Abstract
Objectives
Previous study have shown that the seeds of the tamarind tree (Tamarindus indica) have anti-diabetic properties without any evidence on the processing method. This present study was carried out to investigate the effect of fermentation on antioxidant as well as anti-diabetic properties of Tamarind (Tamarindus indica) seeds.
Methods
The sample was pulverized after five days of typical fermentation alongside raw sample and examined for its antioxidant and anti-diabetic properties. Antioxidant assays such as FRAP, ABTS, NO, and DPPH were carried out to determine the in-vitro inhibitory effects on diabetes-related enzymes α-amylase and α-glucosidase, while amino acids, proximate and minerals content were carried out.
Results
The findings revealed that fermentation changed the antioxidant properties of the seed powder. While total phenols and flavonoids decreased, inhibition was observed with α-amylase and α-glucosidase, although raw has higher results. Interestingly, the analysis of 18 amino acids on raw and fermented samples indicated an increase value with fermentation. Moreover, there were elevations in Zn, Mn, K, and vitamins C, D, E, and B1.
Conclusion
In summary, the study indicates that tamarind fermented seed powder holds promise for exerting anti-diabetic effects, likely due to its rich content of amino acids and vitamins endowed with antioxidant properties.
This is a visual representation of the abstract.
Introduction
Diabetes mellitus (DM) has been found to be a major cause of β-cell destruction in the liver and kidneys and a major contributor to chronic inflammation and apoptosis in the pancreatic islets.1,2 Due to a constant breakdown or wearing away of organ tissues, the development of DM is defined by long-term damage that is coupled with enhanced actions that are oxidative or inflammatory, leading to increasingly severe diabetic consequences.3,4 DM has been identified as one of the largest and most serious health disorders, posing a significant public health threat, according to many reports. 5 The illness results in a chronic metabolic condition that has a substantial influence on people's health, quality of life, and life expectancy, as well as on the public health care system as a whole. According to recent demographic data, an estimated 451 million adults worldwide were diagnosed with diabetes in 2017, with a projected increase to 693 million by 2045 if current trends continue and appropriate management and prevention techniques are not developed. 6
Patients with diabetes who have had the disease for a long time develop a number of comorbidities. It usually starts with macrovascular problems, then progresses to cardiovascular disease and stroke, increased cardiovascular morbidity, and, eventually, mortality. Furthermore, nephropathy, retinopathy, and neuropathy are all possible outcomes of these macrovascular problems. 7
Treatment of hyperglycemia in diabetic patients necessitates a thorough understanding of the numerous critical factors involved in the disease's genesis. Knowing the mechanism by which each available pharmacologic treatment decreases hyperglycemia is critical.8–10 Before administering any anti-hyperglycemic drug, it is important to be aware of the simultaneous effect of each anti-hyperglycemic treatment on cardiovascular risk factors.11,12
Biguanides, inhibitors of DPP-IV, inhibitors of sodium-glucose co-transporter 2 (SGLT2), insulin, glucagon-like peptide 1 (GLP-1) agonists, sulfonylureas (SU), and thiazolidinediones (TZD) are examples of diabetes medications.13,14 Although the efficacy of these treatments has been demonstrated, many research findings have also documented their negative side effects, which are a major source of concern.
Antioxidant compounds can prevent free radicals from attacking cells by stabilizing, scavenging, or deactivating them, metal catalyst chelation, antioxidant enzyme activation, β-tocopherol radical reduction, and oxidase inhibition. 15 Plants with substantial anti-oxidant and anti-diabetic characteristics are gaining popularity as diabetes trends advance. This is due to their greater therapeutic effectiveness and lower biological side effects. Polyphenols are one of several phytochemicals that have been thoroughly researched owing to their considerable antioxidant action. 16
The research conducted by Issahaku et al (2023) 17 and Baru Venkata et al (2023) 18 demonstrates a noticeable trend towards investigating bioactive chemicals such as vitamins and amino acids, which are known to play crucial roles in human health. According to Edwin et al (2008), the medicinal plants’ mechanisms of action include: cyclic adenosine monophosphate (cAMP; secondary messenger) stimulation, pancreatic β-cell K+ ion channel blocking; prevention of oxidative stress; and provision of elements such as magnesium, manganese, calcium, zinc, and copper are employed in β-cells to block β-galactosidase and α-glucosidase.7,19
Recent studies have revealed that tamarind is a medicinal plant, with its fruits, which are its most useful portion, being included in numerous pharmacopeia as a therapeutic agent. It has been used for wound healing and anti-inflammation for centuries, 20 diarrhoea with stomach ache, 21 parasitic infestation, dysentery, 22 fever, malaria, and respiratory issues are all possible. Analysis of T. indica by phytochemistry revealed the presence of phenolic compounds such catechin, procyanidin B2 found in the leaves, bark and fruits, and epicatechin (bark and seed), while tartaric acid, mucilage, pectin, arabinose, xylose, galactose, and glucose were found in the fruits. Uronic acid is a component of cell walls, and triterpenes are found majorly in the bark and seeds of the Tamarind plant. 22
Diabetes, fevers, and intestinal infections have all been treated using tamarind seeds in the past. Despite its historical use in traditional medicine for the treatment of various ailments, scientific knowledge regarding the amino acid composition, phenolic composition, and mode of action of the seeds, particularly in addressing diabetic conditions, remains scarce.
To unlock the nutritional and therapeutic potential of tamarind seeds, we employed a common practice of fermentation carried out by the locals, which makes the seeds suitable for incorporation into meals or as a condiment in their diets, offering a unique perspective on the potential antidiabetic properties of tamarind seeds in its raw and fermented form.
The aim of this study is to bridge the existing gap in scientific understanding shedding light on the composition and mechanisms underlying the antioxidant and anti-diabetic properties of fermented tamarind seed (Tamarindus indica) in vitro.
