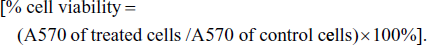Abstract
Background
Research studies have focused on discovering new anti-proliferative and pro-apoptotic agents derived from natural products from which honey constitutes a prominent candidate. The Acacia honey (AH) is known to display anticancer activity, but the mechanisms of action are still not well defined.
Objectives
Using in vitro and computational approaches, we aimed to assess the interaction among selected bioactive compounds derived from AH, with the apoptotic protein p53, which could trigger apoptosis.
Materials and Methods
The phytocompounds of AH were investigated via gas chromatography–mass spectrophotometry analysis. The cytotoxic effect and induced apoptosis on the MCF-7 breast cancer cell line were assessed by 3-(4,5-dimethylthiazolyl-2)-2,5 diphenyltetrazolium bromide and acridine orange-ethidium bromide staining approaches. The molecular docking analysis between AH compounds and p53 was carried out.
Results
The drug-likeness prediction revealed that most of the identified compounds meet Lipinski’s rules. We demonstrate that AH exerts an interesting cytotoxic effect in a dose-dependent manner against the MCF-7 cell line with IC50 5.053µg/mL. Significant cell alterations and notable induced apoptosis were detected when cells were treated with AH. The molecular docking analysis revealed that melezitose is among the most important potential bioactive compounds that interact with p53 leading to apoptosis. The binding affinity was −8.1 kcal/mol, and the closest molecular interactions in the active site included 10 residues, which could explain the potential biological activity.
Conclusion
This work sheds light on AH as a significant source of bioactive chemicals with potential for promoting apoptosis that may be exploited as an alternative therapy for breast cancer.
Introduction
Nowadays, cancers place a huge health burden on healthcare systems, mainly breast cancer, which is the most diagnosed cancer in women. In 2020, the number of diagnosed cases reached around 2.3 million cases with 685,000 deaths. 1 This rate may increase and could top 4.4 million cases by 2070. 2 This increase could be mainly due to epidemiological and demographic transitions the world has faced over the past few decades. Early diagnosis and treatments contribute considerably to overcome breast cancer concern.3, 4 Surgery, radiotherapy and chemotherapy via cytotoxic drugs are among the most common strategies used to treat breast cancer. These approaches remain invasive methods. Their efficiency is mainly related to the stage in which cancer is detected. They become less efficient with metastatic forms. 5
For these reasons, a particular emphasis was placed on the need to explore new alternative treatments. Investigating natural products could provide valuable bioactive compounds to treat cancers. Among natural compounds, honey is a great source of bioactive phytochemicals due to its complex constituents. For a long time, honey has been used in folk medicine and was referred extensively to treat several diseases. Honey’s curative properties have been reported in several diseases including microbial infections and anti-hypertensive, anti-arrhythmic, wound healing, anti-inflammatory modulation, and anticancer effects. 6 These healing properties are mainly due to the prominent antioxidant effect that honey could display through flavonoids and phenolic acids compounds. 7
With the emergence of cancer drug resistance and the decrease of the conventional therapeutics’ efficiency, honey has attracted renewed interest as an alternative treatment provider. Several studies have been conducted, and interesting results have been reported. Recently, research studies have concluded that some honey subtypes could be used as a chemo-protectant or even as an adjuvant during cancer treatment. 8 Moreover, honey could be considered as a prominent chemo-preventive agent and some of its phytochemicals may be therapeutic in treating cancers. 9 Honey’s antioxidant constituents may inactivate the cytotoxic effect of free radicals formed during diverse cellular processes that could lead to the carcinogenic effect.
