Abstract
Background:
Euxanthone is a plant-based flavonoid that is mostly isolated from a Chinese medicinal plant, Polygala caudate. This study was designed to evaluate the anticancer effects of euxanthone against human medulloblastoma cells.
Materials and Methods:
Cell viability was evaluated by CCK-8 and EdU assays. Apoptotic cell percentage was determined by annexin V/PI assay. The mRNA expression was determined by qRT-PCR. Wound healing and transwell assays were used to assess cell migration and invasion.
Results:
The results revealed aberrant activation of RANK/RANKL pathway in human medulloblastoma tissues and cell lines. Euxanthone suppressed the proliferation of the D425 medulloblastoma cells with comparatively lower cytotoxic effects against the normal human cerebellar granule cells. The IC50 of euxanthone against the D425 cells was found to be 10 µM. Interestingly, silencing of receptor activator of nuclear factor kβ (RANK) could also suppress the proliferation of the D425 cells via induction of apoptosis. Nonetheless, overexpression of RANK could abolish the cytotoxic effects of euxanthone on the D425 cells. Finally, wound-heal and transwell assay showed that euxanthone suppressed the migration and invasion of the D425 medulloblastoma cells.
Conclusion:
Collectively, the results revealed the anticancer effects of euxanthone against human medulloblastoma cells via RANK/RANKL pathway. These results suggest the potential of euxanthone as a lead molecule in the development of chemotherapy for medulloblastoma.
Introduction
Medulloblastoma is the most prevalent type of brain tumor among children. 1 This malignancy is quite lethal as medulloblastoma has been shown to exhibit a 5-year survival rate of less than 70%. 2 This signifies the inefficiency of currently applied therapeutic measures against medulloblastoma. The latter include the safe resection of the malignant tissues combined with adjuvant chemo- or radiation therapies. 3 Moreover, the patients surviving over longer periods often exhibit therapeutic side effects which include, but are not limited to, loss of neuro-cognition, impaired endocrine functioning, developmental disorders and development of secondary tumours.4, 5 Pertinent to this, there is a need for the formulation of safer and more efficient chemotherapeutic procedures against human medulloblastoma.
There is strong evidence that targeting the RANK/RANKL signaling pathway might emerge as a therapeutic target for the management of different types of human cancers. 6 This pathway involving the receptor activator of nuclear factor-kB ligand (RANKL) and its receptor (receptor activator of nuclear factor kβ [RANK]) has been shown to play a deciding role in bone-resorbing osteoclast differentiation, 7 RANK/RANKL pathway is reportedly dysregulated in several pathological molecular processes like osteoporosis in post-menopause stages of neoplastic bone destruction. 8 Human tumor cells exhibit significant up-regulation of RANK and RANKL which has also been shown to be important for maintaining their micro-environment. 9 Euxanthone is a plant-derived flavonoid nutraceutical that is usually extracted from a Chinese medicinal plant, Polygala caudate. 10 The former has been traditionally reported to be used against human ailments like anxiety and cough. 11 It was recently shown to possess therapeutic potential against human neurological disorders. 12 Moreover, euxanthone has been reported to exhibit anticancer effects against human cancer cells including glioblastoma cells. 13 This study was undertaken to evaluate the anticancer effects of euxanthone against the human medulloblastoma cells via RANK/RANKL signaling pathway.
Materials and Methods
Clinical Specimens
The collection of medulloblastoma and normal adjacent tissue samples was made from the patients who underwent surgery at the Children’s Hospital, Zhejiang University School of Medicine, National Clinical Research Center for Child Health, Hangzhou, Zhejiang, China. The patients were informed prior to the surgery and written consents were obtained from them before any tissue collection was made. The study was approved by the research ethics committee of the children’s hospital, Zhejiang University School of Medicine under approval number ZU/65HT/2021. Tissues were transported in liquid N2 cans and stored at −80°C till their experimental use.
Culture of Cell Lines
Two different human medulloblastoma cell lines (Daoy and D425) were purchased from the American Type Culture Collection (New York, USA). The human cerebellar granule cells (HCGC) were ordered from 3H Biomedical Company. The cell lines were cultured in Dulbecco’s modified Eagle’s medium (DMEM; Thermo Fisher Scientific, Waltham, MA, USA) carrying the supplementation of 10% fetal bovine serum (FBS; Gibco Inc.) and 100 U/mL penicillin/streptomycin (Hyclone Inc.). The humidified CO2 incubator was used to maintain the cell lines at 37°C with 5% CO2.
