Abstract
Background
Betanin, a natural pigment and glycosidic bioactive compound, possesses anti-inflammatory antioxidant activity.
Objectives
In this study, we evaluated the potential effect of betanin on cigarette smoke extract (CSE)-induced pulmonary inflammation in the zebrafish model.
Materials and Methods
Zebrafish were randomly divided into five groups with control, CSE-exposed and betanin-treated after CSE exposure for 3 and 6 days. Oxidative stress-related parameters like reactive oxygen species (ROS), nitric oxide (NO) and myeloperoxidase (MPO) were analysed, histopathological studies were carried out and gene expression of proinflammatory tumour necrosis factor-alpha, inducible NO synthase and anti-inflammatory interleukin-10 cytokines was determined.
Results
Treatment with betanin reduced ROS, MPO and NO with histopathological improvement, alleviating CSE-induced pulmonary inflammation. Further, decreased expression of the proinflammatory gene and increased expression of the anti-inflammatory gene were observed after 6 days of treatment.
Conclusion
Betanin exhibits a protective effect against CSE exposure by inhibiting oxidative stress and inflammation.
Keywords
Introduction
Chronic obstructive pulmonary disease (COPD) is the third leading cause of death worldwide,1–2 with characteristics of emphysema persistent airflow limitation and impairment of lung function. Cigarette smoking is considered to be the main risk factor for COPD; cigarettes have toxic ingredients that induce inflammation in the respiratory tract. 3 Chronic exposure to cigarette smoke extract (CSE) leads to lung inflammation with infiltration of neutrophil, macrophage, and dendritic cells, which destroys lung tissue and promote remodeling. 4 CSE induces synthesis of reactive oxygen species (ROS), resulting in an imbalance of the oxidant and antioxidant system, which is the main mechanism involved in the onset of COPD. It also promotes proinflammatory cytokines such as tumour necrosis factor-alpha (TNF-α), interleukin-1 (IL-1), interleukin-6 (IL-6), interleukin-8 (IL-8) and granulocyte-macrophage colony-stimulating factor. 5 No treatments are available that can prevent the progressive course of COPD and lung fibrosis. As it includes a cascade of inflammatory events induced by oxidative stress by affecting the activity of enzymes and metabolic pathways, natural compounds can be effective due to their antioxidant activity.
Betanin (betanidin 5-O-b-glucoside), found commonly in beetroot (Beta vulgaris L.), is called betacyanin pigment and shows free radical scavenger activity and induces antioxidant defence mechanisms. 6 It possesses anti-inflammatory, anticancer anti-lipoperoxidation and heme integration activity. 7 Recent research proved the capacity of betanin to suppress NF-κB DNA-binding activity and COX-2 activity with similar or greater effect than anti-inflammatory drugs and some phenolic compounds. 8 However, no study was carried out to study its protective effect on pulmonary inflammation induced by CSE.
The gills of zebrafish exhibit the same gas-exchange function and have the same architecture of mammalian airways, with a mucus-covered respiratory epithelium scattered with immune cells and smooth muscle cells at the base of the lamella. 9 Thus, pulmonary inflammation was induced in the zebrafish model to study the efficacy of betanin for treating lung inflammatory stimuli as observed in COPD cases. The current treatment strategy controls bronchitis, but does not reverse the pathological damage. Hence, there is a great demand for pharmacological agents derived from plants. The current study will enlighten anti-inflammatory and anti-oxidative properties of betanin towards the CSE-induced COPD model.
Materials and Methods
Study Area
This study was carried out at the Public Health Department, School of Health Sciences, Building B, Saudi Electronic University, Jeddah City Sary Road, Faisaliyah Dist, Saudi Arabia from June 2021 to May 2022.