Outstanding in vivo and in vitro anticancer effect of many kinds of honey were disclosed for several cancer forms, mainly breast cancer. This displayed activity involves several mechanisms, mainly cancer cell growth inhibition via anti-proliferative, pro-apoptotic, and anti-metastatic effects. 10 An in vivo experiment showed that Tualang honey alleviates breast carcinogenesis through modulation of hematologic, estrogenic, and apoptotic activities in an experimental breast cancer animal model. 11 Another study revealed that Sidr and wild honey could improve breast cancer treatment by modulating several metastasis–progression gene expression on a human breast adenocarcinoma (MDA-MB-231) cell line. Several studies highlighted the potential of some honey to induce apoptosis in many cancer cell lines as well as breast cancer. The Tualang honey induced apoptosis in two-cell lineage of breast cancer MCF-7 and MDA-MB-231 by modulating the expression of apoptosis-related genes from p53. 12 Another study revealed that treating the breast cancer cell line with Acacia honey (AH) could inhibit cell growth after only 6 h via apoptosis inducing. 13 This observed cell growth inhibition could be the result of apoptosis mediated by cell cycle arrest. We can hypothesize through these observations that honey promptly induces cell cycle perturbation, which may lead to programmed cell death induction. Thus, inducing apoptosis using honey could be a targeted approach and valuable alternative to treat breast cancer.
Apoptosis is known as a programmed cell death that occurs after activation of a series of molecular steps to get rid of abnormal cells and preserve the body integrity. Most cancer cells escape apoptosis by deactivating effector proteins involved in apoptotic pathways, which can promote tumorigenesis and metastasis processes. The apoptosis process is triggered, which involves several proteins, mainly p53. The tumor suppressor protein p53 encoded by the gene TP53 plays a crucial role in inducing apoptosis in potential malignant cells, particularly in breast cancer; for that reason, p53 has been named as the cellular gatekeeper. In breast cancer, 40% of TP53 mutations will lead to instable protein. These mutations are associated with the worst survival in breast cancer patients. 14 Providing bioactive compounds derived from honey that is able to induce apoptosis via p53 activation could be a valuable alternative treatment in breast cancer. These compounds could also be used as potential adjuvants that may enhance response to conventional drugs to provide efficient treatment and ensure better outcomes.
Through this study, we aim to investigate the cytotoxic and apoptosis-inducing effect of AH produced in the Hail region on breast cancer MCF-7 cell lineage using in vitro and in silico approaches. To investigate the pro-apoptotic potential of AH bioactive compounds, we adopt the molecular docking method. It consists of predicting the potential binding modes between a compound and active binding site of a targeted protein.15–17 This method will give an estimation of potential affinity based on conformation and complementarity between AH phytocompounds and apoptotic protein p53 in our case. We investigated the thermodynamically possible interactions between identified compounds with p53 protein that may induce apoptosis in the MCF-7 breast cancer cell line. For that, we assessed the phytochemical profiling of the AH sample and characterize its specific constituents related to the floral source and geographical origin. Moreover, the identified phytochemicals will be evaluated for their pharmacokinetics, drug-likeness, and medical chemistry friendliness by conducting ADME properties.
Materials and Methods
Honey Sampling
AH (Acacia sp.) was collected directly from apiculture apiaries of the Hail region. It was conserved at 4°C in dark, dry, and sterile conditions. The mono floral origin was verified by microscopic observation of the predominant pollens in the honey sample.
Gas Chromatography-Mass Spectrophotometry (GC-MS) Analysis
For honey compound extractions, 5 g of AH was added to 50 mL of ethanol–water solution (50% v/v) and then mixed for 4 h at room temperature. To identify phytochemical compounds from AH, we conducted gas chromatography–mass spectrophotometry analysis. GC apparatus (GC-2030, Nexis, Shimadzu®, Kyoto, Japan) equipped with QP2020 NX-MS was used. Ten microlitre of honey extract was injected into the system, and helium was used as a carrier gas with a maintained flow rate of 1 mL/min for 20 min. The oven temperature was maintained between 60°C and 260°C. The MS of the detected compounds was compared with the National Institute of Standards and Technology (NIST) database and WHILEY libraries.
