Abstract
Background
To enhance the therapeutic potential of Tinospora cordifolia phytosomes, a chloroform extract was prepared.
Objectives
The goal of this study was to create and test the therapeutic potential of a phytosome containing T. cordifolia extract.
Materials and Methods
The extraction of nonpolar compounds was carried out using chloroform as solvent. The active constituent of the extract was subjected to phytochemical and Gas Chromatography–Mass Spectrometry (GC–MS) analyses. Docking studies against multiple targeted proteins confirmed the anti-diabetic, anti-inflammatory, and analgesic properties of T. cordifolia extract. The phytosomes of T. cordifolia extract were prepared by solvent evaporation technique. The phytosomes were characterized for vesicle size, entrapment efficiency, surface morphology, FTIR, in vitro drug release and in vivo anti-diabetic, anti-inflammatory, and analgesic activities.
Results
The highest concentration was found to be 16.58% for epiyangambin. The reported compound inhibits GLUT1 and COX2 with 9.25 and 8.34 kcal/mol binding scores. The optimized TCP4 exhibited 454.2 ± 8.1 nm of vesicle size, –43.1 ± 7.5 mV of zeta potential, and 0.33 of polydispersity index (PDI). The phytosomes exhibited a spherical shape confirmed by TEM analysis. The formulation TCP4 showed a significantly higher release (94.7% ± 1.7%) than pure extract. TCP4 exhibited potent therapeutic potential for anti-diabetic, anti-inflammatory, and analgesic activity.
Conclusion
Chloroform extract loaded phytosomes with cholesterol as lipids exhibited significant therapeutic potential due to the presence of epiyangambin.
Introduction
Herbal formulations consist of individual or combined herbal extracts used to treat various diseases. 1 Due to the rising use of allopathic drugs, herbal remedies have lost their significance. 2 Before introducing the allopathic system, traditional medicine has operated for over two thousand years. 3 The effectiveness of plant-based traditional medicine is gaining attraction due to its cheap cost and minimum adverse effects. 4 India and China reported consuming approximately 1000 tons of Tinospora cordifolia (Willd.) per year.5, 6 The primary active constituents are alkaloids, furano diterpenoids, norditerpenoids, sesquiterpenoids, phenolics, lignin, and sterols. It also contained aliphatic compounds, polysaccharides, essential oils, and fatty acids.7, 8 T. cordifolia (TC) is used as bitter tonics in jaundice, arthritis, diabetes, and other skin ailments.9–11 The whole plant extract is responsible for hepatoprotective, anti-ulcer, and antioxidant properties. The extract of the stem exhibits the hepatoprotective effect. In addition, stems also showed hepatoprotective, anti-pyretic, cytotoxic, anti-diabetic, and immunomodulatory properties.12–17 The roots have emetic, anti-stress, antioxidant, anti-ulcer, anti-diabetic, and visceral obstructions. 18 Hyperbilirubinemia and rheumatic disorders are treated with dried fruits, whereas diabetes is treated with the leaves. 19
The structure-based drug design is widely accepted for determining the binding affinity with specific proteins. Strong protein–ligand interaction indicates a high binding relationship between specific proteins and ligands. The molecular docking approach is applied to simulate a biomolecular interaction, and it provides data on the affinity of each ligand. 20
A lipid-compatible molecular complex between lipids and phytoconstituents is termed phytosomes. 21 It enhanced permeation and intestinal permeability compared to pure phytochemical extracts. It also has a preventive impact on herbal extracts by shielding them from digestive fluids and destroying gut flora. They may readily pass through the bilayer membrane and bloodstream. 22 The lipids and nonionic surfactants generate unilamellar or bi-lamellar structures of phytosomes. Spans and tweens have better entrapment and release characteristics among the nonionic surfactants. 23
Type 2 diabetes is a complex disease associated with genetic defects. Type 2 diabetes is 20 times more common compared to type 1. Patients with prolonged exposure lead to overweight and obesity due to poor lifestyles and uncontrolled diets. The chances of type 2 diabetes increase with an increase in age. Because of their high incidence and common risk factors, type 2 diabetes and arthritis are linked illnesses. Approximately 48% of patients who have type 2 diabetes are associated with the complication of arthritis. Aging is also a critical risk factor for diabetes and arthritis. Different treatment options are available to manage arthritis and diabetes. 24
The present research work involves extracting the phytoconstituents from T. cordifolia using chloroform and identifying the phytoconstituents by Gas Chromatography–Mass Spectrometry (GC–MS). The molecular docking analysis was applied to determine the anti-diabetic, anti-inflammatory, and analgesic effects of the active ingredient. Phytosomes of T. cordifolia chloroform extract was developed by the reverse phase solvent evaporation method. Phytosomes were evaluated for various parameters, that is, physico-chemical characterization, morphological examinations, and in vitro release study. In vivo anti-diabetic, anti-inflammatory, and analgesic effects of phytosomes were also evaluated.
Materials and Methods
Chemicals and Reagents
T. cordifolia hydroalcoholic extract was procured from Herbal Creations (Nainital, India). Span 60, cholesterol, and soyalacethin were purchased from Sigma Aldrich (Bangalore, India). Aspirin, glibenclamide, and indomethacin were procured from Dr. Reddy’s Laboratory (Hyderabad, India). All solvents and other chemical reagents of analytical grade were procured from Fischer Scientific Laboratory (Mumbai, India).
