Abstract
Keywords
Introduction
Acrylamide (ACR) is an important chemical compound that is used extensively in various industries and processes, including sewage treatment, the paper industry, and research laboratories for electrophoresis. 1 ACR is also formed spontaneously at high temperatures (>120°C) in carbohydrate-rich foods, such as the widely consumed potato chips, French fries, and crisp bread.2–4 Additionally, ACR is present in tobacco smoke. 5 Thus, human exposure to ACR through food processing and smoking is unavoidable, and the toxicity of ACR and its lifelong exposure have raised widespread concern.
Accumulating evidence has demonstrated that ACR can cause carcinogenicity, genotoxicity, and reproductive toxicity.6,7 Glycidamide, the epoxide metabolite of ACR, both of them can form DNA adduct, causing gene mutations. 8 Several studies have investigated ACR as a reproductive toxicant. ACR has been shown to decrease the testicular organ index, induce morphological changes and DNA strand breaks in germ cells of male mice and rats.9–11 Chronic exposure to ACR adversely affects fertility rates, mating, and sperm counts and induces sperm-head abnormalities in male rats. 12 Additionally, ACR exposure increases reactive oxygen species (ROS) production and disrupts epigenetic modifications in mouse oocytes.6,13 Moreover, the oral administration of ACR to female mice caused a significant reduction in ovarian weight, fertility rates, progesterone (P4) concentration, and the litter sizes.6,14 However, the regulatory mechanisms of ACR in oocytes have not been fully understood.
Oocyte development in mammals involves the formation of a GV, germinal vesicle breakdown (GVBD), extrusion of polar body and fertilization. The fully grown GV oocytes are arrested in the prophase of meiosis I. 15 GV oocyte quality is essential for female fertility. The mammalian GV oocyte nucleus determines the selective expression or accumulation of dormant maternal transcripts that are essential to sustain the progression of meiosis to the first cleavage division.16, 17 In mice, the diameter, 18 zona thickness, 19 chromatin conformation 20 of GV oocyte are crucial for the meiotic resumption. For example, only fully grown GV oocytes larger than 60 μm in diameter could undergo GVBD. 21 In addition, the GV oocyte is classified into two types according to chromosome configuration, the non-surrounded nucleolus (NSN) and the surrounded nucleolus (SN) oocytes. With the growth of GV oocytes, the chromosome configuration underwent the NSN-to-SN transition. 22 Previous studies showed that NSN oocytes show lower rate of maturation and post-fertilization development than SN oocytes.23–26
In mammals, the mitochondria are maternally inherited organelles, and the mitochondrial reserve is crucial for the developmental competence of oocytes. Previous studies have linked the disruption of mitochondrial function to abnormal oocytes, maturation failure, and pre-implantation embryonic development.27,28 ACR exposure resulted in mitochondrial dysfunction and apoptosis in the mouse microglial BV-2 cell line. 29 However, whether ACR has any effect on oocyte mitochondrial function in mice oocytes remains unknown.
Oocyte quality is also dependent on the intracellular homeostasis between ROS and antioxidants. Previous studies have shown that oxidative stress may be one of the main mechanisms involved in many chemical poisoning-induced cell injuries. 30 A report demonstrated that ACR induced overproduction of ROS in GV oocytes, which is one probable reason for oocyte maturation failure.6,31 Increased levels of ROS can inhibit the quality of oocytes and embryos by inducing autophagy and apoptosis, thus affecting the ratio of pregnancy in females.28,32 Autophagy may cause maternal mRNA degradation and apoptosis in parthenogenetic embryos. 33 Furthermore, autophagy and apoptosis are associated with germ and granulosa cell death and the disruption of pre-implantation development of mouse embryos. 34 However, whether ACR has any effect on autophagy and apoptosis remains unclear.
Although previous rodent studies have demonstrated the effects of ACR on the reproductive system, the mechanism underlying the toxic effects of ACR on mouse GV oocytes is still not completely understood. To further understand the toxic effect of ACR on developmental fate, mitochondrial function, and cytoskeletal organization, we evaluated mitochondrial distribution and Δφm, actin filament assembly, apoptosis, and autophagy in mouse oocytes.
