Abstract
Background:
Silibinin is a bioactive flavonolignan extracted from milk thistle, known as
Method:
The MDA-MB-231 and MCF-7 cell lines were cultured in both monolayer and spheroid cultures. Cells were treated with silibinin at 24, 48, and 72 h of incubation. The 5-bromo-2′-deoxyuridine labeling index was used to determine the cells of the synthesis phase. Poly-ADP-ribose-polimerase immunohistochemical staining and the terminal deoxynucleotidyl transferase dUTP nick and labeling assay were used to determine the death of cells in both the monolayer and spheroid cultures.
Results:
An half maximal inhibitory concentration dose of silibinin in MDA-MB-231 and MCF-7 cells was 100 µM/mL at 24, 48, and 72 h of incubation. Terminal deoxynucleotidyl transferase dUTP nick and labeling positive cells and active poly-ADP-ribose-polimerase were detected after treatment with silibinin in both the monolayer and spheroid cultures. The dead cell count was higher in the MDA-MB-231 and MCF-7 cell lines with silibinin applied than in the controls.
Conclusions:
Our study demonstrated that silibinin applications enhanced terminal deoxynucleotidyl transferase dUTP nick and labeling positive cells and active poly-ADP-ribose-polimerase in comparison to the control in both the monolayer and spheroid cultures.
Keywords
Introduction
Breast cancer is the most frequent malignancy occurring in women, causing approximately 15% of total cancer deaths in female patients. 1 The high rates of morbidity and mortality are mainly attributed to metastasis, which generally includes invasion, intravasation, circulation, extravasation, and colonization in distant organs such as the liver, lung, bone, and so on. 2 –4 Although effective treatment includes surgery, radiation, and chemotherapy, breast cancer frequently shows resistance to those therapies. 5 Therefore, there is a great need for new agents to treat breast cancer. 6
Silibinin is a major bioactive component of silymarin, which is isolated from milk thistle (
Apoptosis has since been recognized and accepted as a distinctive and important mode of “programmed” cell death, which involves the genetically determined elimination of cells. However, it is important to note that other forms of programmed cell death have been described and other forms of programmed cell death may yet be discovered.
18
Apoptosis is critically important during various developmental processes. It occurs normally during development and aging and as a homeostatic mechanism to maintain cell populations in tissues. Apoptosis also occurs as a defense mechanism such as in immune reactions or when cells are damaged by disease or noxious agents.
18,19
Although there are a wide variety of stimuli and conditions, both physiological and pathological, that can trigger apoptosis, not all cells will necessarily die in response to the same stimulus. Irradiation or drugs used for cancer chemotherapy results in DNA damage in some cells, which can lead to apoptotic death through a
In the present study, silibinin was evaluated for its apoptotic effects in vitro in two breast carcinoma cell lines (MCF-7 and MDA-MB-231) via two-dimensional (2-D) monolayer and three-dimensional (3-D) spheroid cultures using immunohistochemical techniques. According to our research, to date, there have been no investigations on the apoptotic effect of silibinin on a spheroid model. Cancer cells grown in vitro as spheroids more accurately mimic the drug sensitivity and/or drug-resistant behaviors of solid tumors in vivo, as compared to cancer cells cultured under monolayer conditions. 20
Methods
Cell culture
The human breast cancer cell lines, MCF-7 and MDA-MB-231, were supplied by the American Type Culture Collection (ATCC; Rockville, USA). These tumor cell lines were cultured in monolayers in RPMI 1640 (Biological Industries, Israel) supplemented with penicillin G (100 U/mL) (Sigma-Aldrich Co., St Louis, Missouri, USA), streptomycin (100 µg/mL) (Sigma-Aldrich Co),
Constitution of a 3-D spheroid model (spheroid culture)
An in vitro multicellular MCF-7 and MDA-MB-231 breast spheroid model were established using a liquid overlay technique. Briefly, once the monolayer cultures became semi-confluent, the cells were trypsinized and single cells with 100% viability were cultured over thin layer of agar-coated (1 mL of a 1,5% (w/v) solution of agar in RPMI-fetal bovine serum) 6-well culture plates containing 5 mL RPMI 1640 medium at a concentration of 5 × 10 5 cells/well. 21 Six well plates were then kept in an electronic incubator (Heal Force, HF90, serial no: 1103K0092CE) under a humidified atmosphere of 5% CO2 and air mix for 2 weeks. Every 7 days, 3 mL of RPMI medium was gently removed from each well and wells were then filled with the same amount of fresh medium.