Materials and Methods
Materials
Sample Gathering and Preparation
Fresh tamarind (Tamarindus indica L.) fruit pods were bought at Abuja's Kwuba Market. The pods were identified and authenticated by the FUTA Centre for Research and Development (CERAD). The tamarind fruit pods were shattered by hand, and the pulp, seeds, and fibres were taken from each pod. The tamarind seeds’ pulp and fibre strands were then steeped in clean water overnight at a weight/volume ratio of 1:3. The cleaned seeds were air-dried in the shade for two weeks before being split in half. One-half of the mixture was crushed into fine flour using a Wonder mill and utilized as a raw sample. The other half of the seeds was processed using the fermentation method developed by Ademiluyi et al (2015), with minor modifications. The seeds were carefully washed before being steeped in clean water at a weight/volume ratio of 1:3 in an airtight container. They were subsequently subjected to three days of wild fermentation at 37 °C, as indicated by. 23 The seeds were taken from the container after fermentation and dried in a 45 °C oven before being milled into fine flour, which was also used as a treatment sample. To keep the flour fresh, it was passed through a 300 µm screen before being stored in an airtight container in a freezer at −4 °C. 24
Chemicals and Reagents
Unless otherwise noted, high-quality chemicals and reagents were utilised throughout the experiment, and the water was filtered using the glass distillation method.
Instruments
Spectrophotometer, cuvettes, conical flask, measuring cylinder, standard flask, micro pipette, amber bottles, centrifuge tubes, and centrifuge.
Methods
Preparation of Aqueous Extracts
For the preparation of the aqueous extracts, 1 g of powdered seed and 100 ml distilled water were vigorously agitated in an extraction container for 4 h. Before centrifuging, the extract was filtered through Muslin and Whatman No. 1 filter paper to get a clear supernatant. This process removed any contaminants or debris from the extract, ensuring a clean solution for analysis. 24
Before bioactivity assays, the extract was kept at −4 °C to ensure stability. This was done to prevent deterioration or denaturation of the extract's active components, which might compromise bioactivity testing’ accuracy and reliability.
Vitamin Analysis, Antinutrient and Phytochemical Analysis, Mineral Analysis, and Proximate Analysis
The tamarind sample was subjected to chemical analysis to determine its nutritional and medicinal properties. This analysis included testing for the presence of vitamins B1, C, D, and E, anti-nutrients, phytochemicals, as well as quantifying the amino acid and phenolic profiles using high-performance liquid chromatography (HPLC). 24
To determine the concentration of vitamin B1 in the tamarind sample, the method described by Okwu and Josiah (2006) 25 was employed. The vitamin C content was determined using the method developed by Benderitter et al (1998), 26 which involved the use of a dinitrophenylhydrazine (DNPH) solution. The vitamin D content was measured using Carr-Price reagent that was free from alcohol, and the concentration was extrapolated from a standard curve using a Vitamin D standard. The vitamin E content was determined using the method developed by 27 Pearson (1976), which involved using a diethyl ether solution.
To determine the levels of oxalate, phytate, saponin, tannin, terpenoids, and glycosides in the tamarind sample, established methods were used. Oxalate levels were determined using the method developed by Day and Underwood (1999), 28 where the solution was titrated against 0.1 M potassium permanganate (KMnO4). The phytate levels were determined using the method developed by 29 Wheeler and Ferrel R (1971). Saponin levels were determined using the spectrophotometric method described by 30 Adetutu (2015) using a 5% iron (iii) chloride (FeCl3) solution. 30 Tannin levels were determined using the method recently described by. 30 The absorbance was read at 725 nm against a reagent blank concentration of the same solution made from a conventional tannic acid curve. Terpenoids and glycosides levels were determined using the methods described by 31 Sofowara, (1993) and 32 Sofowora, (1996), respectively, with alpha-terpineol and Digitoxin serving as standards.
Furthermore, the proximate compositions of the seed powder, including moisture, ash, fat, crude fiber, and crude protein, were determined using the recommended methods of the Association of Official Analytical Chemists. 33
To determine the concentration of certain minerals in the sample, 1 g of the sample was taken and digested in a mixture of 69% HNO3% and 30% H2O2 (v/v: 10 ml) for a short time, and then heated to 120 °C. The resulting digested solutions were then filtered using Whatman No.1 filter paper and diluted with deionized water to a final volume of 50 ml. The concentration of minerals such as K, Mg, Fe, Zn, and Na in the digested solutions was determined using Atomic Absorption Spectrophotometer according to the method described by 34 Kojuncu et al (2004).
HPLC-DAD Analysis
The modified method was used to analyse the samples for phenolics and amino acids via HPLC as mentioned in our previous publication
35
Nwanna et al (2015). In an amber container, 10 g of the sample was measured, then 20 ml of acetonitrile/methanol was added and violently shaken for 30 min. After shaking, the organic layer was transferred to a 25 ml standard flask and prepared for analysis. The analysis was carried out using a Shimadzu Autosampler (Nexera MX) type HPLC with an Ubondapak C18 column and mobile phases of water/methanol and acetonitrile. In a Diode Array Detector, a flow rate of 1 ml/min and an injection volume of 10 µl was employed with a wavelength of 254 nm. The sample and mobile phase were degassed in an ultrasonic bath after passing through a 0.45 m Millipore membrane filter and a pump pressure of 15 mPa. In preparation for the test sample analysis, the HPLC was given a standard analyte profile to generate a window. A chromatogram with a typical peak area and peak profile was obtained by injecting aliquots of the test liquid into the HPLC. Using the supplied formula, the sample analyte concentration was estimated by comparing the peak area of the sample to that of the standard in proportion to the standard concentration.
29
The results were reported as the concentration of the sample. The results were calculated as;
Determination of Total Phenol
The total phenol content of the samples was calculated using the technique of Singleton et al (1999). 36 The reaction mixture consisted of 2 ml of 7.5% Na2CO3, 2.5 ml of 10% Folin-reagent Ciocalteau's, and an aliquot of the extract. After an incubation period of 40 min at 45 °C, a spectrophotometer was used to measure the absorbance at 765 nm. The total phenol concentration was then estimated in mg/g of gallic acid equivalent.
Total Flavonoid Determination
Using the approach described by 37 Meda et al (2005), the total flavonoid content of the samples was determined. The reaction mixture was prepared by adding 0.5 ml of the extract, 0.5 ml of methanol, 50 µl of 10% aluminium chloride, 50 µl of 1 M potassium acetate, and 1.4 ml of distilled water. After which the mixture was incubated for 30 min at room temperature. At 415 nm, the absorbance of the mixture was measured. The total flavonoid concentration was then calculated and expressed in mg/g of quercetin equivalent.
