Abstract
Background
Korean pine nuts (Semen Pinus koraiensis (P. koraiensis)) have been a valuable ingredient in Korean cuisine since ancient times, and the classical literature on traditional medicine states that pine nuts can moisturize skin.
Aims
To confirm the therapeutic efficacy of the ethanol extract of these pine nuts in an animal contact dermatitis (CD) model.
Materials and Methods
Korean pine nuts were extracted using 70% ethanol, and the crude extract obtained was condensed and lyophilized to produce EEPK (the 70% ethanol extract of P. koraiensis). A DNFB (1-fluoro-2,4-dinitrofluorobenzene)-induced BALB/c mouse model of CD was used to investigate the effects of EEPK on skin symptoms, histopathological abnormalities, and cytokine secretion in inflamed tissues.
Results
EEPK reduced skin color changes, lesion severities, and dorsal skin thickening and inhibited epidermis hyperplasia and immune cell infiltration into inflamed tissues. In addition, EEPK suppressed the productions of tumor necrosis factor-α, interferon-γ, interleukin-6, and monocyte chemoattractant protein-1 but did not affect body weights or spleen/body weight ratios.
Conclusion
P. koraiensis has potential as a therapeutic agent for CD and as a treatment for inflammatory skin disease management products. Furthermore, the mechanism underlying its therapeutic effects differs from that of corticosteroids.
Introduction
Korean pine nuts (Semen Pinus koraiensis (P. koraiensis),  , Haesongja in Korean) are the seeds of P. koraiensis Siebold and Zucc. They have an oval shape, an oily, soft consistency, are covered with the white outer skin,[1] and are produced throughout the Korean Peninsula and northeastern China. Pine nuts have been commonly used in Korean cuisine for centuries. For example, finely ground pine nuts are boiled with rice to make porridge and are also used as a garnish for Sujeonggwa, a famous traditional drink prepared using cinnamon and ginger.
, Haesongja in Korean) are the seeds of P. koraiensis Siebold and Zucc. They have an oval shape, an oily, soft consistency, are covered with the white outer skin,[1] and are produced throughout the Korean Peninsula and northeastern China. Pine nuts have been commonly used in Korean cuisine for centuries. For example, finely ground pine nuts are boiled with rice to make porridge and are also used as a garnish for Sujeonggwa, a famous traditional drink prepared using cinnamon and ginger.
Pine nuts have a sweet flavor and are warm in nature; in traditional medicine, these characteristics nourish and regenerate body fluids. For this reason, it is recommended for patients with a dry cough, elderly with a chronic cough, and to “moisten” lungs and colon in patients with chronic or geriatric constipation. Furthermore, classical, traditional medicine literature states that pine nuts can moisturize skin.[2]
Modern studies have reported that P. koraiensis seed oil can reduce weight in obese mice,[3] improve insulin resistance in patients with type-2 diabetes,[4] reduce the risk of arteriosclerosis, and relieve inflammation in rheumatoid arthritis.[5] Pine nut oil is composed of fatty acids, such as palmitoleic, stearic, and pinolenic acids, and pinolenic acid has been shown to be largely responsible for its anti-inflammatory effect.[6]
A study conducted between 2007 and 2017 found that 19.2% and 20.6% of the European and Asian populations had contact dermatitis (CD).[7] Contact dermatitis is caused by irritations produced by detergents, solvents, alcohol, metals, and other substances. Skin lesions are characterized by pain, a burning sensation, itching, fissures, erythema, marginal scales, and skin dryness.[8] In the United States, CD accounts for 90%–95% of all occupational skin diseases (OSDs) that directly or indirectly affect health and productivity and result in disability-related costs, compensation claims, and job changes. These effects result in costs that exceed $1 billion annually, and those affected are forced to take corticosteroids to sustain their careers.
We investigated the anti-inflammatory effects of P. koraiensis nuts in a DNFB (1-fluoro-2,4-dinitrofluorobenzene)-induced mouse model of CD using lesion severity scores, skin and epithelial thicknesses, melanin and erythema indices, degrees of immune cell infiltration, and levels of cytokines and chemokines (tumor necrosis factor (TNF)-α, interferon (IFN)-γ, interleukin (IL)-6, and monocyte chemoattractant protein (MCP)-1).