Animal Acclimatation
Adult zebrafishes were purchased from a local vendor. Before the start of the experiment, the animals were acclimatised to the laboratory condition as per organisation for economic co-operation and development (OECD) guidelines (203). According to the guideline, the static system of water is monitored for the optimum temperature, pH, salinity and dissolved oxygen levels on daily basis. The temperature and pH were maintained as 27°C ± 2 and 6.8–7.4 pH, respectively. The zebrafish were fed with commercially available fish pellets from pet shops twice a day. Both male and female zebrafishes were reared on a 12 h L/12 h D cycle. Adult zebrafishes that were healthy and devoid of any malformations or infections were chosen for the study. Fishes were grouped and separated into various tanks for toxicity assessments.
LC50 Determination of CSE
Zebrafishes of interest were weighed and separated into various tanks for the toxicity study. The cigarette smoke was extracted with the help of a manually designed prototype/apparatus. The number of cigarettes was kept as a dosage for acute toxicity assessment. The dosage given was 0.2, 0.4, 0.6, 0.8 and 1.0 cigarettes per litre. Control fishes were also maintained in a separate tank. The toxicity analysis was performed for up to 7 days. The mortality and abnormal behaviour were noted.
LC50 Determination for Betanin
The drug, betanin, was also checked for its toxicity towards the zebrafish. Gavage administration of betanin was carried out for the toxicity assessment. For this, 2 mg/kg, 4 mg/kg, 8 mg/kg and 16 mg/kg range were given as gavage feed. Then, the dose was reduced to 0.5, 1.0, 1.5, 2.0, 2.5 and 3.0 mg/kg. The acute toxicity study was carried out for up to 7 days.
Experimental Model
The zebrafishes with weights similar to both male and female fishes were chosen for the experiment. The fishes were separated into five groups containing 10 fishes each. The process is duplicated for accurate output.
Group 1: Control fishes/untreated. Group 2: Pulmonary inflammation induction by CSE (0.3 cigarette/litre). Group 3: CSE induction with 0.25 mg/kg of betanin treatment. Group 4: CSE induction with 0.50 mg/kg of betanin treatment. Group 5: CSE induction with 0.75 mg/kg of betanin treatment.
Based on the toxicity study of CSE, 0.3 cigarette per litre concentration was given for the induction of pulmonary inflammation in the zebrafish for 7 days. After the induction period, treatment of betanin was initiated and an oral dose was given once daily for every fish based on the weight. The treatment was given for 7 days. The fishes were fed with commercial feed throughout the study. The fishes were assayed for its biological functions on day 3 and day 6. The fishes were euthanised on day 3 for its oxidative stress enzyme analysis and day 6 for analysis of the level of oxidative stress enzymes, gene expression and morphology of gill tissues through histopathology.
Estimation of Nitric Oxide
The amount of nitric oxide (NO) present in the gill tissues on day 3 and day 6 of treated, untreated and induced groups of zebrafish was estimated by the method given in ref 10 , with slight modification. 100 µl of 100 µM nitrite solution was added to the 96-well plate for the nitrite standard reference curve. 50 µl of the appropriate matrix or buffer was dispensed into the remaining wells. By serial dilution, the following concentrations of 100, 50, 25, 12.5, 6.25, 3.13 and 1.56 µM were obtained. The supernatant of ground gill tissue of untreated, induced and treated groups was added. To the wells, 50 µl of the 1% sulphanilamide in 5% phosphoric acid was added and incubated for 5 min at room temperature (RT) in dark. Then, 50 µl of the 0.1% NED Solution was added and incubated at RT for 5–10 min and protected from light. The absorbance was measured at 520 nm. The values were recorded, and the standard graph was plotted.
ROS Estimation
The reduction process of nitroblue tetrazolium (NBT) assay was performed for tissues taken for day 3 and day 6 to estimate the percentage release of ROS, based on the method given in ref 11 , with slight changes. The gill tissue from untreated, treated and induced was ground. The tissue homogenate was washed with PBS twice, and 100 µl of 0.1% NBT solution was added and incubated for 1 h in dark. Then, NBT was removed and washed with 70% methanol for three times. Then, to the washed pellet, 120 µl of 2M potassium hydroxide (KOH) and 120 µl of DMSO were added. The absorbance was recorded at 630 nm with KOH or DMSO as blank.