Antiproliferative Activity (MTT Assay)
The antiproliferative activity of AH on human cancers breast cell lines (MCF-7) was assessed by 3-(4,5-dimethylthiazolyl-2)-2,5 diphenyltetrazolium bromide (MTT) assay. The doxorubicin was used as a reference standard drug. MCF-7 cell suspension (2 × 105 cells/mL) was placed in 96-well Corning Costar® culture plates and grown in Dulbecco’s Modified Eagle Medium supplemented with 10% fetal bovine serum and then incubated at 37°C, 5% CO2, and 80% humidity. Then, cells were treated for 24 h with respective concentrations of AH: 2%, 4%, 6%, 8%, and 10% in triplicate. After treatment, cells were washed with phosphate buffered saline (PBS) solution, and then MTT solution (5 mg/mL) was added; again, cells were incubated for an additional 4 h. After the second incubation, the supernatant was removed and 100 µL of dimethyl sulfoxide was added in each well. The formazan crystals were dissolved in acidified isopropanol. Cell viability was determined by measuring the absorbance at 570 nm, and the percentage of viable cells was estimated to determine the 50% cytotoxic concentration IC50 value for the respective cancer cell line.
We used the inverted microscope method to evaluate the inhibitory effects of AH on the confluency of the MCF-7 cancer cell line.
Drug-likeness Prediction Analysis
In order to characterize phytochemicals found on AH, we predicted in silico the absorption, distribution, metabolism, excretion, and toxicity (ADMET) parameters using online servers and established commercial packages (
Docking Analysis
The molecular docking study was carried out to estimate the binding energies displayed by the identified phytochemicals toward the apoptotic protein p53 as target. The protein structure of p53 (PDB ID: 3DCY) was retrieved from the Research Collaboratory for Structural Bioinformatics‒Protein Data Bank (RCSB-PDB). 18 To determine the p53 active site for appropriate ligand binding affinity, we referred to the CASTp 3.0 server to predict the best binding pocket. The chemical structures of the honey-identified compounds were obtained from PubChem database in the sdf format. The docking approach was based on the CHARMm force field after processing the receptors by adding polar hydrogens and Kollman charges and removing the crystal water molecules in AutoDock Vina 1.1.2 program 19 and DS visualizer. The phytochemicals and proteins were converted into the PDBQT format by using the AutoDock tools for analysis. The greed box has been targeted by selecting some key residues of the protein pocket region.
Acridine Orange-Ethidium Bromide (AO-EB) Staining
The MCF-7 cells were plated in 6-well plates at an optimum density of 3 × 10 5 cells/per 2 mL. In a CO2 incubator, the cells were incubated for 24 h at 37°C. The spent medium was aspirated, and 1 mL of PBS was added. Following this, the cells were treated with the honey sample (IC50) and further incubated for 24 h before they were removed from the medium. The medium was then removed after the incubation, and a cold PBS was added to it. Afterward, 500 µL of AO-EB staining solution (10 µL of acridine orange [2 mg/mL] and 10 µL of ethidium bromide [2 mg/mL] in 1 mL of PBS) was added to the cells for staining. The mixture was thoroughly mixed and then incubated for 5 min. In the final step, the cells were washed three times with PBS and images were taken immediately under a fluorescent microscope (XDFL series, Sunny Instruments, China) to determine the viability of the cells.