Transfection
The si-RANK and negative control (si-NC) as well as the pcDNA-RNAK were purchased from the RiboBio Co., Ltd. (Guangzhou, Guangdong, China). Lipofectamine™ 3000 reagents (Thermo Fisher Scientific) were used to transfect the D425 medulloblastoma cells with 100 nM si-RANK or si-NC oligos according to the manufacturer’s guidelines. Cell harvesting was carried out within 24 hr of transfection.
Quantitative RT-PCR
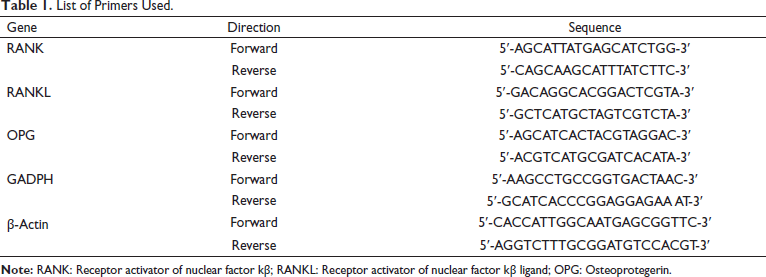
The total RNA was isolated from tissues and cell lines with the help of TRIzol reagent (Thermo Fisher Scientific). Revert Aid First-strand cDNA synthesis kit was used for reverse transcription of RNA (Thermo Fisher Scientific). Using Power SYBR Green PCR master mix (Thermo Fisher Scientific), the quantitative real-time polymerase chain reaction (qRT-PCR) was performed on QuantStudio 3.0 RT-PCR system (Applied Biosystems). The cycling conditions were as follows: 95°C for 20 sec, followed by 40 cycles of 95°C for 15 sec, and 58°C for 1 min. The 2−∇∇Ct method was used for quantifying the relative gene expression levels with GADPH and β-actin genes as endogenous control. The RT-PCR primers used in this study are listed in Table 1.
List of Primers Used.
Cell Counting Kit 8 (CCK-8) Assay
Cell viability was analyzed with the help of CCK-8 kit (Dojindo, Kumamoto, Japan) as per the manufacturer’s method. In brief, the cells were placed at an initial density of 5 × 104 per well of a 96-well plate. Following incubation for varying intervals, 15 µL of CCK-8 solution was added to each well and 37°C incubation was prolonged for another 2 hr. The cells were then harvested and resuspended in 200 µL of DMSO. Finally, the absorbance at 570 nm was recorded for each well with the help of a microplate reader (Bio-Rad, CA, USA).
EdU Incorporation Assay
The EdU analysis kit (RiboBio, Guangzhou, China) was used to assess the proliferative viability of D425 cells. Briefly, the cells were seeded into the 12-well plates and cultured for 24 hr. Next, the wells were added with EdU reagent. After 30 min, the cells were harvested, fixed in 70% ethanol and treated with 0.5 % Triton-X-100 (Sigma-Aldrich). For counterstaining, the nuclei were stained with DAPI (Sigma-Aldrich). Finally, fluorescent microscopy (Olympus, Tokyo, Japan) was used for examining the staining and the relative percentage of EdU-positive cells was determined.
Annexin V-FITC/PI Staining Assay
For Flow cytometry-based apoptosis analysis, the cells were digested with trypsin and then harvested. About 100 µL binding buffer was used for suspending the cells followed by the addition of 5 µL of each of Annexin V-FITC and propidium iodide (PI). After incubating for 15 min at room temperature, the cells were examined through flow cytometry (BD, USA).
Wound Healing Assay
Medulloblastoma D425 cells were seeded in a 6‑well plate at the density of 5 × 105 cells/well. At confluence, the cell monolayer was scraped off and maintained in RPMI‑1640. After 24 hr of incubation, the images were taken using an inverted microscope (magnification ×100).