T. cordifolia Extract Preparation
An amount of 500 ml chloroform was added into 200 g of defatted hydroalcoholic extract and macerated for 24 h with continuous shaking. The chloroform layer was collected and evaporated to dryness to get the semisolid mass. The extract was air-dried and stored in an amber color container for further study. 25
Phytochemicals Analysis
A detailed phytochemical investigation was carried out for chloroform extracts. The chemical tests were performed for alkaloids, glycosides, terpenoids, phenolics, lignans, steroids, aliphatic compounds, polysaccharides, essential oils, and fatty acids. 26
GC–MS Analysis
Agilent 7890A GC System (Model: AccuTOFGCv/JMS-T100GCv, Tokyo, Japan) was used for GC–MS analysis as per previous published work with slight modification. 25 Electron Impact Ionization (EI) positive ionization source mass spectrometry was used to identify the separated chemicals. The structure of the compounds was determined by mass spectral data from the National Institute of Standard and Technology (NIST) library database. 25
Preparation Structures for Docking Analysis
Chemdraw ultra 12.0.2 and Chem 3D Pro 12 were used to create the structure of the identified molecule in mol2 format.
Analysis of Docking
The docking analysis was performed with the Swiss Dock analysis tool (
Formulation of T. cordifolia Loaded Phytosomes
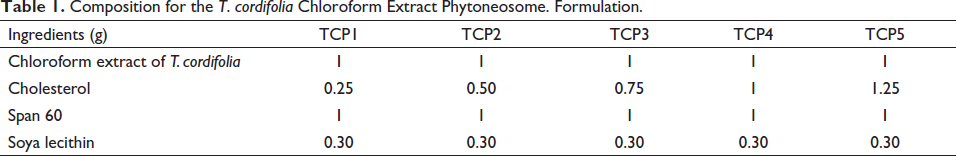
The phytosomes containing T. cordifolia chloroform dry extract were prepared using the reverse phase solvent evaporation method. 27 Table 1 shows the compositions of several phytosome preparations. The varying quantities of cholesterol and soya-lecithin as per given in the Table 1 were transferred in the conical flask and dissolved with ether (15 ml). The required quantity of span 60 (1 g) was dissolved in methanol (5 ml) and mixed with lipid mixture. One gram of extract was added to the lipid surfactant mixture and stirred magnetically at 500 rpm (Remi magnetic stirrer 2MLH, Mumbai, India). Precisely 10 ml of phosphate buffer (pH 7.4) was transferred in a round bottom flask at 1 ml/min. The resultant mixtures were refluxed at 60°C–65°C for 1 h with continuous stirring. A rotary flash evaporator was used to recover the organic solvent at 65°C and 50 rpm (Cyberlab Corporation, CR2001, Delhi, India). The aqueous phase was homogenized at 8000 rpm for 2 h at 4°C (Remi homogenizer, RQT-1274, Mumbai, India). Finally, the phytosomes were sonicated at 8°C using a probe sonicator (Citizen Digital ultrasonicator, CD4820) for 5 min at 30 s intervals. The formulation was stored in an amber glass bottle at 2°C–8°C for further study.
Composition for the T. cordifolia Chloroform Extract Phytoneosome. Formulation.
Entrapment Efficiency (% EE)
The ultracentrifugation technique was used to measure the entrapment efficiency. Two milliliters of freshly prepared phytosomes were centrifuged for 30 min at 10,000 rpm (Remi cooling centrifuge, Remi, India). Carefully pipette out 1 ml supernatant liquid and added 9 ml of buffer and made the final volume 10 ml. The absorbance was analyzed by UV–Visible spectrophotometer (Lab India-UV 3000) at 216 nm. The % EE was calculated by following formula.
28
Each study was carried out in triplicate and data was represented in the form of mean ± SDs.
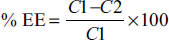
where C1 is the drug concentration in the liquid supernatant and C2 is the total amount of extract in the formulation.
The Size Distribution, Zeta Potential and Polydispersity Index (PDI)
The Malvern Zetasizer (Malvern Instruments, Southborough, UK) was used to study the vesicle size, PDI and zeta potential of phytosomes. 29 The diluted sample was filled in the cuvette and analysis was carried out at 25°C ± 2°C and 90° scattering angle. The measurement was done at a 1.330 refractive index and 0.050 absorptions. The zeta potential of the sample was analyzed using an electrode cuvette. Each study was carried out in triplicate and data was represented in the form of mean ± SDs.
Transmission Electron Microscopy (TEM)
TEM instrument (JEM 2100F, Tokyo, Japan) was used for morphological examination of phytosomes. On a 200-mesh carbon-coated copper grid one drop of a sample dropped. Remove excess solvent by blotting with filter paper. The samples were air-dried and placed into TEM and the image was captured using software.
FTIR Analysis
Bruker instrument (3000 Hyperion Microscope with Vertex 80 FTIR System, Germany) equipped with Micro ATR, Grazing angle was used for FTIR analysis of samples. Approximately 2 mg of the samples were applied to the sample plate and scanned at 4000–500 cm–1 with a spectral resolution of FTIR 1.0 cm–1.