Experimental design
The investigated silibin extract (Sigma-Aldrich Co.) at a volume of 100 μL were applied to monolayer cultures of breast cancer cells at the following final concentrations: 0.1–200 μM, except for the control cells, to which nutrient medium was applied. After 24, 48, and 72 h incubation, the cells were trypsinized and the cytotoxicity was determined by trypan blue dye exclusion test. The concentration of silibin effective on MDA-MB-231 cells and on MCF-7 cells was determined as 100 μM/mL. All experiments were repeated three times and representative data are presented.
DNA synthesis (S phase) analyses of BrdU labeling index in monolayer culture (immunohistochemistry)
MCF-7 and MDA-MB-231 breast carcinoma cells were seeded on glass cover slips and treated with silibinin, as described in the survival studies, every 24 h for a total of 72 h. Cells were washed in phosphate-buffered saline (PBS) and fixed in 70% alcohol for 15-30 minutes at 4 °C. Endogenous peroxidase activity was quenched with 3% hydrogen peroxide (H2O2) in methanol for 10 min. Cells were washed in three changes of distilled water for 2 min each. Double-stranded DNA was then denatured with 4 N hydrochloric acid. Following washing with PBS, a non-specific blocking reagent (Invitrogen) was used to prevent non-specific binding. Monoclonal mouse anti-bromodeoxyuridine (5-bromo-2′-deoxyuridine (BrdU)) antibody (ready to use; Invitrogen, Carlsbad, California, USA) was the primary antibody, and secondary antibody was biotinylated goat anti-mouse (Invitrogen). After washing, peroxidase-conjugated streptavidin (Invitrogen) was applied and diaminobenzidine (DAB) was used as the chromogen. The slides were then counterstained with Mayer’s hematoxylin to enhance nuclear staining. BrdU-labeled cells in S phase showed brown-stained nuclei. Negative control samples were processed in a similar manner but excluding the primary antibodies. BrdU-labeling index (the number of positively stained cells divided by the total number of cells) was calculated by evaluating at least 3000 cells in multiple high-power fields.
DNA synthesis (S phase) analysis of BrdU labeling index in spheroid cultures (immunohistochemistry)
Spheroids were gently removed from the surface of the solidified agarose, fixed %10 neutral buffered formalin, washed tap water incubated with a graded ethanol series (70%, 80%, 90%, 96%, and 100%) and xylene, then embedded in paraffin. Sections were cut by the microtome (Leica SM 2000 R, Serial No: 1985/11.2000 Heerbrugg, Switzerland) and attached to a slide coated with poly-llysine. Sections were deparaffinized with xylene for 5 min. Slides were rehydrated in a series of graded alcohol, treated with 0.1% trypsin for antigen retrieval, and incubated in a solution of 0.3% H2O2. Then the same protocol was applied to spheroid cultures. Approximately 25 spheroids in each slide were selected for calculation of the BrdU labeling index. For negative controls, adjacent sections were processed excluding the primary antibody.
Detection of apoptotic cell death in situ using the terminal deoxynucleotidyl transferase dUTP nick and labeling method in monolayer culture
The in situ apoptosis detection kit (DeadEnd Colorimetric terminal deoxynucleotidyl transferase dUTP nick and labeling (TUNEL) system, Promega G7130, Ca No: S701; Madison, Wisconsin, USA) was used in order to detect apoptosis. Cells fixed 1% paraformaldehyde in PBS for 10 min at room temperature and washed in two changes of PBS for 5 min each wash. Then cells were post-fixed in precooled ethanol:acetic acid 2:1 for 5 min at −20°C, washed in two changes of PBS for 5 min each wash. Endogenous peroxidase activity was inhibited with 3% H2O2. Following washing, sections were then incubated with equilibration buffer for 10–15 s and Terminal deoxynucleotidyl transferase (TdT)-enzyme in a humidified atmosphere at 37°C for 60 min. They were subsequently put into pre-warmed working strength stop/wash buffer at room temperature for 10 min and incubated with antistreptavidin-peroxidase for 45 min. Each step was separated by careful washing in PBS. Staining was performed with DAB and counterstaining was performed in methyl green.
Detection of apoptotic cell death in situ using the TUNEL method in spheroid cultures
Five micrometer sections were cut from the paraffin blocks of the samples. The sections were deparaffinized in xylene, rehydrated, and incubated with 20 μg/mL proteinase K for 10 min and rinsed in distilled water. Endogenous peroxidase activity was inhibited with 3% H2O2. Then the same protocol was applied to spheroid cultures.
