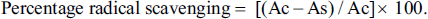Abstract
Background
Kaempferia grandifolia and Kaempferia siamensis are rare plant species in Thailand used for medicinal, food, and ornamental purposes.
Objectives
To compare the phytochemical profiles of leaf and rhizome extracts from wild, in vitro culture, and greenhouse plants of K. grandifolia and K. siamensis.
Materials and Methods
Leaf and rhizome were extracted and analyzed phytochemically by high-performance liquid chromatography (HPLC), gas chromatography mass spectrometry (GC-MS), total phenolic contents (TPC), and total flavonoid contents (TFC). Antioxidant activities were examined using 2,2-diphenyl-1-picrylhydrazyl (DPPH) and 2,2′-azinobis(3-ethylbenzothiazoline6-sulfonic acid) diammonium salt (ABTS) assays.
Results
The results of HPLC analysis showed that the main compounds found in all plants were p-coumaric acid, ferulic acid, and quercetin. GC-MS results revealed 38 compounds in K. grandifolia and 19 compounds in K. siamensis. Major constituents in the leaf and rhizome extracts were 9-octadecenamide, (Z)-, hexadecanamide, and octadecanamide. Total phenolic and total flavonoid contents of leaf extracts decreased from wild to in vitro cultured and greenhouse plants, respectively, whereas rhizome extract contents increased from wild to greenhouse plants. Antioxidant activities of rhizome extracts showed higher activity than leaf extracts in DPPH and ABTS assays.
Conclusion
The present research is the first report to study the phytochemical profiles and antioxidant activities of these plants. Rhizome extracts of K. grandifolia and K. siamensis in greenhouse plants presented the highest phenolic and high free radical scavenging activity compared to rhizome extracts of wild plant. The phytochemical profiles of the compounds found in the K. grandifolia and K. siamensis extracts showed that these plants have potential for future health benefits.
Keywords
Introduction
Kaempferia grandifolia Saensouk and Jenjitt and Kaempferia siamensis Sirirugsa (Zingiberaceae) are both found in Thailand as rare endemic plant species. 1 The genus Kaempferia contains cultivated ornamental, food, and medicinal plants,2–4 and belongs to the family Zingiberaceae with 60 species found in Thailand. Several Kaempferia species are widely used in traditional medicine. Young leaves of K. siamensis are used as food and utilized as ornamental plants, 4 while leaves blended with rhizomes of K. grandifolia and husked rice are used to treat herpes. Furthermore, the young inflorescence is eaten as food. 5 The phytochemical study of Kaempferia plants revealed the presence of phenolic glycosides, flavonoids, monoterpenoids, diterpenoids, cyclohexane oxide derivatives, and essential oils with various biological activity, antimicrobial, anti-inflammatory, and antioxidant properties. 6 Therefore, developing tissue culture techniques for mass micropropagation of Kaempferia sp. would be useful for the conservation of these medicinal plants. Evaluating the potential biological activities of extracts from tissue-cultured plants would be beneficial as an alternative to natural plants. However, there is no data on the phytochemical content of these plants. Thus, this study investigated the phytochemical differences between samples of wild, in vitro cultured, and greenhouse plants. Phytochemical constituents were analyzed by HPLC and GC-MS, while the antioxidant activities of ethanol extracts of K. grandifolia and K. siamensis plants were compared. Results will be helpful for value addition of rare and endemic plant species in Thailand.
Materials and Methods
Chemicals
Chemicals were obtained as follows: gallic acid (Sigma-Aldrich), sodium carbonate (Na2CO3), Folin-Ciocalteu phenol reagent (Sigma-Aldrich), methanol AR grade (Merck), ethanol AR grade (Merck), quercetin (Sigma-Aldrich), aluminum chloride, potassium acetate (Sigma-Aldrich), 2,2-diphenyl-1-picrylhydrazyl (DPPH) (Sigma-Aldrich), ascorbic acid (Sigma Aldrich), 2,2’-azino-bis (3-ethylbenzothiazoline-6-sulfonic acid) (ABTS) (Sigma-Aldrich), dipotassium peroxodisulfate (K2S2O8) (Sigma-Aldrich), and DMSO (Dimethysulfoxide) (Sigma-Aldrich). Solvents and reagents are used in HPLC analysis, including acetonitrile and acetic acid (Merck). Phenolic and flavonoid standards, such as quercetin, catechin, gallic acid, vanillic acid, caffeic acid, syringic acid, p-coumaric acid, ferulic acid, rutin, and kaempferol (Sigma-Aldrich).
