Abstract
Background
Sugreen-120 is one of the famous Indian polyherbal formulations used for the treatment of diabetes. Due to a lack of scientific evidence, the present study is aimed at investigating the phytopharmacology of Sugreen-120 concerning its antioxidant and antidiabetic characteristics.
Materials and Methods
Total phenols and flavonoid content followed by 2,2-diphenyl-1-picrylhydrazyl (DPPH) free radical scavenging activity was estimated in Sugreen-120. α-Amylase and α-glucosidase inhibitory action of Sugreen-120 was estimated to evaluate its antidiabetic potential. Thin layer chromatography (TLC)-bioautography-MS analysis was performed to determine DPPH free radical, α-amylase and α-glucosidase inhibitory phytoconstituents in Sugreen-120. High performance thin layer chromatography (HPTLC)-based quantitative analysis of Sugreen-120 was performed for the simultaneous separation of caffeic acid and kaempferol. In silico docking analysis was performed to determine the effect of Sugreen-120 metabolites against α-amylase and α-glucosidase enzymes.
Results
The results showed that Sugreen-120 is enriched in total phenols and flavonoids and even has good potential to scavenge DPPH free radicals with an inhibitory concentration (IC50) value of 414.59 ± 4.925 µg/mL. In α-amylase and α-glucosidase inhibitory assays, the efficacy of Sugreen-120 was found in a dose-dependent manner and the IC50 values were found as 220.106 ± 1.375 and 441.44 ± 1.992 µg/mL, respectively. TLC-bioautographic analysis showed that 06 constituents were found active against DPPH free radical, 04 constituents active against α-amylase and 03 constituents active against α-glucosidase. A HPTLC quantitative study revealed the content of caffeic acid and kaempferol to be 5.233 ± 0.026 and 16.959 ± 0.036 µg/mg, respectively. In silico docking analysis showed that out of 5 identified metabolites, myricetin, ellagic acid, and kaempferol were found with significant interaction with α-amylase and α-glucosidase proteins. Hence, it can be concluded that Sugreen-120 exhibits not only an antidiabetic effect but also antioxidant potential. It can be a palliative choice and an alternative that can be used for the treatment of diabetes.
Introduction
Diabetes mellitus has been acknowledged as one of the serious metabolic disorders characterized by decreased insulin secretion and sensitivity, decreased glycogenesis, increased gluconeogenesis, disturbed regulation of catabolic function, and so on. Besides, enzymes such as α-amylase and α-glucosidase exert a potential role in the digestion of polysaccharides by breaking down the alpha bonds of alpha-linked polysaccharides, whereas α-glucosidase is responsible for the digestion of postprandial polysaccharides, which results in increased hyperglycemia.1, 2 It has been reported that hyperglycemia or increased sugar in the blood causes severe damage to the body’s systems, particularly the blood vessels and nerves. Along with this, the disturbing catabolic function produces several oxidants or free radicals in the body, which results in the induction of serious oxidative or inflammatory stress. An instantaneous therapy, such as α-amylase, α-glucosidase inhibitors, and antioxidants, is effective in preventing onset of diabetes and oxidative stress. 3
World Health Organization (WHO) reflected that diabetes can be the seventh-lethal disease by 2030, whereas the International Diabetes Federation (IDF 2019), 9th edition, report confined that diabetes is gaining exponential growth in the prevalence of diabetic patients and can be the rapid-growing health emergency of the 21st century. 3
Therefore, to inhibit the prevalence of such metabolic disorders, the modern system of medicine covenants with voluminous classes of oral antidiabetic drugs, such as α-glucosidase inhibitors, biguanides, sulfonylureas, thiazolidinediones, and non-sulfonylureas secretagogues, are strongly hyphenated to reduce the increased blood glucose level. Regardless of the significant progress of these medicines, the treatment of diabetic patients is still far from perfection due to their several constraints, such as adverse effects, drug resistance, and even toxicity. The consideration of such factors prompts healthcare providers and researchers to improve diabetic treatment and develop newer antidiabetic drugs as an alternative for natural sources.4, 5
Since history, herbal medicines and their derivatives have been progressively used for the treatment of several acute and chronic ailments.6, 7 Recently, herbal medicines have gained exponential growth in the treatment of diabetes and are becoming the first choice of people as an alternative and complementary medicine due to their lack of or minimal side effects, easy availability, accessibility, and producibility.8, 9 The complexity of the phytoconstituents present in herbal medicine or herbal products makes them more active therapeutically and synergistically. Besides the analysis of herbal medicine, still there are lacking factors in the assessment of their quality and safety despite the long history of their quality and safety assessment.10, 11 Further, several types of research have been focused particularly on the development of chromatographic and spectroscopic methods to validate herbal medicine or phytoconstituents quantitatively and qualitatively, which steps us toward more formalized analytical and biological investigations. 3
Thin layer chromatography/High performance thin layer chromatography (TLC/HPTLC) techniques are more authentic in fingerprinting, profiling, and quantitative assessment of the phytoconstituents. The hyphenation of HPTLC with spectroscopic techniques such as mass spectroscopy (MS) and nuclear magnetic resonance (NMR) methods, as well as the techniques hyphenated with bio-techniques, comprehend the various phytoconstituents and the target-based screening of phytoconstituents activity.9, 12 Such techniques are being used more and more in the field of herbal medicine or natural products to investigate bioactivity-based screening of phytoconstituents. TLC-bioautography and MS-based identification of bioactive phytoconstituents is being more formalized in the validation of herbal medicine or products with their quality and safety aspects. 13
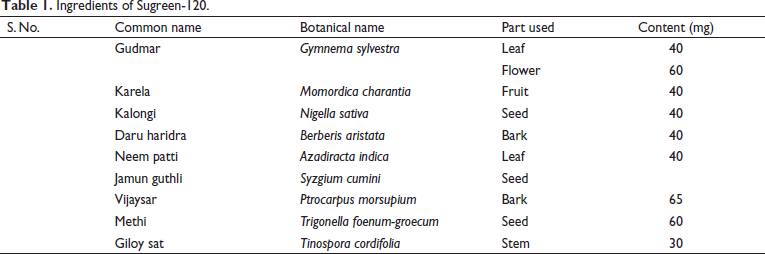
Sugreen-120 is one of the famous polyherbal formulations of India acknowledged, for the treatment of diabetes. It is comprised of Gymnema sylvestra, Momordica charantia, Nigella sativa, Berberis aristata, Azadiracta indica, Syzgium cumini, Ptrocarpus morsupium, and so on. The ingredients of Sugreen-120 are summarized in Table 1.