Immunohistochemical staining with poly-ADP-ribose-polimerase antibody in monolayer culture
MCF-7 and MDA-MB-231 breast carcinoma cells were seeded on glass cover slips and treated with drugs, as described in the survival studies, every 24 h for a total of 72 h. Medium was removed from cells and was wash in several changes of PBS. Cells were fixed in 70% alcohol for 15–30 min at −20°C. Following washing, endogenous peroxidase activity was quenched with 3% H2O2 in methanol for 10 min. Cells were washed in three changes of PBS for 3 min each. Then, a non-specific blocking reagent (Ultra-V-Block, Invitrogen) was used to prevent non-specific binding. Sections were incubated with primary antibodies to anti-poly-ADP-ribose-polimerase (1:200 dilution; Abcam, Ca No: ab32138; Cambridge, Massachusetts, USA). Next, the sections were incubated with biotinylated immunoglobulin G (IgG) and then with streptavidin–peroxidase conjugate (Histostain-Plus Bulk Kits; Zymed, South San Francisco, California, USA). Then, the sections were washed with PBS. Slides were incubated with a solution containing DAB for 5 min to visualize immunostaining. Finally, sections were counterstained with Mayer’s hematoxylin. Control samples were processed in the same manner except that the primary antibodies were omitted.
Immunohistochemical staining with poly-ADP-ribose-polimerase antibody in spheroid cultures
For immunohistochemical analysis, the spheroids were fixed in 4% paraformaldehyde (Sigma) in PBS (pH 7.4) for 24 h at 4°C, then washed in PBS. Following fixation the spheroids were dehydrated through graded ethanol (80%, 95%, and 100% sequentially), cleared in xylene, embedded in paraffin, and 5 μm coronal sections were cut on microtome (Leica SM 2000 R, Heerbrugg, Switzerland). Sections were floated in a sterile bath, collected on poly-
Hematoxylin and eosin staining in spheroid cultures
For hematoxylin and eosin (H-E) staining, the spheroids were fixed in 4% paraformaldehyde (Sigma) in PBS (pH 7.4) for 24 h at 4°C, then washed in tap water. Following fixation, the spheroids were dehydrated through graded ethanol (80%, 95%, and 100% sequentially), cleared in xylene, embedded in paraffin, and 5 μm coronal sections were cut on microtome (Leica SM 2000 R, Heerbrugg). Then sections were stained with H-E.
Statistical analysis
Data were analyzed using SPSS 18 software. One-way analysis of variance and Dunnett’s two-tailed post hoc t test were employed to evaluate the statistical significance of differences between the control and experimental group. Data are presented as mean ± standard error of mean or standard deviation, as appropriate, and at least in triplicate. Results were considered significant with
Results
Cell proliferation
Silibin effective dose on MDA-MB-231 cells and on MCF-7 cells were determined as 100 μM/mL. All experiments were repeated three times and representative data are presented. The effects of silibinin on cell proliferation of MCF-7 and MDA-MB-231 are shown in Figure 1(a) and (b). The cell number of untreated control group showed a proportional increase as a function of incubation time throught the 72 h of culture. In comparison with the control group, silibinin treatment groups inhibit cell proliferation of MCF-7 and MDMB-231 cells for all time intervals (

Effect of silibinin on proliferation of the monolayer culture MDA-MB-231 cells (a). Effect of silibinin on proliferation of the monolayer culture MCF-7 Cells (b). Cells were initially plated at a seeding density of 5 × 105 cells per well. Results are mean of three different experiments and presented as mean ± SEM. SEM: standard error mean.
