Abstract
Background
In ayurvedic practice, the
Objectives
The current study was planned to explore the
Materials and Methods
The aqueous extract of the
Results
The EPM test revealed that treatment with
Conclusion
The results substantiate the
Introduction
Dementia is a condition that chronically and progressively affects memory and learning and consequently affects thinking and comprehension. The most likely cause of dementia is Alzheimer’s disease, a neurodegenerative condition (
Ayurveda, the ancient system of medicine is being practiced in India for centuries. As a time-tested system of medicine, Ayurveda can serve as a rich and potential source for newer therapies in modern medical research. As per the Ayurveda postulation,
Materials and Methods
Crude Drug and Extract
The stem of
The aqueous extract of the
LC-MS Analysis of the Extract
Animal Welfare
The
Experimental Design and Treatment
The animals were randomly distributed into four groups (Groups 1, 2, 3, and 4) to make the mean body weight of all groups nearly equal before the start of dosing. Each group contained nine animals. Group 1 animals were administered purified water orally for the entire study period of 42 days and kept as normal control (NC). Aluminum chloride (Merck, Germany) was used to induce neurotoxicity in experimental rats. Aluminum chloride solution in water (100 mg/kg), which was used to be prepared fresh every day, was orally administered for 42 days to the rats of Groups 2, 3, and 4.
14
Donepezil HCl (Eisai Pharmaceuticals India Pvt. Ltd, Visakhapatnam, Batch No. AR1901) was used as a standard drug. Group 2 was kept as disease control (DC), whereas Group 3, which received Donepezil hydrochloride @ 1 mg/kg orally for 42 days 1 hr after the administration of aluminum chloride, was kept as standard control (SC).
14
In literature, researchers have frequently used doses of
The animals were observed daily for general clinical signs and changes in the skin, fur, eyes, mucus membrane, the occurrence of secretions and excretions, behavior, morbidity, and mortality twice daily.
Elevated Plus Maze
Neurobehavioral changes with reference to the memory due to the treatment were assessed using the elevated plus maze (EPM) test on days 20 and 42 of the experiment. An EPM with walls that were 40 cm high and with closed arms that were traversed by open arms was used. The middle square was 10 × 10 cm2 in size. The maze was kept 50 cm (2 inches) above the ground. During the acquisition stage, the animal was positioned at the end of the arm facing away from the center square. Initial transfer latency (ITL), which measures how long it takes an animal to transition from an open arm to a closed arm, was measured. Each animal was given 20 s to explore the maze after the ITL was recorded before being returned to its home cage.
Following treatment, retention transfer latencies (RTLs) on day 42 and ITLs on day 20 were noted.14, 20
Animal Sacrifice and Organ Collection
Animals were humanely sacrificed after 42 days using the CO2 asphyxia method of euthanasia. The liver, kidney, and brain were collected and weighed after the sacrifice. Brain tissues were collected for biochemical assays as per the standard methods described under each assay. The liver, kidney, and brain were collected in 10% neutral buffered formalin for the histopathological study.
Biochemical Assessment
Estimation of AChE from Brain
The hippocampus and cortex regions of the collected brain tissues were separated to estimate AChE activity. The cortex and hippocampus of the isolated brain were separated immediately and collected in different centrifuge tubes, weighed, and homogenized in an ice-cold phosphate buffer solution. The homogenates were then stored in a −20°C deep freezer until the assay was carried out. The assay was conducted according to the method described by Ellman14, 21 using the colorimetric AChE assay kit (Amplite, AAT Bioquest, USA) as per the procedure provided with the kit. The assay buffer was composed of the sodium phosphate buffer solution (0.1 M, pH 8.0), 0.1% BSA, a working solution of dithiobis-nitrobenzoic acid, and a working solution of acetylthiocholine iodide. The assay mixture was composed of 50 µL tissue sample homogenate and an equal quantity of the assay buffer.
Serially diluted AChE of 50 µL was mixed with 50 µL of the assay buffer to perform the SC assay. The change in the absorbance of the assay mixture was measured for 2 min at 30 s intervals at 410 nm using the enzyme-linked immunosorbent assay (ELISA) Plate Reader (Epoch, Biotek, USA, software version 5.1.11). The observations were expressed as the percentage inhibition of AChE.