Ingredients of Sugreen-120.
Due to the lack of scientific evidence, the facts prompted us to undertake the study aimed at evaluating the antioxidant and antidiabetic potential of Sugreen-120 in terms of DPPH free radicals, α-amylase, and α-glucosidase inhibitory assays. Furthermore, TLC-MS bioautographic studies were performed to identify the phytoconstituents that accounted for its therapeutic potential.
Materials and Methods
Folin Ciocalteu (FC) reagent (2973825), sodium carbonate (A07Z/2006/2512/13), and aluminum chloride (E08Z/0908/2603/13) were procured from S.D Fine Chem Pvt. Ltd, Mumbai, India. α-Amylase (CAS RN 9000-90-2), α-glucosidase (CAS Number: 9001-42-7), pNPG (CAS Number: 2207-68-3), fast blue (CAS Number: 14263-94-6), 2,2-Diphenyl-1-picrylhydrazyl (DPPH) (CAS Number:1898-66-4), Ascorbic acid (CAS Number:50-81-7). HPTLC system (CAMAG, Muttenz, Switzerland), TLC Silica gel 60 F254 (Merck KGaA, 64271 Darmstadt, Germany), caffeic acid (Lot # MKBQ5343V), and kaempferol (Lot # BCBZ6403). Sugreen-120 was obtained as a gift sample from Drug Laboratories, Hajipur, Hapur Road, Ghosipur, Meerut-250002, Uttar Pradesh, India.
Collection and Preparation of Samples
Sugreen-120 tablets were procured from the Drug Laboratories, Hajipur, Hapur Road, Ghosipur, Meerut, Uttar Pradesh, India, as a gift sample to the Bioactive Natural Products Laboratory, Jamia Hamdard, New Delhi, India. Furthermore, one pack of 60 tablets of Sugreen-120 was powdered using a mortar and pestle, and the average weight of the powder was calculated in proportion to the weight of one tablet. The powder was subjected to cold maceration in 380 mL of methanol. The extraction process took 24 h. After the extraction process, the content was filtered using Whatman’s filter paper, and the filtrate was evaporated at reduced pressure to obtain a dried residue. The extractive value of the extract was calculated and stored in a suitable container for further analysis. 3
Total Phenolic and Flavonoid Contents
The total phenol content of the Sugreen-120 was determined using the FC method with some modifications. 11 Briefly, 5 mg/mL of a stock solution of Sugreen-120 extract was prepared in methanol. A volume of 500 µL stock solution from each batch was mixed with 2.5 mL of FC (1:10, v/v). After mixing, 2.5 mL of sodium bicarbonate solution (7.5%) was added and allowed to stand for 30 min with intermittent shaking. The absorbance was measured at 765 nm using a spectrophotometer. The total phenolic content in Sugreen-120 was calculated from a calibration curve of standard gallic acid (31.25–500 µg/mL), and the result was expressed as micrograms (µg) of gallic acid equivalent/mg of extract (µg GAE/mg extract).
The total flavonoid concentration of Sugreen-120 was determined by the aluminum chloride method with some modifications. 11 Briefly, 5 mg/mL of a stock solution of Sugreen-120 extract was prepared in methanol. A volume of 500 µL of stock solution from each batch was mixed with 1.5 mL of methanol. After 5 min, 0.1 mL aluminium chloride (10%), 0.1 mL sodium acetate (1 M), and 2.8 mL water were added. After incubation for 40 min, absorbance was measured at 415 nm using a spectrophotometer. The total flavonoid content in Sugreen-120 was calculated from a calibration curve of standard rutin (31.25–500 µg/mL), and the result was expressed as µg rutin equivalent/mg extract (µg rutin/mg extract).
DPPH Free Radical Scavenging Activity
The antioxidant activity of Sugreen-120 extract was determined by the described protocol with some modifications.
11
In brief, 20 µL of the sample from each concentration was mixed with 180 µL of DPPH solution in methanol (0.01 mM) solution. The obtained mixtures were incubated for 30 min at room temperature in a relatively dark place, and then absorbance was read using a spectrophotometer at 517 nm. Ascorbic acid was used as a positive control to consider the efficacy of Sugreen-120 in the proportion of ascorbic acid. The percent scavenging curve was plotted against the extract concentration and percent scavenging activity. The DPPH scavenging effect was calculated using the following equation:
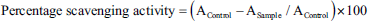
α-Amylase Inhibitory Activity
The α-amylase inhibitory activity of Sugreen-120 was determined by the described protocol with some modifications.