MPO Activity Assay
MPO assay was carried out using the method given in ref 12 , with slight modification. Gill tissues of zebrafish from various groups were collected and weighed on day 3 and day 6. Based on the tissue weight, the ice-cold 50 mM potassium phosphate buffer (pH 6.0) containing 0.5% cetyltrimethylammonium bromide (CTAB) was added, and the tissue was homogenised. The homogenised tissue was centrifuged at 5000 rpm for 5 min, and the supernatant was collected. 7 µl of tissue homogenate was mixed with 200 µl of O-dianisidine and diluted H2O2 mixture. The sample was measured at 450 nm at 30-second intervals up to 90 s spectrophotometrically. The absorbance was recorded, and MPO activity was expressed as MPO activity (U/mg of tissue).
Gene Expression Analysis
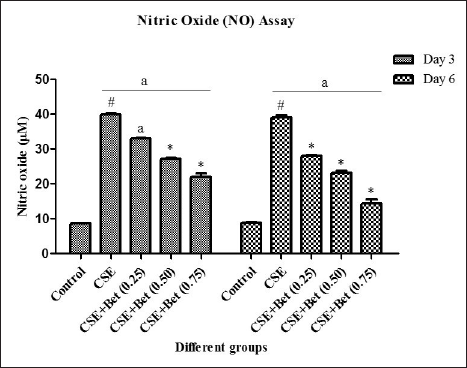
Gill tissues from various groups of fishes were excised on day 6 for the assessment of impairment and rehabilitation of tissue morphology at the molecular level. DNA was extracted from the gill tissues as described in ref 13 , with slight modification. Briefly, the gill tissue was collected and homogenised with digestion buffer, followed by immersion of the tissue in 1.5 ml of digestion buffer with proteinase K at 55°C overnight for lysis. Organic extraction was performed with the phenol: chloroform: isoamylalcohol mixture. The content was centrifuged, and to the aqueous phase, 2X volume of 100% ethanol was added and kept for overnight precipitation. Then, the precipitate was washed and the pellet was air-dried. DNA was diluted with TE buffer and subjected to PCR. Oxidative stress, inflammation and anti-inflammatory genes such as inducible nitric oxide synthase (iNOS), TNF-α and IL-10 respectively were analysed for the level of expression. The primers were designed with the NCBI Primer-BLAST tool. The sequences are given in Table 1.
Primer Sequence.
The PCR mixture was prepared with Emerald master mix, template and forward and reverse primers. The PCR was performed based on optimised PCR conditions for 34 cycles. The PCR products were subjected to qualitative analysis using 1% agarose gel electrophoresis. The gel image was photographed.
Histopathology Analysis
Gill tissues of control, induced and treated groups were taken for the histopathological analysis to study its morphometric alterations and damage. The tissues were stored in 10% formalin, and the tissues were embedded in paraffin wax. Then, the tissue sectioning was subjected to staining with Haemotoxylin and Eosin stains and photographed under a phase contrast microscope (Optica).
Data Analysis
All the experiments were duplicated, and the data were subjected to statistical analysis using GraphPad Prism software, version 5.1. The data are expressed as mean ± SD. The results were analysed with two-way analysis of variance, and the treatments were analysed by Tukey’s multiple comparison test. p < 0.05 was considered as significant.
Results
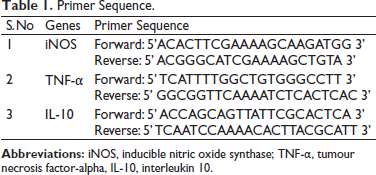
Acute Toxicity Assessment of CSE
Lethal Concentration to kill half of the population (LC50) was studied for CSE between 0.2 and 1.0 cigarette/litre. Figure 1 shows the survival percentage of zebrafishes in CSE. It is evident that there was 100% survival at 0.2 and 0.4 cigarette/litre, whereas at 0.6, 0.8 and 1.0 cigarette/litre, the percentage was decreased in a dose-dependent manner. LC50 was fixed as 0.6 and showed 54.16% mortality, which is indicated as the dotted line. The dose of CSE was fixed as 0.3 cigarette/litre for the induction of pulmonary inflammation based on the result of acute toxicity determination that showed 100% survival at 0.3 cigarette/litre up to 7 days. The result is presented as mean ± SD and error.
