Abstract
We describe a novel nature-derived epoxy resin monomer (ERM) derived from the plant lignan pinoresinol. Epoxy resins are thermosetting materials in global usage owing to their excellent technical properties such as flexibility and durability. However, their adverse health effects are often not considered and affect users of epoxy resins worldwide. Components of epoxy resin systems are strong skin sensitizers and cause allergic contact dermatitis. The reported prevalence attributable to epoxy chemicals is between 11.7 and 12.5% of all cases of occupational allergic contact dermatitis. We are committed to developing epoxy resins with reduced allergenic effect, while maintaining their excellent properties. The novel ERM, pinoresinol diglycidyl ether (PinoDGE), was synthesized in one step from pinoresinol and epichlorohydrin in 88% yield. It was not classified as a skin sensitizer in the in vivo local lymph node assay, at concentrations up to 0.17
There is enormous interest in replacement of conventional plastics with bio-based products from renewable sources. Epoxy resin systems (ERS) are no exception. Epoxy resin systems are commercial thermosetting products used in many diverse applications because of their outstanding performance and resistance. They are particularly useful as adhesives, sealants, inks, paints, and coatings (Allied Market Research, 2020; Royal Society of, 2021). The global epoxy resins market accounted for USD 5.9 billion in 2019 and is expected to reach USD 10.3 billion by 2027, with a global market volume of 3 million tonnes (Allied Market Research, 2020; Royal Society of, 2021). However, the sustainability of fossil-derived ERS is under scrutiny and environmentally friendly options are much sought after (RSC (2021) Natural and bio-based ERS replacements are under investigation, including plant-derived lignins (Ng et al., 2017; Hofmann and Glasser, 1993; Koike, 2012). Biodegradable products that are themselves made from waste would be an excellent alternative to current ERS. We are interested in designing nature-inspired epoxy resins with an additional requirement: reduced capacity to cause skin allergy in users, compared to commercially available resins. It is essential that the health effects of new thermosetting products are considered at the design stage prior to commercial development, so that adverse effects can be minimized or prevented.
Skin contact allergy is the most common form of human immunotoxicity, prevalent in up to a quarter of the European population (Diepgen et al., 2016; Thyssen et al., 2007). It is a type IV delayed-type hypersensitivity reaction to chemicals known as contact allergens. Allergic contact dermatitis, ACD, is the clinical syndrome associated with repeated exposure to a contact allergen and is associated with significant healthcare costs, productivity loss, and patient suffering. Contact allergy to ERS is widespread in dermatitis patients with prevalence ranging from 0.9 to 2.3% (Canelas et al., 2010; Bangsgaard et al., 2012). Epoxy chemicals are implicated in both occupational and non-occupational contact allergy (Majasuo et al., 2012; Amado and Taylor, 2008). The reported prevalence attributable to epoxy chemicals is between 11.7 and 12.5% of all cases of occupational ACD. Workplace studies found exceptionally high rates of ACD from ERS in aircraft manufacturing workers (56%), marble workers (45%), painters (22.6%), metalworkers (10.7%), and construction workers (up to 9.7%) (Higgins et al., 2018). Protective measures, such as use of gloves, are not always effective (Suuronen et al., 2019). Many ERS components cause ACD, most commonly the two epoxy resin monomers (ERMs) diglycidyl ether of bisphenol A (DGEBA, Figure 1) and diglycidyl ether of