase 8 and two hydrogen bonds with Gly41 and Trp59 for cytochrome C, as shown in Figure 6. The hydrogen bonding between plumbagin and PAK1 was shown as two hydrogen bonds with Leu345 and Leu347 in a green dot line. The carbonyl group on positions 4 and 5-hydroxy of plumbagin’s naphthoquinone ring could form hydrogen bond with both cytochrome C and PAK1. The position of 5-hydroxy on the naphthoquinone ring was the good position to form an H-bond with amino group for all three binding sites.
The Docking Results of Plumbagin with Caspase 8, Cytochrome C, and PAK1.
Discussion
Over the past few years, several dietary agents, herbal medicinal plants, and its phytochemical compounds gained significant therapeutic importance with anticancer effects, antiviral activity, anti-inflammatory action, and antioxidant activity. 10
Plumbagin has displayed the strong anticancer effects with growth inhibition, apoptosis induction, and migratory suppression. 3 in many cancer cell types. 11 However, there is still less information of plumbagin ability on bile duct cancer cells or CCA cells. This study indicated that plumbagin effects were also suppressed the growth of two CCA cells with slowly growing (KKU-100) and rapidly growing (KKU-M452) cells. Moreover, the incubation of plumbagin significantly increased apoptotic induction, which began with a low dose of plumbagin and increased with a higher dose while monitoring the percentage of late apoptotic cells. The mechanism of action of plumbagin was significantly detected by the overproduction of ROS levels and reduction of mitochondrial function, especially in slow growth and migratory cells (KKU-100). Finally, this study indicated that plumbagin may be a potential anticancer drug in CCA treatment.
To gain an understanding of growth inhibition was stimulated by plumbagin effects, and the colony formation and cell cycle distribution were performed. Plumbagin significantly suppressed CCA cell proliferation with low IC50 values, similar to the IC50 values in two CCA cells. However, plumbagin inhibited the cells’ proliferation in slowly growing cells (KKU-100) than rapidly growing cells (KKU-M452). In molecular levels, the inhibition of cell growth of plumbagin was controlled through the cell cycle distribution at G0/G1 phase arresting related with the upregulation of p53 and p21 protein levels. 6 On the contrary, the suppressive effect was indicated at the G2/M phase arrest via inactivating the protein levels of Cdc2 and Cdc25C in lung cancer cells. 12 With regard to CCA, plumbagin inhibited CCA cells proliferation with a low dose than fibroblast cells and the selectivity index of 2.28. 13 Moreover, the natural compound, such as glycyrrhizin, induced apoptosis via the activation of caspase 9 and caspase 8 along with arresting the cell cycle at G0/G1 phase, which was associated with the decreased expression of cyclin D1. 14 Such effects were also previously reported in several mechanisms; however, all mechanisms are mentioned to strongly suppress the CCA cells growth as well as the reduction of cells number with morphological changes.
An invasion and metastasis are the crucial role of the secondary tumor with poor prognosis and poor clinical outcome. The study found that plumbagin decreased both CCA cells migration in a concentration-dependent manner after examining the wound healing and Matrigel migration assays. Plumbagin strongly suppressed the migration of KKU-100 (slowly migratory cells) than KKU-M452 (rapidly migratory cells) cells, especially from Matrigel migration study. The migratory suppression was reported in the several natural compounds, such as piperine, 15 berberine, 16 myricetin, 17 and fisetin, 18 in significant effects. The mechanism of migratory suppression of plumbagin was demonstrated in human breast cancer cells, and the result indicated that plumbagin repressed the migration, resulting in a reduction of interleukin (IL)-1α, transforming growth factor (TGF)-β, matrix metalloproteinases (MMP)-2, and MMP-9 mRNA levels with inactivation of STAT3 protein expression. 19 Moreover, plumbagin also reduced the mRNA and protein levels of urokinase-type plasminogen activator (u-PA) and receptor (u-PAR), while increasing tissue inhibitors of metalloproteases (TIMP)-2 and plasminogen activator inhibitor (PAI)-1. 20 Finally, plumbagin suppressed migration with significant effects via suppression of gene- and protein-related migration.
Apoptosis stimulation is one of the crucial roles of anticancer agents. It activates apoptotic pathways to block cancer cells’ growth/proliferation via the stimulation of ROS production and mitochondrial dysfunction. 21 This study found that plumbagin significantly activated late apoptosis, which stimulated ROS generation and decreased mitochondrial function in both CCA cells, especially in slowly growing cells. Mitochondria is membrane-bound cell organelles that are considered a primary intracellular site of ROS generation, importantly, the high levels of ROS can trigger apoptosis, which is what triggers mitochondrial abnormalities.22, 23 Plumbagin data on pancreatic cancer cells showed that it activated apoptosis through the induction of ROS production along with upregulation of cleaved caspase 3 and caspase 9 and downregulation of Bcl-2 protein. 24 The caspase cascade is the key signaling pathway that triggered apoptosis. 25 Therefore, plumbagin activate apoptosis may be beneficial for anticancer treatment including CCA cells. The advantage of computational screening of natural drugs before testing the effects in vitro and in vivo study has been reported in several diseases, 26 including cancer. For computer study of the binding site between plumbagin with caspase 8, cytochrome C and PAK1 were indicated that the position of 5-hydroxy on naphthoquinone ring was the good position to form H-bond with amino group for all three binding sites. Likely Alshahrani et al. indicated that computational screening of natural compounds led to the identification of potential anticancer agents targeting the minichromosome maintenance complex component 7 (MCM7) protein. 27 Therefore, the plumbagin inhibited the CCA cells’ proliferation and induced apoptosis through caspase 8, cytochrome C, and PAK1.
Conclusion
This study found that plumbagin strongly decreased cell growth, cell replication, and cell number, along with suppressing the cell cycle distribution at the G0/G1 phase in KKU-100 and the G2/M phase in KKU-M452 cells. Furthermore, migration was significantly reduced after treatment with plumbagin in a dose-dependent manner. Additionally, apoptotic induction was activated by plumbagin in late apoptosis, especially at 5 µM plumbagin, with induction of ROS generation and reduction of mitochondrial function. The current results suggest that plumbagin functions as an anticancer effects through suppression of cell growth and activation of apoptosis in CCA. Additionally, the deep molecular targets of plumbagin in cell death, antimigration, and apoptosis need to be elucidated.
Footnotes
Summary
Plumbagin has a strong effect on the growth inhibition of CCA cells both in KKU-100 and KKU-M452 cells.
Plumbagin inhibited the cell cycle distribution at the G0/G1 phase for KKU-100 cells and at the G2/M phase for KKU-M452 cells.
Plumbagin decreased cell migration in both CCA cells in a dose-dependent manner.
Plumbagin activated late apoptosis along with the induction of ROS and stimulation of mitochondrial dysfunction.
The position of 5-hydroxy on the naphthoquinone ring of plumbagin was the good position to form H-bond with amino group for all caspase 3, cytochrome C, and p21 binding sites, and these data indicated that plumbagin may suppress CCA cells via modulation of these proteins.
Abbreviations
Acknowledgment
The authors thank Dr. Adrian R. Plant (Mahasarakham University) for language editing the manuscript.
Declaration of Conflicting Interests
The authors declare that there is no conflict of interest.
Funding
This study was financially supported by the Thailand Science Research and Innovation (TSRI) 2021.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