Plant Material
Leaves and rhizomes of Kaempferia grandifolia and K. siamensis; wild, in vitro cultured and greenhouse plants (Figure 1). Wild plants were collected from the Thai National AGRIS Center at Khok Phu Ta Ka, Amphoe Phu Wiang, Khon Kaen Province and Phu Phan National Park, Sakon Nakhon Province. In vitro cultured from shoot explants (wild plant) derived from aseptic culture on MS medium without plant growth regulators (PGR) with 30 g/l of sucrose. The medium was adjusted to pH 5.7. Agar (7 g/l) was used to promote shoot induction at eight weeks of culture. Greenhouse plants from micro shoots on solid MS medium supplemented with 4 mg/l 6-benzylaminopurine (BAP) plus 1 mg/l α-naphthaleneacetic acid (NAA). After eight weeks of culture, the plantlets were transplanted into plastic pots with soil sand (1:1 v/v). The potted plants were maintained for 1 year while growing under greenhouse conditions at Mahasarakham University’s Department of Biology, Faculty of Science, Thailand (Figure 1).
Rhizomes and Leaves of K. grandifolia (A−C), (A) Wild Plant, (B) Greenhouse Plant, and (C) In Vitro Culture; Rhizomes and Leaves of K. siamensis (D−F), (D) Wild Plant, (E) Greenhouse Plant, and (F) In Vitro Culture.

Plant Preparation and Extraction
Leaves and rhizomes were obtained from three types of plants: wild, in vitro cultured, and greenhouse plants. Plant parts were dried in a hot air oven at 60°C for 24 hour 7 and then ground to a fine powder. Five grams of plant parts in powder form were extracted by a soxhlet extraction apparatus with 95% ethanol for 4 hour. The extracts were evaporated and stored in dark containers at room temperature until required.
Using the HPLC method and liquid-liquid extraction, the ethanol extract was separated into ethyl acetate and dichloromethane fractions. A vacuum evaporator was used to evaporate each of the extracts and fractions.
Total Phenolic Content Determination
The folin-ciocalteu colorimetric method was modified from Pothitirat et al. 8 to determine the total phenolic content of the samples. Gallic acid was used as the standard (concentration of 15.625–250 µg). The crude extracts were dissolved in 50% DMSO in methanol. Samples of 200 µl (1 mg/ml) were added to 500 µl of folin-ciocalteu reagent and left for 5 min at room temperature. Then, 800 µl of Na2CO3 was added. The solutions were mixed in volumes of 1,500 µl and stored at room temperature for 5 min in the dark. Samples were measured as three replicates for absorbance at 765 nm wavelength. Total phenolic contents were measured by comparison of standard curves. The values were expressed as mg gallic acid equivalent (GAE)/g of extract sample.
Total Flavonoid Content Determination
Total flavonoid contents were determined following the modified method of Pothitirat et al. 8 Quercetin at a concentration of 15.625–250 µg/ml was used as the standard. Samples of 500 µl (1 mg/ml) were added to 1,500 µl of 95% ethanol and incubated at room temperature for 6 min. Then, 100 µl of 10% aluminum chloride was added to the solutions and mixed. After 5 min, 100 µl of 1 M potassium acetate was added to the solutions and mixed. With distilled water, the volume was adjusted to 5,000 µl and left at room temperature for 30 min in the dark. Samples were measured as three replicates for absorbance at 415 nm. Total flavonoid contents were measured by comparison with standard curves. The values were expressed as mg quercetin equivalent (QE)/g of extract sample.