Results
Compounds Identified
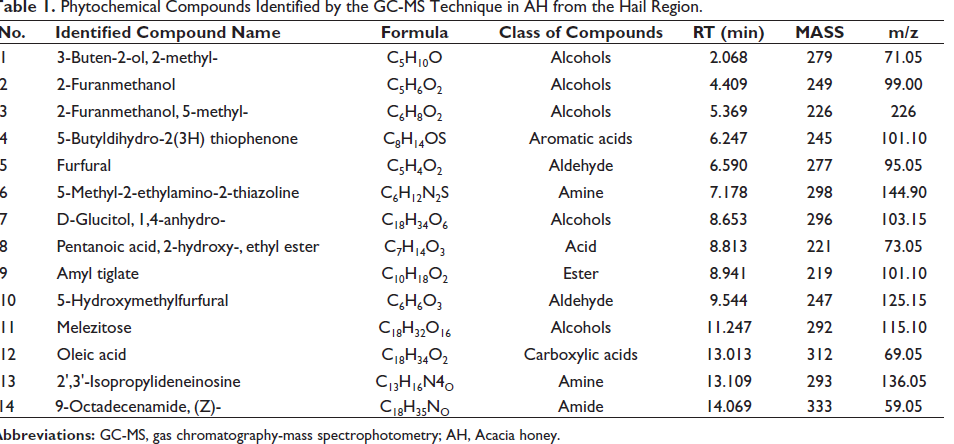
The phytochemical profiling was conducted via GC-MS. The compounds interpretation was achieved relaying on the mass spectrum and retention time. The comparison of the obtained mass spectra with the NIST and WHILEY libraries helped identify 14 peaks of compounds with high spectral matching accuracy (Table 1 and Figure 1).
Phytochemical Compounds Identified by the GC-MS Technique in AH from the Hail Region.
MTT Analysis
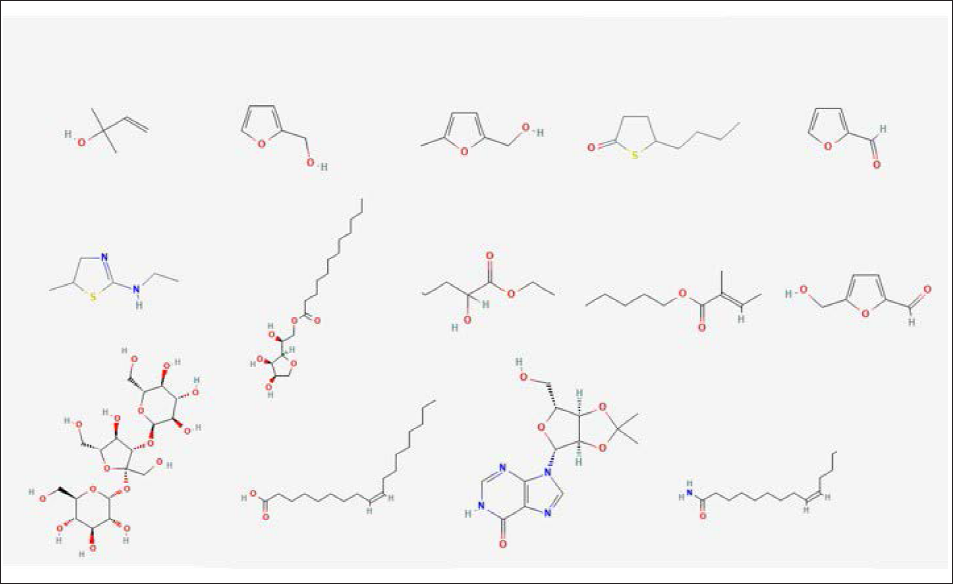
The cytotoxic effect of AH on the breast cancer cell line was assessed by MTT assay. MTT was absorbed into MCF-7 and converted into a purple colored-formazan complex inside mitochondria by succinate dehydrogenase enzyme. A cytotoxic parabolic profile was observed in favor of promising anticancer activity against breast cancer cell lineage (Figure 2). The IC50 value was 5.053 µg/mL. The IC50 of doxorubicin was used as reference against MCF-7 was 3.5 µg/mL. The treatment with increased concentrations of AH caused a gradual decrease in the confluency of the monolayer of MCF-7 cancer cells. Cells also rounded, shrank, and lost their alignment, and the intercellular spaces became larger (Figure 3). These observations highlight the cytotoxic effect displayed by AH.