Lipid Peroxidation, Reduced Glutathione, and Catalase and Superoxide Dismutase Assay from the Brain
The hippocampus and cortex regions of the collected brain tissues were separated to assess oxidative stress parameters. The isolated brain was separated immediately and collected in a centrifuge tube, weighed, and homogenized in an ice-cold phosphate buffer solution. The homogenate was then stored in a −20°C deep freezer till the assay was performed. The extent of lipid peroxidation (LPO) was measured in terms of malondialdehyde (MDA) production using the method described by Ohkawa et al. 22 The reduced glutathione (GSH) level was measured using the method described by Ellman. 23 The catalase assay was performed following the standard method. 24 Superoxide dismutase (SOD) activity was estimated according to the method described by Ukeda et al. 25 using the colorimetric SOD assay kit (Amplite, AAT Bioquest, USA) as per the procedure provided with the kit.
Proteomic Analysis of the Brain
The hippocampus of three animals from each group was collected in the separate centrifuge tubes, weighed, and homogenized in an ice-cold phosphate buffer solution containing the protease inhibitor tablet. The samples were further processed using the ProteaseMAX surfactant (Promega Inc., USA) following the procedure provided along with the product (
The procedure involved the precipitation of the protein from the samples, followed by its quantitation using the Bradford reagent using the ELISA plate reader. The precipitated proteins were digested using the ProteaseMAXTM surfactant, trypsin, and iodoacetamide in the NH4HCO3 buffer solution. In the last step, excessive protein digestion was stopped using formic acid. The mixture was then centrifuged at 12,000 rpm at 4°C for 20 min. The supernatant was thereafter collected from the tubes in LC-MS vials. The samples were analyzed using the UHD Accurate Mass 6538 Q-TOF LCMS system (Agilent Technologies, CA, USA) with a Zorbax extend C-18 analytical column (4.6 × 150 mm2 particle size, 5 µm) and a mobile phase made of water, acetonitrile, and formic acid (H2O:ACN:FA = 90:9.9:0.1). The data obtained after analysis using Agilent Spectrum Mill MS Proteomics Workbench LC-MS software revealed the
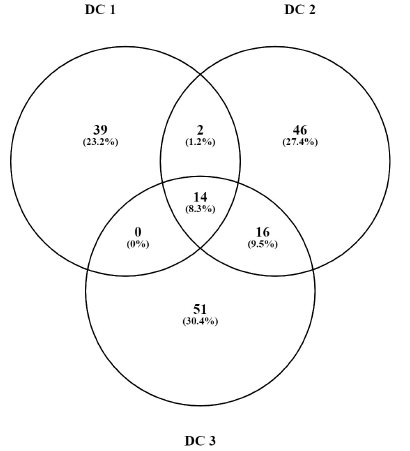
Proteins Commonly Expressed in all Three Animals in DC.
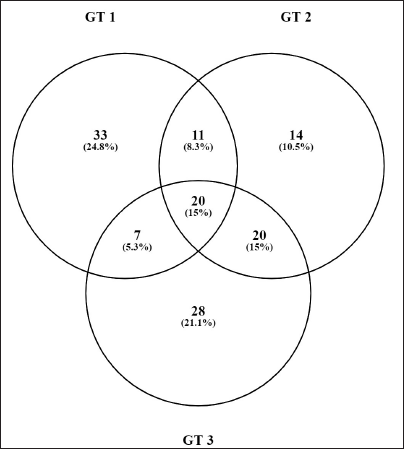
Proteins Commonly Expressed in all Three Animals in GT.
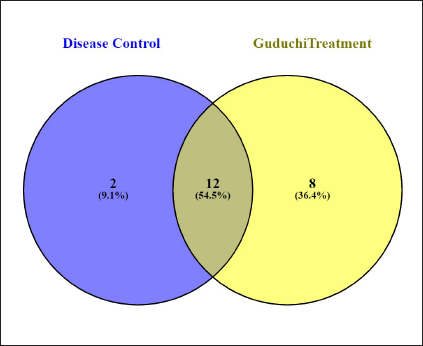
Proteins Commonly Expressed in DC and GT.
Statistical Analysis
Groupwise means of weekly absolute body weights, body weight gain, feed consumption, absolute organ weights, relative organ weights to body weight, antioxidant assay, and AChE assay were statistically analyzed by applying one-way analysis of variance (ANOVA) using SPSS 16.0 software. Elevated plus maze test data were analyzed by two-way ANOVA, followed by Bonferroni’s test, and the significance level was determined within and between groups using Graph Pad Prism V5.0 software.