DPPH Assay
The 2,2-diphenyl-1-picrylhydrazyl (DPPH) assay was investigated following the method described by Likhitwitayawuid et al. 9 Ascorbic acid was used as the standard. The DPPH solution was prepared in methanol at a concentration of 100 µM. Twenty microliters of extracts (1 mg/ml) were added in180 µl DPPH solution. The solutions were incubated in a dark condition at room temperature. After 30 min of preparation, absorbance by microplate reader (ASYS UVM 340) was measured at 517 nm. Methanol was used blank (Ac). Extracts were measured as three replicates. The calculation of percentage inhibition was performed by the following equation;
Percentage Radical Scavenging = [(Ac – As)/Ac] × 100 ABTS Assay
ABTS assay was investigated following the modified method of Payet et al.
10
The reaction between 7 mM ABTS and 2.45 mM potassium persulfate produced the ABTS+ cation radical, which was incubated in the dark at room temperature for 12–16 hours before use. The ABTS solution was diluted with ethanol to obtain an absorbance of 0.700 at 734 nm. Extracts were measured in three replicates. The absorbance of extracts (As), methanol was used as a blank (Ac), and ascorbic acid is used as a standard. The calculation of percentage inhibition was performed by the following equation:
Gas Chromatography-Mass Spectrometry (GC-MS) Analysis
Leaf and rhizome extracts were performed by GC-MS adapted from Suphrom et al. 11 The injector was set at 250°C and carried out in split mode with a 10:1 split ratio (in 1 µl). Hewlett Packard HP-5 (5% phenyl methyl siloxane) uses a fused silica capillary column (30 m × 0.25 mm i.d., 0.25 m film thickness) with helium as the carrier gas at a constant flow rate of 1 ml/min. The GC-MS analysis was detected using GC-MS-QP 2010 Ultra (Shimadzu). The initial oven temperature was 70°C, which was kept for 3 min before being ramped up to 280°C at a rate of 5°C/min and eventually held for 10 min. The transfer line heater was adjusted to a temperature of 280°C. In a complete scan, the mass scanning range was set to 50–550 amu.
HPLC Determination of Phenolic Acids and Flavonoids
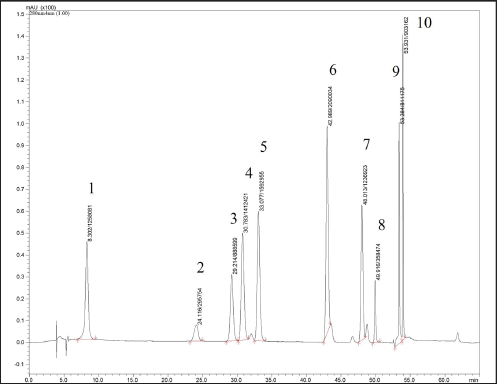
Extracts were analyzed by the procedures of Siriamornpun and Kaewseejan. 12 HPLC analysis was performed using chromatographic conditions as follows: column Inetsil ODS-3, C18 (4.6 mm × 250 mm, 5 µm); ingredient elution acetic acid pH 2.74 (solvent A) – acetonitrile (solvent B); and flow rate of 0.8 ml/min. The operating conditions were as follows: column temperature of 38°C, 20 µl injections of sample solution, and UV-diode array detection at 280 nm for phenolic acids and 370 nm for flavonoids. Peak identification of phenolic and flavonoid compounds in the samples was done by comparing their relative retention times (RT) and UV spectra to those of authentic compounds and was done using external standard procedures. Quercetin, catechin, gallic acid, vanillic acid, caffeic acid, syringic acid, p-coumaric acid, ferulic acid, rutin, and kaempferol were used as standards for analysis (Figure 4).
Statistical Analysis
All values were expressed as the mean ± S.D (obtained from three determinations). Pearson’s correlation coefficient (r) was calculated using Microsoft Excel 2021. Difference among means was analyzed by one-way ANOVA with significance at p < .05.