Materials and methods
Antibodies and chemicals
ACR was purchased from Sigma chemical company (Sigma-Aldrich, St. Louis, MO, USA). Phalloidin-TRITC was purchased from Yeasen Biotech (Shanghai, China). Mouse polyclonal LC3 antibody was bought from Cell Signaling Technology (Danvers, MA, USA). ZP2 rabbit monoclonal antibody was obtained from Abclonal Technology (Wuhan, China). Alexa Fluor 594 goat anti-mouse antibody was from Invitrogen (Carlsbad, CA, USA). MitoTracker Deep Red FM used to stain mitochondria was obtained from Invitrogen (Molecular Probes Eugene, OR, USA). Annexin V-FITC Apoptosis Detection Kits, HRP-conjugated goat anti-rat second antibody and mitochondrial membrane potential assay kit with JC-1 were bought from Beyotime (Shanghai, China). ECL Plus was purchased from Beijing Solarbio Science & Technology (Beijing, China). mRNA Direct Kit, Reverse Transcriptase Kit, and SYBR Green qPCR kit were from TIANGEN Biotech (Beijing, China). Pregnant mare serum gonadotropin (PMSG) was purchased from Ningbo Second Hormone Factory (Ningbo China). All other chemicals were purchased from Sigma chemical company.
Animals and treatment
The Institute for Cancer Research (ICR) strain mice (5-week-old, 20–25 g) were purchased from Nanjing Qinglongshan Animal Breeding Center for all experiments. Mice were housed in a temperature-controlled room (25°C ± 2°C) with 12:12 h dark–light cycles, keeping them in free access to food and water. All animal procedures were performed according to the Ethics Committee of the Ministry of Anhui Normal University, China (ETHICAL CODE: AHNU-ET2021026). The experimental protocols were approved by Anhui Normal University Animal Research Institute Committee.
After the acclimation phase, the 6-week-old female mice (n = 40) were randomly divided into ACR (n = 20) and control groups (n = 20). ACR was dissolved in sterile distilled water and stored at 4°C. As described in previous studies,6,35 ACR was administered daily to the control and ACR-exposed groups at doses of 0 and 10 mg/kg/day, respectively, for four consecutive weeks. After study period, euthanasia of mice was carried out by intraperitoneal injection of 100 mg/kg pentobarbital sodium, followed by cervical dislocation, and reproductive organs were dissected. GV oocytes were harvested from ovaries for the following experiments.
HE staining and follicle counts
Ovaries were fixed in 4% paraformaldehyde for 12 h and then stored in 70% ethanol before being embedded in paraffin. The samples were serially sectioned at 6 μm thickness, and HE staining was performed according to standard histological procedures. 36 Follicles on every fifth section were counted. Follicles were staged based on oocyte diameters and morphological features, as previously described by Camlin et al. 37
GV oocyte collection and in vitro maturation (IVM)
The mice were sacrificed via cervical dislocation 44–46 h after injecting them with 10 IU PMSG, and the large follicles were ruptured to release cumulus–oocyte complexes (COCs) in M2 medium. After three times of washing, the COCs were cultured in 50 μL drops of prepared M16 medium at 37°C in a humidified atmosphere with 5% CO2 for 12 h. The ratio of maturation was detected by the first polar body extrusion.
Oocyte activation, in vitro fertilization, and cell counting
After maturation, oocytes were immersed in 0.1% hyaluronidase for 5 min to remove cumulus cells. Then, oocytes were incubated in Ca2+-free CZB oocyte-activating medium containing SrCl2 and cytochalasin B for 6 h. Spermatozoa for IVF were isolated from the cauda epididymis of 8–12-week-old male mice. The spermatozoa were placed in HTF medium and capacitated for 1 h. Next, approximately 2 × 106 fully capacitated spermatozoa were co-incubated with the oocytes for 6 h. Oocytes with one or two pronuclei were considered as being activated or fertilized. Then, the activated and fertilized oocytes were transferred into KSOM medium for 96 h, respectively. The number of 2-cells, 4-cells and blastocysts were recorded during culture process. Cell number of per blastocyst was counted by counting the number of Hoechst 33342 stained nuclei.