bisphenol F (DGEBF, Figure 1). These ERMs are highly skin-sensitizing and are among the most common causative agents of occupational ACD (Geier et al., 2004). DGEBA is included in the European baseline series for diagnosis of ACD (Wilkinson et al., 2019), DGEBA (Thorgeirsson et al., 1978; Delaine et al., 2011), and DGEBF (Delaine et al., 2011; Pontén et al., 2002) and are classified as strong sensitizers in both mice and guinea pigs according to regulatory guidelines (Basketter et al., 2005). Structures of diglycidyl ether of bisphenol A (DGEBA), diglycidyl ether of bisphenol F (DGEBF), pinoresinol, diglycidyl ether of pinoresinol (PinoDGE), phenylglycidyl ether (PGE), and the analogs 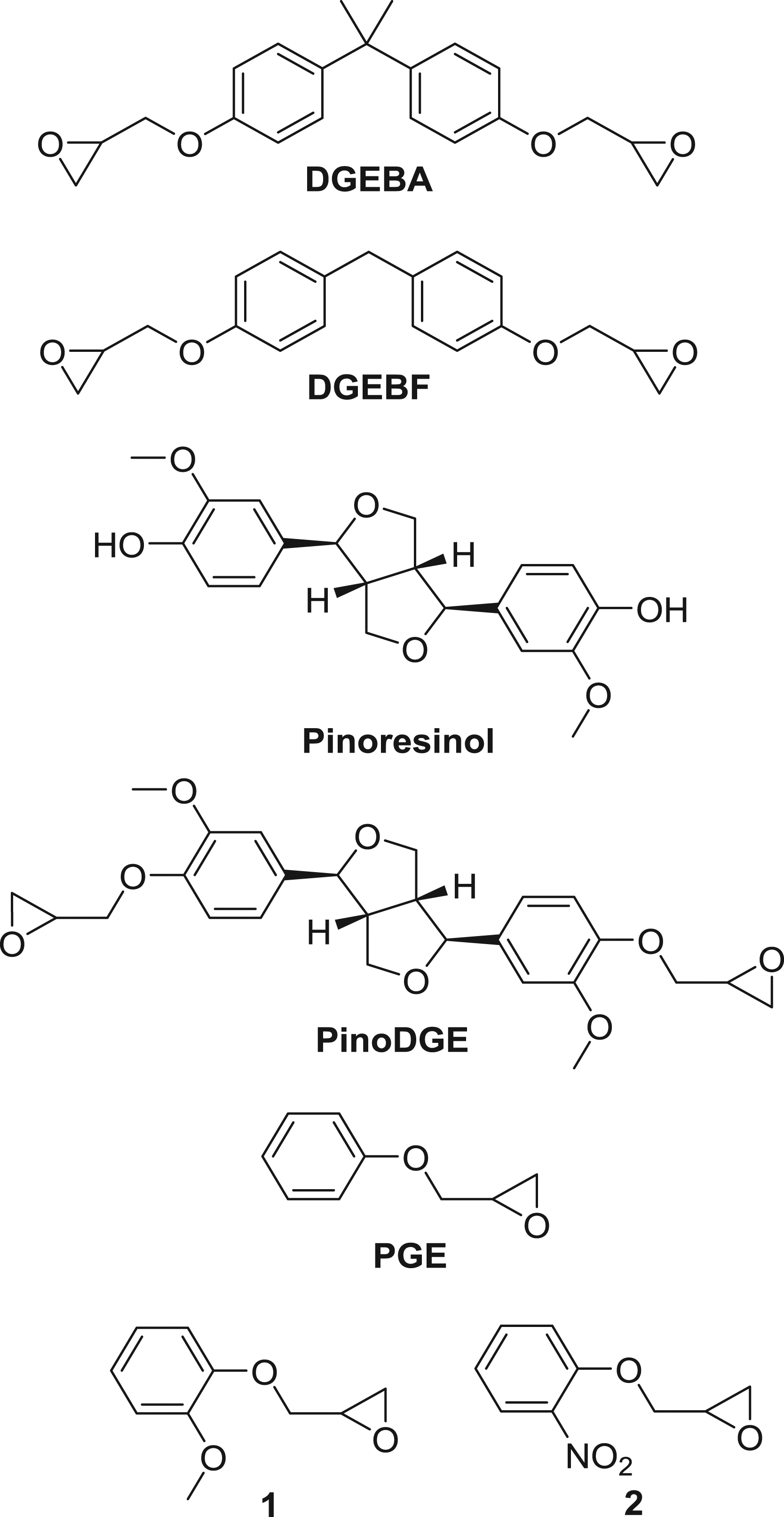
In our search to develop safer ERMs, we identified the plant-derived lignan pinoresinol (Figure 1) as a chemical template for a novel ERM. Pinoresinol is a furofuran lignan found in many plant species, including Forsythia (Oleaceae family) and Cedrus atlantica (Pinaceae family) (Kitagawa et al., 1984; Nam et al., 2011). Based on our previous structure activity relationship (SAR) studies on DGEBA, we considered the structure of pinoresinol suitable for modification to form a nature-derived, bio-based ERM (O’Boyle et al., 2014; Ponting et al., 2019). We synthesized and evaluated the effects of such a novel bio-based ERM, PinoDGE, on skin sensitization in vitro and in vivo. We compared the curing and cross-linking properties of PinoDGE to DGEBA using thermogravimetric analysis (TGA). The monoepoxide phenylglycidyl ether (PGE, Figure 1) is a common reactive diluent used in ERS and is a strong skin sensitizer (Niklasson et al., 2009) It is a monosubstituted aromatic ring bearing structural similarities to DGEBA and was used for further SAR studies of the terminal epoxides.
Materials and methods
Caution: This study involves skin-sensitizing compounds that should be handled with particular care.
Chemistry. General experimental details are included in the supporting information. Ac-Pro-His-Cys-Lys-Arg-Met-OH (AcPHCKRM, 98%) was obtained from Peptide 2.0 Inc. (Chantilly, Virginia, USA). (+)-Pinoresinol was purchased from Separation Research Ab Oy, Turku, Finland, and phenylglycidyl ether (PGE) and hexylcinnamic aldehyde (HCA) (CAS 101–860) from Aldrich and were used as received. Acetone p.a. was purchased from Merck (Darmstadt, Germany) and olive oil from Apoteket AB (Gothenburg, Sweden). Unless otherwise indicated, reagents were obtained from commercial suppliers and used without further purification. Microwave reactions were carried out using a Biotage Initiator™ Sixty in 10–20 mL capped microwave vials with fixed hold time, normal or high absorption, and 10–30 sec pre-stirring. Column chromatography was performed using Merck silica gel Geduran Si 60 (0.063–0.200 mm) or using an automatic Biotage SP4 Flash+® instrument with prefabricated NH silica columns (50 μm irregular silica) (Biotage). TLC was performed using silica gel coated aluminum plates (Merck, 60 F254).
Synthesis of pinoresinol diglycidyl ether (PinoDGE) (Scheme 1) Synthesis of PinoDGE
a
.
a
Reagents and conditions: (±)-Epichlorohydrin (22 eq.), NaOH (4 eq.), EtOH, 80°C, microwave irradiation, 40 min, 88%.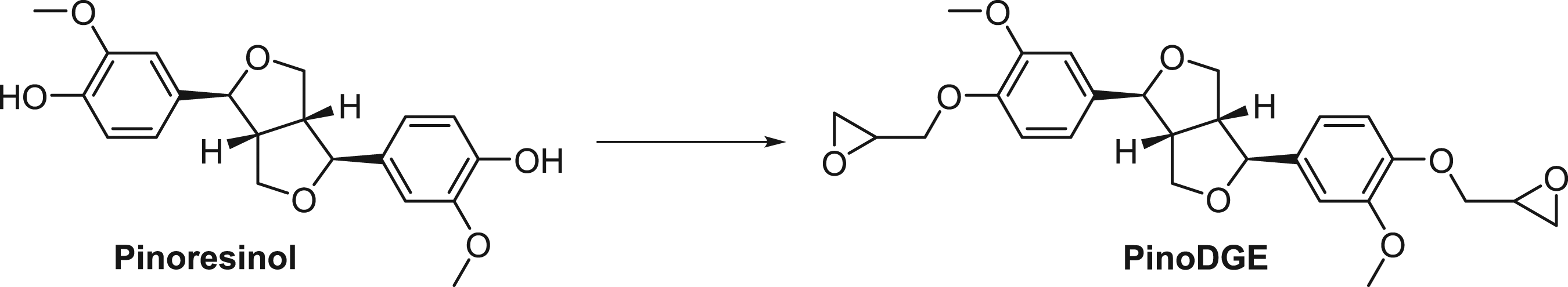
(±)-Epichlorohydrin (0.72 mL, 9.20 mmol) was added to a solution of (+)-pinoresinol (150 mg, 0.42 mmol) and NaOH (73 mg, 1.68 mmol) in EtOH (99.7%, 4 mL). The mixture was heated in a microwave cavity to 80°C for 40 min. The mixture was filtered and concentrated to yield colorless oil. The crude was purified by automated flash column chromatography on an NH column using dichloromethane as eluent to afford
Synthesis of 2-((2-methoxyphenoxy)methyl)oxirane ( Synthesis of Monoepoxides 1 and 2
a
.