Cell cycle kinetics
DNA synthesis (S-phase) analysis of monolayer cultures
DNA synthesis (S-phase) analysis of breast cancer cells was assessed by BrdU staining. Cells with brown-stained nuclei represent BrdU uptake and considered BrdU-positive. As shown in Figure 2(a), MDA-MB-231 cells nuclei were positive for BrdU in the control group with BrdU-LI values of 60% at 24 h and 62% at 72 h. Silibinin treatment reduced BrdU-labeled cells (
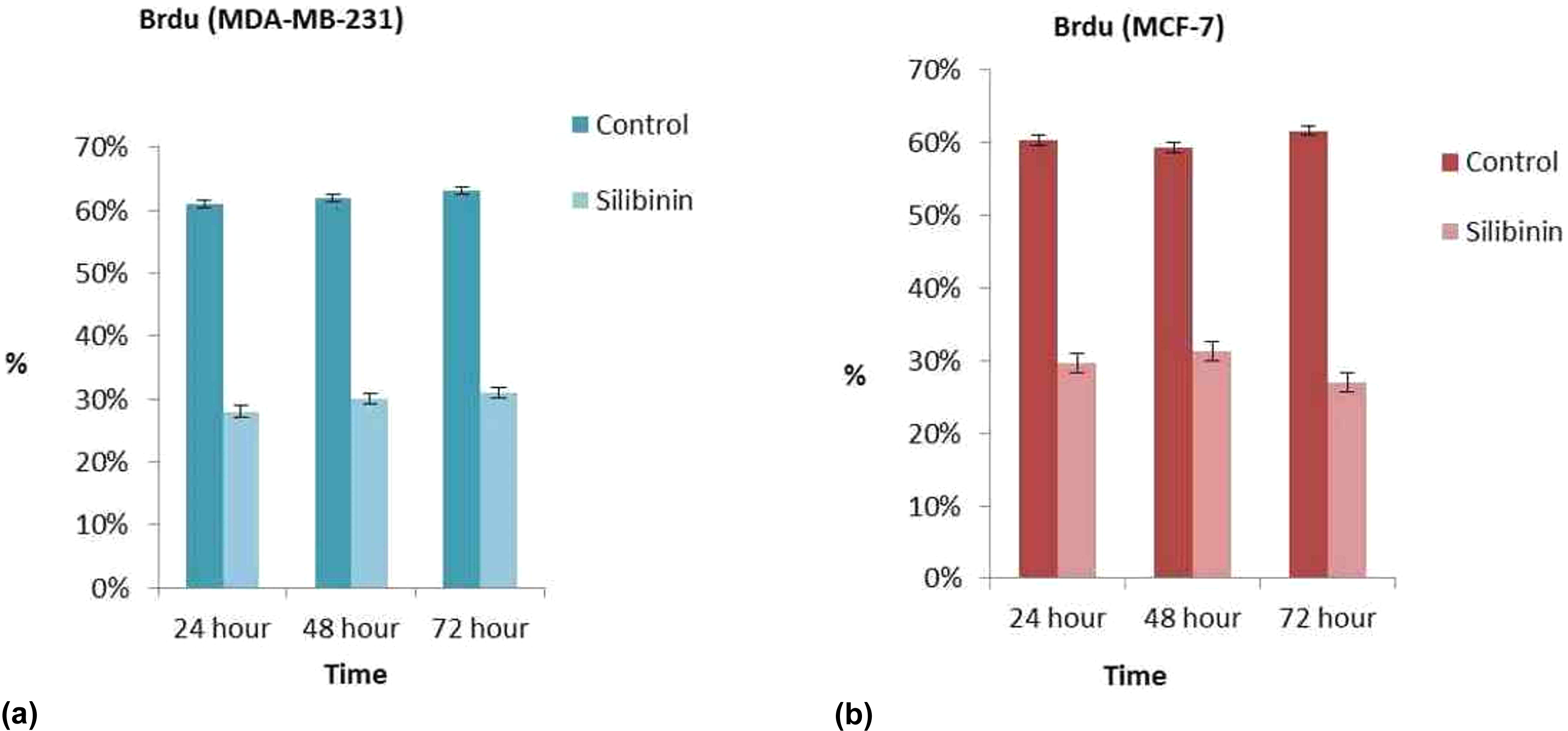
Two dimentional cell culture BrdU staining in MDA-MB-231 cells (a). Monolayer culture BrdU staining in MCF-7 cells (b). Results are mean of three different experiments and presented as mean ± SEM. SEM: standard error mean.

BrdU immunohistochemistry staining in monolayer culture. Control group was high staining with BrdU in MDA-MB-231 cells (a1), high staining with BrdU in MCF-7 cells (b1). The other all groups were light stainied with BrdU (A2-24h-MDA-MB231, A3-48h-MDA-MB231, A4-72h-MDA-MB231, B2-24h-MCF-7, B3-48h-MCF-7, B4-72 h- MCF-7) (×40).
DNA synthesis (S-phase) analysis for spheroid cultures
Twenty-five spheroids from all experimental groups, including the control group, were evaluated for 24, 48, and 72 h. Cells with brown-stained nuclei considered BrdU-positive. The control group had a healthy pattern of S-phase fraction and many of the MCF-7 and MDA-MB-231 cell nuclei were observed to be positive for BrdU with BrdU-LI values of 20%, 18%, and 19%, respectively. In comparison the controls, silibinin reduced the BrdU-LI values of MCF-7 spheroids to 7.6% for 24 h, 7.5% for 48 h, and 7.2% for 72 h. Also, in MDA-MB-231 spheroids, silibinin reduced the BrdU-LI values to 7.3% for 24 h, 7.4% for 48 h, and 7.7% for 72 h (Figures 4(a) and (b) and 5(a) and (b)).