Sperm‐oocyte binding assays
Caudal epididymal sperms were collected from mature untreated male mice and capacitated in HTF medium for 1 h at 37°C. Matured MII oocytes were immersed in M2 medium supplemented with 0.1% hyaluronidase to strip their cumulus cells. For performing zona pellucida–free binding assays, the zona pellucida was digested by 0.5% streptomycin protease. Both sperm–zona and sperm–oolemma binding oocytes were incubated with capacitated sperms at a concentration of 1 × 106 per droplet in HTF medium for 15 min. Oocytes were fixed in 2% paraformaldehyde for 5 min, and the bound sperms were quantified using phase microscopy (Ni-E, Nikon, Japan).
Immunofluorescence and confocal microscopy
For actin and LC3 staining, oocytes were fixed in 2% paraformaldehyde (w/v) for 15 min, then incubated with 0.25% Triton X-100 (v/v) for 15 min at room temperature, blocked in PBS containing 1% BSA for 1h, then incubated them in 1:100 dilution of mouse polyclonal LC3 antibody or 100 nM of Phalloidin-TRITC at 4°C overnight. After three washes in PBS, oocytes were incubated in Alexa Fluor 594 conjugated goat anti-mouse secondary antibody for 1 h. Then, they were co-stained with Hoechst 33342 (10 μg/mL in PBS) for 15 min. Finally, oocytes were mounted on glasses and measured by confocal microscope (Leica TCS SP8, Germany).
Annexin-V staining to detect early apoptotic oocytes
According to the manufacturer’s instructions, oocytes were stained with the Annexin-V staining kit, Briefly, after three washes in PBS, oocytes were then transferred into 195 μL of binding buffer, which contained 5 μL of Annexin-V-FITC for 10 min in the dark. Subsequently, fluorescent signals were measured by confocal microscope.
Distribution of mitochondria and Δφm staining
To determine the evenness of mitochondrial distribution, oocytes were incubated in 200 nM MitoTracker Red at 37°C for 30 min. After three washes in PBS, oocytes were co-stained with Hoechst 33342 for 15 min. Finally, oocytes were mounted on glasses and measured by confocal microscope.
To analyze Δφm, oocytes were evaluated using a JC-1 assay kit. Oocytes were incubated with 10 μM JC-1 at 37°C for 20 min, washed three times in PBS, and then mounted on glass slides for measurement by confocal microscope. According to the manufacturer’s instructions, ratios of red to green fluorescence were used to analyze the Δφm. Fluorescence intensity of oocytes were analyzed by ImageJ software V1.8.0 (National Institutes of Health, Bethesda, MD, USA).
Western blotting
Cumulus-free oocytes (n = 50) obtained by in vitro maturation were lysed in a microfuge tube containing RIPA buffer and PMSF for 10 min on ice, and the tubes were heated at 95°C for 5 min. Total proteins were separated on 12% SDS-polyacrylamide gel and transferred onto PVDF membranes. After three washes in TBST, the membranes were blocked with 5% non-fat milk in TBST for 1 h at room temperature. The membranes were incubated with ZP2 rabbit monoclonal antibody (1:500 dilution) in TBST containing 5% non-fat milk. After washed three times in TBST, membranes were incubated with HRP-conjugated goat anti-rat second antibody (1:1000 dilution) for 2 h at room temperature. The signals were measured by ECL Plus. For internal controls, β-actin was also measured.
RNA extraction and quantitative PCR
Primer sequences for qPCR.

Statistical analysis
All experiments were repeated at least three times, and each sample had three replications. Data are expressed as the mean ± Standard Error of Mean (SEM). Student’s t-test analysis of variance was used to compare differences between the control and treatment groups by using Prism V8.0.2.263 (GraphPad software Inc., San Diego, CA, USA), and a value of p < 0.05 was considered to be a statistically significant.