In vitro Release and Release Kinetic Studies
The USP dissolution apparatus (Dissolution tester, USP, TDT-08L, Elctrolab, Mumbai, India) was used for the release study using a dialysis bag. 30 The 900 ml of freshly prepared phosphate buffer (pH 6.8) was filled into basket as release media and maintained at 37°C ± 0.5°C in a pretreated dialysis bag (Dialysis Membrane-60, Av. flat width 25.27 mm, Av. diameter 15.9 mm, approximate capacity 1.99 ml/cm, Mumbai, India) 5 ml of the sample (250 mg equivalent extract) was filled and fitted with pad of dissolution apparatus and rotated at 50 rpm. The 5 ml sample was collected at predefined intervals and replaced with 5 ml of fresh medium to maintain a constant volume. The concentration of the drug was analyzed by UV-spectrophotometric at 216 nm. The absorbance data were used to calculate the percentage of cumulative drug release (% CDR). The release profile fitted to zero order, first order, Higuchi diffusion, and Korsmeyer–Peppas models. The correlation coefficient (R2) was calculated to determine the best-fitted releases model and mechanism of drug release. Each study was carried out in triplicate and data was represented in the form of mean ± SDs.
In vivo Study
Animals
Wister rats (180 and 200 g) of either sex were used for anti-diabetic, anti-inflammatory, and analgesic activity. Wister mice of 20–25 g of either sex were selected for analgesic activity. The animals were housed in standard climatic conditions for 24 h before the experiment. The Institutional Animal Ethical Committee (IAEC) approved all of the experimental protocols of Translam Institute of Pharmaceutical Education and Research, Meerut (Reg. Number 1207/PO/RE/S/08CPCSEA, Ref. no. TIPER/CPCSEA/21/247).
Anti-diabetic Properties
Induction of Diabetic Condition
Insulin-dependent diabetes is induced by intraperitoneal administration of a single dose of streptozocin (25 mg/kg) and continuous supply of sugar solution for drinking. 28 The animal was standing for 72 h for stabilization of blood glucose level. Blood glucose level was determined by taking blood samples from the dorsal vein. Fasting blood glucose levels of 270–310 mg/100 dl is considered a diabetic condition.
Dose Schedule and Experimental Procedure
The rats were dived into four groups and each group contained six rats. Group 1 is a normal control (10 ml/kg normal saline), group 2 is disease control, group 3 is treated with glibenclamide (50 mg/kg), group 4 is treated with pure T. cordifolia extract (250 mg/kg), and group 5 is treated with phytosomes (TCP4, 250 mg/kg). Treatment was done for 21 days and at different time intervals (1, 7, 14, and 21 days) and the blood glucose level was measured using glucose oxidase-peroxidase (GOD-POD) kit (Accurex, India).
Biochemical Parameters
All biochemical parameters like total cholesterol, high-density lipoprotein (HDL), low-density lipoprotein (LDL), very low-density lipoprotein (VLDL), triglycerides, creatinine, urea, and uric acid were measured using published methods. 31 Commercially available kits were used to carry out the estimations by Erba diagnostic kit Mannheim GmbH, Mallaustr, Germany. The concentrations of different biochemical parameters were determined using the auto-analyzer Erba Chem-7. 32
Histological Examination
A semi-automated microtome (RM2155 Leica Microsystems) was employed to slice the paraffin-embedded pancreas.34 The tissue pieces were placed on a glass, slide and deparaffinized with xylene and 70% ethanol. Hematoxylin and eosin were used to stain the sections. All slides were viewed at a magnification of X200 using light microscopy (Yamtoo 100, Japan).
Anti-inflammatory Activity
Carrageenan-induced hind paw edema technique was used to evaluate the anti-inflammatory effect.
34
Rats were divided into four different groups and each group having six rats. Group 1 was treated with normal saline (10 ml/kg), group 2 treated with indomethacin (50 mg/kg), group 3 treated with pure T. cordifolia extract (250 mg/kg), and group 4 treated with TCP4 (250 mg/kg). After one hour of therapy, 1% carrageenan was injected into the left hind leg to induce edema. The rats were given water at a 2 ml/100 g body weight to limit variance in edema development. Paw volume measured up to 5 h at 1 h of interval. A decrease in paw volume is considered an anti-inflammatory action.

Acetic Acid-Induced Writhing in Mice
This study was conducted on mice. The mice were divided into four groups and each group contain six mice. Group 1 received normal saline 10 ml/kg, group 2 received aspirin 100 mg/kg, group 3 treated with pure T. cordifolia extract (250 mg/kg), group 4 received TCP4 (250 mg/kg). Glacial acetic acid solution (IP injection (0.6% v/v, 1 ml/kg glacial acetic acid solution) was injected 30 min before receiving the treatments. The number of writhes was counted for 3 h with 1 h of interval. The % inhibition was calculated by the given formula. A decrease in the number of writhes indicates the presence of analgesic properties.
35

Statistical Analysis
The data were analyzed using Graph Pad Prism 5 (GraphPad Prism 5.00.288). The data were presented as mean and standard error mean (SEM), with a one-way analysis of variance (ANOVA). Dunnett’s test was used to compare the control and test substances (p ≤ .05 considered significant).