Materials and Methods
Preparation of P. koraiensis Extract
The Korean pine nuts were purchased from a public market (Sunny Mart, Gyeongnam, South Korea) and certified by Professor Jeong-Hoon Kim (Voucher no. MS-2017017). Chopped nuts were extracted using 70% ethanol (the ethanol extract of P. koraiensis, EEPK), as previously described (yield, 6.5%).[9]
Animals
The animal study was conducted using BALB/c mice (male, 6-week-old, Samtaco, Gyonggi, Republic of Korea). The study protocol was preapproved by our Institutional Animal Care and Use Committee (PNU-2019-2269).
Experimental Design and the Induction of Contact Dermatitis
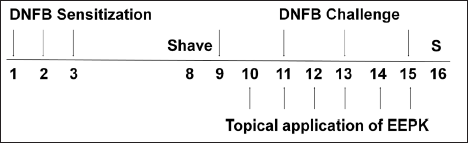
Experimental CD was induced using standardized methods, as previously described.[10] The shaved backs of normal (NOR) animals were treated with vehicle topically (n = 7). CD controls (CTL) were treated with DNFB (CD induction) and then with vehicle (n = 8). EEPK-treated animals were treated topically with DNFB and treated with EEPK (25, 75, or 250 µg/day) (n = 8/group). Dexamethasone (DEX; the positive control) was applied topically at 150 µg/day for 6 days. The experimental process is summarized in Figure 1.
Assessment of Skin Lesion Severities
Skin lesions (roughness, excoriation, scab, and erythema) severities were assessed on day 16 using a 4-point scale (0 = no, 1 = slight, 2 = mild, and 3 = severe symptoms), as previously described.[9]
Erythema and Melanin Indices
Dorsal skin color was evaluated on day 16 at three different locations per mouse using a skin colorimeter (DSM II, Cortex Technology, Denmark). The representative value of each mouse was presented as the average of three values.
Skin Weights and Thicknesses
Dorsal skins were removed, and skin samples were obtained using a biopsy punch (diameter 5 mm). Skin thicknesses were measured using a Vernier caliper (Mitutoyo, Tokyo, Japan).
Histopathological Examination
Inflamed tissues were dissected, embedded in paraffin, stained with hematoxylin and eosin (H&E), and observed under an optical microscope (100×). Epidermal hyperplasia and immune cell infiltration were evaluated as previously described.[9, 11]
Measurement of Cytokine Levels
Cytokine levels in skin samples were assessed using a cytometric bead array mouse inflammation kit (BD, San Jose, CA, USA).
Calculation of Spleen Bodyweight Ratios
Changes in body weights between day 1 and day 16 are expressed as percentages of body weights. Spleens were weighed after sacrifice on day 16.
Statistical Analysis
The significance of intergroup differences was determined by one-way analysis of variance (ANOVA) and Dunnett’s multiple comparisons. The analysis was performed using Prism 5 (version 5.01) software. Results are expressed as means ± standard deviations, and statistical significance was accepted for P values < .05.
Results
The Effects of EEPK on Skin Lesions and Thickness in CD Mice
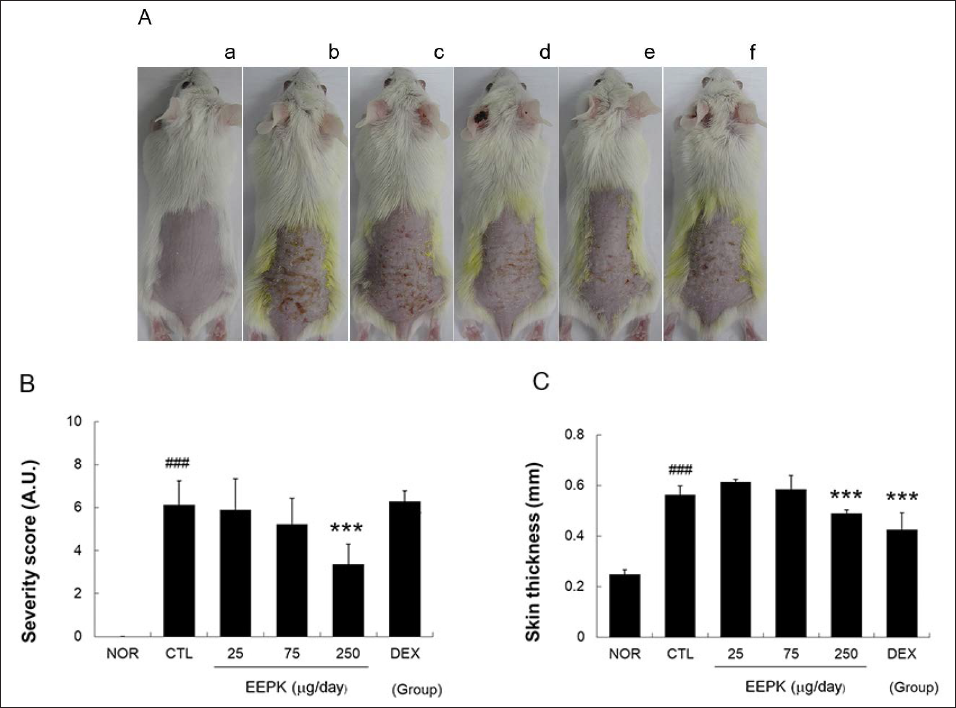
Local application of 0.15% DNFB to shaved skin caused induration, fissure, scale, erythema, and petechiae. The topical application of EEPK for 6 days reduced these symptoms (Figure 2A). Skin thickness in the CTL group was more than twice that in the NOR group, and skin thickness in the 250 µg/day EEPK group was 13% lower than that in the CTL group. Skin thickness in the DEX group was significantly lower than that in the CTL group, but no improvement in skin symptoms was observed (Figures 2B and 2C).
