Abstract
Background:
The fruit of Schinus terebinthifolia Raddi is used in ethnomedicine for the treatment of different ailments.
Materials and Methods:
We studied the phytochemical profile of S. terebinthifolia fruit and evaluated its properties, including its antioxidant, anti-inflammatory, analgesic, cytotoxic and antimicrobial activity.
Results:
Phytochemical investigation of S. terebinthifolia fruit showed an estimated total phenolic content of 67 mg of gallic acid equivalent (GAE)/g, with respect to gallic acid, while the flavonoid content was 26 mg of quercetin equivalent (QE)/g, in terms of quercetin. Bioassay fractionation of methanolic extract of the fruit resulted in the isolation of a new molecule for the first time from this natural source: 4′,methoxy-6-vinyl-7-O-rhamnosyl-dihydromyricetin or 4′,methoxy-6-vinyl-7-O-rhamnosyl-ampelopsin (Compound 8). Seven other phenolic compounds were isolated, such as (1) gallic acid; (2) afzelin; (3) quercetin 3-O-β-
Conclusion:
The new compound shows immunomodulatory effects that can be utilized to augment host responses, which is important in both cancer and infectious diseases and thus worth further pharmacological investigation.
Keywords
Introduction
Schinus terebinthifolia is a species of the genus Schinus that belongs to the Anacardiaceae family and is native to South America, Central America and Africa. It also exists in Egypt as an ornamental tree known for its decorative foliage and edible fruits. This plant has been used widely for different purposes, such as dyeing, tanning, and decoration as well as a common spice. 1 In traditional medicine, different portions of S. terebinthifolia, including the roots and the leaves, are used for the treatment of diseases including hypertension, ulcers, diarrhea, gastroduodenal disorders, urinary and respiratory problems, cancer, arthritis, skin problems and mucous membrane injuries. In addition, it is traditionally used in the treatment of sexually transmitted diseases and endometriosis, and it has been found to combat microorganism growth, oxidative stress and inflammation. 2
The genus Schinus is rich in different chemical classes; for example, volatile oils (α- and β-pinene and germacrene are major components of the fruit), flavonoids, tannins, coumarins and sterols.3-6 Several scientific studies have brought out the biological importance of this plant, such as its antioxidant, antimicrobial, antiallergic, anticancer, and insecticidal properties.7-10
Given the need to discover potential new immunomodulators, and the potential of S. terebinthifolia to be used for this purpose, we here document the isolation, characterization and structural profiling of a new compound from the fruit, in addition to presenting data showing the in vitro anti-inflammatory, antioxidant, antimicrobial and cytotoxic properties of the plant.
Materials and Methods
Extraction and Isolation
After air drying the plant materials and grinding them, 400 g of powder of S. terebinthifolia was immersed and refluxed in hot 80% EtOH to achieve exhaustive extraction. The 60 g of obtained residue was extracted using chloroform and the 55 g of aqueous residue was separated using a polyamide column, with elution with water and water/methanol, to obtain several fractions. After concentrating the collected fractions under pressure, they were purified using sephadex LH-20 and/or cellulose columns.
Instruments and Materials
One-dimensional (1D) and two-dimensional (2D) (1H- and 13C-) NMR spectra were obtained from Varian Mercury (500 MHz and 125 MHz for 1H and 13C, respectively). Chemical shifts (δ) were shown as parts per million (ppm) and J-values are given in Hz. UV analysis of solutions in methanol with various UV shift reagents was recorded on a Shimadzu UV 240 spectrophotometer. The negative ESI Mass spectra were recorded on Agilent.
Phenolic Content Assay
The whole amount of phenolic content in the extract was measured using the Folin Ciocalteu colorimetric method, as outlined previously. 11 Ethanolic extract was diluted to a final concentration (1 mg/mL) and a calibration curve of gallic acid was constructed over a range of 0.5 to 2.5 mg/mL. About 10 mL of each stock solution and standard was mixed with Folin Ciocalteu reagent and water (10 mL) and then diluted to 25 mL using sodium carbonate solution. The absorbance level was detected after 30 min at 730 nm, and then at 760 nm, and the whole amount of phenolic content was reported as g/100 g gallic acid equivalent (GAE).