Results and Discussion
Percentage Yield and Phytochemical Analysis
Extraction was carried out by the cold maceration method using chloroform as solvent. The percentage yield was found to be 29.8%. Phytochemical screening showed the presence of alkaloids, flavonoids, steroids, phenolic compounds, lignin, terpenoids, and aliphatic chemicals in the extract.
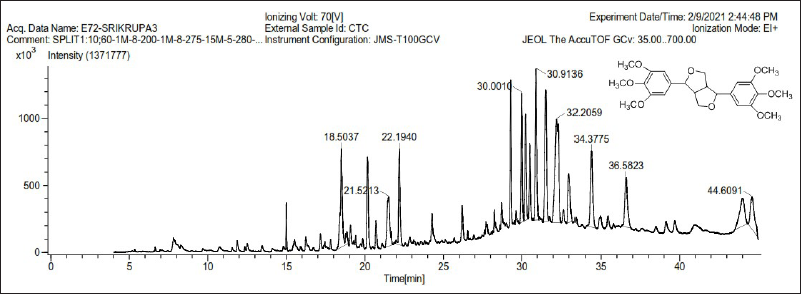
GC–MS analysis
The GC–MS chromatogram of the T. cordifolia extract is shown in Figure 1. Epiyangambin exhibited in highest concentration as compared to another compound (16.58% at 32.50 min). 25 Chemically it is a furofuran lignan with PAF antagonist activity. 36
Docking Analysis
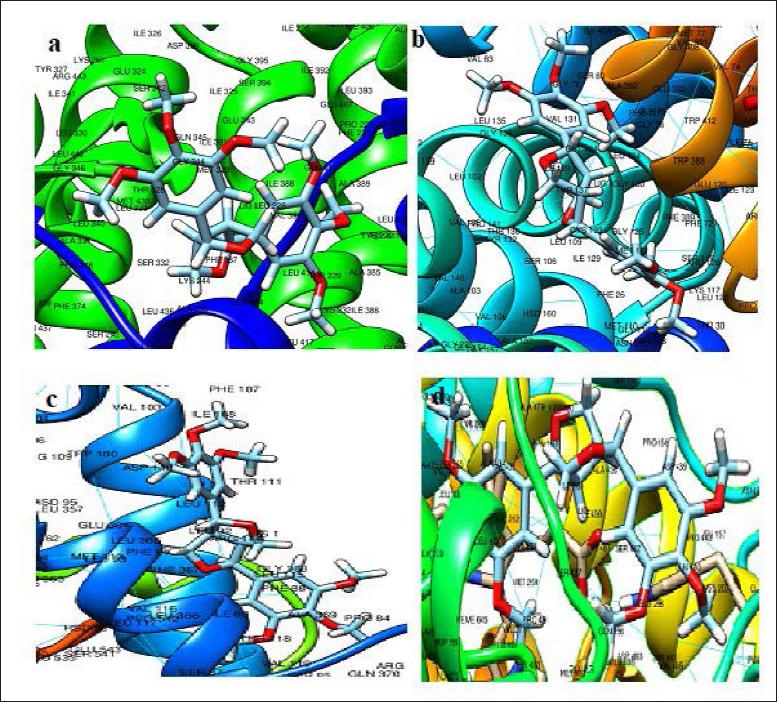
The ligand–protein interactions with strong binding affinity are represented in Figure 2. The epiyangambin exhibited good binding affinity against peroxisome proliferator-activated receptor gamma ligand (PPAR-γ), glucose transporter 1 (GLUT-1), cyclo-oxygenase-1 (COX1) and cyclo-oxygenase-2 (COX-2) inhibition, and compared to standards (rosiglitazone, glibenclamide, aspirin, and celecoxib). The binding energies (∇G) were found to be –8.84, –9.25, –8.24, and –8.34 kcal/mol, respectively. The molecule epiyangambin has more GLUT-1 and COX-2 inhibitory action. 25 High blood sugar levels promote the formation of reactive oxygen species (ROS), which are chemicals that stimulated the release of pro-inflammatory cytokines (substances secreted by specific immune cells). It also causes the formation of chemicals known as advanced glycation end products (AGEs), which accumulate in and destroy joints. 37 Lignins have limited medicinal applications because of the lack of solubility in aqueous and organic phases. 38 Phytosomes are considered an ideal approach to improving the therapeutic efficacy of water-insoluble phytoconstituents. 21 The solubility profile of lignin epiyangambin and its GLUT-1, and COX-2 inhibitory action suggests further formulation of phytosomes of the extract to increase oral bioavailability and therapeutic efficacy.
Ligand Protein Interaction of CEC7 with 5ycp (a), 4pyp (b), 1eqg (c), and 3ln1 (d).
Formulation Development
The extract-loaded phytosomes were successfully prepared by the solvent evaporation method. The composition of all phytosomes was given in Table 1.
Vesicle Size, PDI, and Zeta Potential
Table 2 represented the result of vesicle size, PDI, and zeta potential of all phytosomes formulation. The vesicle size, PDI, and zeta potential of all phytosomes were found in the range of 265.4–814.5 nm, 0.23–0.57, and 40.8–58.1 mV (negative), respectively. It was observed that cholesterol concentration increased the vesicle size because it interferes with the closed packing of the lipid bilayer and increases the fluidity of the membrane. 39 In addition, the PDI of phytosomes increases on increasing the cholesterol concentration because of vesicle size increases. 40 Zeta potential of phytosomes increases by increasing the cholesterol concentration. A high value of zeta potential indicated the high stability of formulation (more repulsion between vesicles) and that vesicles did not coagulate with each other.