a
Reagents and conditions: (i) (±)-Epichlorohydrin (1.5 eq.), K2CO3 (1.2 eq.), acetonitrile, reflux, 24 h, 47%; (ii) (±)-Epichlorohydrin (3.8 eq.), Cs2CO3 (1.9 eq.), acetonitrile, reflux, 3 h, 28%.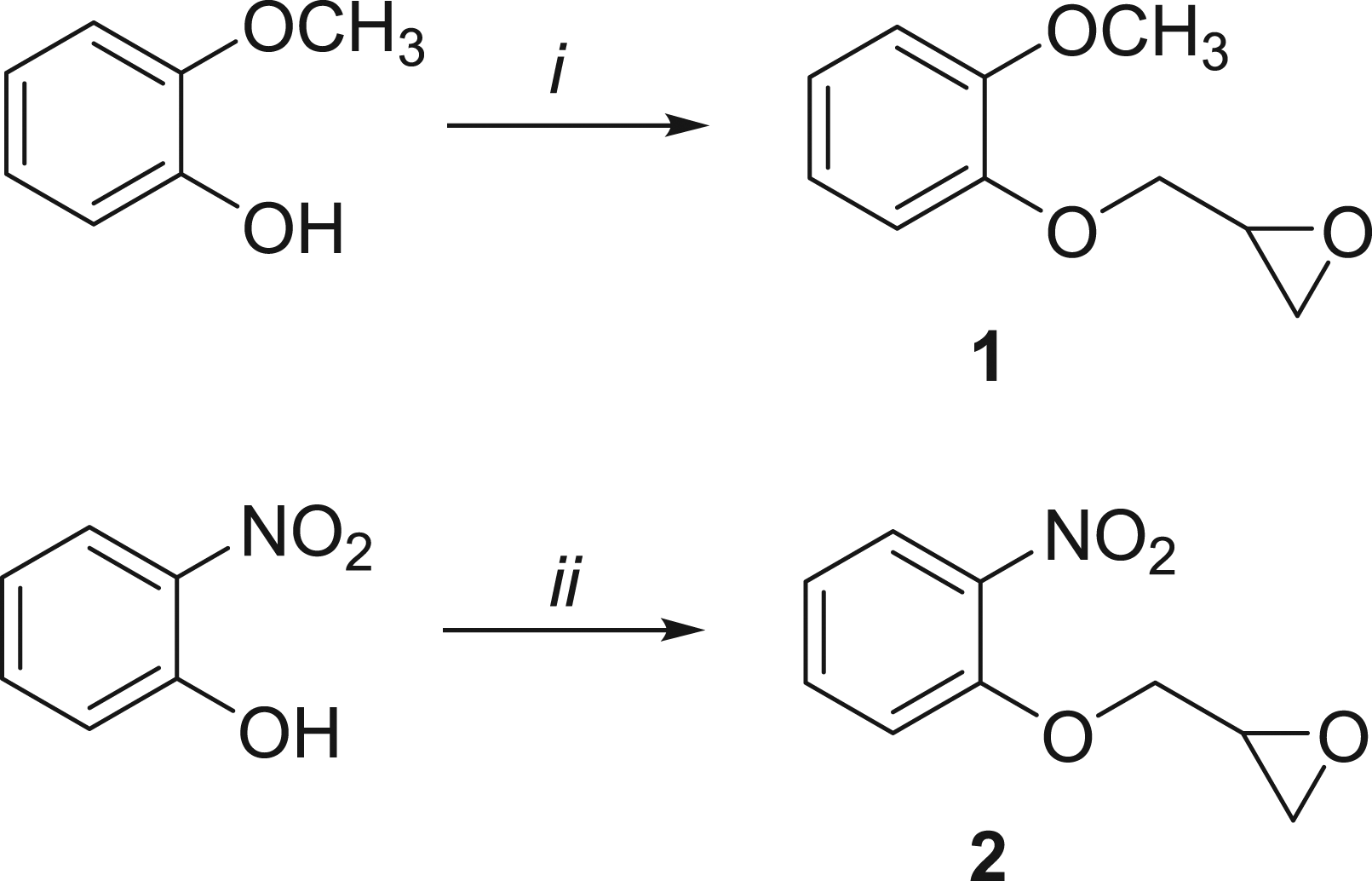
(±)-Epichlorohydrin (1.2 mL, 15 mmol) was added dropwise to a reaction mixture of 2-methoxyphenol (1.24 g, 10 mmol) and K2CO3 (1.66 g, 12 mmol) in freshly distilled acetonitrile (50 mL). The reaction mixture was refluxed under nitrogen atmosphere for 4 h and epichlorohydrin (1 mL) and K2CO3 (1.2 g, 8.7 mmol) were then added. The mixture was refluxed overnight. The reaction mixture was allowed to cool to room temperature, water (50 mL) was added, and the mixture was extracted with ethyl acetate (50 mL × 2). The solvent was evaporated and the mixture was purified by column chromatography (hexane: ethyl acetate 7:3) to give
Synthesis of 2-((2-nitrophenoxy)methyl)oxirane (
(±)-Epichlorohydrin (1.5 mL, 19 mmol) was added dropwise to a reaction mixture of 2-nitrophenol (0.69 g, 5 mmol) and Cs2CO3 (3.36 g, 9.5 mmol) in freshly distilled acetonitrile (90 mL). The reaction mixture was refluxed for 3 h under nitrogen atmosphere. After 3 h, the reaction mixture was allowed to cool to room temperature and filtered through Celite. The solvent was evaporated and the mixture was purified by column chromatography (hexane: ethyl acetate 7:3) to give
Reactions of the Epoxides DGEBA, PinoDGE, PGE,
KeratinoSens Assay for Sensitization and Cellular Viability. The KeratinoSens™ assay was performed according to the OECD guidelines (OECD, 2018). Compounds were incubated at 12 concentrations (0.98–2000 μ
Experimental Animals. Female CBA/Ca mice, 7 or 9 weeks of age, were purchased from NOVA SCB Charles River, Germany. The mice were housed in HEPA filtered airflow cages and kept on standard laboratory diet and water ad lib. The regional ethics committee, Jordbruksverket, approved the protocol and the procedure was performed in accordance with the guidelines. Sensitization Potential of PinoDGE in Mice. The murine local lymph node assay (LLNA) (Gerberick et al., 2007) was used to assess the sensitizing potency of PinoDGE (purity >99%), using a slight modification to the validated protocol to enhance the predictive capacity of the assay, using five groups of three mice (one group exposed to each concentration) and a further control group of four mice exposed to vehicle alone (Delaine et al., 2011). Computational