Ferric Reducing Antioxidant Property (FRAP) Determination
The reducing property of the extracts was determined by evaluating their ability to reduce FeCl3 solution as indicated by Oyaizu, (1986). 38 0.25 ml of 200 mM sodium phosphate buffer (pH 6.6) and 0.25 ml of 1% potassium ferricyanide were added to a 0.20 ml of aliquot of the sample. After 20 min of incubation at 50 °C, 0.25 ml of 10% trichloroacetic acid, 1 ml of distilled water and 0.20 ml of 0.1% ferric chloride were added. The absorbance was read at 700 nm and the ascorbic acid equivalent (AAE) was used to calculate the ferric-reducing power.
Radical Scavenging Capability of 2-Azinobis (3-Ethylbenzothiazoline-6-Sulfonate) (ABTS)
The ABTS scavenging capacity of the extracts was evaluated using the technique described by Re et al (1999). 39 The ABTS aqueous solution (7 mmol/l) and K2S2O8 (2.45 mmol/l, final concentration) was subjected to a dark reaction for 16 h. Following this, ethanol was added to adjust the absorbance to 0.700 at 734 nm. Then, 0.1 ml of an appropriate dilution of each extract was added to 2.0 ml of ABTS solution The absorbance was then measured at 734 nm, 15 min later after being placed in the dark, with trolox being used as a reference. Following that, the trolox equivalent antioxidant capacity (TEAC) was computed.
Radical Scavenging Capability of 1, 1 Diphenyl-2-Picryl-Hydrazyl (DPPH)
The extracts’ radical scavenging capacity against DPPH radicals was assessed as specified by Gyamfi et al (1999). 40 1 ml of extracts was combined with 1 ml of DPPH-containing 0.4 mM methanol solution. The absorbance of the remaining non-scavenged DPPH was measured at 516 nm after 30 min of dark storage. The percentage DPPH scavenging capacity of the extracts was then calculated as a percentage of the control.
Determination of Nitric Oxide Scavenging Assay
The Griess reaction was used to determine the aqueous-ethanolic samples’ nitric oxide radical scavenging capability. 41 0.3 ml of the aqueous-ethanolic mixture was combined with 2.7 ml of 10 mM sodium nitroprusside in phosphate-buffered saline (PBS) and left to incubate at 25 °C for 150 min. Afterwards, 0.5 ml of the incubated mixture was mixed with 0.5 ml of Griess reagent (consisting of 1% (w/v) sulfanilamide, 2% orthophosphoric acid, and 0.1% (w/v) naphthyl ethylene diamine hydrochloride that was made in an amber container and kept away from light). The absorbance was then measured at 546 nm. By comparing the absorbance values of the control and samples, the percentage of inhibition of nitric oxide formation was calculated.
Lipid Peroxidation and Enzyme Inhibition Assays (In vitro)
Fruit fly Tissue Homogenate Preparation
Before being homogenised in 0.1 M sodium phosphate buffer, pH 7.4, the flies were anaesthetized in ice. The homogenates were centrifuged for 10 min at 10,000 X g, 4 °C in a Kenxin refrigerated centrifuge Model KX3400C. (KENXIN Intl. Co., Hong Kong). The supernatant from the pellet was then collected and placed in Eppendorf tubes for enzyme inhibition tests.
Assay for Lipid Peroxidation
The lipid peroxidation experiment was carried out using a method invented by Ohkawa et al (1979). 42 100 µl of fly tissue homogenate, 30 µl of 0.1 M Tris HCl buffer (pH 7.4), extract (0-75 µl), and pro-oxidant solutions were mixed (250 M FeSO4 or 7 mM SNP). The volume was raised to 300 µl with distilled water before incubation at 37 °C for an hour. 300 µl of 8.1% sodium dodecyl sulphate (SDS) was added to the reaction mixture to commence the colour reaction. The next step involved adding 500 µl of acetic acid/HCl (pH 3.4) and 500 µl of 0.8% thiobarbituric acid (TBA) to the solution. The mixture was then incubated at 100 °C for 60 min. The amount of TBARS produced was measured at 532 nm.
α-Amylase Inhibitory Activity
The inhibitory activity of α-amylase was measured using the technique of Worthington, Von. (1993). 43 A 500 µl diluted aqueous sample was mixed with 500 µl of 0.02 M sodium phosphate buffer (pH 6.9 with 0.006 M NaCl) containing 0.5 mg/ml pig pancreatic α-amylase (EC 3.2.1.1) for 10 min at 25 °C. Following that, 500 µl of 1% starch solution in 0.02 M sodium phosphate buffer was added to each reaction mixture (pH 6.9 with 0.006 M NaCl). After 10 min of incubation at 25 °C, the reaction mixtures were stopped using 1 ml of DNSA colour reagent. After incubating for 5 min in a hot water bath, the mixture was cooled to room temperature. The absorbance at 540 nm was measured after adding 10 ml of distilled water to the reaction mixture. All the reagents and the enzyme except for the test sample were added to the reference samples, and the percentage of enzyme inhibitory activity of the extract was then calculated.
Inhibitory Activity of α–Glucosidase
The approach to measuring α-glucosidase inhibitory activity used was by Apostolidis et al (2007). 44 The procedure involved incubating the sample (50 µl) and 100 µl of -glucosidase solution (EC 3.2.1.20; 1.0 U/ml) in 0.1 M phosphate buffer at 25 °C for 10 min (pH 6.9), after suitable dilution. The solution was then mixed with 50 µl of a 5 mM pnitrophenyl-D-glucopyranoside solution in 0.1 M phosphate buffer (pH 6.9). After 5 min of incubation at 25 °C, the absorbance at 405 nm was measured with a spectrophotometer. The inhibitory activity of α-glucosidase was represented as a percentage inhibition.