Vesicle Size, Zeta Potential and PDI of Optimized Phytosomes.
Entrapment Efficiency
The entrapment efficiency is an important parameter because of its direct effect on the concentration of the drug for pharmacological activity. The results of entrapment efficiency were given in Table 2. All the formulations exhibited % EE in the range of 55.8%–92.1%. The entrapment efficiency of the extract in phytosomes increases by increasing cholesterol concentration because cholesterol increases the rigidity and permeability of lipid bilayer and prevents the leakage of the drug, hence increasing the entrapment efficiency. 41
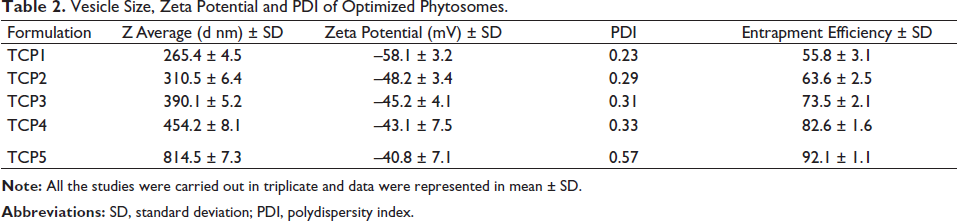
On the basis of optimum vesicle size, PDI, zeta potential and entrapment efficiency, the formulation TPC4 was selected as the best formulation. The optimized phytosomes TPC4 have a vesicle size of 454.2 ± 8.1 nm (Figure 3a), PDI of 0.33, zeta potential of –43.1 ± 7.5 mv (Figure 3b) and entrapment efficiency of 82.7% ± 1.6%, respectively. TPC4 is used for further evaluation.
(a) Vesicle Size Distribution Graph of TCP4 and (b) Zeta Potential Analysis TCP4.
TEM Analysis
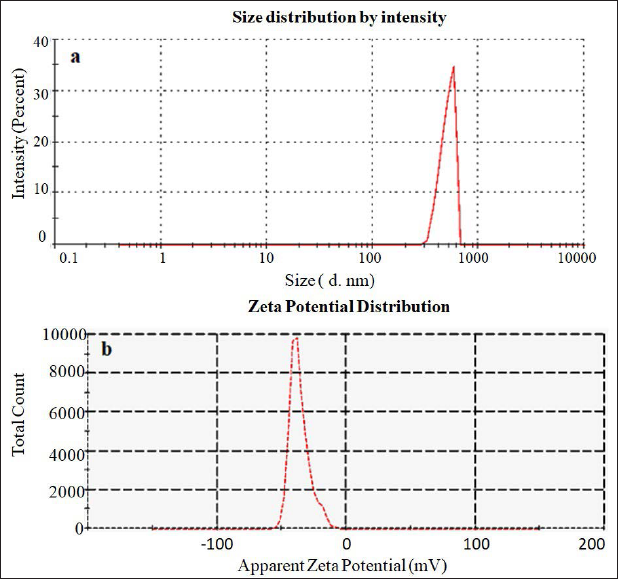
The morphology of the optimized TCP4 formulation was analyzed by TEM instruments and the image was shown in Figure 4 and found to be a spherical shape. The concentric bilayer vesicle was formed due to hydrophobic and hydrophilic interaction between lipid and drugs. The nonionic surfactants reduce the interfacial tension between both surfaces. 42 The shape of the phytosomes depends on the nature of the lipid and the temperature of the hydration medium. The hydrogen bonding interaction of lipid with surfactant is essential for encapsulation efficiency and release rate. 43 The stronger interaction reduces vesicle size and release rate. The asymmetric vesicle morphologies are stable; promote cellular polarity and consequently biological effectiveness. 44
HR-TEM Image of TCP4.
FTIR Analysis
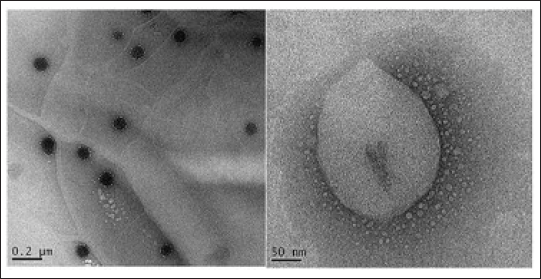
The FTIR spectra of pure T. cordifolia extract, cholesterol, span 60 and optimized formulation (TCP4) were recorded and spectra were depicted in Figure 5. FTIR spectra of pure TC extract exhibited characteristic peaks at 3391 (–OH, str), 1620 (C=O, str), 1384 (–C–O–, str), and 1153 (–C–N, str). The cholesterol exhibited characteristic peaks at 3423 (O–H, str), 2934 (CH3, str), 1712 (C–H, str), and 1672 (C=C, str). The span 60 exhibited characteristic peaks at 3392 (O–H, str), 2956 (–CH3, str), 2850 (–CH2–, str), 1736 (C=O, str), and 1415 (–C–O–, str). However, the TCP4 exhibited characteristic peaks at 3380 (O–H, str), 2956 (CH3, str), 2850 (–CH2–, str), 1600 (C=C, str), 1737 (C=O, str), and 1467 (–C–O–, str). In TCP4 all the characteristic peaks of pure T. cordifolia extract present without significant shift revealed that there is no interaction between drug and excipients. The change in intensity of the characteristic IR peaks also confirmed the formation of van der Waals forces, repulsive forces, entropic repulsive forces and short-acting repulsive forces, and so on. among surfactant lipid and extract molecules. The van der Waals forces provide stability in the phytosomes. The repulsive forces played an important role in drug release characteristics. 45
In vitro Incompatibility Study by FTIR Analysis for Optimized Phytosomes.