Techniques. Reactivity of cysteine residues toward the compounds under investigation was modeled as reactivity toward methanethiolate. Reactivity calculations were carried out at the B3LYP-D3/6–31+G**(Stephens et al., 1994; Becke, 1988; Slater, 1974; Goerigk and Grimme, 2011; Grimme et al., 2010; Ditchfield et al., 1971) level of theory in Jaguar (Schrodinger LLC, N. Y. Jaguar, version 7.6, 2009). Further experimental details are contained in the Supplementary Information. Cross-Linking Procedure and Thermogravimetric Analysis (TGA)
Results
Chemical Synthesis. The diglycidyl ether of pinoresinol (PinoDGE) or structures derived from it are potential alternative ERMs for the construction of epoxy resin polymers. Bis-epoxide PinoDGE was synthesized following a previously reported procedure (Chen et al., 1994) (Scheme 1). In our case, microwave irradiation and purification using an amine column were efficacious in obtaining PinoDGE from pinoresinol and epichlorohydrin in high yield (88%). (+)-Pinoresinol was reacted with racemic epichlorohydrin, so there is potential for PinoDGE to form as a mixture of isomers.
Epoxides
Reactivity of PinoDGE, 1, and 2 toward the model peptide AcPHCKRM
Based on our previous studies regarding the chemical reactivity of the epoxide-containing compounds (O’Boyle et al., 2014; Ponting et al., 2019), the depletion of the nucleophilic hexapeptide Ac-Pro-His-Cys-Lys-Arg-Met-OH (AcPHCKRM) was analyzed in reaction mixtures at pH 7.4 with a 10-fold excess of the respective test chemical in a mixture of phosphate buffer and DMSO. Bis-epoxides DGEBA and PinoDGE showed similar reactivity profiles, but the results indicated that PinoDGE was more reactive than DGEBA. The largest depletion of peptide occured in the first 45 minutes for both bis-epoxides. DGEBA reacted rapidly with the peptide, with 37% of peptide remaining after 45 min, 18% after 85 min, and 7% after 205 min. PinoDGE depleted the peptide more rapidly, with only 8% of peptide remaining after 45 min. The amount of peptide remaining unreacted with PinoDGE after 85 minutes was only 5% (Figures 2(a) and (b)). A. and B. Depletion curves of peptide AcPHCKRM in presence of DGEBA (--●--) and PinoDGE (–■–); C. Depletion curves of peptide AcPHCKRM in the presence of PGE (--■--), 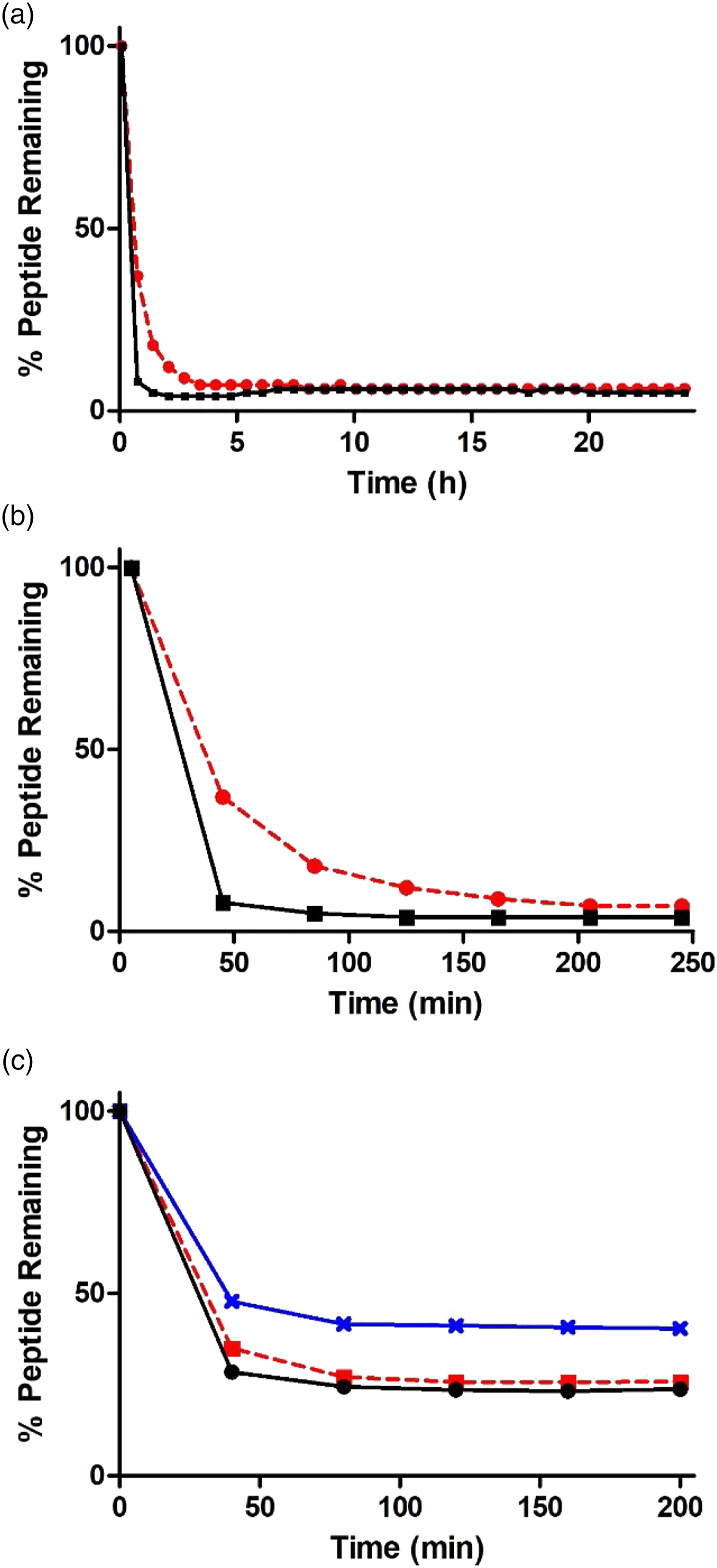
For the mono-epoxides, a decrease in reactivity between
Evaluation of compounds in the in vitro KeratinoSens Assay a .