14
Briefly, 40 µL of the sample from each concentration was mixed with 40 µL of amylase solution (4 units/mL in sodium phosphate buffer pH 6.7) in each defined well of the 96-well plate. The obtained mixtures were incubated at 37°C for 30 min. After incubation, 40 µL of starch solution (0.1%) was added to the mixture. After 10 min, 20 µL of hydrochloric acid (IM) was added to stop the enzyme and substrate reaction, and 100 µL of iodide solution (5 mM iodine + 5 mM potassium iodide in distilled water) was added, and the absorbance was measured at 580 nm. Acarbose was used as the standard.
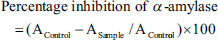
α-Glucosidase Inhibitory Activity
The α-glucosidase inhibitory activity of Sugreen-120 was determined by the described protocol with some modifications.
14
Briefly, 120 µL from each concentration sample was mixed with 20 µL of α-glucosidase solution (1 U/mL in 0.1 M potassium phosphate buffer, pH 6.8) in each defined well of 96-well plate. The obtained mixtures were incubated for 15 min at 37°C. The reaction was initiated by adding 20 µL of para-nitrophenyl-α-d-glucopyranoside (5 mM) and the mixtures were further incubated for 15 min. The reaction was terminated by adding 80 µL of 0.2 M sodium carbonate, and then absorbance was measured at 405 nm. Acarbose was used as the standard. The percent inhibition of α-glucosidase was calculated using the equation:
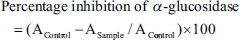
TLC-Bioautographic-MS Analysis
TLC-Bioautography assays were performed to determine free radical scavenging, α-amylase and α-glucosidase inhibitory compounds through DPPH free radical scavenging, and enzyme and substrate reaction over the developed TLC plate. For bioautographic analysis, each plate was developed in a pre-saturated TLC development chamber containing toluene:ethyl acetate:glacial acetic acid (6:3:2 drops, v/v/v) as a solvent system. After development, bioautography analysis was performed to screen the bioactive constituents followed by the MS analysis as performed in our earlier report 3 using a mass spectrometer of Water’s ACQUITY UPLC (TM) system (Waters Corp., MA, USA) equipped with a tunable MS detector and monolithic capillary silica-based C18 column (ACQUITY UPLC(R) BEH C18 1.7 µm, 2.1 × 100 mm) for identification of the active principles in Sugreen-120.
TLC-Bioautographic Assay for Antioxidant Activity
DPPH free-radical scavenging compounds in Sugreen-120 were determined as per the standard protocol with some modifications. 3 In brief, the developed TLC plate was dried and cut from two separate tracks, which were sprayed/dipped in a methanolic solution of DPPH (5 mM). Thereafter, the yellowish color bands appeared against the purple background, indicating that the bands are active against free radicals of DPPH. The images of the developed chromatogram were captured for record purposes.
TLC-Bioautographic Assay for α-Amylase Activity
α-Amylase inhibitory compounds in Sugreen-120 were determined by enzyme and substrate reaction per the standard protocol with some modifications. 3 Briefly, the developed TLC plate was dried and cut from two separate tracks, which were sprayed/dipped in a prepared enzyme solution (10 mg) and incubated for 1.5 h in a humid desiccator with proper adjustment of humidity under the desiccator. After the period of incubation, the plate was dipped in of the starch solution (1%) as substrate and further incubated for 20–30 min for enzyme and substrate reaction. Finally, Gram’s iodine solution was used as an indicator for observation of active bands against α-amylase. The active bands appeared as a violet spot against a dark brown color background. The images of the developed chromatogram were captured for record purposes.
TLC-Bioautographic Assay for α-Glucosidase Activity
α-Glucosidase inhibitory compounds in Sugreen-120 were determined by enzyme and substrate reaction per the standard protocol with some modifications. 3 Briefly, the developed TLC plate was dried and cut from two separate tracks, which were sprayed/dipped in a prepared enzyme solution (100 U/10 mL) and incubated for 1.5 h in a humid desiccator with proper adjustment of humidity under the desiccator. After the period of incubation, the TLC plate was further dipped in the substrate mixture of Fast-Blue B salt solution (2.5 mg/mL) and 2-naphthyl-α-d-glucopyranoside (2 mg/mL) in 1:1 (v/v) ratio for the completion of enzyme and substrate reaction. The plate was then incubated for another 5 h to complete the reaction. Glucosidase activity was visible on the TLC plate by the appearance of the white spot on a purple/violet background, and the images of a developed bioautogram were captured.
Quantitative Analysis of Marker Compounds by using HPTLC
A volume of 30 mg of Sugreen-120 methanolic extract and 1 mg of caffeic acid and kaempferol were dissolved individually in HPLC grade solvent, vortexed, and then filtered using a 0.22 µ PTFE membrane filter. Furthermore, from the stock solution of each standard, 0.5 µL was mixed to get a 500 µg/mL concentration of each marker. Afterward, CAMAG Linomat-V (CAMAG, Switzerland) was used to apply 6 µL from the sample and 0.2–4 µL of mix standard to obtain a linearity curve. Each applicant was applied simultaneously with a 6-mm wide band length to pre-washed and activated Silica gel 60 F254 pre-coated HPTLC plates (20×10 cm; Merck, Germany). The nitrogen flow was set up for a delivery speed of 150 nL/s. Thereafter, the TLC plate was developed in a pre-saturated TLC development chamber containing toluene:ethyl acetate:glacial acetic acid (6:3:1 v/v/v) as a solvent system. The developed plate was visualized and scanned at 254 nm and 366 nm, respectively. The quantitative analysis for each marker was performed using the CAMAG TLC scanner III WinCATS 1.2.3 software and the obtained peak intensity at 254 nm. 3
In silico Docking Analysis
In silico docking analysis was performed using Autodock Vina software for estimation of the biological interaction of identified metabolites in Sugreen-120 against α-amylase and α-glucosidase enzymes. The analysis was performed as per the standard protocol with some modifications.