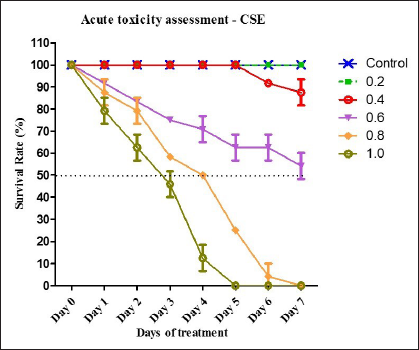
Lethal Concentration Determination for Betanin
The toxicity analysis was performed for betanin. Initially, the dose was taken as 2, 4, 8 and 16 mg/kg for the study. The 100% mortality was observed for 8 and 16 mg/kg within 24 h. Data were not shown. Then, the dose was reduced to 0.5, 1.0, 1.5, 2.0, 2.5 and 3.0 mg/kg. Figure 2 shows the acute toxicity analysis of betanin. Betanin showed 100% survival up to 1.5 mg/kg. The mortality rate slightly increased at 2.0 mg/kg, whereas at 2.5 and 3.0 mg/kg, there was a moderate increase in the mortality rate. The LC50 value was fixed as 3.0 mg/kg, which showed 50% mortality on day 6, as indicated by the dotted lines. The betanin dose for treatment is fixed as 0.25, 0.50 and 0.75 mg/kg based on the acute toxicity analysis. The result is presented as mean ± SD and error.
Lethal Concentration Determination for Betanin. LC50 Determination of Betanin was Plotted against the Days of Treatment based on the Different Oral Dosage Range of Betanin. The Survival Percentage was Shown Up to 7 Days. The Results were Represented as Mean ± SD and Error.
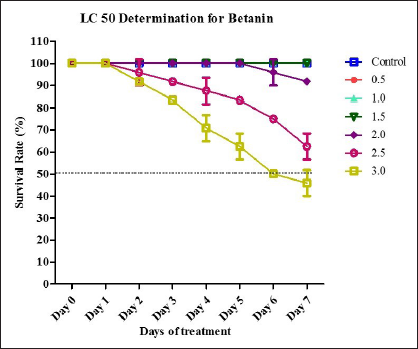
NO Estimation
The synthesis of NO level was determined on day 3 and day 6, which is shown in Figure 3. An elevated level of NO was observed in an induced group such as 39.9 ± 0.16 for day 3 and 39 ± 0.01 for day 6 compared to the control. The treatment of pulmonary inflammation was given with betanin at 0.25, 0.5 and 0.75 mg/kg. The figure shows the slight reduction in the NO level in a dose-dependent manner on day 3. In day 6, the moderate reduction of NO was observed when compared to day 3 in a dose-dependent manner. The alleviation of NO was significant in high doses of betanin, and there was no significance between the treatment periods, that is, day 3 and day 6.
ROS Determination
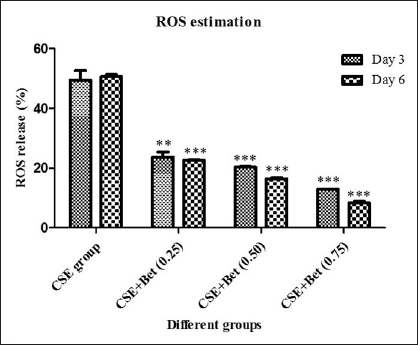
The percentage of ROS was estimated. Figure 4 represents the percentage of ROS levels in induced, control and treated groups. From the figure, it is clearly indicated that the CSE induction escalates the percentage of ROS compared to the control as 49.53 and 50.64 for day 3 and day 6, respectively. Betanin treatment showed a significant reduction in the percentage of ROS in a dose-dependent manner. There was an appropriate reduction was observed in day 3 and day 6 compared to the induced group. 0.75 mg/kg dosage showed a high impact in the reduction of ROS percentage as 12.97 and 8.35 for day 3 and day 6, respectively.