Flavonoids Content Assay
The total flavonoid amount in the sample was evaluated as previously described. 12 Ethanolic extract was prepared to an initial concentration (1 mg/mL). After evaporating 1 mL in a test tube, 5 mL of 0.1 M aluminum chloride was added. The yellow color produced was measured at 415 nm, where its intensity is proportional to the flavonoids, due to the complexes with aluminum chloride. The whole flavonoid amount was recorded as mg of quercetin equivalent (QE)/g dry matter from herbs.
Animals
Albino mice of both sexes and 25 to 30 g body weight were used to determine the median lethal doses (LD50) for antioxidant, anti-inflammatory and analgesic activity.
Determination of Median Lethal Dose (LD50)
In order to determine the LD50, graduated amounts between 100 mg and 3 g of the dried extract/kg by weight were reconstituted in 1 mL saline and injected subcutaneously into the mice, which were divided into three groups of six. For 24 hr after this, any clinical signs of toxicity or mortality in the animals were noted. Determining the LD50 of the extract in this way meant that fewer animals were needed for the experiment. 13
Antioxidant Activity
Antioxidant activity was evaluated and compared with that of ascorbic acid as standard, by applying the previously described method to determine glutathione levels in alloxan-induced diabetic rats’ blood. 14 A single intraperitoneal injection of alloxan in phosphate-buffered saline (PBS) (100 mg/kg body weight) was used to artificially induce diabetes in adult male albino mice. 15 The diabetic rats were divided into four groups: untreated animals, those treated with the reference drug (ascorbic acid, 10 mg/kg body weight), those treated with aqueous extracts and those treated with alcoholic extracts of S. terebinthifolia fruit. Doses of 100 mg/kg body weight were administered orally at regular intervals of 12 hr for a total of 28 days. Later, blood glutathione levels were determined using blood glutathione tests from Wak-Chemie Medical, Germany, as directed by the manufacturer.
Anti-inflammatory Activity
Anti-inflammatory activity was measured using the mice paw edema method. 16 Male albino mice were classified into four groups of six animals, the untreated group receiving 1 mL of saline, one of the treated groups receiving 20 mg/kg of the reference drug (indomethacin) and the other two treated groups receiving 100 mg/kg and 300 mg/kg body weight of the extract, respectively. After 1 hour, all animals were injected with 1% carrageenan solution in saline (0.1 mL) in the right hind paw and saline (0.1 mL) in the left hind paw. Four hours after drug administration, both hind paws of sacrificed rats were excised and weighed separately.
Analgesic Activity
The acetic acid-induced writhing method was applied to evaluate the analgesic effect of the extract. 17 Twenty-four mice were divided into four groups of six animals each, the control group receiving acetic acid (1 mL), the positive control treated group receiving 20 mg/kg body weight of the reference drug (indomethacin) and two treated tested groups receiving 100 mg/kg body weight of the aqueous and alcoholic extract orally, respectively. Later after 30 min, 0.6% acetic acid was injected intraperitoneally (0.2 mL/mouse) and the total number of writhes/30 mins was counted for each mouse.
Antimicrobial Activity
The inhibitory effect of the alcoholic extract on the microbial growth of Staphylococcus aureus, Escherichia coli, and Candida albicans, obtained from the National Committee for Clinical Laboratory Standards (NCCLS), Cairo, Egypt, was determined by applying the modified disc diffusion method. 18 Microbial suspensions were prepared and spread onto agar plates, in addition to standard discs of positive controls (tetracycline and amphotericin B). Negative controls were also used as filter discs with 10 µL of various solvents (distilled water, chloroform, DMSO) and another filter disc loaded with the tested sample were placed on agar. Plates with Gram +ve bacteria S. aureus and Gram −ve bacteria E. coli were cultured at 35 to 37°C for 24 to 48 hr and yeast in the form of C. albicans was incubated at 30°C for 24 to 48 h. Following this, the diameters of the inhibition zones around the disc were measured.
Proliferation of Immune Cells
The effect of the extract and the isolated compound on the growth of raw macrophage 264.7 was evaluated by the 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide (MTT) assay. 19 The tetrazolium salt was metabolized by mitochondrial dehydrogenase if cells were metabolically active, to form insoluble detected formazan crystals (purple). Around 5×104 cells were seeded and incubated at 37°C with a range of concentrations of the compounds in an FBS-free medium. After 48 hr of incubation, the MTT solution was added to the 96 wells in the plate, the cells were then incubated, and solubilizing agent was added. The absorbance was measured with a plate reader at 570 nm to detect any change in the color of added reagent. The percentage of cell viability was determined by changes detected in the amount of formazan salt, and data were expressed as the percentage of live cells as compared to DMSO-treated cells. A 1000 U/mL recombinant macrophage colony-stimulating factor was applied as a positive control in the experiment.