Results
ACR exposure alters the ovarian index and follicle population
In this study, we first examined ovarian variations in ACR-exposed female mice. The ovarian index of ACR-exposed mice was significantly lower than those of control mice (5.98 ± 0.53% vs 4.21 ± 0.42%, Figure 1(a)). The histological assessment showed that the number of follicles was reduced in ACR-exposed mice (58.67 ± 4.33 vs 37.00 ± 2.19, Figure 1(b)−(d)). ACR-exposed mice contained fewer antral follicles (12.78 ± 1.71 vs 6.95 ± 0.99, Figure 1(e)) and corpora luteum (6.27 ± 1.44 vs 2.53 ± 0.13, Figure 1(e)) than the control mice, but primordial, primary, and secondary follicle populations were unchanged. These results indicate that ACR exposure alters follicular development and corpus luteum formation (Figure 1). Effects of ACR on ovarian index and follicle population. (a) Ovarian index of control and ACR-exposed mice. (b) Representative image of ovaries in control and ACR treated mice by HE staining. Bar = 500 μm. (c) Different classes of follicles in the ovaries of mice. (d) Average number of follicles per ovary. (e) Different classes of follicles in ovaries of control and ACR-exposed mice. Each treatment was repeated at least four times and each replicate containing one mouse. Dates are expressed as mean ±SEM. *p < 0.05 vs control, **p < 0.01 vs control
ACR exposure affects the diameter, zona thickness, and chromatin configuration of GV oocytes
Considering ACR-exposed reduced the number of follicles in the ovaries, we further evaluate some quality indexes of GV oocyte. The ACR-exposed mice had a widely reduced diameter of fully grown GV oocytes (101.10 ± 1.001 μm, vs 95.32 ± 1.062 µm, Figure 2(a)). We also found that zona thickness of GV oocyte was decreased in ACR-exposed mice (9.48 ± 0.25 µm, vs 8.68 ± 0.26 µm, Figure 2(a)). Two mainly patterns of chromatin configuration were classified into NSN-type and SN-type (Figure 2(a)). The percentage of SN-type oocytes was decreased (78.54 ± 3.41 vs 63.33 ± 3.29, Figure 2(a)) and NSN-type oocytes was increased (21.46 ± 3.41 vs 36.67 ± 3.29, Figure 2(a)) in ACR-exposed mice. These results suggest that ACR exposure impairs the oocyte diameter, zona thickness, and chromatin configuration patterns. Effects of ACR on the diameter, chromatin configuration, sperm binding, and the developmental potential of oocytes. (a) Representative image of oocytes and chromatin configurations from control and ACR-exposed mice. Graphical representation of the average diameter, zona thickness, and percentages of chromatin configurations of GV oocytes. Bar = 20 μm. (b) Representative image of sperm binding to the surface of MII oocytes. Graphical representation of average number of sperm bound. Bar = 50 μm. (c) Representative image of sperm binding to the surface of zona-free MII oocytes. Graphical representation of the average number of sperm bound. Bar = 50 μm. (d) Protein levels of ZP2 in ovulated MII oocytes in control and ACR-exposed groups. (e) Percentage of pre-implantation embryonic development following Sr2+ activation and the cell number of per blastocyst. (f) Percentage of pre-implantation embryonic development after IVF and the cell number of per blastocyst. Each experiment was repeated four times, and in each replicate, 15–30 oocytes were observed. Dates are expressed as mean ±SEM. *p < 0.05 vs control, **p < 0.01 vs control, ***p < 0.001 vs control.
ACR exposure weakens sperm–oocyte binding ability
The first step in successful fertilization is the interaction of the sperm with the zona pellucid surrounding the oocytes, so we followed by a sperm–oocyte binding assay in both sperm–zona and sperm–oolemma of unfertilized oocytes. The oocytes from ACR-exposed females showed a significant decrease in both sperm–zona (50.25 ± 1.93 vs 21.44 ± 1.13, Figure 2(b)) and sperm–oolemma binding (24.58 ± 0.88 vs 12.42 ± 0.54, Figure 2(c)), suggesting that ACR exposure reduces the sperm–oocyte binding sites in unfertilized oocytes. In addition, we also detected the protein level of ZP2 which provides the sperm-binding site on the oocyte. In ACR-exposed unfertilized oocytes, the total protein level of ZP2 was markedly reduced compared to controls (Figure 2(d)). Together, these results suggest that ACR exposure inhibits the expression of ZP2, which may explain why the loss of sperm–oocyte binding sites occurs in MII oocytes.