15
The three-dimensional crystal structures of alpha-amylase (PDB ID: 3BAI) and alpha-glucosidase (PDB ID: 2QMJ) with a resolution of 1.9 Å) were downloaded from the Research Collaboratory for Structural Biology (RCSB) protein data bank (
Statistical Analysis
Data are represented statistically as mean ± SD (n = 3) using one-way ANOVA analysis followed by the Tukey test to compare all the columns of the respective data. The p-value and summary were considered the significance level, while p-value of 0.05 was considered statistically significant.
Results and Discussion
Methanolic extract of Sugreen-120 was prepared using shacking method successively. The extractive yield of methanolic extract was found to be 2.796 ± 0.193%. The obtained extractive yield was processed for further analysis.
Total Phenols and Flavonoids
The total phenol and flavonoid content of the Sugreen-120 was determined by using the FC aluminum chloride method successively. Sugreen-120 is rich in phenols and flavonoids, with concentrations of 73.026 1.657 and 56.4 0.995 g/mg, respectively, equivalent to gallic acid and rutin/mg of extract. The phenolic and flavonoid content of Sugreen-120 was found to be higher than its major single herbal ingredient, Gymnema sylvestra. 11
Polyphenols such as phenols and flavonoids are highly abundant throughout the plant, and such phytoconstituents provide self-defense to the plant. Besides, polyphenols are reported to have strong antioxidant activity through the suppression of biological free radicals generated by the dysregulated catabolic function of the body. In the advent of polyphenols, several studies have been conducted to evaluate the antidiabetic potential of polyphenols, and the great chunk is that polyphenols such as gallic acid, ferulic acid, and rutin are strong candidates as antidiabetic or even antioxidants.3, 16, 17 Some of the studies reported on polyphenols or rich fractions suggest that plant polyphenols act as dietary antioxidants in human health and potentiate a defensive counterpane against deleterious diseases including diabetes. 18 Moreover, Ahangarpour et al. reported that phenols have a multidimensional role in the management of diabetes. Such phytoconstituents suppress hepatic gluconeogenesis by inhibiting glucose-6-phosphatase and increasing insulin sensitivity. Besides, these regulate the calcium channels, which play a crucial role in insulin secretion, stimulate glucose uptake, inhibit lipid peroxidation, and scavenge superoxide anion and hydroxyl radicals. Polyphenols exert a protective action as a guard for pancreatic cells, which inhibits damage and further fatality or death.3, 16, 17
DPPH Free Radical Scavenging Activity
The antioxidant activity of Sugreen-120 extract was determined by DPPH free radicals scavenging of Sugreen-120 successively. The resulted data were expressed statistically as mean ± SD, which reveals dose-dependent inhibition of DPPH free radicals by Sugreen-120. The highest concentration (500 µg/mL) of Sugreen-120 inhibits 58.423 ± 1.375% of DPPH free radicals, while the IC50 of Sugreen-120 was found as 414.59 ± 4.925 µg/mL. The experimental outcomes of Sugreen-120 against DPPH free radicals scavenging activity have been represented in Figure 1.
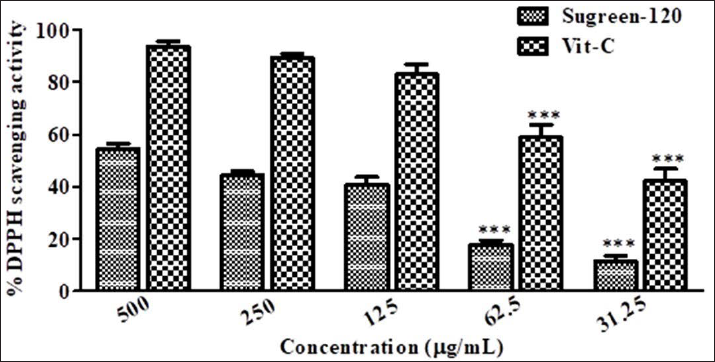
The assessment of the antioxidant capacity of natural products or herbal medicines through the DPPH method is gaining adventurism in the research field of herbal medicines. It is the most widely used method throughout the globe. Based on the spectrophotometric techniques and measurement of stable color dark purple to yellow is directly the proportion of free radicals quenched by the phytoconstituents, which potentiates antioxidant activity. Sugreen-120 is rich in secondary metabolites and includes phenol and flavonoids that have antioxidant activities, owing to its redox characteristics. Despite this, polyphenols strengthen the efficacy of antioxidant enzymes such as superoxide dismutase (SOD), glutathione peroxidase (GPx), glutathione reductase (GR), and catalase (CAT), which are responsible for the management of the redox system against reactive oxygen or nitrogen species (ROSN) that cause oxidative stress. In the cited study, it has been reported that dietary polyphenols have enough efficacy to exhibit antioxidant potential against ROSN-induced oxidative stress. As far as, polyphenols are known for their essential starring role in subduing the efficacy of excessive ROS by contravention of quenching the trace elements, autoxidative chain reaction, and averting the complete cellular injury.