In vitro Release Study
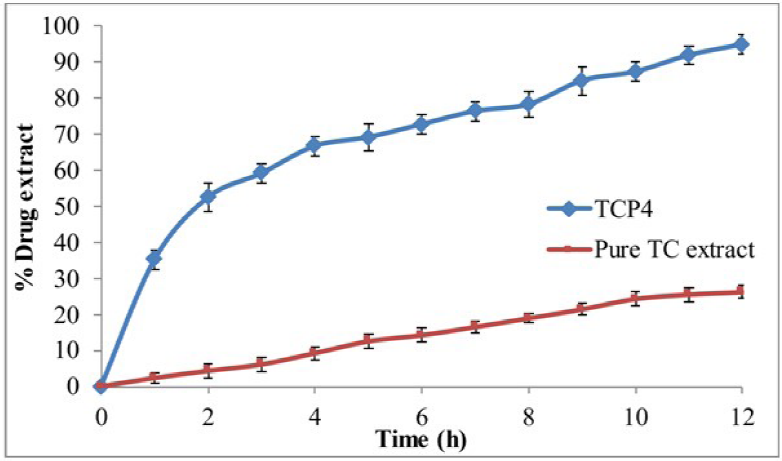
In vitro release study of pure T. cordifolia extract and optimized formulation TCP4 dispersion was analyzed by dialysis bag and data were represented in Figure 6. The in vitro release study was done to determine the release behavior of drugs from the formulation in body circulation. The optimized formulation TCP4 showed initial fast-release (49.43% in 1 h) and later sustained-release (94.7% ± 1.7% in 12 h). The higher release of extract from phytosomes is due to the surface deposition of a drug as well as increased solubility due to the presence of lipid and surfactant in the formulation. The surfactant and lipid reduced the interfacial tension between the aqueous and lipid phase. However, the pure extract exhibited only 26.2% ± 1.8% in 12 h, which is due to the low aqueous solubility of the extract.
In vitro Drug Release Study of Phytosomes and Pure T. cordifolia Extract.
Drug Release Kinetics
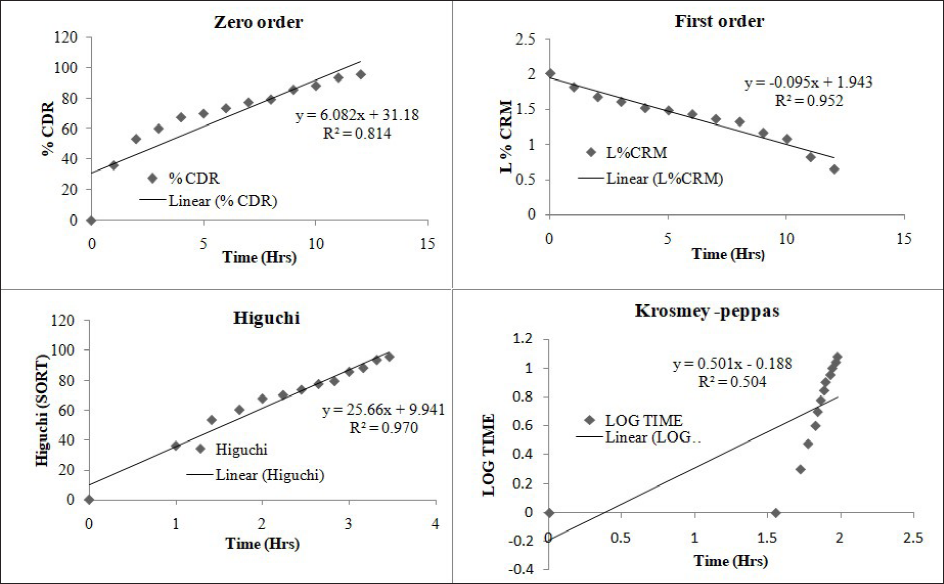
Drug release kinetics of optimized formulation (TCP4) was assessed by plotting zero-order, first-order, Higuchi, and Korsmeyer–Peppas model through Microsoft Excel as shown in Figure 7. The mean correlation coefficient (R2) is 0.814 for zero-order, 0.952 for first order, 0.970 for Higuchi and 0.504 for Korsmeyer–Peppas model. The maximum R2 was found to be more maximum for the Higuchi model (R2 = 0.97) than in other models. So, Higuchi is the best fit kinetic release model. The release exponent is 0.532 indicates correlation coefficient value of drug release.