Statistical Analyses
The results of triplicate experiments were collated and expressed as mean ± standard deviation (S.D). The results were analysed using One-way Analysis of Variance (ANOVA) and the Tukey post hoc test, with p < 0.05 levels of significance accepted. GraphPad PRISM (V.8.0) was used for all data analysis. 24
Results
Proximate and Mineral Composition
Table 1 shows proximate and mineral analyses of fermented and raw tamarind seeds. The results showed that the fermented sample had (p < 0.05) lower percentage of crude fibre (7.59 ± 0.36), higher crude protein (43.48 ± 0) and less carbohydrate content (20.3 ± 0.0) due to the fermentation process. The reduction in crude fiber content in the fermented seeds may be attributed to the breakdown of complex carbohydrates during fermentation, resulting in the release of soluble and insoluble fiber. A reduction in percentage moisture (9.83 ± 0.04) and ash content (2.71 ± 0.24) in the fermented sample was also observed because of the fermentation process. The sample lost Fe, Na, and K following fermentation, according to Table 2. The significant (p < 0.05) reduction in Fe (1.948 ppm), Na (424 ppm), and K (505 ppm) content observed after fermentation of tamarind seeds can be attributed to the leaching of these minerals into the brine or their uptake by microorganisms during the fermentation process. However, Mn (1.264 ppm) and Zn (4.242 ppm) concentrations increased with fermentation when compared to the raw sample that was not fermented.
Showing the Proximate Analysis Results of the Fermented and Raw Tamarind Seed Flour.

The values indicate the mean ± SD of triplicate readings. Mean values in the same row with different superscript letters (a,b) differ significantly (p < 0.05).
Showing the Mineral Content Results of the Fermented and raw Tamarind Seed Flour.

The values indicate the mean ± SD of triplicate readings. Mean values in the same row with different superscript letters (a,b) differ significantly (p < 0.05).
Vitamins, Antinutrients and Phytochemical Constituents
Table 3 illustrates how fermentation alters the chemical makeup of tamarind seed by outlining the chemical make-up of the vitamins in the seed. This proved that following fermentation, the quantities of vitamins C, D, and E greatly rose, as observed by Nwanna et al (2022). From the result, vitamin C (32.06 ± 2.35 mg AAE/g of dry sample), vitamin D (0.65 ± 0.01 mg unit/g of dry sample), and vitamin E (1.65 ± 0.0001 mg unit/g of dry sample) of the fermented were significantly (p < 0.05) higher than the raw sample. Table 4 provides a chemical breakdown of the antinutrients and phytochemicals in the seed. It revealed that fermentation significantly (p < 0.05) lowered the content of saponin (25.64 ± 2.06 mg/g), tannin (5.21 ± 0.07 mg/g), terpenoids (3.07 ± 0.06 mg/g), and glycosides (25.98 ± 0.18 mg/g), while significantly (p < 0.05) increasing oxalate content (1.53 ± 0.13 mg/g) and stabilizing the phytate content (11.95 ± 0.58 mg/g) of sample. 29
Effects of Fermentation on Some Vitamins in the Composition of T. indica Seeds.

Note: Values represent the mean ± SD of triplicate experiments. Abbreviations: AAE, ascorbic acid equivalent. Values are significantly different at * p < 0.05.
Effects of Fermentation on the Anti-Nutrient and Phytochemical Composition of T. indica Seeds.

Note: Values represent the mean ± SD of triplicate experiments. Values are significantly different at * p < 0.05
Total Phenolic, Flavonoid and HPLC Characterization of Constituents in the Tamarind Seeds
Table 5 shows the total phenolics as well as the total flavonoid content of the fermented forms of the seed. While the overall flavonoid content did not change much (Raw = 13.47 ± 3.01 mg QUE/g, Fer = 9.36 ± 1.70 mg QUE/g), there was a significant (p < 0.05) difference in total phenol content (Raw = 53.64 ± 8.37 mg GAE/g, Fer = 25.68 ± 2.75 mg GAE/g).
Raw and Fermented Total Phenol and Flavonoid Content of T. indica.

The values indicate the mean ± SD of triplicate readings. The mean values in the same column with different superscript letters (a,b) differ significantly (p < 0.05).
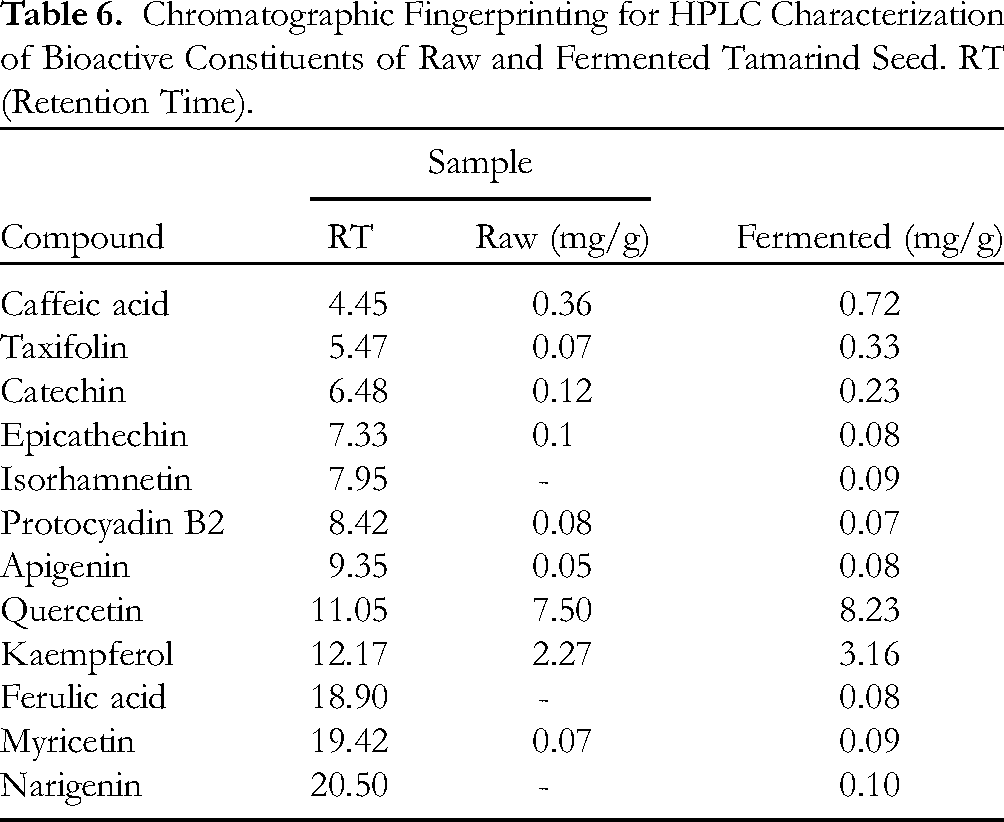