Effects of EEPK on skin lesions and thickness in CD mice. Skin lesions were observed using a digital camera on day 16. (A) a, NOR; b, CTL; c, 25 µg/day EEPK; d, 75 µg/day EEPK; e, 250 µg/day EEPK; f, DEX. (B) NOR, non-treated-naïve group; CTL, non-treated CD group; EEPK, ethanol extract of P. koraiensis seed treated CD groups; DEX, dexamethasone-treated CD group. A. U. means arbitrary units. (C) Skin thicknesses were measured using a Vernier calipers. All values are expressed as means ± standard deviations. ###P < 0.001 versus the NOR group (non-treated-naïve controls); ***P < 0.001 versus the CTL group (non-treated CD group).
The Effects of EEPK on Erythema and Melanin Indices
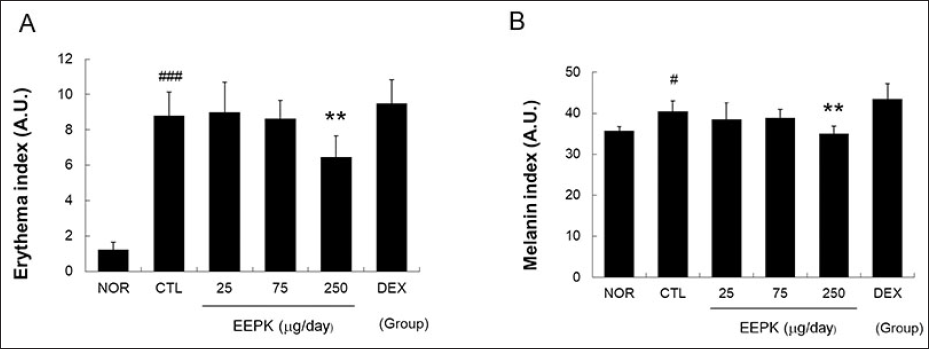
The mean erythema index in the CTL group was more than 7 times that in the NOR group. Administration of EEPK at 250 µg/day reduced erythema by 26% as compared with the CTL group. Likewise, DNFB increased melanin indices in the CTL group, and EEPK administration reduced melanin indices. However, DEX did not reduce DNFB-induced increases in erythema or melanin indices (Figures 3A and 3B).
Effects of EEPK on erythema and melanin indicis. Melanin (A) and erythema indices (B) were measured using a dermo-spectrophotometer. Results are expressed as means ± standard deviations. #P < 0.05 and ###P < 0.001 versus the NOR group; **P < 0.01 versus the CTL group. Abbreviations are as defined in the legend of Figure 2.