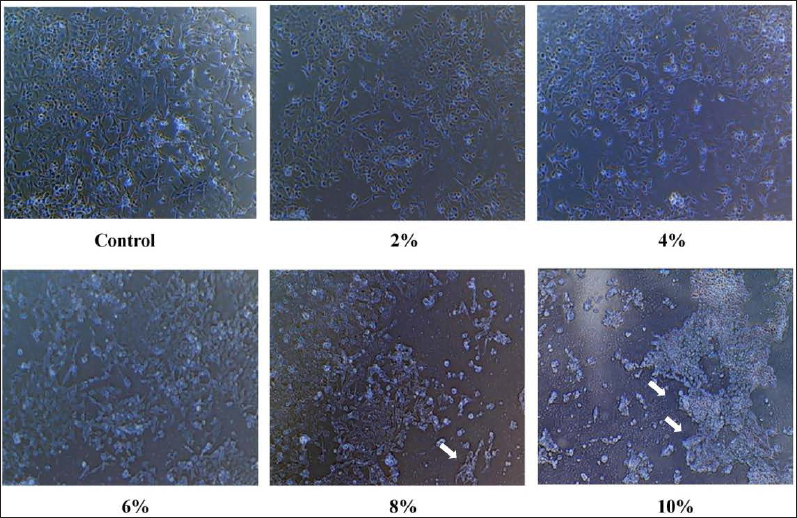
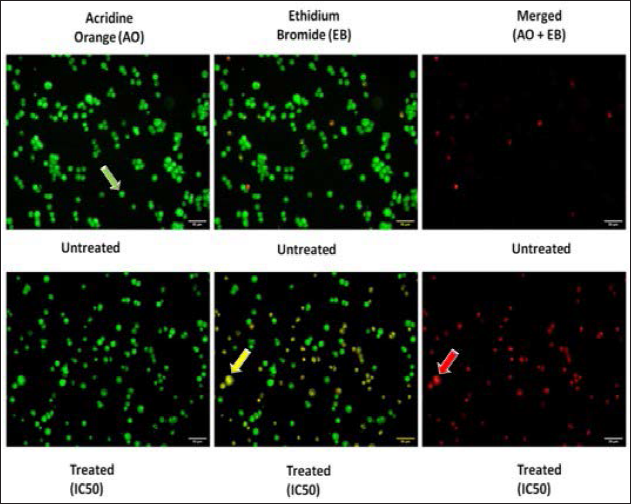
AH Induces Apoptosis in the MCF-7 Breast Cancer Cell Line
Fluorescent microscopy analysis with AO-EB-stained cells was carried out to determine whether the honey sample causes the apoptosis or not. Based on the results of using the staining method with these cells, it was clear that the morphology of the cancer cells treated with this method shows clear signs of apoptosis based on the staining method. There are two types of cells: green cells that are stained with only AO and orange cells that are stained with EB. The green cells are viable cells, while the orange cells are late apoptotic cells. Contrary to this, the green and orange cells with condensed chromatin that are stained with both AO and EB indicate that they are early apoptotic cells. According to the results of the experiments performed on MCF-7 cells that were treated with a honey sample, apoptosis is the most common mode of cell death, characterized by both chromatin condensation and membrane damage, which is a death caused by the loss of membrane integrity (Figure 4).
ADMET Analysis
We assessed the ADMET profiling for all identified compounds from AH. By applying Lipinski’s rule of five, 20 we can deduce that most of the identified compounds can be qualified as biologically active with good absorption and permeation except for compound 11, melezitose (Table 2). Despite that this compound presents exception to Lipinski’s rules, no alert has been detected similar to the other identified compounds.
Drug-likeness, Pharmacokinetics, and Physicochemical Properties of the Honey-identified Compounds (1−14) Based on the ADMET Properties.