Three-dimentional cell culture BrdU staining in MDA-MB-231 cells (a). Three-dimentional cell culture BrdU staining in MCF-7 cells (b). Results are mean of three different experiments and presented as mean ± SEM. SEM: standard error mean,

BrdU immunohistochemistry staining in spheroid culture. Control group was high staining with BrdU in MDA-MB-231 cells (a1), high staining with BrdU in MCF-7 cells (b1). The other all groups were light staining with BrdU (A2-24h-MDA-MB231, A3-48h-MDA-MB231, A4-72h-MDA-MB231, B2-24h-MCF-7, B3-48h-MCF-7, B4-72h-MCF-7) (×40). BrdU: 5-bromo-2′-deoxyuridine.
Tunel analysis for monolayer cultures
To confirm the cell death mechanism on MCF-7 and MDA-MB-231, monolayer cultures were used to determine differences in their sensitivity in the molecular process triggered by silibinin for 24, 48, and 72 h. At all incubation periods, TUNEL-positive cell indices of silibinin were found to be significantly different from the TUNEL-positive cell indices of control (Figures 6(a) and (b) and 7(a) and (b)).
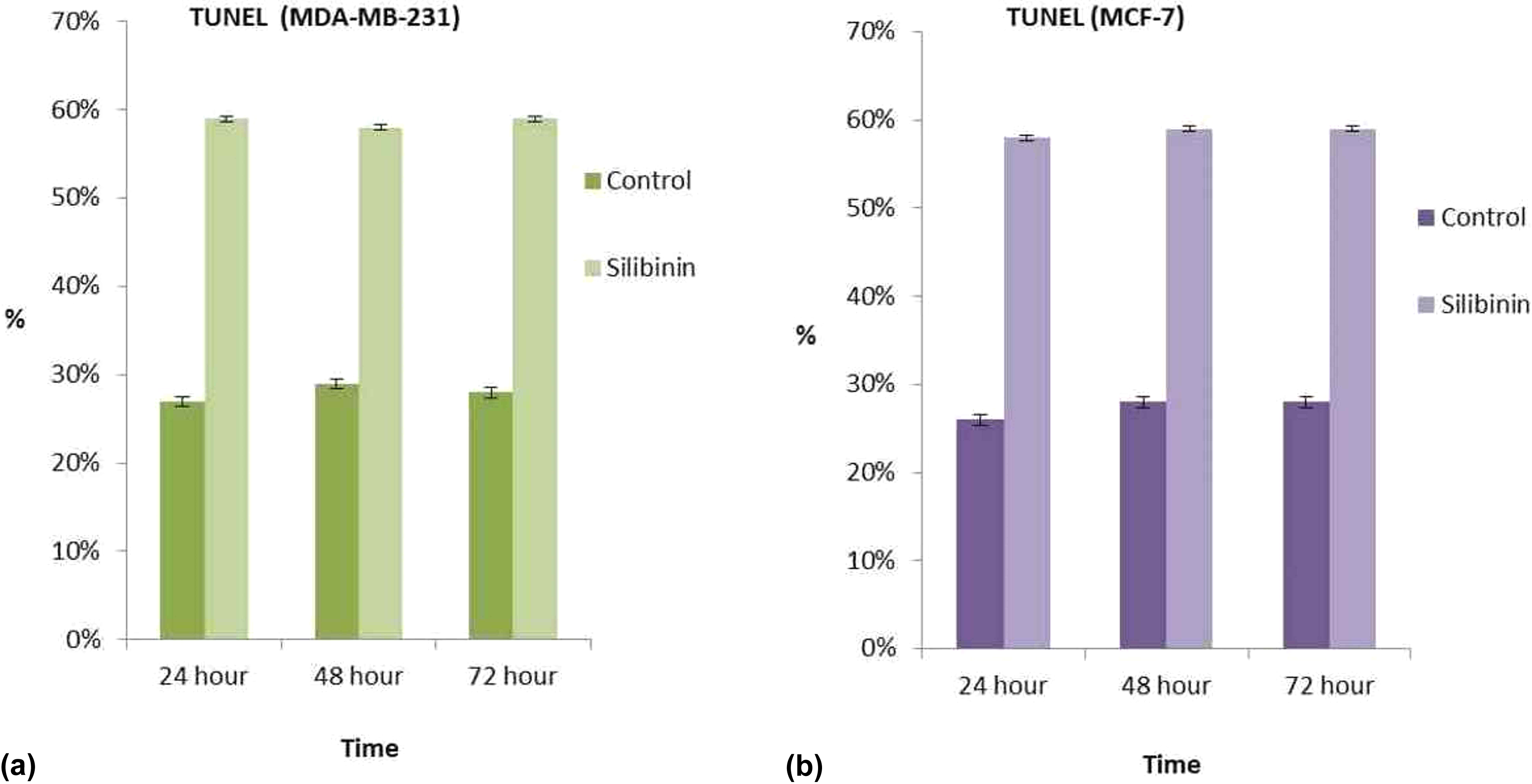
Two-dimentional cell culture TUNEL staining in MDA-MB-231 cells (a). Two-dimentional cell culture TUNEL staining in MCF-7 cells (b). Results are mean of three different experiments and presented as mean ± SEM. TUNEL: terminal deoxynucleotidyl transferase dutp nick and labeling; SEM: standard error mean.