α-Amylase Inhibitory Activity
The enzyme α-amylase is a calcium-containing metalloenzyme accountable for the breakdown of polysaccharides to monosaccharides in the oral cavity from their α-1, 4 linkages. The excessive metabolism of carbohydrates and inhibition of α-amylase can significantly reduce the elevated level of postprandial blood glucose so that it can be an important strategy for the regulation of hyperglycemia or even management of type-2 diabetes. Thus, it is necessary to invent a new alternative for the inhibition of such enzymes in nature to mimic the further side effects of modern medicines. Therefore, the antidiabetic potential of Sugreen-120 was determined by its α-amylase inhibitory potential. The resulted data were expressed statistically as mean ± SD, which reveals that the average inhibition of α-amylase at the highest concentration (500 µg/mL) of Sugreen-120 was found as 77.35 ± 2.844%, while the IC50 of Sugreen-120 was found as 220.106 ± 1.375 µg/mL. Acarbose was used as positive control that showed 64.4145 ± 4.314% inhibition at the highest concentration (500 µg/mL), while the IC50 of acarbose was found to be 300.82 ± 1.864 µg/mL. It has been found that Sugreen-120 has significant inhibitory potential against α-amylase and thus can be a strong candidate in the management of diabetes (Figure 2).
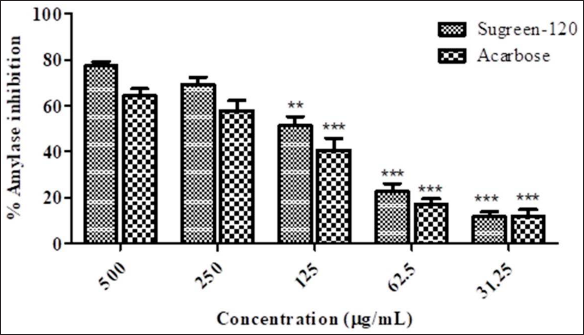
Previous studies reported that herbal medicine such as G. sylvestra is known for its strong antidiabetic activity via inhibiting α-amylase activity. 11 Poovitha et al., evaluated α-amylase and α-glucosidase inhibiting activities of two different varieties of bitter gourd (M. charantia) protein extracts and reported significant inhibition of α-amylase and α-glucosidase activity along with both the protein extracts significantly decrease peak blood glucose and area under the curve in streptozotocin-induced diabetic in Wistar albino rats. Furthermore, it was confirmed that protein extract of bitter gourd can be accounted to normalized the level of increased hyperglycemia. 19
Moreover, natural ingredients, especially acarbose, have been used in the management of diabetes. It regulates the function of PPG and HbA1c, which are altered in cases of hyperglycemia. Furthermore, it is reported that α-amylase inhibitors block or shutdown intestinal absorption of starch and other oligosaccharides into the gastrointestinal tract (GIT) mainly by blocking the hydrolysis of 1,4-glycosidic linkages. Besides, diabetes-induced oxidative stress causes slow advancement in the management of diabetes due to the development of free radicals produced by glucose oxidation and the consequent glycated proteins oxidative degradation.1, 3 Sugreen-120 are phytoconstituents that may be responsible for preventing such complications.
α-Glucosidase Inhibitory Activity
The enzyme α-glucosidase is a vital enzyme that plays a crucial role in the final step of digestion. Typically, it works on the breakdown of polysaccharides to di/monosaccharides by breaking glycosidic linkage, reducing postprandial plasma glucose levels, delaying glucose absorption, and reversibly suppressing postprandial hyperglycemia. 20 Many of the natural compounds are strongly responsible for the treatment of hyperglycemia by reducing blood glucose levels, inhibiting the breakdown of polysaccharides to monosaccharides, and decreasing or inhibiting α-glucosidase sensitivity. Therefore, the α-glucosidase inhibitory potential of Sugreen-120 was determined through enzyme and substrate reaction. The resulted data were expressed statistically as mean ± SD, which reveals that the average inhibition of α-glucosidase at the highest concentration (500 µg/mL) of Sugreen-120 was found to be 50.261 ± 2.819%, while the IC50 of Sugreen-120 was found to be 441.44 ± 1.992 µg/mL. Acarbose was used as a positive control, which showed 58.919 ± 3.457% inhibition at the highest concentration (500 µg/mL), while the IC50 of acarbose was found to be 348.22 ± 1.447 µg/mL. Hence, it has been found that Sugreen-120 has significant inhibitory potential against α-glucosidase and thus can be a strong candidate in the management of diabetes (Figure 3).