In vitro Drug Release Kinetics Diagrams of Optimized Formulation (TCP4).
Anti-diabetic Activity
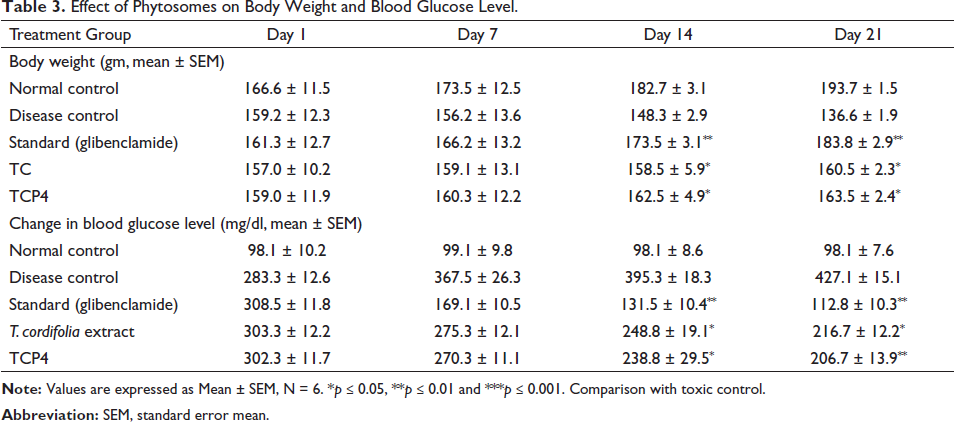
The anti-diabetic activity for 21 days of the study were given in Table 3. The body weight of toxic control rats was 136.6 ± 1.9 g at 21 days. However, the body weight of the glibenclamide and TPC4 treated groups increases significantly i.e., 183.8 ± 2.9 g for the glibenclamide group, 160.5 ± 2.3 g for T. cardifolia extract and 163.5 ± 2.4 g for TCP4 group on 21 days treatment. The blood glucose levels of disease control groups was 427 ± 15.10 mg/dl at 21 days. The glibenclamide group and TCP4 treated group showed significant decreases (p < .05) in blood glucose levels up to 21 days of study which is 112.8 ± 10.31 mg/dl (glibenclamide), 216.7 ± 12.2 mg/dl (TC) and 206.7 ± 13.9 mg/dl (TCP4) respectively. T. cordifolia exhibits stimulatory glucose absorption. The TPC4 demonstrated anti-hyperglycemic action owing to epiyangambin inhibition of GLUT1. GLUT1 and GLUT3 do not require insulin to translocate glucose, and these are abundant in human tissues. These are the most prevalent glucose transporter isoforms expressed in the adult brain and are responsible for baseline glucose absorption. 46
Effect of Phytosomes on Body Weight and Blood Glucose Level.
Biochemical Parameters
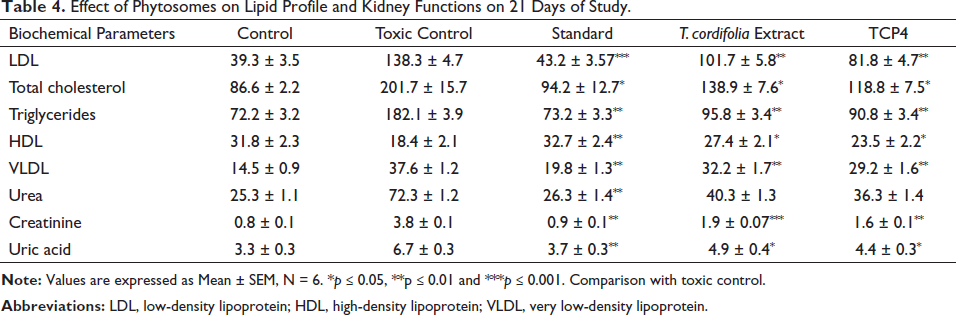
The biochemical evaluation was done on the 21st day of treatment and data was expressed in Table 4. The result showed that glibenclamide, pure T. cordifolia extract and TCP4 caused a significant (p < 0.05) decrease in lipid levels, for instance, cholesterol, LDL, VLDL, and triglycerides, except for HDL, as compared to disease control. Similarly, kidney function tests, for instance, creatinine, urea, and uric acid also exhibited a significant decrease (p < 0.05) in the levels as compared to disease control. Destruction of pancreatic β cells is responsible for the decrease in glucose-stimulated insulin release. Due to the reduction of plasma insulin, a change in lipid profile occurred. The movement of fatty acids from peripheral tissues is also responsible for the increase in lipid levels. This condition is caused by insulin-sensitive lipase inhibition. 47 Similarly, urea, creatinine, and uric acid are important renal parameters, especially in the case of type 2 diabetic patients. 48 Different types of complications like hyperlipidemia, edema, obesity, and diabetic nephropathy are evoked due to increased lipid levels and alteration in kidney functions, which are crucial factors in developing arthritis, gout inflammation, and pain.
Effect of Phytosomes on Lipid Profile and Kidney Functions on 21 Days of Study.