Furthermore, the samples’ qualitative HPLC evaluation for phenolic constituents (Tables 6) revealed the presence of chloramphenicol, β-Amyrin, caffeic acid, phenolic monomers (catechin, epicathechins, and taxifolins), protocyadin, apigenin, lupeol, lupanone, β-Sitosterol, and myricetin, while the anti-inflammatory monophenolic compound isohamnetin was found in the fermented sample along with ferulic acid and narigenin, as observed in our previous studies by Nwanna et al. (2022). The phenolic content was revealed to be increased on fermentation of the seed with the phenol quercetin showing the highest concentration (8.23 mg/g) in the fermented seed as against the raw seed (7.49 mg/g). A total of 12 phenolics were identified with fermentation showing the release of 3 more phenolics when compared with the raw sample. Table 7 shows the amino acid profile, which reveals the existence of many structural proteins which are beneficial for growth and metabolic functions. Like the result of the HPLC evaluation of the phenolic constituents, 17 essential amino acids were identified in both the raw and fermented samples HPLC amino acid profile. In the amino acid profile, it was discovered that glycine had the highest concentration (3.37 mg/g) while the raw had a lower concentration (2.19 mg/g). This was followed by histidine which also increased concentration from 2.30 mg/g (raw) to 2.95 mg/g (fermented). Phenolic compounds are important components of plants with redox properties responsible for antioxidant activity while flavonoids are secondary metabolites that possess antioxidant activities. 24
Chromatographic Fingerprinting for HPLC Characterization of Bioactive Constituents of Raw and Fermented Tamarind Seed. RT (Retention Time).
Chromatographic Fingerprinting for Amino Acid Profile of Constituents of Raw and Fermented Tamarind Seed .RT (Retention Time).

In vitro Antioxidants
Table 8 displays the antioxidant investigative values in vitro of the processed forms of Tamarind (Tamarindus indica) seed in vitro which gives us a very good insight of how rich the seed is in antioxidants gotten by the values of ABTS and FRAP. The In vitro antioxidant analysis of the processed tamarind seed showed that all the samples exhibited ABTS radical scavenging abilities, as seen in Table 8. According to the findings, the raw tamarind seed (14.56 ± 1.38 µmol TEAC/g) was significantly (p < 0.05) higher than the fermented sample in terms of scavenging abilities (11.03 ± 0.07 µmol TEAC/g). This result also correlated with the ferric-reducing antioxidant properties as presented in Table 4. The raw seed had a significantly (p < 0.05) higher reducing property (2.44 ± 0.53 mgAAE/100 g) than the fermented (0.48 ± 0.31 mgAAE/100 g).
Ferric Reducing Antioxidant Property (FRAP) and 2, 2′-Azino-bis (3-Ethylbenzothiazoline-6-Sulfonic Acid) (ABTS) Radical Scavenging Ability of Aqueous Extracts of T. indica.

The values indicate the mean ± SEM of three readings. The mean values in the same column with different superscript letters (a,b) differ significantly (p < 0.05).
The aqueous extract of Tamarindus indica considerably inhibited Nitric Oxide (NO) buildup in a concentration-dependent manner, and the effect of fermentation was significantly different from that of the raw (Figure 1a). The EC50 values of the NO inhibition for both values were not significantly different. The raw and fermented forms had EC50 values of 1.1 ± 0.01 mg/ml and 1.05 ± 0.01 mg/ml respectively (Table 9).

Nitric oxide (NO*) aqueous extracts of raw and fermented tamarind seed have radical scavenging abilities. Value stands for mean ± SD.
EC50 Values for Raw and Fermented Tamarind Seed Aqueous Extracts’ 2,2-Diphenyl-1-Picrylhydrazyl (DPPH*) and NO Radical Scavenging.

The values indicate the mean ± SD of triplicate readings. Mean values in the same column with different superscript letters (a,b) differ significantly (p < 0.05).
In addition, T. indica powder aqueous extracts (raw and fermented) were examined for antioxidant activity utilizing the DPPH free radical assay technique. Figure 1b depicts the antioxidant activity of the extracts. T. indica extracts’ DPPH free radical scavenging capabilities were concentration-dependent, with EC50 values of 0.702 ± 0.01 mg/ml and 1.243 ± 0.02 mg/ml for both raw and fermented samples, respectively, showing the raw sample having significantly (p < 0.05) inhibitory power than the fermented sample (Table 9).
Figure 2 demonstrated the effect of processed tamarind seed aqueous extracts on lipid peroxidation generated by pro-oxidants in Drosophila melanogaster tissue homogenate. The fly tissue homogenate was treated with 7 mM Sodium nitroprusside (SNP) after pre-incubation. This treatment caused a significant (p < 0.05) increase in TBARS production, indicating lipid peroxidation in the fly tissue homogenate. However, the addition of the processed sample extract considerably (p < 0.05) reduced the high TBARS generated in a concentration-dependent manner (0.5-2 mg/ml). It would be observed that the fermented sample was able to reduce TBARS production more effectively when compared to the raw.