ACR exposure affects pre-implantation embryonic development
The mature oocytes from IVM were activated or fertilized by IVF and then the embryos were cultured in KSOM medium for 96h. The parthenogenetic activation rate was significantly reduced in ACR-exposed oocytes (77.32 ± 5.26% vs 57.61 ± 3.12%, Figure 2(e)). Furthermore, the number cells of per blastocyst of activation (41.46 ± 0.69 vs 33.96 ± 0.76, Figure 2(e)) and IVF (38.97 ± 0.66 vs 36.19 ± 0.68, Figure 2(f)) was significantly reducing in ACR-exposed group compared with the controls. Collectively, these results suggest that ACR exposure impairs the developmental potential of GV oocytes (Figure 2).
ACR exposure disrupts the mitochondrial distribution of mouse oocytes
To further explore the mechanism through which ACR reduces the developmental potential of oocytes, we assessed the mitochondrial distribution of GV oocytes. In mammals, the mitochondria are maternally inherited organelles, and the function of the mitochondria is crucial for the developmental competence of oocytes. The mitochondria of control oocytes were distributed uniformly in the cytoplasm, while they were more clustered and asymmetrically distributed in the ACR-exposed group (Figure 3(a)). The fluorescence intensity curves of typical oocytes were consistent with those described in Figure 3(a) and (b). Additionally, ACR exposure reduced the fluorescence intensity of MitoTracker in GV oocytes (1 vs 0.20 ± 0.03, Figure 3(c)). These results indicate that ACR impairs the developmental potential of oocytes by disrupting the distribution of mitochondria. Effects of ACR on mitochondrial distribution, quantity and Δφm in GV stage oocytes. (a) Representative image of abnormal distribution of mitochondria in control and ACR-exposed GV oocytes. White arrow indicates aggregation. Bar = 25 μm. (b) Representative image of aberrant mitochondrial distribution pattern and the fluorescence intensity curves of GV oocytes from control and ACR-exposed mice. (c) Relative fluorescence intensity of MitoTracker Red in control and ACR-exposed GV stage oocytes. (d) Representative image of GV oocytes from control and ACR-exposed mice for Δφm staining. Bar = 25 μm. (e) Fluorescence emitted by each GV oocyte was analyzed using ImageJ software (red/green). Each experiment was repeated three times, and in each replicate, 15–30 oocytes were observed. Dates are expressed as mean ±SEM. **p < 0.01 vs control. ***p < 0.001 vs control.
ACR exposure alters the mitochondrial Δφm of mouse oocytes
We also elucidated the effect of ACR on mitochondrial Δφm values in GV oocytes. As shown in Figure 3(d), ACR decreased the magnitude of Δφm. Statistical analysis suggested that ACR exposure induced a significant reduction of Δφm values (1.07 ± 0.03 vs 0.96 ± 0.02, Figure 3(e)), suggesting that ACR weakens the quality of oocytes by decreasing the Δφm levels. (Figure 3).