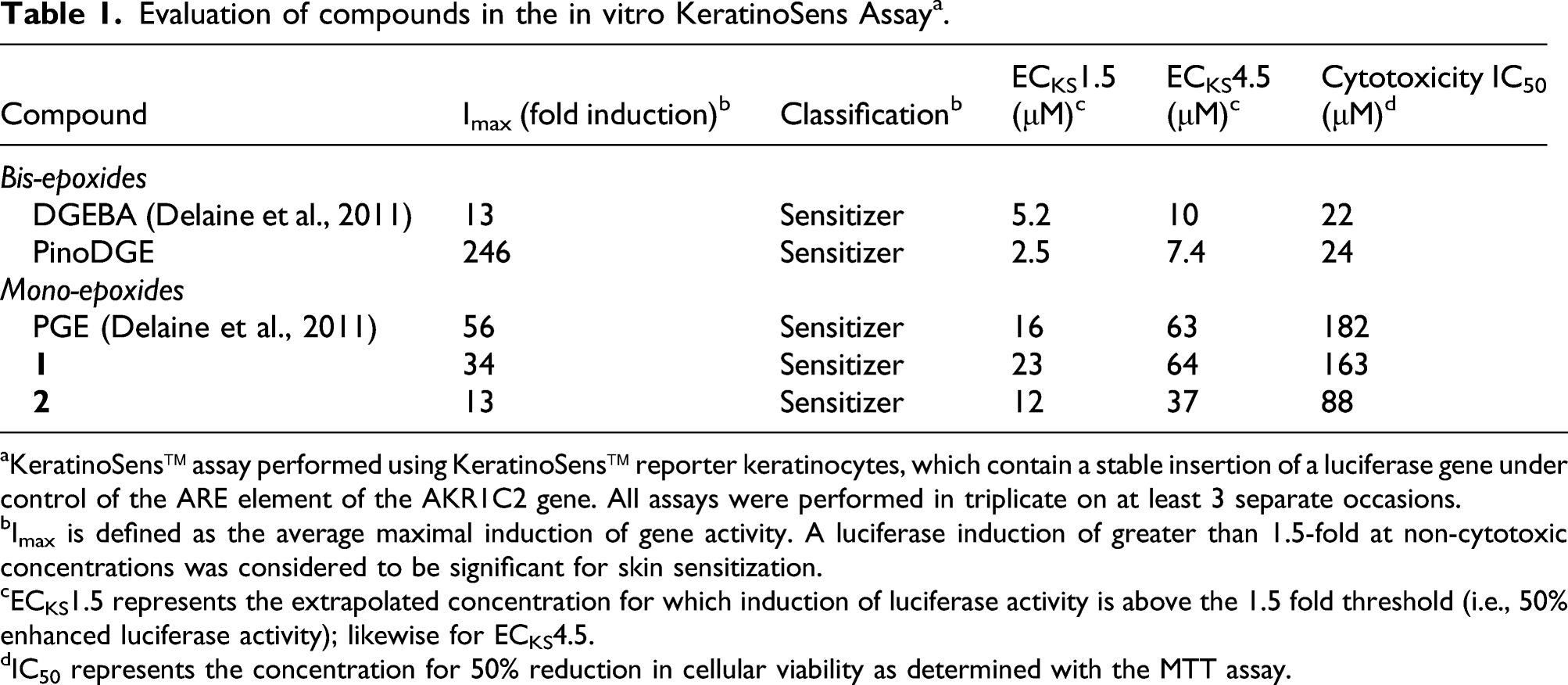
aKeratinoSens™ assay performed using KeratinoSens™ reporter keratinocytes, which contain a stable insertion of a luciferase gene under control of the ARE element of the AKR1C2 gene. All assays were performed in triplicate on at least 3 separate occasions.
bImax is defined as the average maximal induction of gene activity. A luciferase induction of greater than 1.5-fold at non-cytotoxic concentrations was considered to be significant for skin sensitization.
cECKS1.5 represents the extrapolated concentration for which induction of luciferase activity is above the 1.5 fold threshold (i.e., 50% enhanced luciferase activity); likewise for ECKS4.5.
dIC50 represents the concentration for 50% reduction in cellular viability as determined with the MTT assay.
Detailed results from the LLNA of pinoresinol diglycidylether (PinoDGE) a in AOO 4:1 and in AOO 16:1.