Histopathology
Brain, kidney, and liver samples were trimmed, processed in ascending grades of alcohol and xylene, embedded in paraffin blocks, sectioned, and stained with routine hematoxylin and eosin, and subjected to histopathology evaluation.
Results
Crude Drug and Extract
Physico-chemical properties such as a loss on drying, ash value, and extractive values of the
Physico-chemical Properties of the Guduchi Stem.

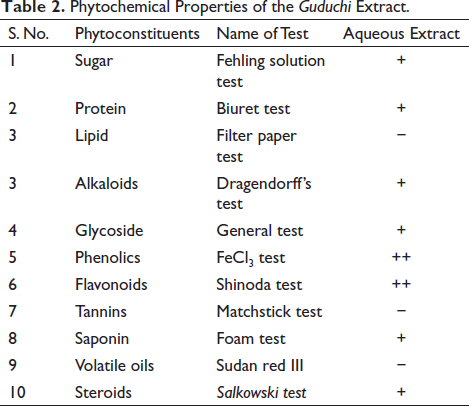
Phytochemical Properties of the Guduchi Extract.
LC-MS Analysis of Extracts
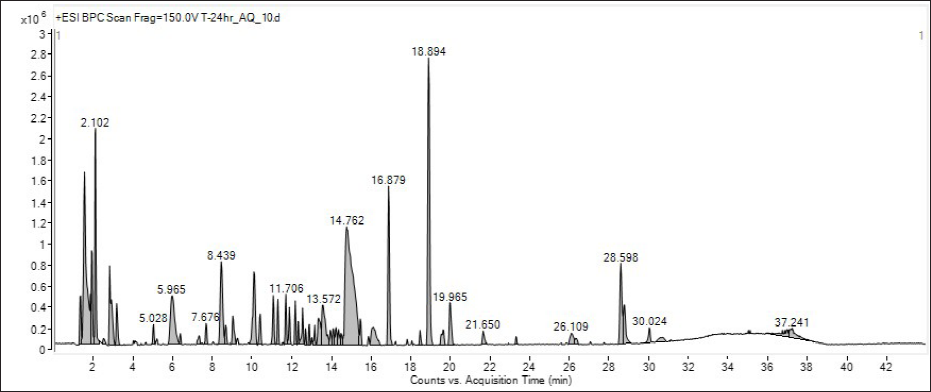
The LC-MS chromatogram of the aqueous extract of the
Important Phytoconstituents Found on LC-MS Analysis of the Guduchi Aqueous Extract.
Chromatogram of LC-MS Analysis of the Guduchi Aqueous Extract.
Clinical Signs, Mortality, and Other In-life Phase Observations
The cage-side observation showed no significant change in the clinical signs of the animals across the groups. No significant change in feed consumption and the body weight gain was found between the groups.
Elevated Plus Maze
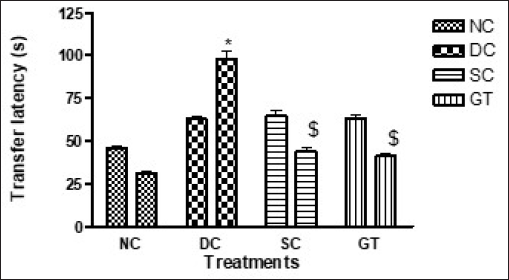
In the EPM test, ITL on day 20 was significantly increased in the DC group compared with other groups, including the NC group. Furthermore, the ITL in the SC and GT groups was comparable with the DC group. However, on day 42, RTL in the SC and GT groups was significantly reduced compared with the DC group. Therefore, the treatment with the
Elevated Plus Maze Test.
Biochemical Assessments
AChE Assay
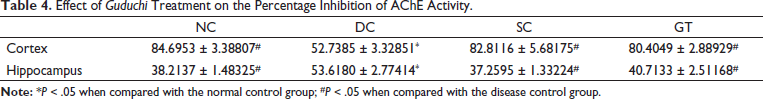
Cortical inhibition of AChE in the DC group was significantly lower than in other groups, including the NC group. Therefore, treatments in the SC and GT groups induced significant inhibition of cortical AChE in comparison with that in the DC group. Moreover, the AChE inhibition in the GT and SC groups was comparable with that in the NC group. The hippocampal AChE inhibition in the DC group was significantly higher than that in other groups, including the NC group. Thus, treatment with donepezil in the SC group and with the
Effect of Guduchi Treatment on the Percentage Inhibition of AChE Activity.