Transwell Assay
The transwell chambers with 8-µm pores and transwell membranes pre-coated with Matrigel (BD Bioscience) were obtained from Corning. The D425 medulloblastoma cells were collected and suspended in RPMI with 10% FBS at concentrations of 1 × 105 cells (0.5 mL). The cells were subsequently placed in the upper chamber of a 24-well plate. RPMI media (1.6 mL) having 10% FBS was placed in the lower chamber. At the end of the procedure, D425 cells that invaded the lower side were washed, fixed with methanol, stained with Giemsa, which had invaded the reverse side of the transwell membrane, and finally observed under an inverted light microscope.
Statistical Analysis
The data are presented as mean ± standard deviation (SD). The student’s t-test was performed to analyze the difference between the two treatments. p values less than 0.05 were considered to represent a statistically significant difference. The GraphPad Prism 7.0 offline software (GraphPad Software, La Jolla, CA, USA) was used for statistical analysis.
Results
Overexpression of RANK/RANKL Pathway in Medulloblastoma
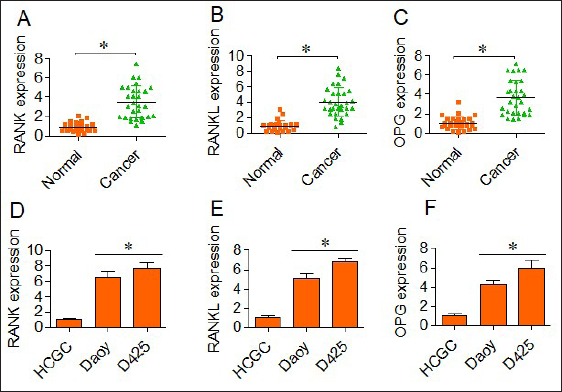
Whether medulloblastoma shows dysregulation of RANK/RANKL/OPG pathway, the expression analysis of signaling components namely the RANK, RANKL and osteoprotegerin (OPG) was examined. The qRT-PCR results showed that RANK, RANKL and OPG are significantly upregulated in medulloblastoma tissues in comparison to the normal adjacent tissues (Figure 1A−C). The RANK, RANKL and OPG protein expression was also examined in the medulloblastoma cell lines (Daoy and D425) and HCGC, normal HCGCs. The results showed that the expression of all these proteins was upregulated in medulloblastoma cell lines relative to that of the HCGC cells (Figure 1D−F). Together, the results are indicative of aberrant activation of RANK/RANKL pathway in human medulloblastoma.
Euxanthone Inhibits Medulloblastoma Proliferation by Suppressing RANK/RANKL Pathway
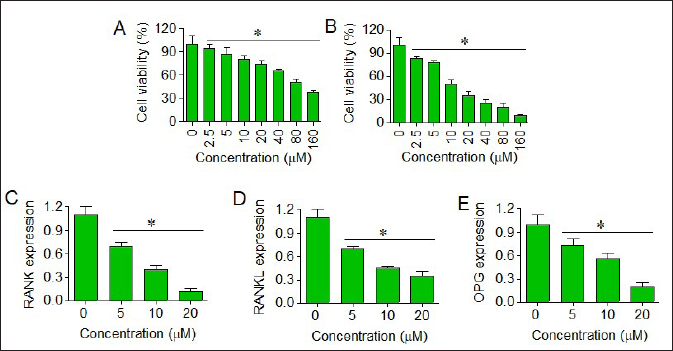
The human medulloblastoma cell line (D425) along with HCHC cells was administered with different concentrations of euxanthone (0–160 µM) and the effect on respective cell viabilities was evaluated by performing the CCK-8 assay. HCGC cells showed dose-dependent loss of viability by euxanthone administration with an estimated IC50 of 145 µM (Figure 2A). Interestingly, the viability of the D425 medulloblastoma cells was shown to be affected more severely by euxanthone than HCGC cells and the IC50 of euxanthone was estimated to be 18 µM against D425 cancer cells (Figure 2B). Moreover, the D425 cells treated with 0, 5, 10 or 20 µM euxanthone were shown to exhibit dose-dependent repression of RANK, RANKL and OPG genes at the transcriptional level by euxanthone (Figure 2C–E). Taken together, the results were suggestive that euxanthone selectively inhibits the viability of human medulloblastoma cells through RANK/RANKL pathway repression.