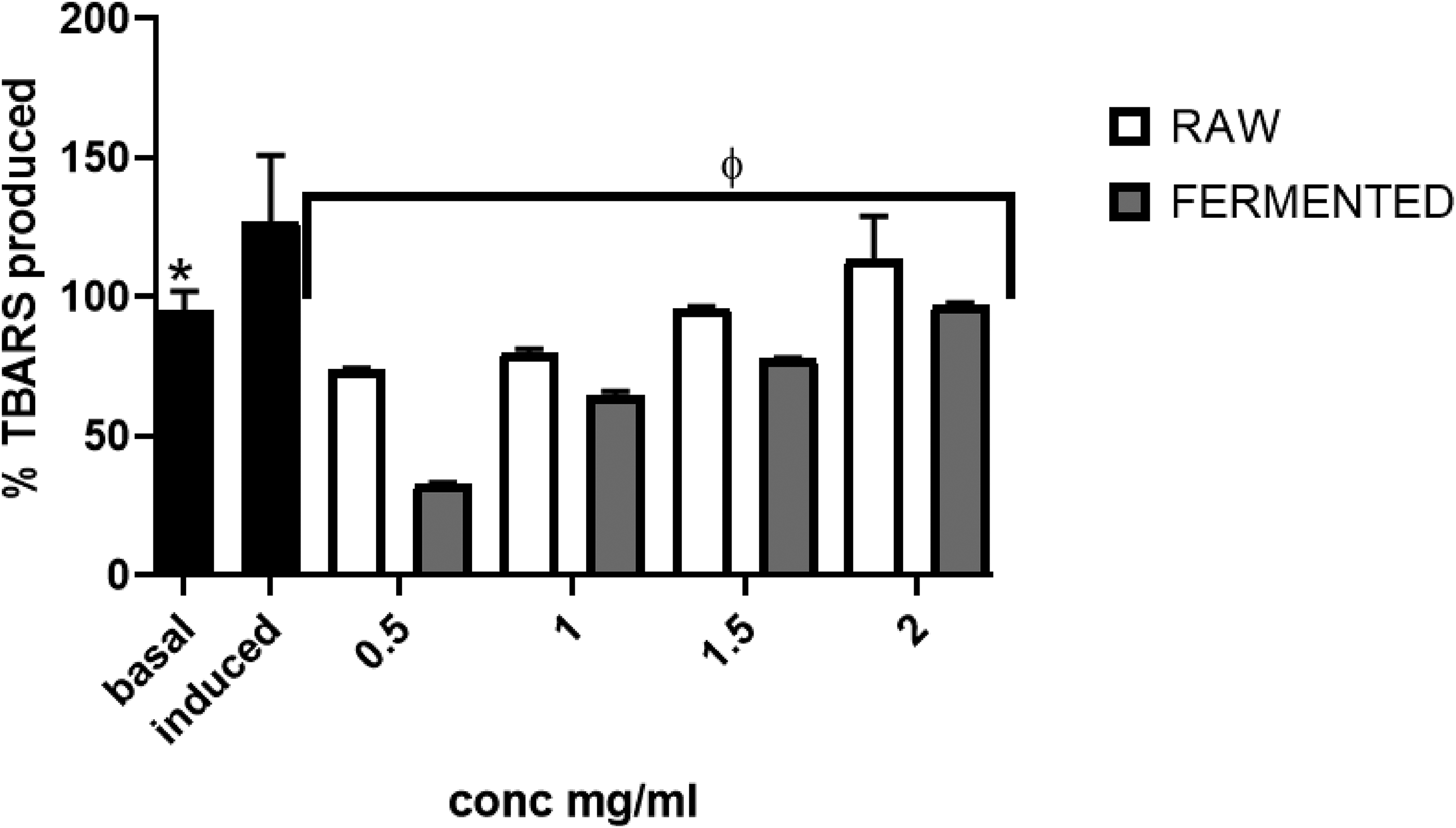
Raw and fermented tamarind seed inhibited SNP-induced thiobarbituric acid reactive substance (TBARS) production in Drosophila melanogaster tissue homogenate. Value stands for mean ± SD. * Mean values differ considerably (p < 0.05) from Induced values. ¶ Mean values differ considerably (p < 0.05) from Basal values.
Enzyme Inhibitory Activity
Furthermore, in this study, the results indicated that T. indica extract showed significant effect on α-amylase but showed a weak significant effect on α-glucosidase at all the tested concentrations (Figures 3 & 4). At the highest concentration (2 mg/ml) investigated, both extracts displayed appreciable effect on α-amylase inhibition by having values above 75%. Likewise, for α-glucosidase, at the highest concentration (10 mg/ml), both extracts displayed ameliorative effects on α-glucosidase inhibition, but their values were not as high as their α-amylase inhibitory activity.
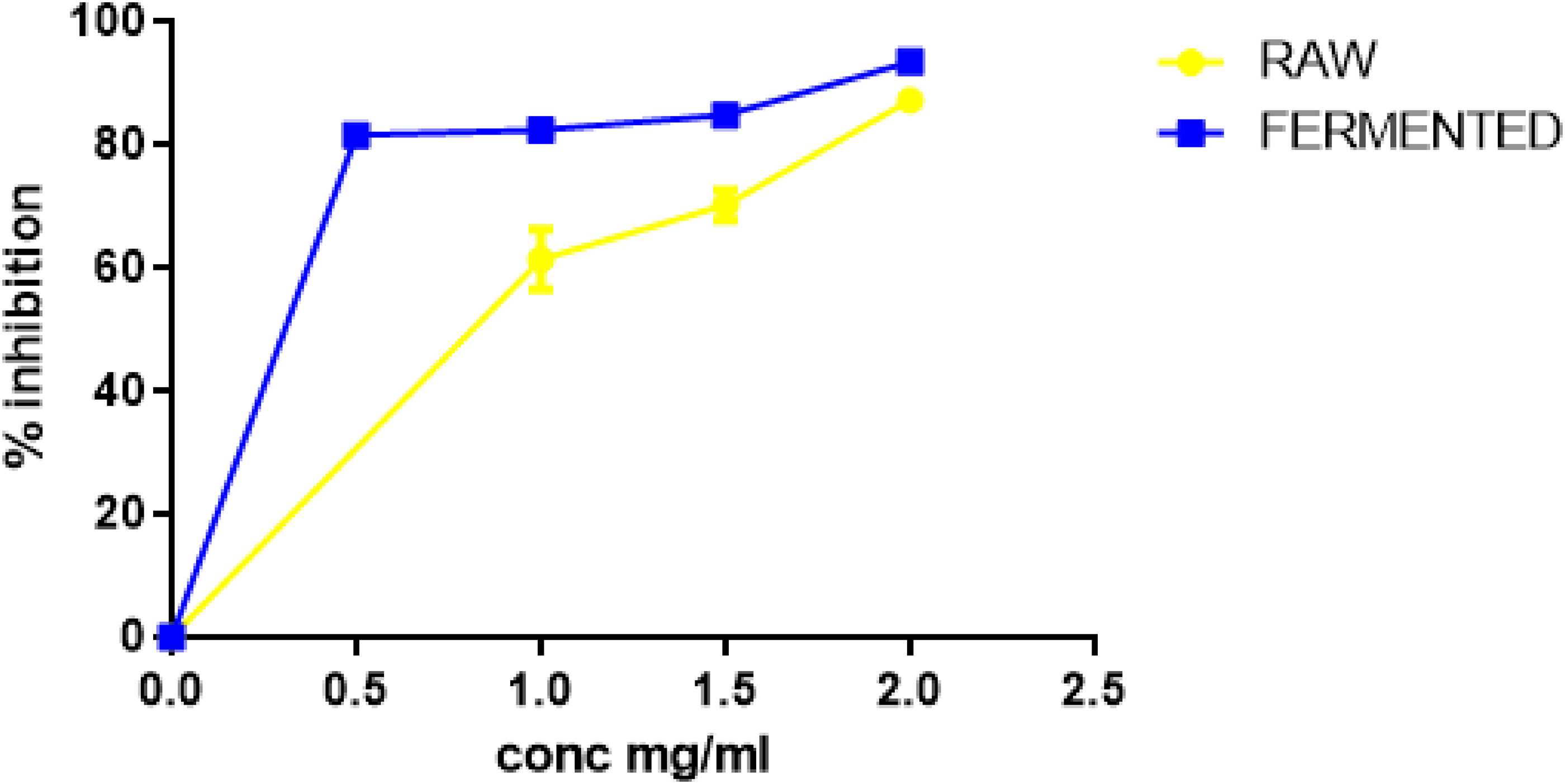
The significant inhibitory effect of aqueous extracts from raw and fermented tamarind seeds on α-amylase activity. Values represent mean ± SD.

The significant inhibitory effect of aqueous extracts from raw and fermented tamarind seeds α-glucosidase activity. Values represent mean ± SD.
Discussion
Bioactive phytochemicals originating from plants and plant products, such as seeds, fruits, and vegetables, have anti-diabetic effects that are at least as powerful as, and frequently much greater than, those of currently available oral hypoglycemic drugs.45,46
Recent studies have revealed that various processing techniques and culinary practices can significantly modify the nutritional and therapeutic properties of functional foods.47,48 Depending on whether the foods are cooked, boiled, fermented, or roasted, the bioactive components of plant foods may either be increased or decreased. Therefore, it is crucial to investigate the health benefits of plant foods and how different preparation methods affect the bioactive compounds of these foods, as people often consume processed versions of these foods.47,48
The proximate composition of fermented and raw tamarind seeds underwent significant changes due to fermentation. The fermented seed has a lower amount of crude fiber and a higher amount of protein. Adams et al. (2018) 49 discovered that dietary fiber facilitates the absorption of trace elements by the body, hence promoting regular bowel movements and perhaps decreasing the occurrence of diverticulosis. Nevertheless, our study indicates that fermentation has the capability to utilize fiber as a source of energy, hence decreasing its overall concentration. Fermentation has been shown to enhance the protein content of seeds through microbial protein production and the degradation of intricate proteins. The decreased crude fiber and higher protein content of the fermented seeds, as revealed by our study, therefore align with previous studies on variations caused by the fermentation of plant-based foods. 50