Results
Total Phenolic Contents
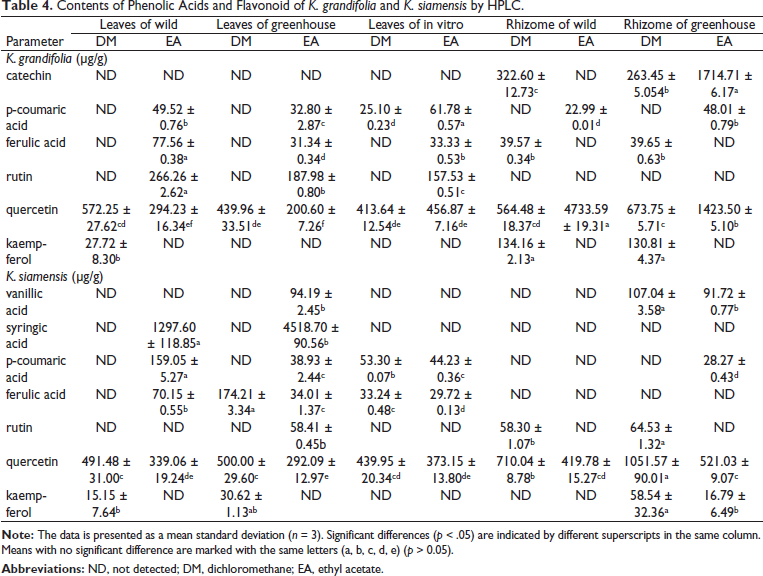
Total phenolic contents of K. grandifolia ranged from 70.42 to 177.65 mg GAE/g of extract. Total phenolic contents of wild, greenhouse, and in vitro culture ranged from 164.38 to 170.24, 169.84 to 177.65, and 70.42 mg GAE/g extracts, respectively. The total phenolic contents of K. siamensis ranged from 100.42 to 233.51 mg GAE/g extracts. Total phenolic contents of wild, greenhouse and in vitro culture were range (183.00–233.51, 180.07–187.59, and 100.42 mg GAE/g extracts, respectively) (Table 1). Leaf extract of K. siamensis wild had the highest total phenolic contents, with 233.51 mg GAE/g extracts. Results showed that phenolic contents of rhizome extracts increased from wild to greenhouse plants.
Total Phenolic Contents, Total Flavonoid Contents, and Free Radical Scavenging Activity (DPPH and ABTS) of K. grandifolia and K. siamensis Extracts by Ethanol Solvent.
Total Flavonoid Contents
Total flavonoid contents were calculated from the standard curve of quercetin. Total flavonoid contents of K. grandifolia ranged from 22.91 to 70.24 mg QE/g of extracts. Total flavonoid contents of wild, greenhouse, and in vitro culture ranged from 22.91 to 70.24, 24.88 to 61.73, and 55.66 mg QE/g extracts, respectively. The total flavonoid contents of K. siamensis ranged from 41.28 to 137.35 mg QE/g extracts (Table 1). The total flavonoid contents of wild, greenhouse, and in vitro culture ranged from 41.28 to 137.35, 73.20 to 109.22, and 65.52 mg QE/g extracts, respectively). The highest total flavonoid contents were found in leaf extracts from wild plants of K. siamensis (137.35 ± 8.11 mg QE/g extracts). Results showed that flavonoid contents of leaf extracts decreased from wild to in vitro cultured and greenhouse, respectively.
DPPH and ABTS Assays
The DPPH results indicated that the rhizome extracts from greenhouses of K. grandifolia had the highest percentage of radical scavenging with 38.10%, whereas leaf extracts from in vitro cultured showed the lowest percentage radical scavenging compared to the other extracts. For the ABTS assay, results showed that the leaf extracts from wild plants of K. siamensis had the highest percentage of radical scavenging with 25.52%, whereas leaf extracts from in vitro cultures showed the lowest percentage of radical scavenging (Table 1). The DPPH assays of K. grandifolia and K. siamensis had higher correlations with TPC (r = 0.734 and r = 0.696, respectively), while the ABTS assays of K. grandifolia and K. siamensis had higher correlations with TPC (r = 0.713 and r = 0.876, respectively).