Euxanthone Exerts Antiproliferative Effects by Targeting RANK
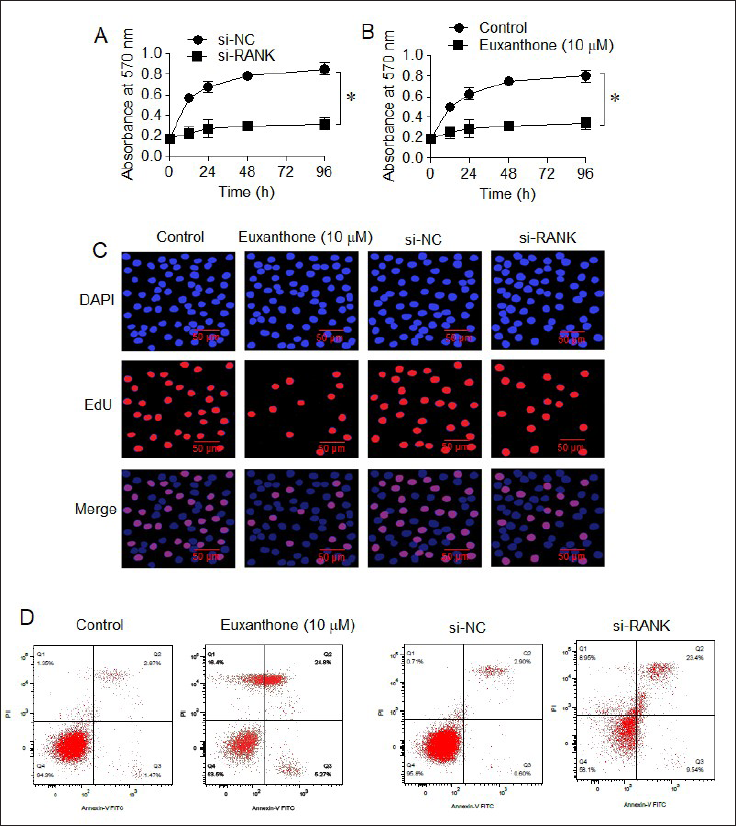
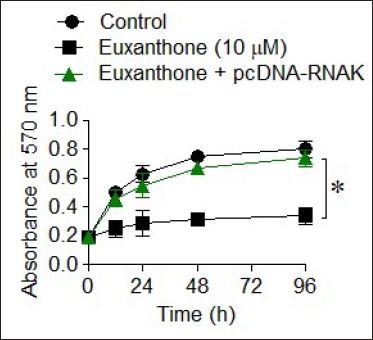
To further characterize the mechanism of the antiproliferative action of euxanthone against the medulloblastoma cells, D425 cells were first incubated without or with 10 µM euxanthone and their proliferation was assessed at 0, 12, 24, and 96 hr of incubation. As expected, the results showed that D425 cells treated with euxanthone suffered a significant decline in proliferation in vitro with reference to control untreated cells at different intervals of growth (Figure 3A). Noteworthy, the knockdown of RANK gene in D425 was shown to imitate the anti-proliferative effect of euxanthone against the D425 medulloblastoma cells (Figure 3B). Moreover, both the euxanthone administration (10 µM) and silencing of RANK were shown to diminish EdU incorporation in D425 cells with respect to the corresponding negative control cells (Figure 3C). The Annexin V-FITC/PI staining followed by flow cytometry confirmed that apoptosis was induced in D425 cells by both the euxanthone treatment and RANK knock-down (Figure 3D). Interestingly, overexpression of RNAK could abolish the anti-proliferative effects of euxanthone on D425 cells (Figure 4). Collectively, the results signify that euxanthone-induced apoptosis in medulloblastoma cells by targeting RANK gene to inhibit their proliferation, in vitro.
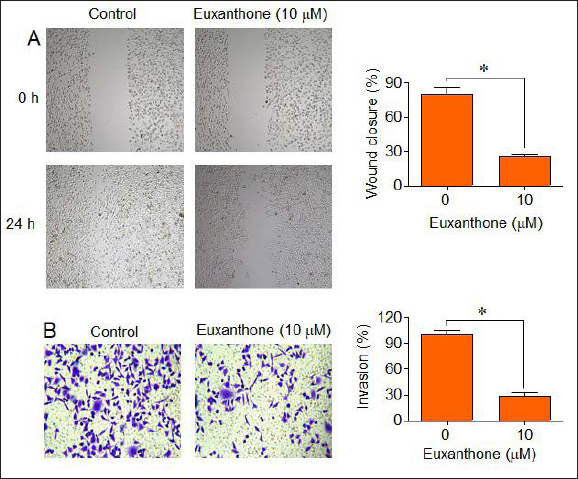
Euxanthone Inhibits the Migration and Invasion of Medulloblastoma Cells
The effects of euxanthone were also evaluated on the migration and invasion of the D425 cells. The results of the wound heal assay showed that the wound closure was 81% in control as compared to 25% at 10 uM euxanthone (Figure 5A). These results indicate that euxanthone inhibits the migration of the D425 cells. Moreover, the results of the transwell invasion assay showed that the D425 cells were inhibited by 72% (Figure 5B). Thus, these results suggest that euxanthone inhibits the migration and invasion of the D425 medulloblastoma cells.