Fermentation is a process that transforms carbohydrates into organic acids, this process therefore resulted in a decrease in the amount of carbohydrates present in fermented seeds, as seen in our studies, which also aligns with some previous studies. 24 The process of fermentation results in the production of organic acids, which offer advantages such as improved gastrointestinal health and enhanced mineral absorption. 51 Nevertheless, the reduction in mineral content following fermentation may raise concerns, particularly in less developed nations where there is a prevalent deficiency of minerals.52,53 The ash content of fermented seeds is higher due to the mineralization of organic materials that occurs during the fermentation process. This higher ash content in fermented seeds indicates increased mineralization, balancing the reduction in Fe, Na, and K levels. Akajaiku et al. (2014) 54 discovered that the fermentation of tamarind seeds resulted in a higher ash content. The presence of ash in a substance indicates a higher mineral content, and a higher ash content suggests a higher nutritional value. 55 This study affirms that fermentation enhances the nutritional quality of tamarind seeds, particularly in terms of crude fiber and protein.
Vegetable fats and oils possess a hypolipidemic action that can potentially avert conditions such as hypertension, coronary artery disease, myocardial infarction, and cerebrovascular accidents. 56 Eziuche et al. (2016) 57 discovered that diets deficient in these plants can decrease the levels of NADPH and NADH, which are two cofactors essential for fat metabolism. The study revealed that the fermented samples had a lower fat content compared to the raw samples, suggesting that the fermented seeds may have a minimal impact on fat accumulation.
For the body's many metabolic activities, minerals are essential in terms of nutrition and dietary inclusion. 58 According to the research, the fermented sample's levels of Fe, Na, and K were (p < 0.05) lower while those of Mn and Zn were higher. Conversely, the increase in Mn and Zn content in the fermented seeds may be due to the synthesis of these minerals by microorganisms during fermentation. 59 Similar reductions in Fe, Na, and K content after fermentation have been reported in other fermented food products as well. 60 Overall, the fermentation of tamarind seeds appears to enhance the nutritional quality of the seeds, particularly in terms of crude fiber and protein content.