The Effects of EEPK on Histopathological Changes in Inflamed Tissues
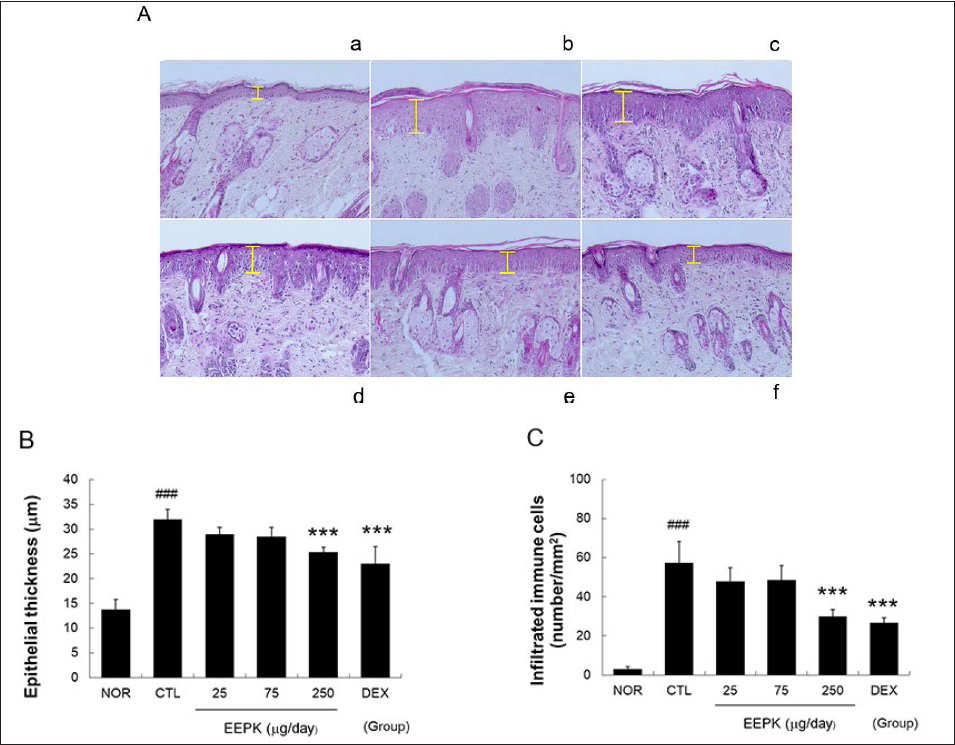
The topical application of DNFB destroyed the basal cell layer and produced an unclear boundary between epidermis and dermis, immune cell infiltration, papillary dermal edema. EEPK treatment at 250 µg/day inhibited these changes (Figure 4A). Furthermore, 250 µg/day treatment of EEPK reduced epidermal hyperplasia significantly in inflamed tissues by 21% and reduced immune cell infiltration by 48% as compared with the CTL group. DEX also significantly reduced epidermal hyperplasia and immune cell infiltration (Figures 4B and 4C).
Effects of EEPK on histopathological changes in inflamed tissues. The skin tissues were stained with H&E and observed under a light microscope (100×). (A) Yellow bars indicate epidermal thicknesses. (B) Distances between basal lamina and the upper surface of stratum granulosum were measured. Results are expressed as means ± standard deviations. ###P < 0.001 versus the NOR group; ***P < 0.001 versus the CTL group. Abbreviations are as defined in the legend of Figure 2.
The Effects of EEPK on Cytokine and Chemokine Levels in Inflamed Tissues
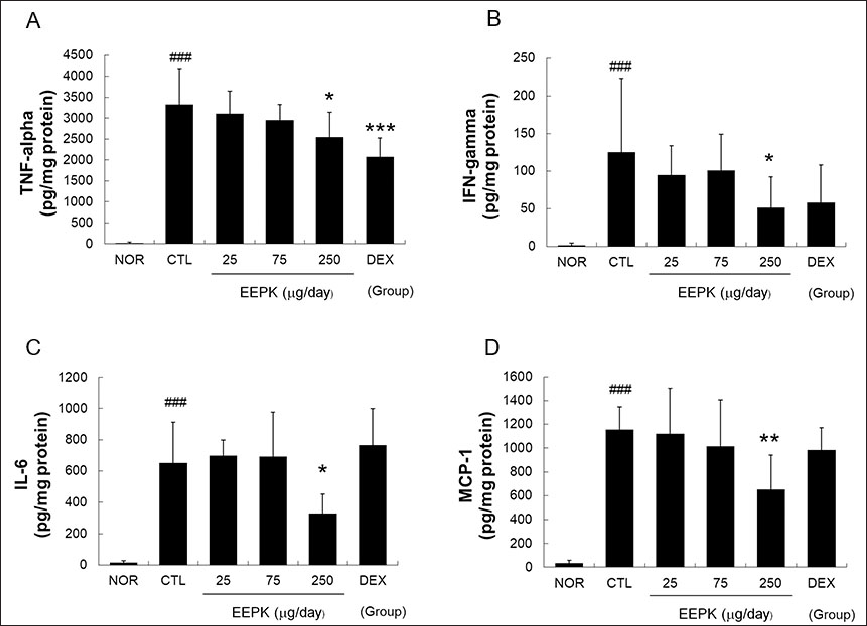
Significant increases in cytokine (TNF-α, IFN-γ, and IL-6) and chemokine (MCP-1) levels were observed in the CTL group. EEPK and DEX significantly lowered the TNF-α levels (Figure 5A). The topical application of EEPK at 250 µg/day also significantly reduced the IFN-γ, IL-6, and MCP-1 levels (Figures 5B–5D).