Cytotoxic Activity Against Breast Carcinoma Cell Lines
The tested cell line was a human breast cancer cell line (MCF-7) cultured in RPMI 1640 medium containing 2 mM L-glutamine that was supplemented with 10% fetal bovine serum, 100 units/mL penicillin G sodium, 100 units/mL streptomycin sulphate and 250 ng/mL amphotericin B, at 37°C in an incubator, with 5% CO2. For sub-culturing, monolayer cells of the third or fourth generation were harvested when confluence had reached 75%, after examination under an inverted microscope, and after trypsin/EDTA treatment at 37°C. Around 5×10 4 cells/well in fresh medium cells were cultured in a multiwell plate and left overnight for attachment. Then, the seeded cells were treated in triplicate with a range of concentrations of 6.25 to 50 µg/mL of the extract solution prepared in DMSO. The cultured cells were incubated for 18 h at 37°C and the cell concentration was determined by counting the MCF-7 cells using a hemocytometer after the addition of trypan blue. 20
Statistical Analysis
Data are reported as the mean results of three individual experiments. Results were considered significant when a p value of <0.05 was obtained as there is statistical significance between the treated and control groups. GraphPad Prism 9.2.0 was used (GraphPad, San Diego, CA, USA).
Results
Phytochemical Results
The total polyphenol content was found to be the major constituent in addition to the flavonoids of the fruit. The phenolic content was determined to be 67 ± 2.43 mg of GAE/g, using the Folin Ciocalteu colorimetric method, with gallic acid as a reference. The whole flavonoid content was determined to be 26 ± 3.52 mg of QE/g of the dry material, by measuring the strength of the developed color of the flavonoid complex, using quercetin as a reference. The results obtained in this study were compared with several other reports on the total phenolic content of this plant, such as one in which the leaf was reported to contain phenol and flavonoid amounts of 145.45 ± 4.5 mg GAE/g, 12.46 ± 1.6 QE/g of extract, respectively. 14
Seven phenolic compounds (Figure 1) were isolated from the aqueous-alcoholic fruit extract and were identified using different spectroscopic techniques. Their structures were confirmed by comparing our findings with that of previously published research.15, 21-26 In addition, our research has resulted in a new compound being isolated for the first time from this plant. It is here termed Compound 8.
Structures of Isolated Compounds from Fruits of S. terebinthifolia: (1) Gallic Acid; (2) Afzelin; (3) Quercetin 3-O-β-d -glucopyranoside; (4) Genistein 7-O-α-l -Rhamnopyranoside; (5) Ellagic Acid; (6) Kaempferol and (7) Quercetin.
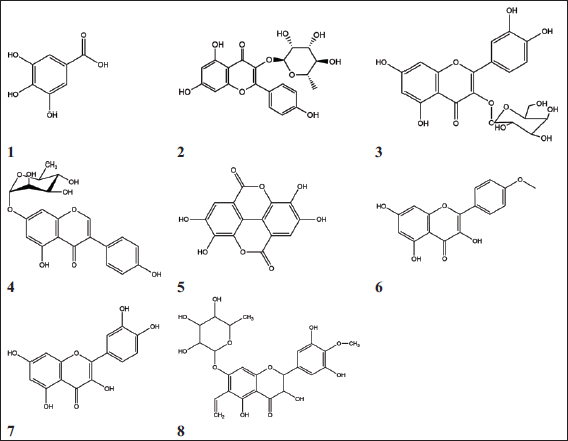
Compound 8
Compound 8 was obtained as a pale yellowish brown amorphous powder (9 mg) showing a number of chromatographic properties: Rf values; 0.33 (S1), 0.51 (S2); dark purple spot under UV light showed a green color with FeCl3 and reddish-orange with Naturstoff’s spray reagents. UV–spectral data λmax (nm) (MeOH): 225, 285, 302; (+NaOMe): 260, 301, 320; (+NaOAC): 225, 284, 303; (+AlCl3): 277, 303(sh), 347; (+AlCl3/ HCl): 278, 303(sh), 345, mp 226-230°C.