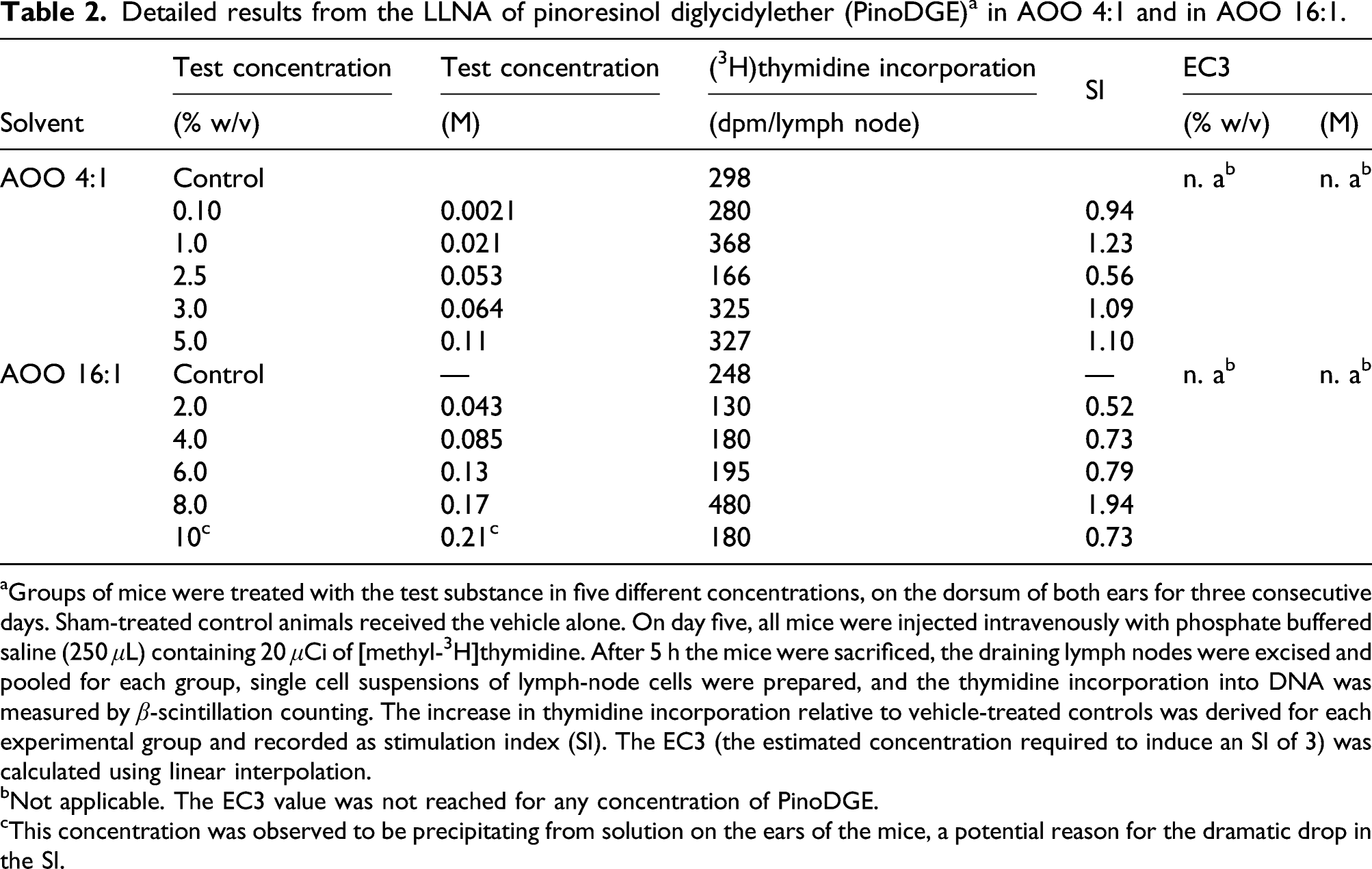
aGroups of mice were treated with the test substance in five different concentrations, on the dorsum of both ears for three consecutive days. Sham-treated control animals received the vehicle alone. On day five, all mice were injected intravenously with phosphate buffered saline (250 μL) containing 20 μCi of [methyl-3H]thymidine. After 5 h the mice were sacrificed, the draining lymph nodes were excised and pooled for each group, single cell suspensions of lymph-node cells were prepared, and the thymidine incorporation into DNA was measured by β-scintillation counting. The increase in thymidine incorporation relative to vehicle-treated controls was derived for each experimental group and recorded as stimulation index (SI). The EC3 (the estimated concentration required to induce an SI of 3) was calculated using linear interpolation.
bNot applicable. The EC3 value was not reached for any concentration of PinoDGE.
cThis concentration was observed to be precipitating from solution on the ears of the mice, a potential reason for the dramatic drop in the SI.