Effects of EEPK on cytokine and chemokine levels in inflamed tissues. Cytokine and chemokine levels were assessed using a cytometric bead array method. (A) Tumor necrosis factor-alpha, (B) interleukin-6 and (C) monocyte chemotactic protein-1. N.D. means undetectable. Results are expressed as means ± standard deviations. ###P < 0.001 versus the NOR group; *P < 0.05, **P < 0.01, and ***P < 0.001 versus the CTL group. Abbreviations are as defined in the legend of Figure 2.
The Effects of EEPK on DNFB-induced Changes in Body Weights and Spleen/Body Ratios
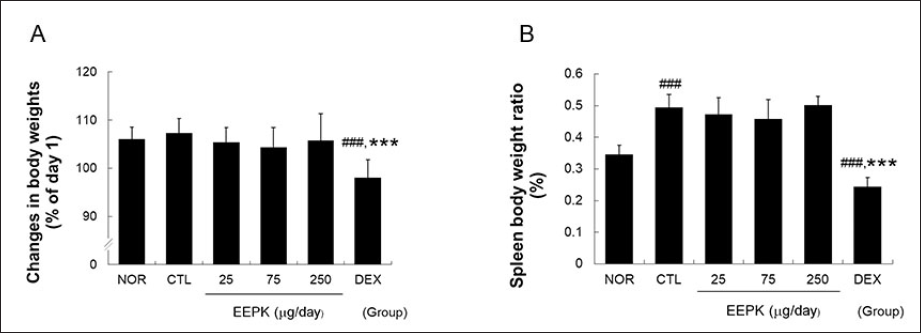
Contact dermatitis induction did not affect body weights but significantly increased spleen/weight ratios (Figure 6). On the other hand, weight gains and spleen/weight ratios were the same in both the EEPK and CTL groups but significantly lower in the DEX group than that in the CTL group. Furthermore, the mean spleen/weight ratio in the DEX group was significantly lower than that in the NOR group (Figure 6).
Effects of EEPK on DNFB-induced changes in body weights and spleen/body ratios. Body and spleen weights were measured on day 15, and spleen/body weight ratios were calculated: (A) body weights and (B) spleen/body weight ratios. Results are expressed as means ± standard deviations. ###P < 0.001 versus the CTL group and ***P < 0.001 versus the CTL group. Abbreviations are as defined in the legend of Figure 2.
Discussion
In Bencao Gangmu (本草纲目), a famous herbal text written by Li Shizhen (李时珍) during the Ming (明) Dynasty, P. koraiensis is referred to as Xinluosong (新罗松). Xinluo (新罗) is an ancient dynasty on the Korean Peninsula. Korean pine nuts have been used medicinally for a very long time in China, Korea, and Japan, and classical literature states that they moisturize skin and enrich hair.[2]
Essential oils are often used in skincare products and as natural treatments for skin diseases, and patients with atopic dermatitis currently use essential oils as complementary treatments or as alternatives to modern treatments (e.g., corticosteroid treatments). In Korea, oils extracted from pine nuts, sesame seeds, camellia, and arborvitae fruits are used for skin care.[12] Jung et al.[13] recently studied the anti-atopic dermatitis effects of five essential oils and reported that pine nut (semen P. koraiensis) essential oil inhibited thymus secretion, macrophage-derived chemokine levels, and IL-6 levels in HaCaT keratinocytes stimulated with TNF-α and IFN-γ.
Based on the above discussions, we hypothesized that the topical application of EEPK might alleviate the symptoms of CD. In our mouse model, topical stimulation by DNFB caused skin thickening and lesions typical of CD (induration, fissure, scale, erythema, and petechiae (Figure 2)) and skin color changes (Figure 3), and these abnormalities were effectively ameliorated by the topical application of EEPK (Figures 2 and 3).
The inflammatory process within skin tissues is accompanied by pathological changes in tissues that include basal cell layer destruction, an unclear boundary between epidermis and dermis,[14] activated keratinocyte-induced epidermal hyperplasia and hyperkeratosis,[14] and infiltrations of various immune cells into the perivascular region.[15] In our animal model, tissue damage was caused by repeated DNFB stimulation, and EEPK significantly ameliorated these abnormalities (Figure 4). These results indicate that EEPK can alleviate skin lesions and restore normal skin color by preventing histopathological abnormalities.