Oxidative Stress Assessment
The cortical and hippocampal MDA production in the DC group was significantly higher than in other groups, including the NC group. Thus, the treatment in the SC and GT groups led to a significantly decreased production of MDA in the cortex and hippocampus compared with that in DC group (Table 5).
Effect of Guduchi Treatment on Oxidative Stress Parameters in the Cortex.
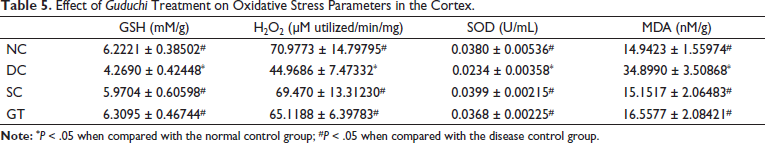
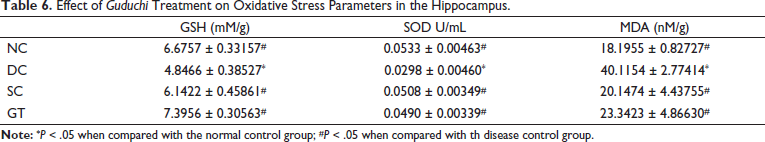
The cortical and hippocampal GSH and SOD concentration in the DC group were significantly lower than that in other groups, including the NC group. Thus, the treatment in the SC and GT groups led to a significant increase in the concentration of GSH and SOD in the cortex and hippocampus compared with that in the DC group. Similarly, the H2O2 concentration was found to be increased in the cortex in the SC and GT groups, which can be attributed to the treatment (Table 6). Overall oxidative stress was found to be reduced in rats treated with donepezil (SC) and Guduchi (GT).
Effect of Guduchi Treatment on Oxidative Stress Parameters in the Hippocampus.
Histopathology
Histopathological evaluation of the brain, kidney, and liver revealed that no aluminum chloride treatment was performed in the DC group compared with the NC group. No histomorphological changes were observed in the SC and GT groups as well. The results were consistent with earlier reports.30, 31
Proteomic Analysis
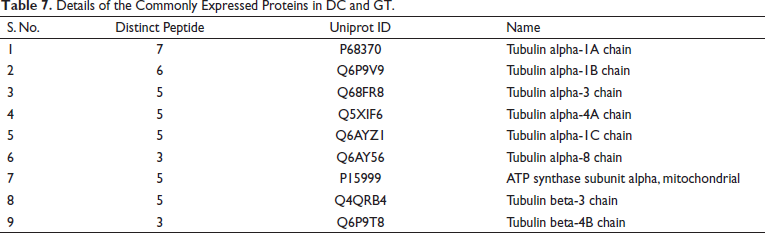
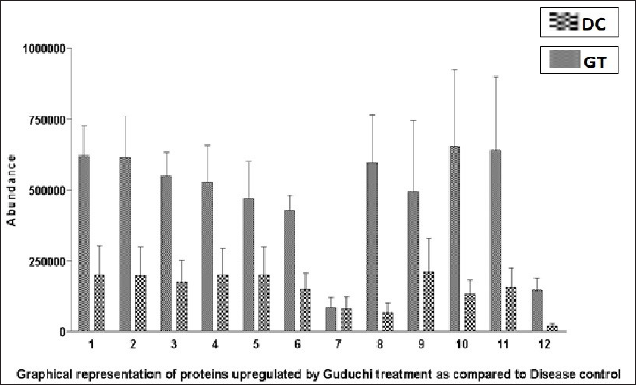
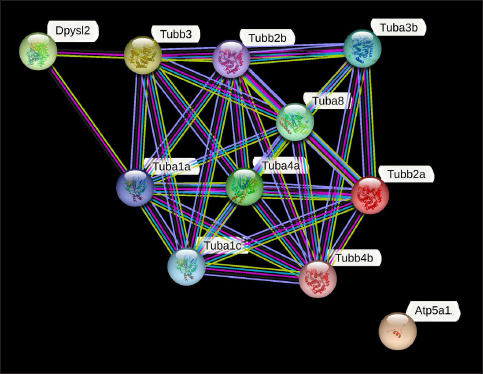
The data showed the expression of 12 proteins, which were commonly expressed in the DC and GT groups. The details of the proteins are given in Table 7. The groupwise abundance of proteins and their graphical representation are presented in Figure 6, which shows the upregulation in the GT. Both α and β tubulins were the major proteins expressed. Furthermore, protein–protein interaction was predicted using the string database that indicated that proteins are highly interrelated and co-expressed. 28 The string plot is presented in Figure 7.