Computational Calculations of Chemical Reactivity. Chemical reactivity was calculated for the structurally simplified analogues PGE (comparable to DGEBA), Reactivity parameters (free energy of activation, ΔG‡, kJ/mol and ΔrG, free energy change of reaction, kJ/mol) for the attack of a model nucleophile on PGE, 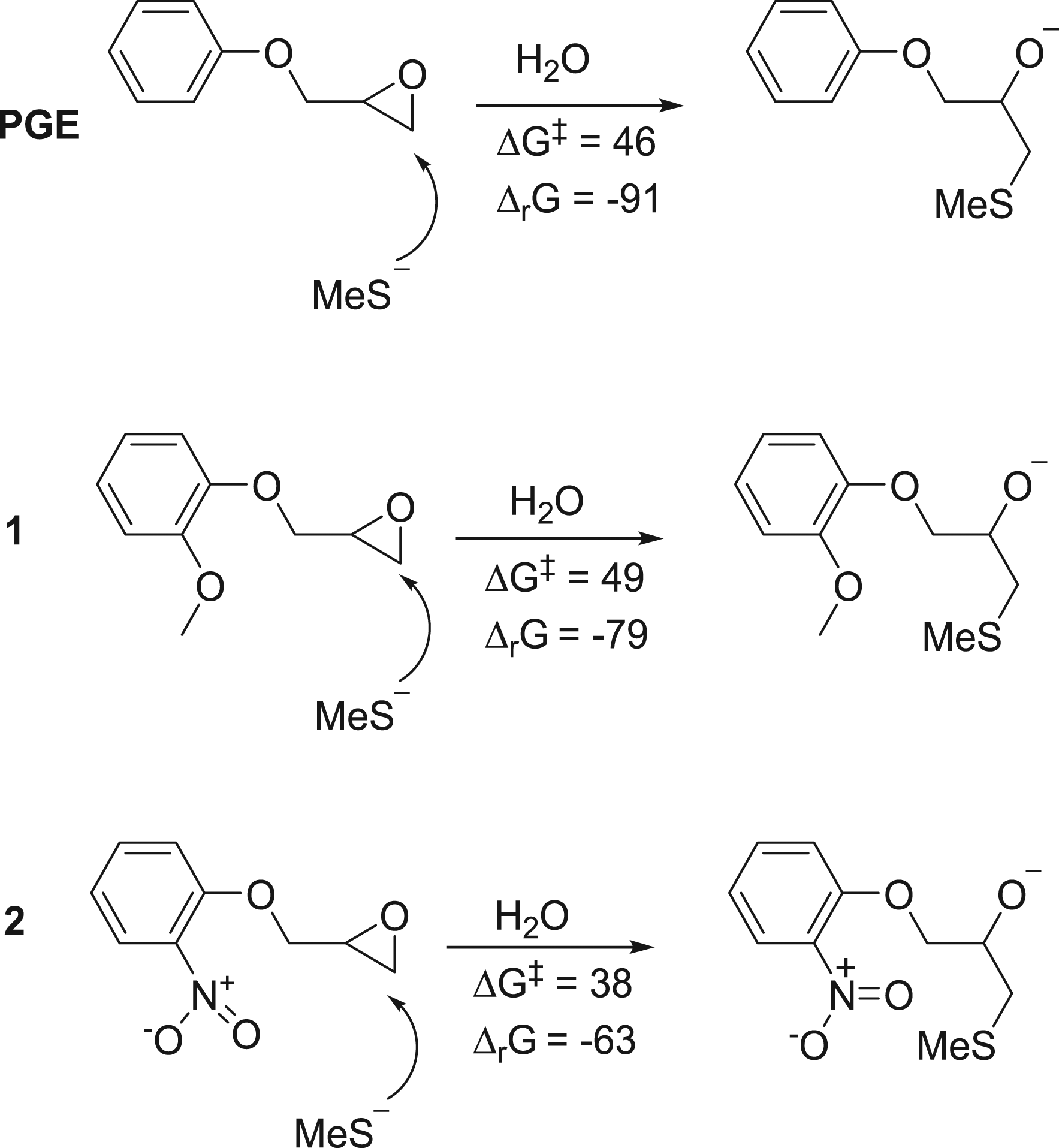
Thermal stability and degradation data of PinoDGE under nitrogen atmosphere determined by TGA.

a
b
c
d
eData taken from our previous publication.(O’Boyle et al., 2014).
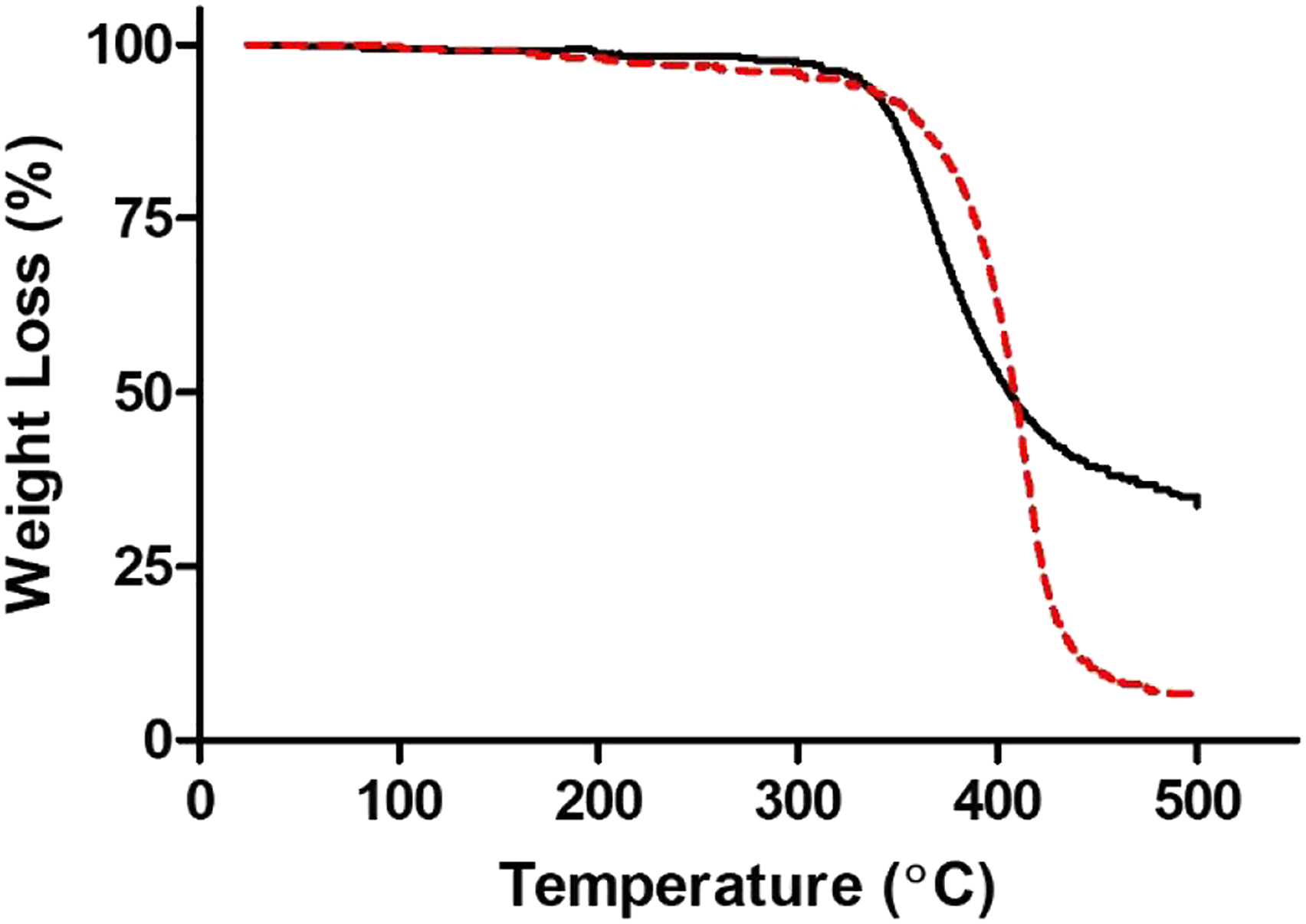
Thermogravimetric thermograms showing % weight loss at increasing temperatures of epoxy resins based on different ERMs in N2. DGEBA (dashed red line---) and PinoDGE (solid black line–). Data shown are the mean for three independent experiments.