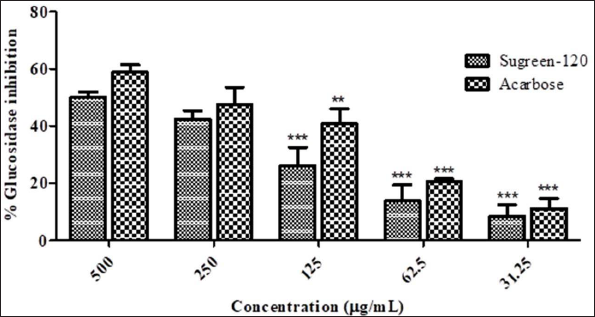
α-Glucosidase inhibitors (AGIs) are drugs that prevent or delay the absorption and digestion of polysaccharides from the gut, which results in the reduction of hyperglycemia or the management of type 2 diabetes. It has been reported that such enzyme inhibitors exhibit significant effects on glycated hemoglobin. The treatment effect of natural α-glucosidase inhibitors is strongly hyphenated with the management of diabetes with no liable side effects as typically observed with modern medicines.21, 22
TLC Bioautographic-MS Methods for Identification of Antidiabetic and Antioxidant Compounds in Sugreen-120
TLC-bioautographic-MS methods such as DPPH free radicals, α-amylase, and α-glucosidase inhibitory assay were evaluated to investigate key bioactive constituents responsible for antidiabetic and antioxidant activity. The observations were made based on the color obtained post-treatment of the developed TLC plate. The TLC bioautographic assay for the determination of antioxidant compounds was done successively through the DPPH free radical scavenging method. The observations were made based on the yellow color spots visible against the dark purple color background, which represents those compounds as antioxidants. The observation revealed a total of 6 antioxidant compounds as myricetin (m/z: 318.85), elagic acid (302.98), caffeic acid (m/z: 181.54), kaempferol (m/z: 287.14), trans-cinnemic acid (m/z: 148.62), and one unidentified compound (m/z: 271.02) at 0.13, 0.28, 0.31, 0.48, 0.57, and 0.68 R f , respectively. TLC bioautographic assay for determination of α-amylase inhibitory compounds was done successively through enzyme and substrate reaction method. The observations were made based on dark violet color spots visible against the brown color background, which represents the compounds are active against the α-amylase enzyme. The observation revealed a total of four compounds such as elagic acid (m/z: 302.98), caffeic acid (m/z: 181.54), kaempferol (m/z: 287.14), and cinnemic acid (m/z: 148.62) at 0.28, 0.37, 0.48, and 0.57 R f , respectively, were active against the α-amylase enzyme activity. Furthermore, TLC bioautographic assay for the determination of α-glucosidase inhibitory compounds was done successively. The observations were made based on a light creamish or white color against a purple color background. The observation reveals a total of 3 compounds such as caffeic acid (m/z: 181.54), quercetin (m/z: 302.42), and kaempferol (m/z: 287.14) at 0.31, 0.48, and 0.68 R f , respectively, were active against the α-glucosidase enzymatic activity. TLC bioautogram of DPPH free radicals, α-amylase, and α-glucosidase activity is summarized in Figure 4 and Table 2.
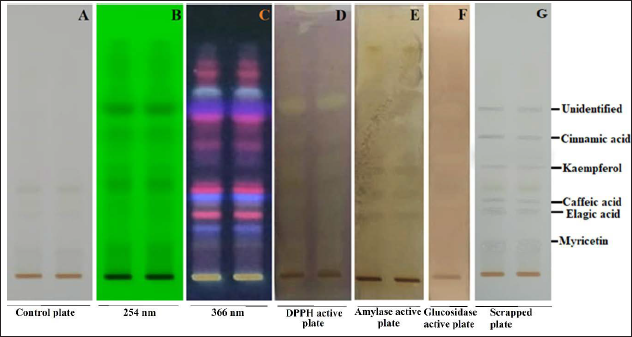
DPPH, α-Amylase, and α-Glucosidase Active Phytoconstituents Present in Sugreen-120 at Different R f .
The TLC-bioautographic assay is one of the emerging techniques used for screening target bioactive constituents based on their bioactivity on a silica phase. There are many acting techniques such as DPPH, α-amylase, and α-glucosidase used to identify the targeted constituents responsible for antioxidant and antidiabetic activity. A recent study conducted by Gaurav et al. evaluated several antioxidant, α-amylase, and α-glucosidase inhibitor compounds such as gallic acid, quercetin, chlorogenic acid, palmatine, vanillic acid, and ellagic acid in a polyherbal formulation BGR-34 containing herbal ingredients such as G. sylvestre, B. aristata, and Trigonella foenum-graecum. 3 Since numerous studies have proven that hyperglycemia-induced oxidative stress causes slow progression in the treatment of diabetes. 23 Sugreen-120 exhibits high potential as antioxidants and antidiabetic, which can be used potentially not only as antidiabetic but also against hyperglycemia-induced oxidative stress. The experimental outcome revealed several phytoconstituents, which may possess not only antioxidants but also antidiabetic activity.
Quantitative Analysis of Marker Compounds in Sugreen-120 by using HPTLC
HPTLC profiling and quantitative estimation for the simultaneous separation of caffeic acid and kaempferol in Sugreen-120 were performed successively. The resulted data reveals numerous numbers of major and minor metabolites in Sugreen-120 (Figure 5 and Table 3), while validation analysis for simultaneous separation of caffeic acid and kaempferol was found linear, accurate, and robust in the wide range of 200–4,000 ng/spot. The regression equation and coefficient of the validated method for simultaneous separation of caffeic acid and kaempferol were found to be y = 1.9074x + 12.553, 0.9986, and y = 2.7535x + 169.14, 0.9965, respectively. The limit of detection (LOD) and the limit of quantitation (LOQ) were found to be 24.223, 73.405 ng/spot for caffeic acid and 21.204, 57.598 ng/spot for kaempferol. The intraday and interday precision were determined as percentage relative standard deviation (%RSD) or the coefficient of variation, and the results were expressed in the range 0.239–1.995 (0.352–2.199 for caffeic acid and 0.418–2.276, 0.083–2.380 for kaempferol). The accuracy of the developed method was determined as the percentage of drug recovered by percentage spiking 0%, 50%, 100%, and 150% of the standard to the sample, which exhibited recovery in the range of 97.978–99.952% for caffeic acid 99.956–106.125% for kaempferol. After all, the content of caffeic acid and kaempferol in Sugreen-120 was determined, which were found to be 5.233 ± 0.026 and 16.959 ± 0.036 µg/mg, respectively. HPTLC plate view at 254 nm and chromatograms of Sugreen-120 have been displayed in Figure 5, and the metabolites found at different R f have been mentioned in Table 3.