ACR exposure affects actin filament assembly in mouse oocytes
Actin filaments are essential for polar body extrusion and cytokinesis. As shown in Figure 4(a), the accumulation of actin signals significantly decreased after ACR treatment. The actin fluorescence intensity from the lineation also confirmed that ACR exposure inhibited actin filament assembly both in cortex (1 vs 0.46 ± 0.03, Figure 4(b)) and cytoplasm (1 vs 0.71 ± 0.05, Figure 4(b)) of GV oocytes. These results suggest that defects of actin distribution may be one reason for the failure of meiosis and embryo cleavage in ACR-exposed mice. (Figure 4). ACR exposure affects the distribution of actin in mouse GV oocytes. (a) Representative image of GV oocytes from control and ACR-exposed mice for Phalloidin-TRITC staining (Red). The fluorescence intensity curve was constructed based on the samples depicted in the left panel. Bar = 25 μm. (b) Relative fluorescence intensity of actin in the membrane and cytoplasm of GV oocytes. Each experiment was repeated at three times, and in each replicate, 15–30 oocytes were observed. Dates are expressed as mean±SEM. **p < 0.01 vs control, ***p < 0.001 vs control.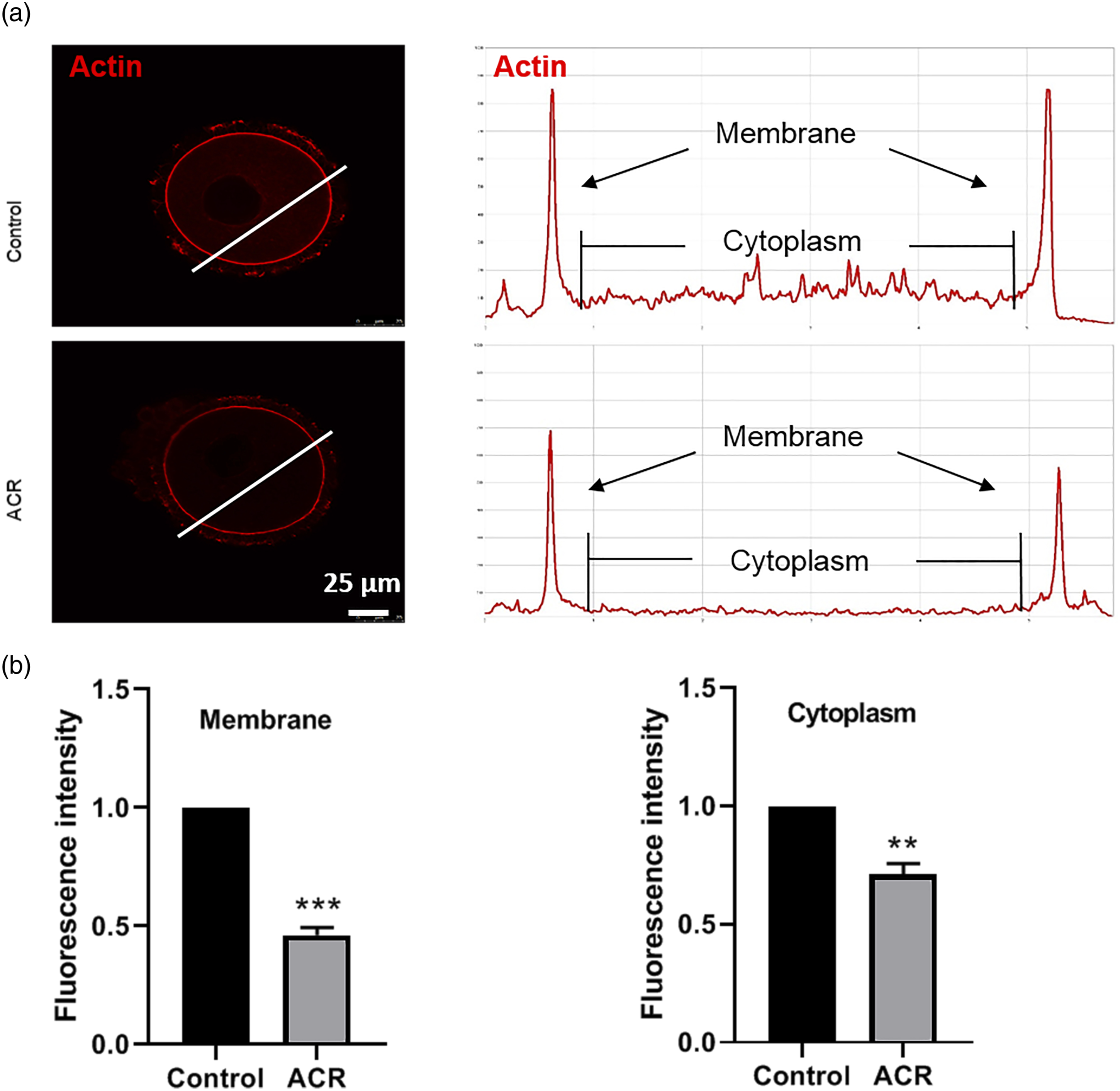
ACR exposure induces early apoptosis and autophagy of GV oocytes
To examine the mechanisms underlying the effects of ACR exposure on the developmental potential of oocytes, we detected whether autophagy was occurred in the oocytes of ACR-exposed mice. There were more clearly red dots, representing LC3 fluorescent signals, which appeared in ACR-exposed oocytes than that in controls (Figure 5(a)). The relative fluorescence intensity of LC3 (1 vs 21.131 ± 0.056, Figure 5(b)) and ratio of autophagy (6.25 ± 1.38% vs 22.50 ± 3.38%, Figure 5(c)) also confirmed that ACR exposure could lead to autophagy. Our results also found that ACR-induced autophagy was associated with an increase in the mRNA expression of LC3, ATG3, and ATG5 and a decrease in that of Beclin1 and mTOR, which play critical roles in autophagy (Figure 5(d)), suggesting that autophagy may be one of the mechanisms underlying the reduction of oocyte quality. Effects of ACR on mouse GV oocyte autophagy and apoptosis. (a) Representative image of LC3 staining in control and ACR-exposed GV oocytes. Bar = 25 μm. (b) Relative fluorescence intensity of LC3 in control and ACR-exposed GV oocytes. (c) Percentage of LC3-positive oocytes in control and ACR-exposed mice. (d) Relative expression levels of genes associated with autophagy in control and ACR-exposed GV oocytes. (e) Representative image of GV oocytes from control and ACR-exposed mice for Annexin-V staining. Bar = 25 μm. (f) Ratio of early apoptosis oocytes in control and ACR-exposed mice. Each experiment was repeated three times, and in each replicate, 15–30 oocytes were observed. Dates are expressed as mean±SEM. *p < 0.05 vs control, **p < 0.01 vs control, ***p < 0.001 vs control.