GC-MS Analysis
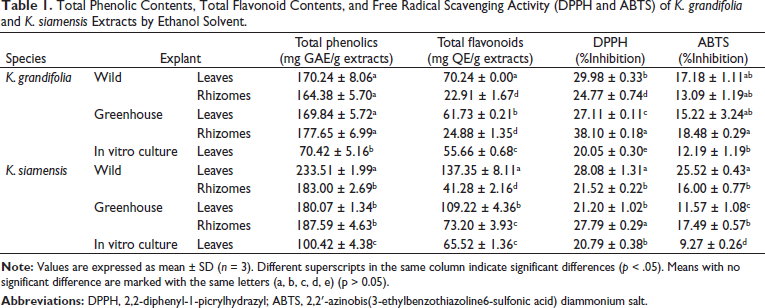
The extracts were subjected to GC-MS for identification of the phytochemical constituents. Ethanol extracts of K. grandifolia revealed the presence of 38 compounds, with 12 compounds identified from wild leaf extracts, 7 compounds from in vitro cultured, and 5 compounds from greenhouse plants (Table 2). Many of the compounds from wild plants showed greater similarity to greenhouse plants than in vitro cultured plants. Four similar compounds were common to wild, in vitro, and greenhouse extracts. Ethanol extracts of K. siamensis included 19 compounds. Five compounds were identified from wild leaf extracts, 5 compounds from in vitro cultured plants, and 5 compounds from greenhouse plants (Table 3). Rhizome extracts of K. siamensis included 10 compounds from wild and 10 compounds from greenhouse extracts, while six similar compounds were found in both extracts. The compounds identified from extracts; neophytadiene, hexadecane, octadecanamide, lupeol, and betulinaldehyde 3,7,11,15-tetramethyl-2-hexadecen-1-ol are normally reported for their antimicrobial, anti-inflammatory, anticancer, antioxidant, antitumor, chemopreventive, lipoxygenase-inhibitor, and pesticidal abilities.13–17
Phytocomponents Detected in Ethanol Extracts of Leaves and Rhizomes
from K. grandifolia.