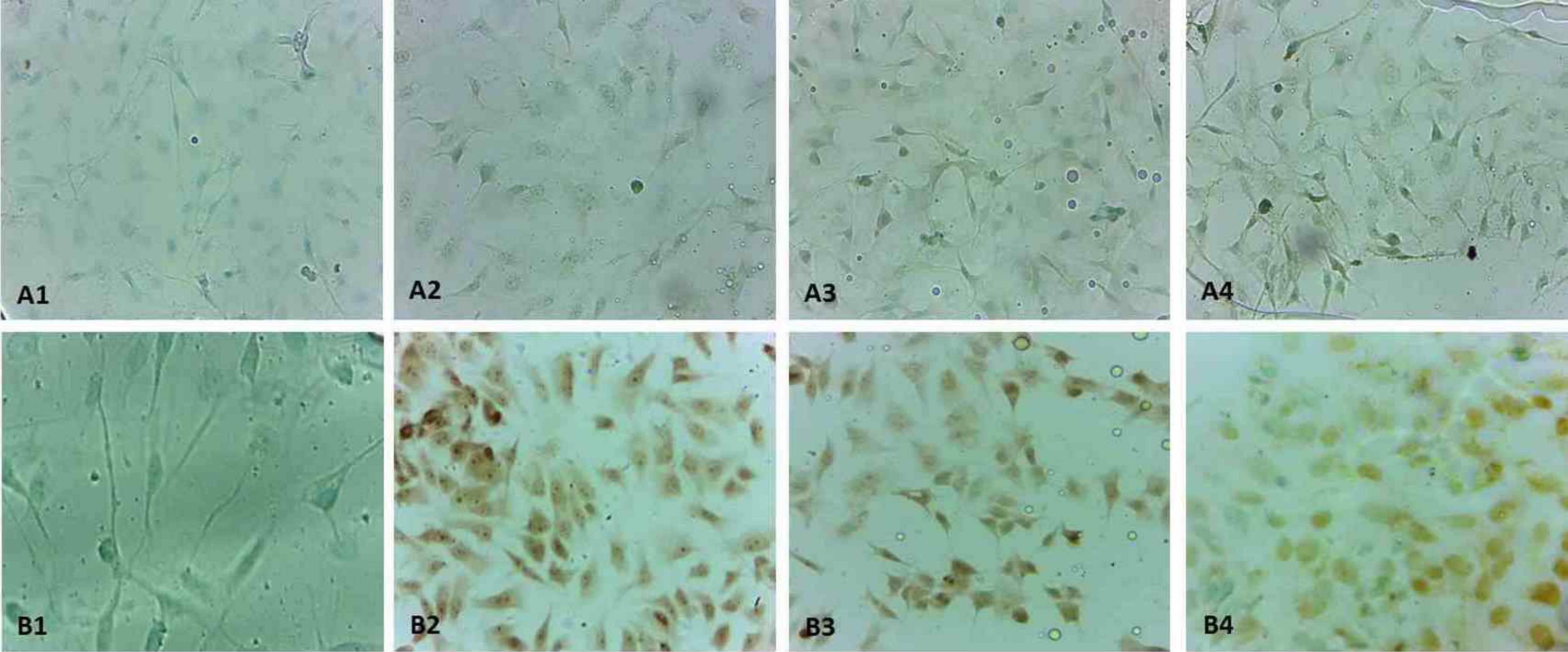
TUNEL immunohistochemistry staining in monolayer culture. Control group was light staining with TUNEL in MDA-MB-231 cells (a1), light staining with TUNEL in MCF-7 cells (b1). The other all groups were high staining with BrdU (A2-24h-MDA-MB231, A3-48h- MDA-MB231, A4-72h- MDA-MB231, B2-24h-MCF-7, B3-48h- MCF-7, B4-72 h- MCF-7) (×40). TUNEL: terminal deoxynucleotidyl transferase dutp nick and labeling; BrdU: 5-bromo-2′-deoxyuridine.
TUNEL analysis for spheroid cultures
While TUNEL-positive cells were detected weakly in the control, there were more TUNEL-positive cells in the silibinin-treated spheroids. This finding supported our monolayer culture results that the spheroids of silibinin group showed more TUNEL-positive MCF-7 and MDA-MB-231 cells (Figures 8(a) and (b) and 9(a) and (b)).