Additionally, we detected early apoptosis of mouse oocytes by the Annexin V-FITC assay. As shown in Figure 5(e), Annexin-V green signals were observed in the membrane and zona pellucida of ACR-exposed oocytes compared with controls. The proportion of early apoptotic oocytes was significantly increased in the ACR-exposed group (26.08 ± 2.89%, vs 54.14 ± 5.21%, Figure 5(f)). These results indicate that ACR exposure induces early apoptosis in mouse oocytes. (Figure 5).
Discussion
Contamination of food with ACR is a serious global problem, affecting human and animal reproductive health. In the present study, we examined the toxicity of ACR in GV oocytes and elucidated the potential mechanisms.
The Joint FAO/WHO Expert Committee on Food Additives (JECFA) estimates a 5.1 μg ACR intake/BW kg/day for large consumers. 38 Owing to many uncertainty factors in the assessment, the actual intake may be higher than the estimated values. Thus, considering the different dietary habits and ACR levels in food, we used an oral dose of 10 mg/kg/day of ACR in this study, which is equal to 0.81 mg/kg/day in humans, 39 approximately 150 times the intake of large consumers, although this is still lower than the dose administered in previous studies.6,14
Previous studies have associated ACR exposure, not only with the carcinogenicity and genotoxicity on somatic cells, but also with reproductive toxicity.1,3,8 Several studies have reported that the oral administration of ACR to female mice causes a significant reduction in ovarian weight and follicle number.6,14 These results indicate that ACR exposure directly affects the reproductive system, which is consistent with our results of reduced ovarian index to body weight and follicle populations. Histological examination showed that the number of antral follicle and corpora lutea were reduced in ACR-exposed females, which indicates that the toxic effects of ACR may be on fully grown follicles and corpus luteum formation. The ACR exposure decreased the diameter and zona thickness of GV oocyte, and these parameters change can impair the GVBD and fertilization competent of oocyte.21,40 Before NSN-type oocytes transit to SN-type, sufficient materials must be accumulated for GVBD, meiotic resumpution and blastocyst formation.41,42 The SN-type GV oocyte have more capacity than NSN-type oocyte in meiotic process and pre-implantation development.23,25 Here, we found that big proportion of NSN-type oocytes could not transition to SN-type oocytes in ACR-exposed females. In addition, the NSN-to-SN transition of chromatin configuration occurs in antral follicles, which is consistent with the deceased of antral follicles in ACR-exposed females. These results suggests that ACR exposure impairs the follicle development and oocyte quality in mice, but the time information for these parameters changes require further investigation.