Discussion
We here describe a novel nature-derived ERM PinoDGE, derived from the plant lignan pinoresinol. PinoDGE was synthesized in high yield for experimental investigations of its skin sensitizing and polymerization properties. Components of ERS are highly skin-sensitizing and are among the most common causative agents of occupational ACD. The most commonly used ERMs diglycidyl ether of bisphenol A (DGEBA, Figure 1) and diglycidyl ether of bisphenol F (DGEBF, Figure 1) are highly skin-sensitizing. We are committed to developing epoxy resins from natural materials and with reduced allergenic effect, while maintaining their excellent technical properties. The reactivity of the terminal epoxides of DGEBA is the basis of the polymerization process (O’Boyle et al., 2012). As the terminal epoxides also are the major cause of the skin-sensitizing effect, there is a delicate balance between skin allergy and polymerization properties (Ponting et al., 2019). However, by modification of the core structure, it has been possible to decrease the epoxide reactivity enough to maintain a favorable polymerization profile combined with a desirable reduction in skin-sensitization (O’Boyle et al., 2014). Based on this experience, we considered pinoresinol with two phenolic groups at opposite ends of the molecule separated by a rigid structure, suitable for modification.
The peptide reactivity of PinoDGE showed it to be chemically reactive. In vitro studies in cell culture according to the KeratinoSens method indicated a sensitization potential for PinoDGE. The threshold ECKS1.5 used for hazard identification is not always the best parameter for potency prediction and comparison. Based on our previous experience, we used instead the ECKS4.5 values for comparison between DGEBA and PinoDGE since they were more predictive for quantitative evaluation of the sensitization potency due to the wide dynamic range of the epoxides (Delaine et al., 2011). The results from the KeratinoSens assay predicted that PinoDGE and DGEBA had similar sensitizing capacities with ECKS4.5 values of 7.4 μM (PinoDGE) compared to 10 μM for DGEBA (Table 1). The skin sensitizing potency of PinoDGE was also evaluated in vivo in mice according to the LLNA method in two experiments with different vehicles and concentrations. None of the concentrations of PinoDGE tested gave an SI of greater than 3, indicating that the compound was not sensitizing at these concentrations. The result obtained in the second LLNA experiment was in accordance with the result obtained in the first experiment indicating an EC3 of PinoDGE >0.17
From the polymerization perspective, the IDT for polymers based on PinoDGE was 20°C lower than that of DGEBA and the Tmax was 12°C lower. Despite these small differences, the results indicated that the polymer obtained from PinoDGE shows promise and could also be investigated for mechanical strength and other material properties.
PinoDGE contains aromatic rings with ortho-methoxy substituents, which potentially could modify the reactivity of the terminal epoxides. Therefore, the monoepoxide PGE and structural analogues
Nitro-substituted epoxide
In conclusion, the present study showed a nature-derived alternative to the traditionally based DGEBA epoxy resins, which are common occupational skin sensitizers (contact allergens). Three different methods to determine the sensitization potential of the new chemical were used: peptide reactivity studies, cell studies using the KeratinoSens method, and the murine LLNA for in vivo studies. The data from peptide reactivity and KeratinoSens indicated a sensitization potential for PinoDGE. However, PinoDGE was much less sensitizing than DGEBA in in vivo testing, indicating a reduced sensitization potential. The results from cross-linking with TETA indicated that the polymer obtained from PinoDGE has promising properties when compared with DGEBA. PinoDGE is a lead compound for further development of alternative ERMs based on natural and renewable sources.
Supporting information available
The Supporting Information is available free of charge online. This includes general experimental details, experimental details for calculations, cross-linking procedure, and thermogravimetric analysis. Additional LLNA information (LLNA of the positive control substance hexyl cinnamic aldehyde); 1H and 13C NMR spectra of PinoDGE are also given.
Supplemental Material
Supplemental Material - Nature-derived epoxy resins: Synthesis, allergenicity, and thermosetting properties of pinoresinol diglycidyl ether
Supplemental Material for Nature-derived epoxy resins: Synthesis, allergenicity, and thermosetting properties of pinoresinol diglycidyl ether by Niamh M O’Boyle, Ida B Niklasson, David J Ponting, Miguel A Ortega, Tina Seifert, Andreas Natsch, Kristina Luthman and Ann-Therese Karlberg in Toxicology and Industrial Health
Footnotes
Acknowledgments
We thank Susanne Exing, Gabriella Wendt, and Anders Eliasson for assistance with the LLNA experiments. Bjarne and Thomas Holmbom, Åbo Akademi University, Turku, Finland and Romain Bordes, Chalmers Technical University, Gothenburg, Sweden, are acknowledged for valuable discussions.
Declaration of conflicting interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) received no financial support for the research, authorship, and/or publication of this article.
Supplemental material
Supplemental material for this article is available online.
References
Supplementary Material
Please find the following supplemental material available below.
For Open Access articles published under a Creative Commons License, all supplemental material carries the same license as the article it is associated with.
For non-Open Access articles published, all supplemental material carries a non-exclusive license, and permission requests for re-use of supplemental material or any part of supplemental material shall be sent directly to the copyright owner as specified in the copyright notice associated with the article.