Docking Analysis
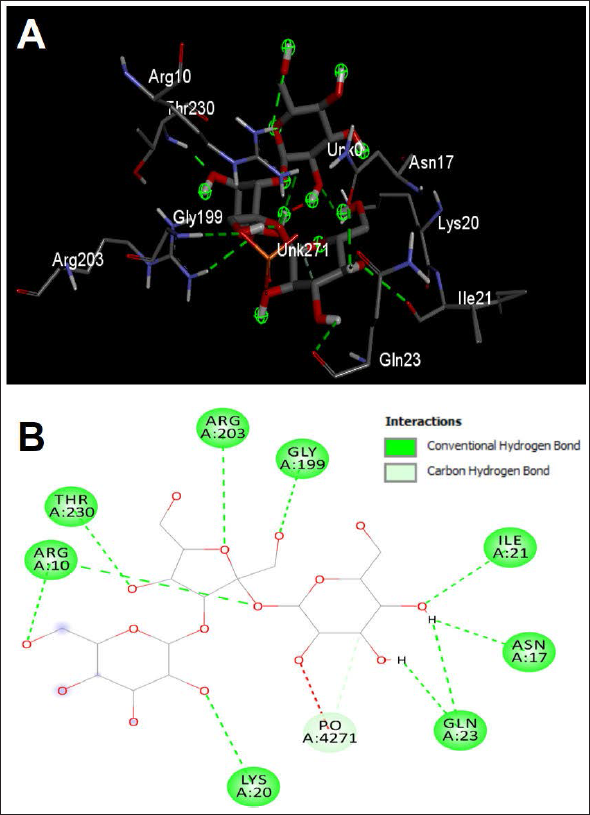
To investigate mechanistic effects of AH-identified compounds leading to the biological effects, we assessed the binding affinities and molecular interactions to p53. The selection of the reported positions was based on the best binding score and RMSD equal to zero. 21 The identified constituent compounds of AH exhibited negative binding energies (ranging from −4.1 to −8.1 kcal/mol) with the p53-targeted receptor (Table 3). Considering the number of conventional hydrogen bonds, the number of the closest interacting residues and the closest distance during the ligand–receptor complex, the best scores were obtained for the Melezitose compound (Figure 5).
Binding Affinity, Conventional Hydrogen Bonds and Interacting Residues of the Honey-identified Compounds with p53.
Microphotograph of Ligand_11 Bounded to the Pocket Region of the Targeted Protein (p53). (A) 3D Illustration and (B) Diagram of Interactions. Note that this Complex Presented the Highest Number of H-bonds, the Best Free Binding Energy, and the Deepest Embedding.
Discussion
Challenging cancer by proposing new alternative treatments derived from natural products, mainly honey, brings new effective therapeutics. This huge demand on phytocompounds to treat cancers is mainly due to the failure of conventional chemotherapeutic agents related to drug resistance as well as its multiple side effects. This highlights the potential role of honey as a source provider of potent bioactive compounds that could be used for therapeutic purposes in cancers. Bio-active compounds derived from honey are known for their negligible side effects, as they have been used for many years in traditional medicine. AH has been widely studied in the cancer field due to its prominent results observed in many cancer cell lineages. To identify chemopreventive and chemotherapeutic agents from AH, we performed the GC-MS analytical method. The analysis of the mass spectrum and retention time identified 14 elements belonging to different classes of compounds, including alcohols, aldehydes, aromatic acids, amine, acid, esters, carboxylic acids, amine, and amid. The volatile compound 3-buten-2-ol, 2-methyl- has been previously reported in several kinds of honey and designated as a potential marker of the botanical origin. 22 The 2-furanmethanol is previously reported in Malaysian Tualang honey and Sudanese polyfloral honey.24, 25 This compound contributes to antioxidant activity displayed by honey. 23 Studies investigating the furfural in several kinds of honey revealed that it may range between 0.34 and 2.23 mg/kg with the highest concentration recorded in thyme honey.26, 27 5-hydroxymethylfurfural (HMF) is a natural compound found in honey at the low level, and it is an indicator of honey quality. This compound is formed during the decomposition of monosaccharides via Maillard’s reaction. According to the Codex Alimentarius Commission, the HMF must not exceed 40 mg/kg. 28 Previous studies reported that HMF measured in several samples of AH do not exceed 5 mg/kg. 29 The melezitose is a trisaccharides reported in several kinds of honey, mainly in honeydews with a high amount. This compound is characterized by low solubility in water. A study reported a low acid hydrolysis and low α-glucosidase hydrolysis susceptibility, which may fulfil the prebiotic criterion of human indigestibility for melezitose. 