Three-dimentional cell culture TUNEL staining in MDA-MB-231 cells (a). Three-dimentional cell culture TUNEL staining in MCF-7 cells (b). Results are mean of three different experiments and presented as mean ± SEM. TUNEL: terminal deoxynucleotidyl transferase dutp nick and labeling; SEM: standard error mean.

TUNEL immunohistochemistry staining in spheroid culture. Control group was light staining with TUNEL in MDA-MB-231 cells (a1), light staining with TUNEL in MCF-7 cells (b1). The other all groups were high staining with TUNEL (A2-24h-MDA-MB231, A3-48h-MDA-MB231, A4-72h-MDA-MB231, B2-24h-MCF-7, B3-48h- MCF-7, B4-72 h- MCF-7) (×40). TUNEL: terminal deoxynucleotidyl transferase dutp nick and labeling.
Poly-ADP-ribose-polimerase level analysis for monolayer cultures
In this step, we investigated whether silibinin activate caspase-dependent apoptosis by measuring the the cleavage of anti-poly-ADP-ribose-polimerase. We observed that silibinin induced the cleavage of poly-ADP-ribose-polimerase (PARP) in MCF-7 and MDA-MB-231 cells in all intervals (Figure 10(a) and (b) and 11(a) and (b)).

Two-dimentional cell culture PARP staining in MDA-MB-231 cells (a). Two-dimentional cell culture PARP staining in MCF-7 cells (b). Results are mean of three different experiments and presented as mean ± SEM. SEM: standard error mean; PARP: poly-ADP-ribose-polimerase.

Anti-PARP immunohistochemistry in monolayer culture staining. Control group was light staining with anti-PARP in MDA-MB-231 cells (a1) and light staining with anti-PARP in MCF-7 cells (b1). All other groups were high staining with anti-PARP (A2-24h-MDA-MB231, A3-48h-MDA-MB231, A4-72h-MDA-MB231, B2-24h-MCF-7, B3-48h-MCF-7, B4-72 h-MCF-7) (×40). PARP: poly-ADP-ribose-polimerase.
PARP level analysis for spheroid cultures
We observed silibinin caspase-dependent apoptosis by measuring the anti-PARP in 3-D culture. This finding supported our monolayer culture results that the spheroids of silibinin group showed more anti-PARP-positive MCF-7 and MDA-MB-231 cells (Figures 12(a) and ( b) and 13(a) and (b)).

Three-dimentional cell culture PARP staining in MDA-MB-231 cells (a). Three-dimentional cell culture PARP staining in MCF-7 cells (b). Results are mean of three different experiments and presented as mean ± SEM. PARP: poly-ADP-ribose-polimerase; SEM: standard error mean.

Anti-PARP immunohistochemistry staining in spheroid culture. Control group was light staining with anti-PARP in MDA-MB-231 cells (a1), light staining with anti-PARP in MCF-7 cells (b1). All other groups were high staining with anti-PARP (A2-24h-MDA-MB231, A3-48h-MDA-MB231, A4-72h-MDA-MB231, B2-24h-MCF-7, B3-48h-MCF-7, B4-72h-MCF-7) (×40). PARP: poly-ADP-ribose-polimerase.
H-E staining in spheroid cultures
Twenty-five spheroids from all experimental groups, including the control group, were evaluated for 24, 48, and 72 h. Spheroid structure of control group was observed normally. We observed both normal spheroids and separated spheroids in treated with silibinin group. Spheroid structures were also degenerated (Figure 14(a) and (b)).

H-E staining in spheroid culture. Spheroid structure of control group was observed normally in MDA-MB-231 cells (a1) and in MCF-7 cells (b1). Group of treated with silibinin was observed both normal spheroids and separated spheroids. Spheroid structures were also degenerated (A2-24h-MDA-MB231, A3-48h-MDA-MB231, A4-72h-MDA-MB231, B2-24h-MCF-7, B3-48h- MCF-7, B4-72 h-MCF-7) (×40). H-E: hematoxylin and eosin.