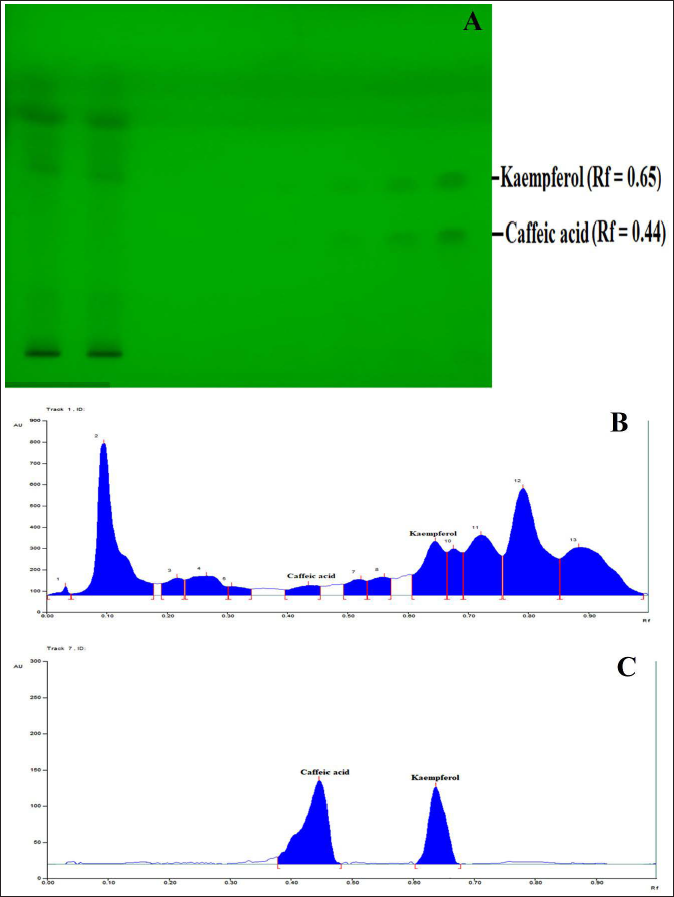
HPTLC Fingerprinting Profile of Sugreen-120 at 254 nm and 366 nm
Previous studies reveal that caffeic acid and kaempferol are the major phytoconstituents of G. sylvestra N. sativa, T. foenum-groecum, and Tinospora cordifolia, which act as the potent inhibitor of α-amylase and α-glucosidase activities that delays the digestion of starch and disaccharides to absorbable monosaccharides, resulting in a reduction of postprandial hyperglycemia.24–28
In Silico Docking Analysis
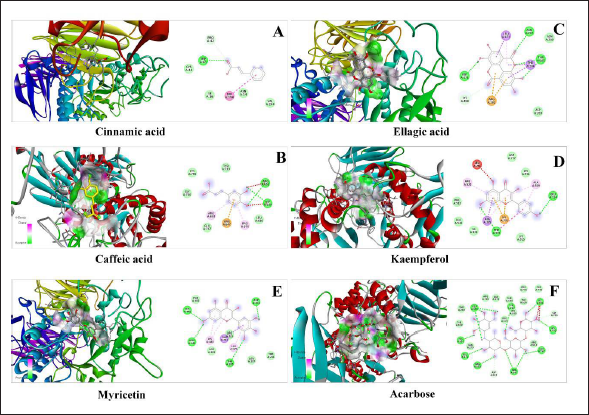
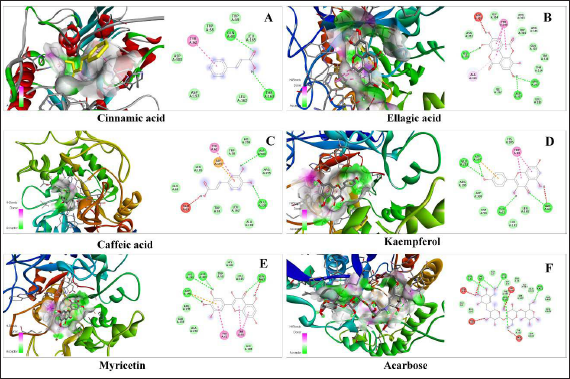
In silico docking analysis for estimation of the antidiabetic effect of the metabolites identified in Sugreen-120 was conducted successfully. The conventional hydrogen bonding and the binding energy of each compound with the protein of α-amylase and α-glucosidase enzyme were considered as the biological interaction activity of each compound. Acarbose was used as the standard drug to validate the results of the identified molecules in proportion to the standard drug. The outcome of the study showed that ellagic acid, kaempferol, and myricetin showed a prominent interaction with both proteins with high binding energy and conventional hydrogen bonding. The binding energy of ellagic acid, kaempferol, and myricetin with α-amylase was found to be −8.6, −8.3, and −8.5, respectively, while the binding energy with glucosidase was found to be −8.8, −8.0, and −8.1, respectively, under the grid box dimension center_x = 9.995, center_y = 30.374, and center_z = 51.813 and size_x = 72, size_y = 72, and size_z = 72 for α-amylase protein and center_x = −28.316, center_y = 6.055, and center_z = −18.368 and size_x = 90, size_y = 90, and size_z = 90 for α-glucosidase. The outcome of the study showed that among the 5 molecules, 3 molecules were found prominent even comparable with acarbose which was used as the standard drug for validation of the present findings. The outcome of the study has been summarized in Table 4, Figures 6 and 7.
In silico Docking Analysis of Sugreen-120 Compounds with Amylase Glucosidase Protein.