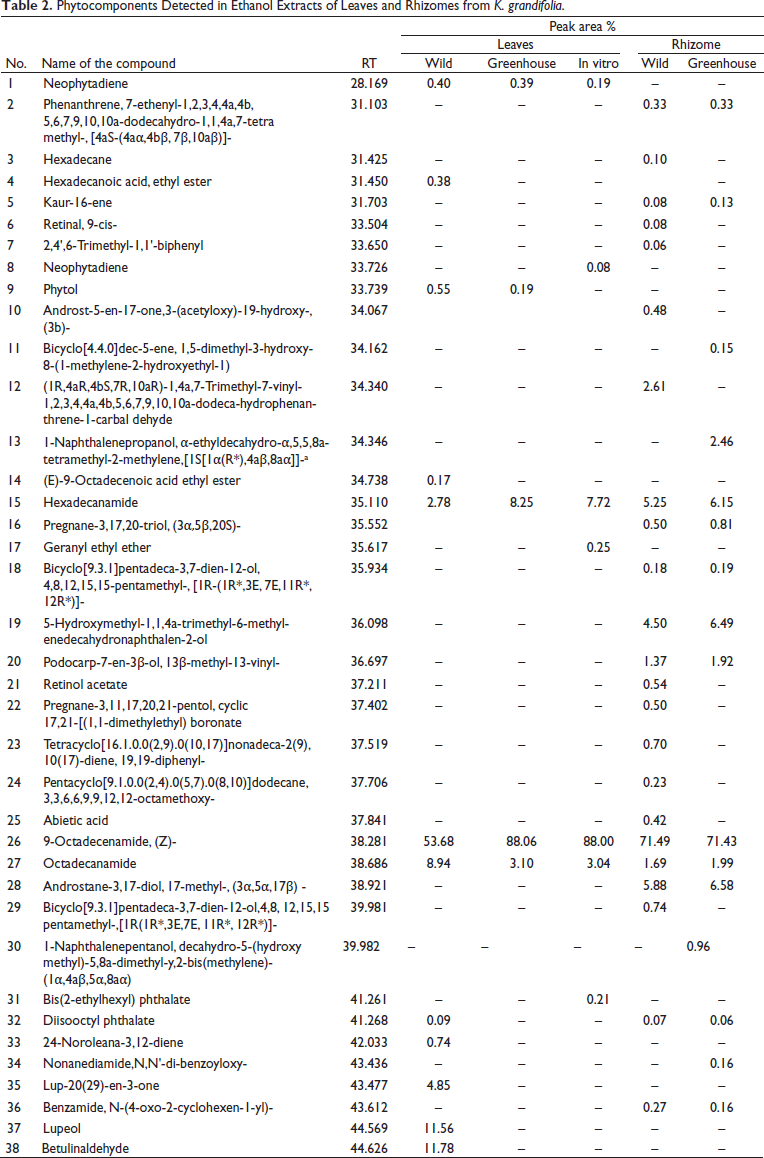
Phytocomponents Detected in the Ethanol Extract of Leaves and Rhizome of
K. siamensis Plant.
The Quantifications of the Phenolic Acid and Flavonoids by HPLC
The HPLC chromatograms of leaf and rhizome extracts from wild, in vitro, and greenhouse-grown K. grandifolia and K. siamensis plants are shown in Figure 2. The results of the HPLC analysis are presented in Table 4 and Figure 3. Quercetin was found in all extracts; the ethyl acetate rhizome extract from wild plants of K. grandifolia had the highest content of quercetin. Vanillic acid was found in the leaf and rhizome of a greenhouse plant of K. siamansis, which is not found in the leaves of in vitro or wild plants. In rhizome extracts of K. grandifolia, catechins were identified. Vanillic acid, syringic acid, p-coumaric acid, ferulic acid, rutin, and quercetin were revealed in leaf extracts from K. siamensis greenhouse plants. The quantifications of the phenolic acid and flavonoids based on calibration curves of authentic standards are presented in Figure 4.
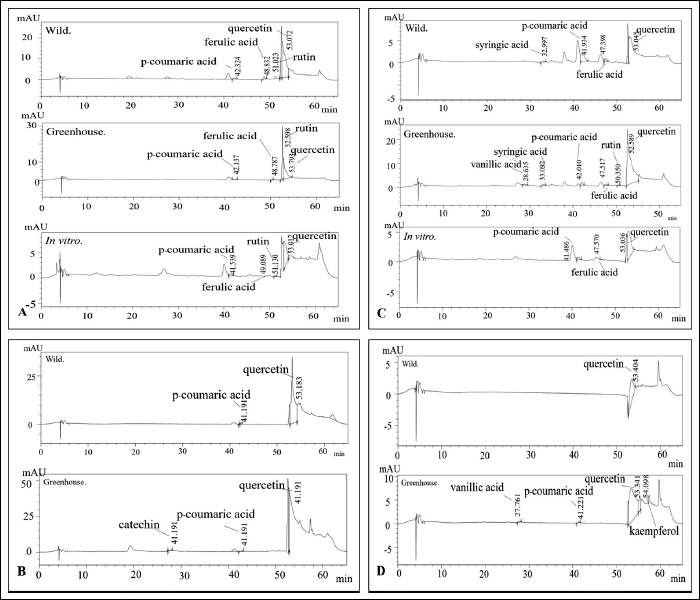
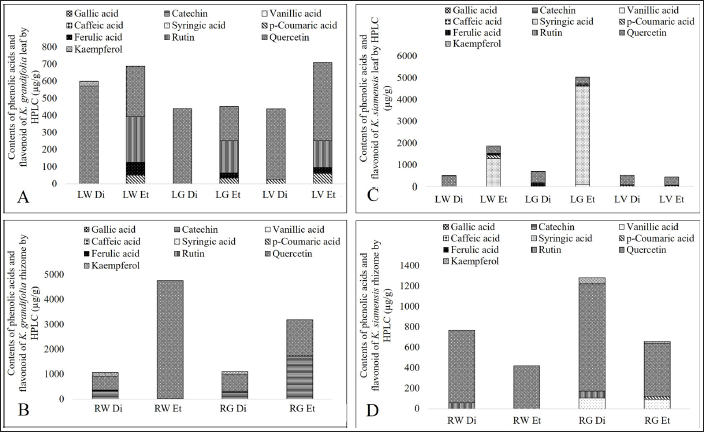
Contents of Phenolic Acids and Flavonoid of K. grandifolia and K. siamensis by HPLC.