In the 1H-NMR (500 MHz, DMSO-d6) spectrum of Compound 8, the proton signals of the compound were determined at 4.58, d, J = 1.4 Hz (1H-2), 4.50, d, J = 1.4 Hz (1H-3), 6.35, s (1H-8), 6.56, s (1H-2′/6′), 5.59, d, J = 1.5 Hz (1H-1″), 3.33, d, J = 1.5 Hz (1H-2″), 3.14, d, J = 1.5 Hz (1H-3″), 3.15, t, J = 1.7 Hz (1H-4″), 3.16, t, J = 1.7 Hz (1H-5″), 1.43, d, J = 1.4 Hz (3H-6″), 6.59, t, J = 11 Hz (1H-1′″), 4.91 and 5.92, d, J = 11 Hz (exomethine 2H-2′″), 3.84, s (3H, OMe-4′).
13C-NMR (125 MHz, DMSO-d6) peaks were detected at δ ppm: 86.03 (C-2), 79.64 (C-3), 188.76 (C-4), 159.18 (C-5), 104.16 (C-6), 163.61 (C-7), 97.14 (C-8), 134.02 (C-1′), 108.23 (C-2′), 151.85 (C-3′), 137.28 (C-4′), 159.18 (C-9), 151.85(C-5′), 108.23(C-6′), 104.16 (C-10), 102.35 (C-1′′), 72.04(C-2′′), 70.54 (C-3′′), 73.81 (C-4′′), 70.52(C-5′′), 17.13 (C-6′′), 115.75 (C-1′′′), 128.03 (C-2′′′), 60.68 (OMe-4′).
Negative ESI-MS: m/z 319.2471 (ampelopsine-H), 345.1243 (vinyl ampelopsin-H), 359.2431 (methoxy, vinyl ampelopsine-H), 491.1281 (rhamnosyl-vinyl-ampelopsine), 505.1253 (methoxy, vinyl, rhamnosyl, ampelopsine-H), 1011.3445 (Dimer-H).
This compound was expected to be 4′-methoxy, 7-glycosyl ampelopsin and the UV spectrum in MeOH showed two distinctive bands at λmax (nm) 225 nm (band II) and 285 nm (band I) of the apigenin nucleus. Once sodium acetate was added, a bathochromic shift of band II was not observed, which is diagnostic of a 7-derivative group. The remaining diagnostic shift reagents were in complete agreement with the 4′-methoxy,7-glycosyl ampelopsin structure. 27 Negative ESI-MS spectrum exhibited the molecular ion peak at m/z 505.1253 [M–H]– which corresponds to a molecular weight of 506 and a molecular formula of C24H26H12. In addition, a fragment ion at m/z 319.2471 indicated ampelopsin-H, m/z 345.1243 indicated vinyl ampelopsin-H, m/z 359.2431 indicated methoxy-vinyl ampelopsin-H, m/z 491.1281 indicated rhamnosyl-vinyl-ampelopsin, m/z 505.1253 indicated methoxy-vinyl-rhamnosyl-ampelopsin-H and m/z 1011.3445 indicated dimer-H. Mild acid hydrolysis and COPC showed the presence of rhamnose in the aqueous phase. 1H-NMR spectrum showed an AX coupling system of singlet at δ 6.56 ppm integrated to two protons, accounting for two protons; these were assigned to H2′/6′. In addition, the doublet signal at δ 4.58 was assigned to H-2, the doublet signal at δ 4.50 was assigned to H-3, the singlet at 6.35 ppm was assigned to H-8 and the absence of signal of H-6 indicated the presence of a derivative at this position. The UV spectrum indicated the presence of 7,4′-derivatives. All 1H and 13C chemical shifts were allocated by comparing the detected values of related compounds with previously published data and that was confirmed by two dimensions NMR (COSY and HMBC).26-33 Therefore, Compound 8 was identified as 4′,methoxy-6-vinyl-7-O-rhamnosyl-dihydromyricetin or 4′,methoxy-6-vinyl-7-O-rhamnosyl-ampelopsin.