The profile of inflammatory cytokines expressed in the skin tissues of CD patients is well known. Silvestre and Reis, in a small-scale clinical study, found that TNF-α, IFN-γ, IL-4, IL-13, and IL-17 were overexpressed in the skin tissues of CD patients.[16] TNF-α and IFN-γ play major roles in the pathogenesis of CD and directly stimulate keratinocytes to promote proliferation and differentiation, and, as a result, induce epidermal hyperplasia and hyperkeratosis in inflamed tissues.[17] In addition, TNF-α and IFN-γ can increase the keratinocyte secretions of chemokines. For this reason, the inhibitions of TNF-α and IFN-γ are important targets for the treatment of skin diseases associated with the hyper-proliferation and abnormal differentiation of keratinocytes.[18] TNF‐α is a primary cytokine that causes the release of secondary cytokines and chemokines and can induce the secretions of IL-6 and MCP-1.[19] In addition, it is well known that IL-6 and MCP-1 are involved in the lifespan extension and infiltration of immune cells.[20, 21] In a recent study, IL-6 and MCP-1 were identified as the factors that aggravate skin inflammatory diseases in certain environments.[22] In the present study, EEPK significantly prevented the production of TNF-α, IFN-γ, IL-6, and MCP-1 in the skin tissues of CD mice (Figure 5), and our results show that the anti-inflammatory properties of EEPK can prevent basal cell layer destruction and histopathological abnormalities, such as epidermal hyperplasia, and immune cell infiltration by suppressing cytokine and chemokine production.
Korean pine nuts contain several fatty acids, and its pinolenic acid (a major component) has known anti-inflammatory effects. For example, it was reported to reduce IL-6 and TNF-α release in LPS-stimulated peripheral blood mononuclear cells obtained from rheumatoid arthritis patients,[5] and when applied topically to mice, it suppressed 12-O-tetradecanoylphorbol-13-acetate-induced pro-inflammatory cytokine production.[23] The present study also shows that EEPK inhibits inflammatory cytokine production, suggesting that pinolenic acid suppresses skin inflammatory diseases.
Although many researchers have been studying the treatment of skin diseases for many years, the choice of patients with chronic inflammatory skin disease, especially in the case of patients with OSD, is still difficult to get out of the category of corticosteroids. In addition to the well-known systemic adverse effects such as immunosuppression and growth retardation, corticosteroids can cause skin atrophy even when applied topically.[24]
We found that DEX did not improve skin surface symptoms or ameliorate skin color changes but significantly reduced DNFB-induced skin thickness increases. These observations mean that the reduction in skin thickness observed in the DEX group was not only caused solely by its anti-inflammatory effect but also by skin atrophy. Weight gain was suppressed in the DEX group, and the spleen/body weight ratios were significantly lower than that in the CTL group (Figure 6), and these are typical of the effects of systemic corticosteroid administration in mice.[9, 10] Unlike DEX, topical EEPK application did not influence body weights or spleen/body weight ratios (Figure 6). These results imply that the mechanisms responsible for the effects of EEPK and DEX differ and that EEPK does not possess the general immunosuppressive effects of DEX.
Conclusion
In this study, the therapeutic effects of P. koraiensis on skin lesions in our murine DNFB model of CD were investigated. We found that the topical application of EEPK reduced DNFB-induced induration, fissure, scale, erythema, and petechiae, and changes in skin color. The therapeutic effects of EEPK were attributed to the suppression of abnormal histopathological findings, such as epidermal hyperplasia and immune cell infiltration. In addition, EEPK ameliorated CD abnormalities in skin tissues and suppressed the production of pro-inflammatory cytokines and chemokines. Furthermore, although the efficacies of EEPK and DEX were similar, their action mechanisms appeared to differ. These results indicate that semen P. koraiensis has potential use as a treatment for CD or as a raw material in inflammatory skin disease management products.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
The study protocol was preapproved by our Institutional Animal Care and Use Committee (PNU-2019-2269).
Funding
The authors disclosed the following financial support for the research, authorship, and/or publication of this article: This research was supported by a grant from the Korea Health Technology R&D Project through the Korea Health Industry Development Institute (KHIDI), funded by the Ministry of Health and Welfare, Republic of Korea (grant number: HF21C0089).