Discussion
The study is the first report on the phytochemical profiles from K. grandifolia and K. siamensis. Rhizome extracts of K. grandifolia in greenhouse plants presented the highest phenolic and high free radical scavenging activity compared to different extracts of wild and in vitro cultured. The highest TPC, TFC, DPPH, and ABTS were found in leaf extracts of K. siamensis. These results show that wild and greenhouse plants had the highest levels of phenolic, flavonoid contents, and free radical scavenging activity. Wild and greenhouse plants do not have a huge statistical difference, so if plants in the greenhouse will be helpful to replace plants in the wild that is considered the most useful.
The bioactive compounds were identified, and their bioactive applications are listed in Table 5. Bioactive compounds are normally reported for their antioxidant, anti-inflammatory, anticancer, and antimicrobial properties.
Bioactivity of Phytocomponents Identified in the Extract of Leaves and Rhizomes of K. grandifolia and K. siamensis.

GC-MS analysis of leaf and rhizome extracts in wild, greenhouse, and in vitro cultured extracts of K. grandifolia and K. siamensis revealed 9-octadecenamide, (Z)-, hexadecanamide, and octadecanamide as the major constituents. 9-octadecenamide, (Z)- is a fatty acid amide known as oleamide. Oleamide, which is found in many species of Zingiberaceae plants, such as the root and fruit of Zingiber vuquangensis, Zingiber castaneum, and Curcuma cochinchinensis. 28 It is reported that these compounds have bioactivity such as antioxidant, anti-inflammatory, and antimicrobia. 28 HPLC analysis found that the main compounds found in all extracts were p-coumaric acid, ferulic acid, and quercetin, while quercetin content in rhizome extracts from wild plants was higher than that of greenhouse plants. Vanillic acid is not found in the wild plants of these plants, but it is found in greenhouse plants. It possesses anticancer, antiobesity, antidiabetic, antibacterial, anti-inflammatory, and antioxidant activity. 25 Krishnakuma et al. 18 studied phytochemical compounds from Kaempferia rotunda. They reported that the rhizome extracts contain quercetin, ferulic acid, p-coumaric acid, gallic acid, myricetin, caffeic acid, kaempferol, and syringic acid. The fractions of K. grandifolia had higher contents of the phytochemical compounds than K. siamensis fractions. Different plant parts, different conditions of plant, and types of extraction solvent are important factors that impact the phytochemical compounds and bioactivity of plant extracts. K. grandifolia and K. siamensis phytochemical components were shown to have considerable antioxidant activity, whereas greenhouse extracts had potential antioxidant activity.
Conclusion
Extracts of Kaempferia from greenhouse plants and tissue-cultured plants were found to be similar to wild plants and exhibited good medicinal activity. When compared to wild plants, rhizome extracts from greenhouse plants have higher phenolic and antioxidant activity. Biological activities such as anti-inflammatory activity, anti-microbial activity, and anti-cancer activity should be further investigated. This study may be helpful to increase the value of commercial production of ornamental plants, cosmetics, and supplementary foods and lead to future developments in industrial pharmacy.
Summary
According to HPLC results, the main compounds found in all plants were p-coumaric acid, ferulic acid, and quercetin.
The GC-MS results revealed 38 compounds in K. grandifolia and 19 in K. siamensis. Major constituents in the leaf and rhizome extracts were 9-octadecenamide, (Z)-, hexadecanamide, and octadecanamide.
Total phenolic and total flavonoid contents of leaf extracts decreased from wild to in vitro cultured and greenhouse plants, respectively, whereas rhizome extract contents increased from wild to greenhouse plants.
Antioxidant activities of rhizome extracts showed higher activity than leaf extracts in DPPH and ABTS assays.
Footnotes
Abbreviations
GC-MS, gas chromatography‑mass spectrometry; HPLC, high-performance liquid chromatography; K., Kaempheria; QE, quercetin equivalent; Rt, retention time.
Acknowledgment
The first author is grateful to the Science Achievement Scholarship of Thailand for support during the study. This research project was financially supported by Mahasarakham University. The authors thank the Department of Biology, Faculty of Science, Mahasarakham University, for providing facilities and support during this research.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical approval is not applicable for this article.
Funding
This research project was financially supported by Mahasarakham University.
Informed Consent
Not applicable, because this article does not contain any studies with human or animal subjects.