According to the study, the plant samples’ aqueous extracts contained high levels of soluble phytochemicals, which included various polyphenols. These polyphenols are considered excellent antioxidants due to their structure's presence of one or more OH groups, enabling them to participate in antioxidant mechanisms of proton donation and electron transfer.61,62 From our results both samples had a high concentration of phenols and flavonoids. These results suggest that Tamarind seeds have potential as a source of natural antioxidants, which could be useful in developing functional foods or supplements. The in vitro antioxidant results of the current study agree that both raw and fermented extracts of Tamarind seeds contain considerable amounts of phenols and flavonoids, which have been reported to have excellent antioxidant properties due to their ability to donate hydrogen. This is consistent with prior research on Tamarind seeds and leaves, which have also demonstrated their antioxidant properties in several studies.62–64
According to this work and previous studies on T. indica seed, the hydrogen-donating capacity of compounds included in the extracts is assumed to be responsible for their antioxidant character. When DPPH accepts an electron from antioxidant chemicals, the color of the solution changes from violet to yellow, which may be detected spectrophotometrically at 517 nm. 65 Much research has demonstrated that the seed and leaves of T. indica have antioxidant capabilities. 20 Siddhuraju identified antioxidant properties not only in the phenolic features of fresh seeds, but also in heat dried seeds throughout his research. 66 The seeds of T. indica contain a high amount of polyphenols, which have been shown to have a regulating effect on neutrophils. 67 This was proven using various in vitro techniques, including ABTS, 68 this is consistent with the current study's findings. These results suggest that Tamarind seeds have potential as a source of natural antioxidants, which could be useful in developing functional foods or supplements.
Additionally, from our results, the fermentation of tamarind seeds resulted in an increase in vitamin content, terpenoids, glycosides, and a reduction in anti-nutrients, ultimately leading to an increase in overall palatability. The fermented seeds were revealed to be a rich source of various nutrients, including amino acids, terpenoids, and glycosides. Amino acids have been shown to stimulate insulin release from primary islet cells and β-cell lines, which may help regulate blood sugar levels.69,70 Terpenoids and glycosides are known to have various health benefits, including anti-inflammatory, anti-cancer, and anti-diabetic properties. Vitamin C, a strong antioxidant, can reduce the harmful effects of free radicals, 71 while vitamin D has been found to impact the liver's response to insulin and insulin production in islet cells. 72 Studies have reported that amino acids like glutamate, arginine, alanine, and glutamine may stimulate insulin production in primary islet cells and ß-cell lines73,74 Therefore, it could be suggested that the fermentation process helped to increase the antidiabetic potentials of the seeds as evidenced from the various nutritional potentials seen in the results.
The presence of polyphenols in the fermented seeds may contribute to anti-diabetic properties by inhibiting alpha-amylase and alpha-glucosidase. Polyphenols have been shown to inhibit alpha-amylase and alpha-glucosidase, two enzymes involved in the digestion and absorption of carbohydrates. This inhibition can help regulate blood sugar levels and prevent spikes in blood glucose after meals as described by Sun et al (2019). 75 The results show that fermented tamarind seed flour has significant therapeutic potential for the control and prevention of diabetes. Its high nutrient content, including amino acids, vitamins, and polyphenols, can block key diabetes-related enzymes and enhance the antioxidant capacity of pancreatic cells, leading to improved insulin sensitivity and regulation of blood sugar levels. T. indica seed extract has been proven to preserve pancreatic β-cell islets due to its anti-inflammatory characteristics, blood glucose control, and reversal of pancreatic tissue damage. The effects of fermented tamarind seeds on insulin metabolism and secretion from pancreatic cells are thought to be due to the increase in intracellular Ca2 + and plasma insulin activity. Additionally, the seeds may influence the expression of GLUT-2, GLUT-4, and SREBP-1c by increasing mRNA content in liver cells, as demonstrated in a study by Sole, Srinivasan and Akarte (2013). 76 The fermented forms of T. indica seed extract were shown to be able to inhibit both α- amylase and α-glucosidase in this investigation, though the fermented samples of T. indica had a weaker ability to inhibit α-glucosidase. Fermented tamarind seed flour is a great option for the therapeutic control or prevention of diabetes since it has the therapeutic potential to block key diabetes-related enzymes and enhance the antioxidant capacity of pancreatic cells. The in vitro results obtained in this study support the potential use of Tamarind seeds in the management of Type 2 diabetes mellitus.
The fermented Tamarind seeds had high levels of antioxidant capabilities due to the amino acids such as histidine arginine and lysine with potential antioxidant property as well as vitamins C, D, E and B1. This proves that processing of the seed did not reduce its antioxidant potential but increases it even though the phenolic, anti-nutrients reduce during the fermentation. Amino acids may, under the correct conditions, stimulate the release of insulin from primary islet cells and β-cell lines. Recent studies have shown a correlation between the polyphenol content of functional foods and nutraceuticals and their ability to inhibit alpha-amylase and alpha-glucosidase. 76 It is conceivable that the phenolic interactions between the components of the fermented seed give it its anti-diabetic capabilities. Suggestions entail performing animal experiments to validate the discovered antidiabetic characteristics and investigating other processing techniques for possible improvements. Moreover, it is imperative for future studies to investigate the bioavailability of the found chemicals and conduct clinical trials to assess the effectiveness of the seeds in human subjects. This will contribute to a fuller comprehension of their therapeutic potential in managing diabetes.
Conclusion
In summary, our comprehensive investigation revealed that fermented Tamarind seeds are rich in essential nutrients, including vitamins, phytochemicals, and crucial amino acids positioning it as a potentially effective therapeutic candidate for diabetes mellitus and malnourish person. Our findings were investigated through a range of in-vitro techniques, including assessments of total phenolic and total flavonoid levels, as well as alpha-amylase and alpha-glucosidase inhibitory activities. This multifaceted approach provides strong evidence supporting the potential use of Tamarind seeds as functional food resources.
Supporting Information
Footnotes
Acknowledgments
We are grateful for the technical assistance of staff of Functional Food Nutraceuticals and Phytomedicine Unit FUTA, Nigeria. We are also grateful to the Journal of Food Processing and Preservation for publishing the in vivo aspect of this project from which some of the data used for this research article were derived from.
Authors’ Contribution
Availability of Data and Materials
All data and databases used and/or analysed during the current study are available from the corresponding author on reasonable request.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Ethical Approval is not applicable for this article.
Funding
The authors received no financial support for the research, authorship, and/or publication of this article.
Statement of Human and Animal Rights
This article does not contain any studies with human or animal subjects.
Statement of Informed Consent
There are no human subjects in this article and informed consent is not applicable.
Consent for Publication
Not applicable.