Details of the Commonly Expressed Proteins in DC and GT.
Graphical Representation of Abundance of Commonly Expressed Proteins in DC and GT.
String Plot of Proteins Commonly Expressed in DC and GT Indicating that Proteins are Highly Interrelated.
Discussion
The results of qualitative and quantitative phytochemical analysis of the
Acetylcholine is the principal neurotransmitter in the nervous system, which plays a vital role in various functions such as memory, learning, and coordination of motor activity.
40
The marked decrease in the level of ACh is an important pathophysiological change observed in various neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease.2, 3 High levels of aluminium have been reported in the brains of AD patients.3, 7, 41, 42 Aluminum is acetylcholinotoxic in experimental animals and causes apoptotic loss of neurons, eventually resulting in AD.
43
The decrease in the level of ACh is also associated with damage to the cholinergic neurons, possibly due to increased oxidative stress in the brain.3, 44 Thioacetylcholine is hydrolyzed by the enzyme AChE, which stops the transmission of nerve impulses. In diseases like Alzheimer’s disease, where large amounts of AChE are seen in AD patients’ brains, the degree of memory loss is significantly correlated with the decline of cholinergic transmission.
14
In the present study, a significant increase in AChE activity due to its lower inhibition in the cortex was observed in the Aluminum chloride-treated group compared to the NC group, which is in line with published literature.
14
The present study also showed the significantly decreased activity of hippocampal AChE due to higher inhibition in the Aluminum chloride treated group when compared to the NC group. The results were found to be as per the literature published.
45
Treatment with
EPM test assesses memory-related behavior by measuring transfer latency from open arm to close.20, 47 The aluminum chloride-induced neurotoxicity leads to progressive memory loss, which is reflected in the increased RTL compared to the ITL, as rats could not memorize and recollect the path on the maze. The present study showed that AlCl3 administration to the rats resulted in progressive deterioration of memory and consequent neurobehavioral deficits. Therefore, rats of the DC group showed an increase in RTL compared to ITL and a significant increase in RTL compared to NC. Therefore, the results of the EPM test in the neurotoxicity DC group show that the model was established as reported in earlier reports.14, 20, 30 Treatment with
A decrease in catalase, SOD, GSH, and an increase in MDA/LPO level, as well as neuronal damage in the cortex and hippocampus regions of the brain, were all signs indicating a considerable increase in oxidative stress in the brain following aluminium chloride therapy in the current study. According to earlier research, aluminium stimulates and activates LPO when there is iron present in the brain.
50
Aluminium enhances iron-based oxidation in the brain, which alters iron homeostasis mainly via the Fenton reaction, increasing iron-induced oxidative injury.
51
Aluminium is reportedly responsible for modulation in brain amyloidosis through oxidative damage.
52
An increase in oxidative stress due to aluminium chloride treatment in rats leads to exhaustion of the body’s various endogenous antioxidant enzymes such as catalase, SOD, and GSH resulting in their decreased concentrations in the tissues.
53
Thus, the findings of antioxidant assays in DC are as per the reported literature. In the present study, treatment with an aqueous extract of
These results of the proteomic study revealed the increased expression of tubulin proteins in GT, which are involved in the generation, migration, and differentiation of neurons.
56
α and β–tubulins are reported to play a significant role in the survival and positioning of neurons.
57
α- and β-tubulin heterodimers form microtubules which are required for axonal transport of molecular motors in neurons.
58
The concept of
Conclusion
The results suggest that
Summary
The present article reports the study of an aqueous extract of the
Footnotes
Acknowledgement
The authors are thankful to the Director General, Central Council for Research in Ayurvedic Sciences, New Delhi for the funding support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship and/or publication of this article.
Ethical Statement
The approval for the conduct of the animal experimentation study was obtained from the Institutional Animal Ethics Committee vide approval number IAEC/2019_3/05 (dated: October 15, 2019).
Funding
The study was carried out under the Intra Mural Research Scheme of the Central Council for Research in Ayurvedic Sciences, New Delhi vide project sanction no. 3-110//2019-CCRAS/Admin./IMR4/3750 (dated: September 30, 2019).