Euxanthone Inhibits the Migration and Invasion of the Medulloblastoma Cells. (A) Wound Healing Assay Showing Effects of Euxanthone on the Migration of D425 Cells. (B) Transwell Assay Showing Effects of Euxanthone on the Invasion of D425 Cells.
Discussion
The cancer cells evade the normal program of cell death, thus exhibiting prolonged survival and proliferating at much-elevated levels. 14 At the molecular level, the malignant behavior of human cancer cells is attributed reportedly to the anomalous expression of multiple signaling pathways. 15 Exploration of the aberrantly expressed pathways in cancer cells and their possible subsequent restoration might prove vital in our fight against the devastating disease of human cancer. The RANK/RANKL signaling pathway is over-active in different types of human cancers such as breast cancer and colorectal cancer.16, 17 The molecular triad RANKL, RANKL-receptor (RANK) and decoy receptor, OPG influence diverse cellular, physiological and pathological processes. 18 The over-expression of RANK, RANKL and OPG has been shown to frequently surface the tumor micro-environments and dominantly affect the process of human tumorigenesis. 19 The over-expression of RANK, RANKL and OPG was reported from the medulloblastoma tissues and cell lines in comparison to the corresponding normal tissues and cells in the present study. Of interest, the medulloblastoma cells treated with euxanthone showed significant repression of RANK, RANKL and OPG at both transcriptional and translational levels. Moreover, euxanthone selectively inhibited the proliferation of medulloblastoma cells while it showed minimal effect on normal human neural cells. This suggests the potential utility of this compound to serve drug development in future against the human medulloblastoma. Previous studies have also made similar inferences regarding the selective cytotoxic effects of euxanthone against human cancer cells such as osteosarcoma cells and colorectal cancer cells.20, 21 The silencing of RANK gene mimicked the anti-proliferative effects of euxanthone against the medulloblastoma cells, in vitro and overexpression of RANK nullified the antiproliferative effects of euxanthone which confirmed that euxanthone targeted RANK/RANKL pathway at the molecular level to inhibit the growth of medulloblastoma cells. Moreover, the euxanthone-driven inhibition of RANK/RANKL signaling pathway in medulloblastoma cells was found to trigger the induction of apoptotic cell death. The apoptosis-inducing potential of euxanthone has already been established against human cancer cells.21, 22
Conclusion
Collectively, the results of this study indicate that medulloblastoma exhibits significant over-expression of RANK/RANKL pathway. Euxanthone blocked the RANK/RANKL pathway to inhibit the growth and metastasis of medulloblastoma cells by inducing cell apoptosis. Thus, euxanthone might serve as a lead molecule in drug development against medulloblastoma and its efficacy might be enhanced through semi-synthetic chemistry approaches.
Footnotes
Abbreviations
CCK-8: Cell counting kit 8; qRT-PCR: Quantitative real-time polymerase chain reaction; FBS: Fetal bovine serum; PI: Propidium iodide.
Acknowledgment
We acknowledge the Children’s Hospital, Zhejiang University School of Medicine, National Clinical Research Center for Child Health, Hangzhou, Zhejiang, China, for providing laboratory support.
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethics Approval
The study was approved by the ethics committee of Zhejiang University School of Medicine, National Clinical Research Center for Child Health, Hangzhou, Zhejiang, China.
Informed Consent
The patients were informed prior to the surgery and written consents were obtained from them before any tissue collection was made.
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Summary
Euxanthone inhibits proliferation of medulloblastoma cells via induction of apoptosis.
Euxanthone blocks RANK/RANKL pathway in medulloblastoma cells.
Euxanthone suppresses migration and invasion of medulloblastoma cells.