Histopathological Study
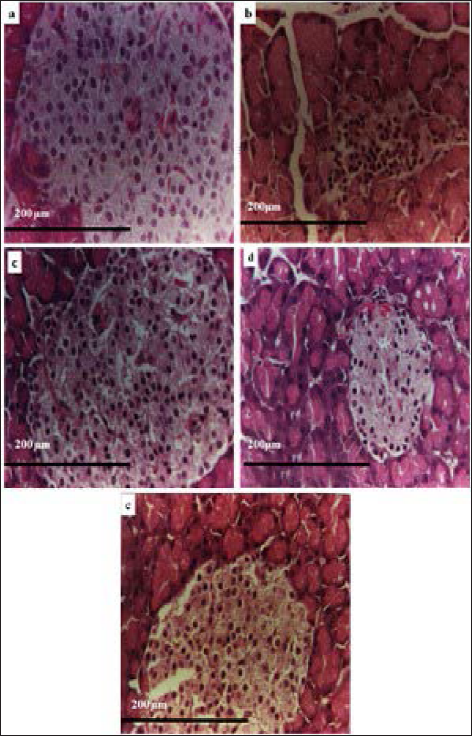
The histopathological examination of rat pancreatic cells is depicted in Figure 8. The normal control group of rats showed normal cellular morphology in islet cells (Figure 8a). There was necrosis and shrinkage of islet cells with dilated acini of the pancreas observed in diabetic rats (Figure 8b). The treatment group with glibenclamide showed regular pancreatic architecture, increasing islet size, along with the proliferation of β-cells (Figure 8c). However, pure T. cordifolia extract and TCP4 also exhibited increases in the islet cells with dilated acini than extract and disease control (Figure 8d and e). It revealed that T. cordifolia extract-loaded phytosomes have good anti-diabetic activity due to an increase in the solubility of lipid and surfactant.
Histopathological Image of (a) Normal Control, (b) Disease Control, (c) Treated with Glibenclamide, (d) Treated with T.cordifolia Extract, and (e) Treated with TCP4 of Pancreas.
Anti-inflammatory Activity
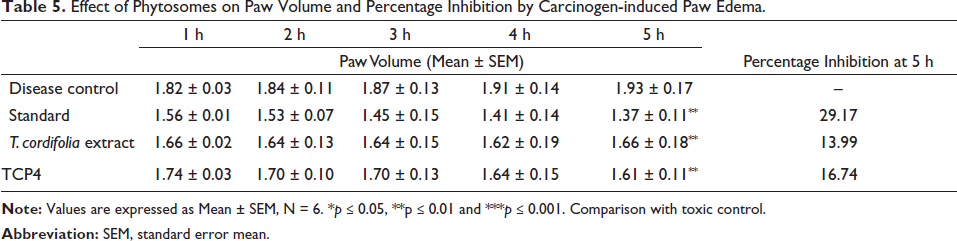
Table 5 shows the results of the anti-inflammatory activity of indomethacin and TCP4. It was observed that indomethacin, T. cordifolia extract and TCP4 exhibited significant inhibition in paw edema (29.17%, 13.99%, and 16.74%) as compared to disease control. The significant anti-inflammatory activity of TCP4 is due to the increased solubility of extract as well as the high release of extract.
Effect of Phytosomes on Paw Volume and Percentage Inhibition by Carcinogen-induced Paw Edema.
Analgesic Activity
The result of analgesic activity was depicted in Table 6. TCP4 exhibited good analgesic effects (43.19%) than disease control. However standard (aspirin) exhibited analgesic activity (78.26%) than T. cordifolia extract (37.55%) and TCP4 (43.19%). The analgesic and anti-inflammatory activity of T. cordifolia-loaded phytosomes (TCP4) are due to the inhibition of COX-2. 49 The significant activity of T. cordifolia in the formulation may be due to high solubility in the lipid system.
Conclusion
The phytochemical analysis reported the presence of lignin as one of the major phytoconstituents. GC–MS chromatogram reported the highest concentrations of lignin compound epiyangambin. The reported compound also exhibited good anti-diabetic, anti-inflammatory, and analgesic potential by GLUT-1 and COX-2 inhibition. Phytosomes were prepared by a reverse-phase solvent evaporation method using span 60, soyalecithin, and cholesterol as formulation ingredients. Formulations exhibited high entrapment efficiency and nanoscale of vesicle size. The morphological behavior of vesicles with bilayer membranes validates their spherical form. The FTIR peaks show no interaction between the extract and excipients. The physico-chemical and morphological studies revealed that formulation at the equal ratio of drug lipid and surfactant demonstrated sustained-release behavior with the Fickian diffusion mechanism. Phytosomes loaded with T. cordifolia extract showed significant anti-diabetic, anti-inflammatory, and analgesic activity compared to extract alone. This might be due to the presence of epiyangambin in the formulation and enhanced solubility in the lipid and surfactant of the formulation.
Footnotes
Acknowledgments
All the authors would like to provide sincere thanks to Principal SY Manjunath and the management of Srikrupa Institute of Pharmaceutical Sciences for their continuous support to complete the work successfully. We would also like to acknowledge SAIF IIT Bombay for providing FTIR and TEM analysis facilities. We would also like to give our heartiest thanks to NIPER Hyderabad for conducting particle size and zeta potential analysis. Finally, we would like to express our appreciation to the Director of the Translem Institute of Pharmaceutical Education and Research in Merrut for approving the animal study and carrying out all of the experiments.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