Myeloperoxidase Enzyme Estimation
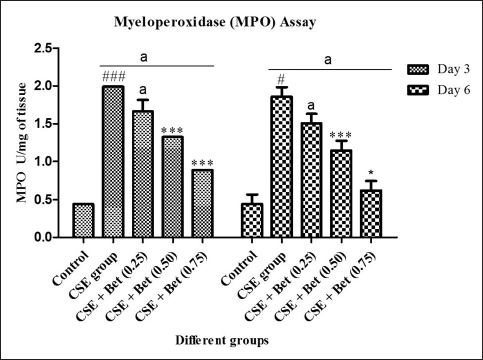
Myeloperoxidase (MPO) activity was determined to know the oxidative stress responses in inflammatory diseases, and the impact of the treatment on MPO activity is shown in Figure 5. The MPO activity of the induced group was significantly high, 1.99 and 1.85 U/mg on day 3 and day 6, respectively, compared to the control group. Betanin treatment provides dosage dependent attenuation in the MPO activity, 1.65, 1.32 and 0.88 U/mg of tissue was observed for day 3 and 1.50, 1.15 and 0.61 U/mg of tissue was observed for day 6 for 0.25, 0.50 and 0.75 mg/Kg of betanin respectively.
Gene Expression Analysis
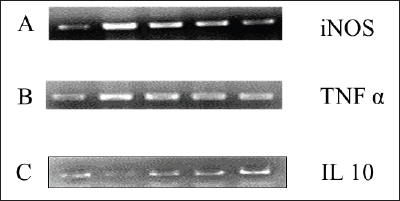
The molecular level of analysis was carried out for untreated, induced and treated groups. The inflammatory genes TNF-α and iNOS showed upregulation in the induced group, and the expression was reduced based on the betanin dose range in treated groups (Figure 6A and 6B). The anti-inflammatory gene IL-10 showed downregulation in the induced group, whereas in treated groups, the level of gene expression increased in a dose-dependent manner (Figure 6C).
Histopathological Examination
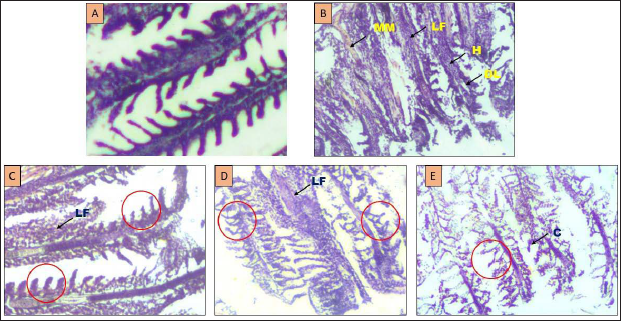
Figure 7 shows the histopathological analysis of gill tissues from control, induced and treated groups of day 6. The control group showed normal architecture with proper gill filament, primary and secondary lamella. The induced group indicates the massive disintegration of tissue with lamellar fusion, hyperemia and abrasion of tissue cells. After treatment, the damaged tissue architecture was significantly restored in a dose-dependent manner.
Discussion
Cigarette smoking contributes to the imbalance of oxidant/antioxidant due to exogenous ROS and results in the inflammatory process and mitochondrial dysfunction due to endogenously released ROS, providing the path for disease progression. Betanin are red pigments found in beetroot (B. vulgaris), amaranths, rose and cactus pear. It contains phenolic and cyclic amine groups, which are good electron donors with exceptionally high free radical scavenging ability. Several studies have shown that betanin possesses antioxidant and anti-inflammatory activity. In this study, pulmonary inflammation was established in the zebrafish model in order to investigate the effect of betanin on cigarette smoking-induced COPD.