30 This compound has promising pharmaceutical applications. It is used as an excipient to optimize pharmaceutical formulations and to stabilize proteins during recombinant protein therapy by protein glycosylation. 31 Oleic acid is among fatty acids mainly found in olive oil but also revealed in honey. 32 A great antioxidant potential has been reported in oleic acid with interesting therapeutic relevance. An in vivo study revealed that oleic acid could protect efficiently against cadmium-induced cardiac and hepatic tissue injury. 33 Moreover, many studies highlighted anti-tumor and anticancer effects for hepatocellular carcinoma cell lines and tongue squamous cell carcinomas with apoptosis inducing potential.34–36 Among the identified honey compounds in our AH sample, 9-octadecenamide was the only one amide revealed. Previous studies revealed that 9-octadecenamide might inhibit the spontaneous metastasis of BL6 skin melanoma cells. 37
Previous studies highlighted prominent anticancer effects displayed by AH. This activity is mainly due to the potent bioactive compounds, which are considered promising pharmacological agents to treat cancers. 38 Studies attributed anticancer properties essentially to flavonoids and phenol acids that could lead to cancer prevention and treatment by interfering in several cancer-related pathways. 39 Thus, honey could exhibit cytotoxic effects and inhibit cancer cell proliferation, even induce apoptosis. 40 Aliyu M et al. showed that AH could inhibit lung cancer cell line NCI-H460 proliferation through cell cycle arrest and downregulation of genes involved in apoptosis. 41 They also showed that AH could induce apoptosis in prostate cell line PC-3 through modulation of G0/G1 phase, pro-inflammatory cytokines, calcium ion secretion, and downregulation of prostate specific antigen. 40
In our study, we investigate the anticancer effect against breast cancer cell line MCF-7 via using an MTT assay approach. After treatment with several AH concentrations, we noticed that cancer cell line proliferation was inhibited in a dose-dependent manner. The obtained IC50 value was 5.053 µg/ml, while for doxorubicin, it was 3.5 µg/mL. Previous studies investigating AH derived from the Asir region (Southwest of Saudi Arabia) reported interesting anticancer properties against several cancer cell lines from breast cancer MCF-7. 7 Another interesting study revealed that AH derived from Malaysia could inhibit breast cancer cell MCF-7 with IC50 of 5.49 µg/mL. They reported that honey could induce apoptosis after 2 h of treatment and observed apoptotic bodies within 6 h. 13
The tumor suppressor gene p53 is among the most key apoptotic genes. This gene is involved in many complex metabolic processes, including repairing DNA damage, promoting cell cycle arrest, and inducing programmed cell death. It has been revealed that p53 is silenced in most cancer cells, leading to the loss of the protective effect, resulting in apoptosis resistance and sustained proliferation. 42 For that, several alternative therapeutic approaches have been established to discover pro-apoptotic phytocompounds. Thus, increasing the p53 expression level or reactivating the mutated form of p53 and inducing apoptosis are strategies to treat cancers.