Discussion
Cancer may be defined as a disease characterized by uncontrolled multiplication, which spreads within the body through abnormal formation of the body’s own cells. There are three main treatments available to deal with established cancer: surgical excision, irradiation, and chemotherapy. Most anticancer drugs are only antiproliferative; they have no specific effect on invasiveness or the tendency to metastasize. Furthermore, they affect all rapidly dividing cells, including normal tissues, and show dose limiting toxic effects. Hence, an alternative approach would be to use pharmacokinetic principles to optimize drug administration such as targeted drug delivery. 22
Breast cancer is a major public health problem worldwide, and about 70% of primary breast tumors in women are ER-positive (ERα). 23 Phytochemicals, such as flavonoids, have good potential as anti-cancer agents because of their antiproliferative activity against human tumor cell lines, safety, and ability to target multiple cell-signaling pathways. 24,25 Silibinin is a polyphenolic flavonoid antioxidant and the major biologically active compound of milk thistle. It is well-known that silibinin is safe and well tolerated, and it protects the liver from drug or alcohol-related injury. 26,27 Also it has been used as both an antihepatotoxic and an anti-carcinogenic agent. 28 Recently, silibinin has been shown to have substantially higher cytotoxicity to tumor cells and significantly lower toxicity toward normal cells, as determined by the proliferation assay. 29 Silibinin has been shown to exert antitumor activity in various cancer types such as lung, bladder, hepatoma, and prostate carcinoma. 30 Lee et al. have shown that silibinin suppresses PMA-induced matrix metallopeptidase 9 expression in MCF-7 cells by inhibiting the activation of activator protein 1 via the mitogen-activated protein kinases signaling pathways. 17 In this study, we investigated the apoptotic effect of silibinin on MDA-MB-231 and MCF-7 breast cancer cells in both 2-D (monolayer) and 3-D (spheroid) cultures.
The mechanisms of apoptosis are highly complex and sophisticated, involving an energy-dependent cascade of molecular events. Apoptosis is also necessary to rid the body of pathogeninvaded cells and is a vital component of wound healing in that it is involved in the removal of inflammatory cells and the evolution of granulation tissue into scar tissue. 18
The TUNEL method is used to assay the endonuclease cleavage products by enzymatically end-labeling the DNA strand breaks. Terminal transferase is used to add labeled UTP to the 3′-end of the DNA fragments. The dUTP can then be labeled with a variety of probes to allow detection by light microscopy, fluorescence microscopy, or flow cytometry. 18
PARP, the plasma membrane cytoskeletal protein alpha fodrin, the nuclear protein NuMA, and others, which ultimately cause the morphological and biochemical changes seen in apoptotic cells. Due to the dual role of PARP-1 in both DNA repair and apoptosis, the pharmacological use of PARP-1 inhibitors may be able to attenuate ischemic and inflammatory cell and organ injury or may be able to enhance the cytotoxicity of antitumor agents. 31
We determined that the half maximal inhibitory concentration (IC50) values of silibinin in both the MDA-MB-231 and MCF-7 cells were 100 µM/mL at 24, 48, and 72 h of incubation. One-hundred micromoles per milliliter silibinin (IC50 dose) was applied on breast cancer cells (MDA-MB-231 and MCF-7 cells) in both monolayer and spheroid cultures. Then the TUNEL assay and PARP immunohistochemistry staining were used to determine the death of the cells in both the monolayer and spheroid cultures. At all incubation periods (24, 48, and 72 h), TUNEL-positive cell indices of silibinin were found to be significantly different from the TUNEL-positive cell indices of control in monolayer cultures. Silibinin increased the number of TUNEL-positive cells (MDA-MB-231 and MCF-7 cells) and increased the number of PARP-positive cells (MDA-MB-231 and MCF-7 cells) in the monolayer culture. Also, DNA synthesis (S-phase) analysis of breast cancer cells was assessed by BrdU staining. Cells with brown-stained nuclei represent BrdU uptake and considered BrdU-positive. Silibinin reduced the number of cells marked with BrdU (MDA-MB-231 and MCF-7 cells) when compaired with untreated control group. Wang et al. showed that silibinin induces the occurrences of both apoptosis and ROS generation, urging us to focus on mitochondria that play a bifunctional role in apoptosis and oxidative stress. Silibinin-induced MCF-7 cell apoptosis is in concomitance with ROS producing from mitochondria and this process is further augmented by cytochrome
In this study, silibinin applications enhanced the TUNEL-positive cells (MDA-MB-231 and MCF-7 cells) and anti-PARP positive cells (MDA-MB-231 and MCF-7 cells) in comparison to the controls in the multicellular spheroid model.
Conclusions
As a result of our study, it was observed that silibinin inhibits the proliferation and proapoptotic effect on MDA-MB-231 and MCF-7 cell lines by immunohistochemistry staining in both the monolayer and spheroid models.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: The present study was supported by the Research Fund of Mugla Sıtkı Koçman University. Some parts of this manuscript were presented at the 5th International Congress on Cell Membranes and Oxidative Stress: Focus on Calcium Signaling and TRP Channels 9-12 September 2014, Isparta.