With the purpose of establishing the model of CSE-induced COPD, the zebrafish was exposed to CS extract. LC50 dosage was kept constant and fixed for establishing the lung inflammation model. 0.3 cig/L dosage was given to induce pulmonary inflammation for up to 7 days. Similarly, drug toxicity was also determined up to 16 mg/kg and the dosage of 0.25, 0.50 and 0.75 mg/kg was fixed for the study.
Our investigation of CSE resulted in increased oxidative stress via increasing ROS, NO and MPO, which was associated with increased levels of TNF-α and iNOS and decreased levels of IL-10 gene expression in gill tissue. Betanin was able to reduce ROS levels in a time-dependent manner and dose-dependent manner, which was highly significant on the 6th day (0.25, 0.50 and 0.75 mg/kg). When the delicate balance between ROS and antioxidant production surpasses the limit after exposing to CSE, it induces oxidative stress resulting in cell injury. The MPO level is also increased with ROS, which is the main enzyme involved in phagocytes to produce ROS, leading to macrophage activation along with increase in NO production.14, 15 Thus, the CSE-exposed group showed a significant increase in ROS, MPO, and NO, which decreased with treatment based on dosage and time duration. As flavonoids are antioxidants, they can scavenge free radicals and no longer react with NO and result in less damage.16, 17 Our finding coincides with pre-treatment of quercetin, which reduced ROS and NO to baseline levels due to the high rate of diffusion through cell membranes and on the surfaces of lipid bilayers and also in the aqueous phase. 18
Betanin could enhance IL-10 production, an anti-inflammatory compound, by combined inhibition of TNF-α and iNOS in gill tissue. The expression of cytokine TNF-α plays an important role in driving the expression of iNOS in inflammatory conditions. 19 In the present study, a consistent increase in TNF-α has been observed, which in turn increases IkB-α degradation, leading to NF-κB activation and transcription of several inflammatory genes, including iNOS, which produces a large amount of NO, 20 which is consistent with a possible role of this cytokine.21–23 Many flavonoid shaves proved inhibition effect on TNF-α and iNOS,24, 25 in inflammatory condition. Catechin and quercetin showed similar results with a concomitant increase of IL-10 expression with a decrease in TNF-α. 26
We found damages in gill lamella and filaments after exposure of zebrafish to CSE for 6 days. It also showed neutrophil infiltration in the CSE-exposed group. Gills absorb toxic substances dissolved in the aquatic environment due to direct water and gaseous exchange.27, 28 After treatment with betanin, lamellae and filament damage reduced in a dose-dependent manner with restoration of the morphological architecture of gills. A decrease in neutrophil infiltration was also observed, indicating the potency of betanin against pulmonary inflammation.
Conclusion
In conclusion, our study confirms that CSE exposure enhances the release of ROS, MPO and NO in the gill tissue of the adult zebrafish model. Betanin acted on oxidative stress by elevating anti-inflammatory cytokines and reducing proinflammatory cytokines in pulmonary inflammation induced by CSE. Extensive studies are needed to explore the mechanisms involved in inhibition of pulmonary inflammation by betanin to substitute as potential herbal medicine. Though the effect has been studied and confirmed in zebrafish model, it is very important to duplicate a similar effect in animal and human trials to elaborate the mechanism of action.
Summary
Cigarette smoking is considered to be the main risk factor of COPD caused by toxic ingredients which induces inflammation in respiratory tract. This study mainly focused on the potential effect of betanin on CSE induced pulmonary inflammation in zebrafish model.
Betanin showed significant reduction in oxidative stress conditions induced by CSE that was confirmed by NO, ROS and MPO assays.
Further, the rehabilitation of gill tissues by betanin treatment was confirmed with histopathological analysis.
Down regulation of proinflammatory gene (TNF-α), NO synthase gene (iNOS) and up regulation of anti-inflammatory gene (IL-0) confirms the anti-inflammatory and antioxidant potency of betanin at molecular level.
Footnotes
Abbreviations
Declaration of Conflicting Interests
The author declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