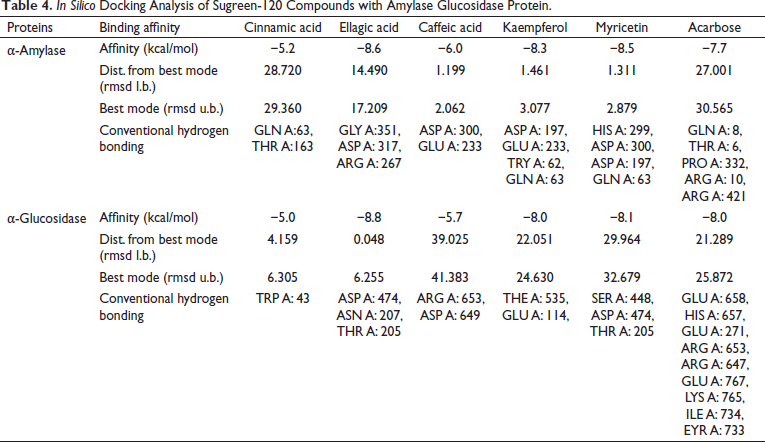
In silico Docking Analysis of Sugreen-120 Compounds with Amylase Protein.
In silico Docking Analysis of Sugreen-120 Compounds with Glucosidase Protein.
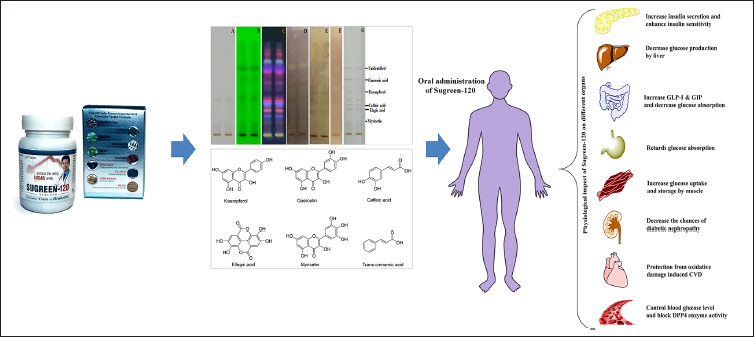
Furthermore, the probable mechanistic approach of Sugreen-120 has been represented (Figure 8) based on the several antioxidant and antidiabetic compounds identified in the formulation that are reported to have antidiabetic effects as reported in previous reports.3, 20, 27 Further Sugreen-120 proceeded for the identification of free radical scavenging compounds, α-amylase, and β-glucosidase inhibitors through the TLC-bioautographic method coupled with mass spectrometry. Several phytoconstituents were identified in the methanolic extract of Sugreen-120 having antioxidants, α-amylase, and β-glucosidase inhibitory activity. The quantitative validation of Sugreen-120 was done using caffeic acid and kaempferol as major markers of Sugreen-120. The outcomes revealed that Sugreen-120, its ingredients, and their phytochemical constituents were found to be responsible for a physiological actions in the body, including decreased glucose production by the liver, increased GLP-1 & GIP and decreased glucose absorption in the gut, increased insulin secretion and enhanced insulin sensitivity, delayed gastric glucose absorption, increased glucose uptake and storage by muscle, decreased the feeling of hunger, and decreased the risk of diabetes.3, 27
Conclusion
The present study concludes that Sugreen-120 exerts excellent antidiabetic and antioxidant potential due to the presence of varieties of phytoconstituents that are potentially active as antioxidant and antidiabetic agents with respect to α-amylase and α-glucosidase enzymatic activity. TLC-bioautography-MS analysis showed that myricetin, ellagic acid, caffeic acid, kaempferol, and cinnamic acid were found to be the active principle against DPPH free radicals, α-amylase, and α-glucosidase enzymes. In silico docking analysis showed that out of five identified metabolites, myricetin, ellagic acid, and kaempferol were found with significant interaction with α-amylase and α-glucosidase proteins. Hence, it can be concluded that Sugreen-120 has the potential to be both an antioxidant and an antidiabetic. It can be a palliative choice and an alternative that can be used for the treatment of diabetes. Moreover, further experimental investigations based on molecular biology are necessary to validate and claim the antidiabetic phytopharmacological role of Sugreen-120.
Summary
Medicinal plants and their derived polyherbal formulations have been playing an immense role in alleviating several acute and chronic ailments due to the multi-targeted therapeutic approach exhibited by the varieties of constituents present in them. Sugreen-120 is a polyherbal formulation developed for the mitigation of diabetes and its associated problems. Due to a lack of phytopharmacological evidence, several major phytochemicals were identified based on their biological contribution as an antidiabetic and antioxidant. Antidiabetic and antioxidant activity of Sugreen-120 was determined, followed by an assessment of TLC-bioautographic analysis and qualitative and quantitative validation of its phytochemicals. In silico analysis was performed to investigate the molecular-based interaction of identified metabolites in diabetes. The study showed that Sugreen-120 possesses several varieties of antioxidant and antidiabetic compounds which are potentially active against α-amylase and glucosidase enzymes. Hence, the generated scientific evidence contributes to the quality, safety, and regulatory purposes of Sugreen-120. The developed formulation might be the better alternative that can occupy the opacity of potent herbal drugs in the healthcare system for the treatment of diabetes and even promote the sustainable development of the nation.
Footnotes
Acknowledgment
The authors would like to thank the Ministry of AYUSH for providing the facilities at the Centre of Excellence in Unani Medicine (Pharmacognosy and Pharmacology), Bioactive Natural Product Laboratory, School of Pharmaceutical Education and Research, Jamia Hamdard, New Delhi, India, and Drugs Laboratories, Khasra No. 4, Hajipur, Hapur Road, Meerut-250004, India, for providing gift sample of Sugreen-120.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The authors received no financial support for the research, authorship and/or publication of this article.
Statement of Informed Consent and Ethical Approval
Necessary ethical clearances and informed consent was received and obtained respectively before initiating the study from all participants.