In recent years, the ability of honey to inhibit cell proliferation, alter the cell cycle, and induce apoptosis in diverse cancer cell lines has been widely studied. 43 The aim is to discover new therapeutic phytocompounds that are able to interact with oncogenes and tumor suppressor genes that display prominent functions in cell cycle regulation. Evidence of promoting p53 in the cancer cell line when treated with honey has been highlighted. 43 Previous reports revealed that treating the cancer cell line with honey could increase p53 expression, which may induce apoptosis.44, 45 As previously reported by Aliyu et al., AH can induce apoptosis by downregulating Bcl-2 and p53 genes. 41
The anticancer potency of AH to the breast cancer cell line has been described,7, 13 but the molecular basis is still not well defined. The complexity of AH and the incomplete knowledge of its constituents and their mechanism of action constitute the main limits in using honey in conventional treatment for breast cancer. The absence of standardization of biological activities of AH compounds remains the major limit despite the prominent in vitro and in vivo results. For that reason, the identification of each compound, characterization of its pharmacological properties, and understanding its mechanism of action to treat cancer are important requirements. In our investigation, we tried to explore the potential of AH derived from the Hail region to induce apoptosis in the breast cancer MCF-7 cell line. Through our established assays, we observed interesting cytotoxic effects and induced apoptosis when cells were treated with IC50 concentrations of AH honey compared with those untreated ones. Among identified compound in our honey sample, two have been known for their anticancer activity, mainly oleic acid and 9-octadecenamide.36, 37
Through the computational study, we investigated the binding affinities and molecular interactions of AH-identified compounds to p53 protein. This approach provides information on the binding energy between the protein and ligands to identify potential compound that could trigger apoptosis by activating p53. Among the 14 identified compounds, the best docking results were observed with melezitose that displayed the highest binding affinity (−8.1 kcal/mol). The closest molecular interactions in the p53 active site included 10 residues: Arg10, Arg10, Lys20, Ile21, Gly199, Arg203, Thr230, Asn17, Gln23, and Gln23. These residues established 10 conventional H-bonds, which were supported with an extra carbon H-bond. Such bonds are commonly searched for drug design and development, as they are associated with significant pharmacological effects, including anticancer.22, 46, 47 Melezitose was also deeply embedded in the active site of p53 with a distance of only 1.85Å from Thr230. This could confirm the potential biological activity, as deeper embedding is usually associated with better activity. 48 A previous report showed that melezitose could be used for the development of anticancer therapeutics. 31 Ghazarian and his collaborators performed quantitative kinetic profile assays and revealed that D-melezitose is the best inhibitor of yeast binding to lectin microbeads used as a model system to identify agents that might block cancer cell clumping. 49 They explained that this activity was mainly due to the presence of more than one binding residue constituting the melezitose structure. 31 In this study, melezitose interacted with nine different close residues. These observations are in concordance with our findings. The overall biological effects of tested phytocompounds in AH from the Hail region appeared to be thermodynamically feasible, particularly for melezitose. We observed interesting interactions between melozitose and p53 in favor of anticancer activity. These computational results in parallel with the findings of in vitro analysis could explain the promising cytotoxic and apoptosis-inducing effects observed in the MCF-7 breast cancer cell line.
Conclusion
AH has been widely studied for its medical virtue mainly against cancers. It prompted us to explore its phytocompounds, and we discovered potential anti-apoptotic agents that are able to induce apoptosis in breast cancer. Through this study, we showed interesting cytotoxic effects and induced apoptosis through in vitro approaches in MCF-7 breast cancer cells. Among the identified phytocompounds, oleic acid and 9-octadecenamide have been already known for their anticancer activity. Melozitose displays the highest binding affinity to p53, which could trigger apoptosis and enhance the apoptotic effect of AH to MCF-7 cell line. Although drug-likeness prediction showed that melezitose presents an exception to Lipinski’s rule of five, no violation regarding this product has been detected. Further investigation of melozitose derivatives in induced apoptosis could be of interest. Investigation of melezitose or AH-derived compound on other apoptotic pathway proteins such as Bcl‐2, Bcl‐xL, and MDM2 could be also of interest.50 These findings provide valuable information on using AH as a source of potent compounds that could be used for therapeutic purposes in breast cancer.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
This research has been funded by Scientific Research Deanship at the University of Hail, Hail, Saudi Arabia through project number RG-21 109.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.