Additionally, the sperm-binding ability of MII oocytes was severely impaired in ACR-exposed mice, and interestingly, the zona thickness of GV oocytes becomes thinner. The zona pellucida (ZP) of mouse oocytes is composed of ZP1, ZP2, and ZP3 proteins. 43 The deletion of these three genes results in abnormal oocyte development, early embryo loss, and even infertility in mice.44–46 Before fertilization, ZP2 is intact and provides the sperm–oocyte binding site, once fertilization is completed, ZP2 will be cleaved.47,48 In this study, we found that the protein level of ZP2 was reduced in ACR-exposed oocytes, suggesting that the decreased sperm-binding ability may be related to the inhibition of ZP2 expression. Furthermore, the development of pre-implantation embryo obtains from parthenogenetic activation and IVF was also impaired, indicating that ACR exposure attenuates the quality of oocytes may be before ovulation. The decline of fertilization and embryonic development may contribute to the decreased numbers of offspring of ACR-exposed females. 6 However, to verify these results and corroborate our understanding of how ACR exposure affects fertilization and offspring numbers, more studies utilizing female ACR mouse models at the fertilization and embryonic stages should be conducted.
Mitochondria are the powerhouse of cells, which generate ATP through oxidative phosphorylation. Mitochondria play a vital role in oocyte maturation, fertilization, and embryonic development. 28 A previous study reported that mitochondrial distribution is essential for human oocyte maturation and early embryonic development. 49 Furthermore, strong associations between abnormal mitochondrial distribution and meiosis spindle assembly or polar body extrusion have been reported. 50 In the present study, the abnormal mitochondrial distribution was observed among oocytes from ACR-exposed females, which might explain why polar body extrusion was reduced. Additionally, the mitochondria undergo a significant increase in Δφm during oocyte maturation, and the absence of an increase in Δφm results in decreased developmental potential of oocytes. 51 As the basis of mitochondrial respiration, Δφm is crucial for the conversion of ADP to ATP in the electron transfer chain. Our results showed that ACR induced greater depolarization of mitochondria. These results suggest that ACR affects the developmental competence of oocytes by disrupting mitochondrial distribution and Δφm.
Actin microfilament organization acts as a critical factor for oocyte meiotic maturation and fertilization. In our study, actin organization was significantly disrupted in both the membrane and cytoplasm of ACR-treated mice. ACR has been reported to disrupt the localization of γ–tubulin and p-MAPK, leading to aberrant meiotic spindle morphology. 6 However, normal mitochondrial function is crucial for actin assembly and spindle formation. Thus, these results indicate that ACR-disrupted oocyte actin organization may be related to mitochondrial dysfunction in mouse oocytes, which may be another reason why polar body failed to be extruded.
According to previous studies, autophagy and apoptosis are two important cellular processes for determining cell fate. 52 ROS accumulation can induce oxidative damage, which is accompanied by apoptotic and autophagic cell death. An abnormal mitochondrial Δφm can induce ROS production by affecting electron transfer. 53 In a previous study, oral ACR administration to female mice induced excessive ROS generation, 6 which echoes our result of abnormal Δφm. The present results indicate that an increased early apoptosis rate may contribute to mature and activated embryonic development failure. Additionally, autophagy plays an essential role during maturation and fertilization, which affects maternal mRNA degradation and apoptosis in parthenogenetic embryos. 34 Furthermore, autophagy affects apoptosis in mouse embryos and is regarded as a cellular response to oxidative stress. 54 Here, we found that autophagy was occurred in the oocytes of ACR-exposed mice. Thus, induction of apoptosis and autophagy in oocytes may be involved in the decreased developmental potential.
In conclusion, we used an orally administered mouse model to provide evidence that ACR exposure adversely affects the quality of GV oocytes. Our results indicate that ACR exposure causes a reduction in the developmental potential of GV oocytes, by impairing chromatin configuration, sperm-binding ability, and embryonic development through the induction of mitochondrial dysfunction, abnormal actin assembly, and autophagy/apoptosis. (Figure 6). These findings elucidate the possible mechanisms underlying the developmental limitation of oocytes in ACR-exposed mouse. The effect of ACR exposure on the developmental potential of germinal vesicle oocytes. ACR is spontaneously produced through the Maillard reaction and widespread in a variety of carbohydrate-rich foods. When adult female mice were exposed to ACR, the developmental potential of GV oocytes were reduced by inducing abnormal actin assembly, mitochondrial dysfunction, and autophagy/apoptosis.
Footnotes
Acknowledgments
The authors are thankful to the Wannan Medical College for providing necessary research facilities to carry out the present research work.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This study was supported by the National Natural Science Foundation of China (No. 31970499) and Key Scientific Research Project of Wannan Medical College (No. WK2020Z08).