Biological Activities
The oral administration of the alcoholic extract of S. terebinthifolia with up to 3 g/kg body weight did not show any signs of toxicity or death in the animals within 24 h of its administration. Neither were there any signs of toxicity after further 14 days of observation. The results revealed the antioxidant effect of the extract, in which restoration of the blood glutathione in diabetic rats was detected once the plant extract was administered orally at both concentrations of 100 and 300 mg/kg (Table 1). Furthermore, the ability of the extract to restore glutathione is strongly linked to the high phenolic content, as mentioned earlier (67 ± 2.43 mg of GAE/g), and the high flavonoid content (26 ± 3.52 mg of QE/g) of the dry material.
Antioxidant Activity of Alcoholic Extract of S. terebinthifolia Fruit via Measuring the Glutathione Level in the Blood of Diabetic Rats (n = 6 in Each Group).

Our study showed that administration of the fruit extract caused anti-inflammatory and analgesic activity in the tested animals. As given in Table 2, the extract reduced the mice paw edema in a dose-dependent manner (p < .05). The percentage inhibition of edema was found to be 50.24% to 53.73% for different doses of extract (100 and 300 mg/kg). Also, dose-dependent analgesic effects of the extract were reported in this research (Table 3). The results of doses 100 and 300 mg/kg were significant (p < .05) and the number of writhes was reduced at both concentrations to 25.67 and 23.73, respectively.
Anti-inflammatory Activity of Alcoholic Extract of Fruits of S. terebinthifolia on Mice Paw Edema (n = 6 in Each Group).
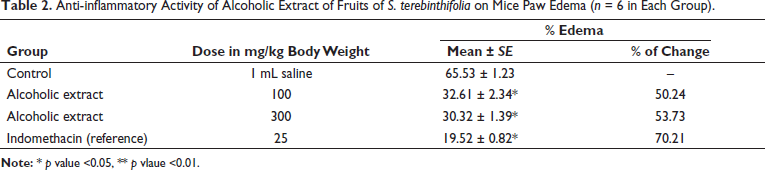
Analgesic Activity of Alcoholic Extracts of Fruits of S. terebinthifolia on Acetic Acid-induced Writhing Response (n = 6 in Each Group).
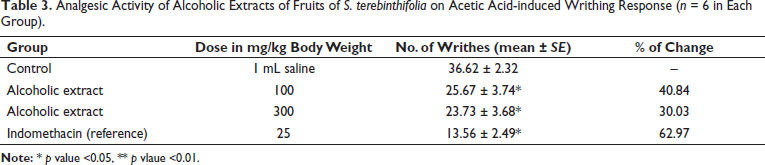
In this study, the cytotoxicity assay of the ethanol extract of S. terebinthifolia fruit showed a significant inhibitory effect against the MCF-7 cell line (half maximal inhibitory concentration (IC50) =18.3 ± 1.12 µg/mL) and the dosing results that demonstrated an inhibition effect were considered active (Figure 2). As reported by the National Cancer Institute, the extract inhibits 50% of cell growth and an IC50 of <20 µg/mL is considered to have effective in vitro cytotoxic activity. 34
Cytotoxic Activity of Ethanolic Extract of S. terebinthifolia Fruit via Measuring Cell Viability Against the MCF-7 Cell Line (n = 3).
Antimicrobial assay with the fruit extract revealed an inhibitory effect on E. coli and S. aureus growth as an example of both Gram +ve and Gram −ve in different ratios in relation to the standards of 58.6% and 43.8%, respectively (Table 4). The antifungal assay revealed that C. albicans growth was inhibited by the fruit extract by 63.2% in comparison with the standard (amphotricin B).
Antimicrobial Activity of Alcoholic Extract of Fruits of S. terebinthifolia (% Related to Standard).

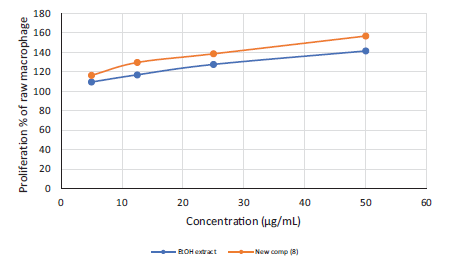
Results of the effect of the incubation with the tested samples (80% alcoholic fruit extract and the new compound) for 48 hr on the proliferation of RAW 264.7 macrophage cells was estimated by MTT assay and revealed a significant increase in cellular proliferation (p < .05) at the higher dose. This elevation in cell proliferation was dose-dependent (Figure 3), starting from the lowest tested dose and reaching 1.41- and 1.56-fold of the control cells, respectively, at the higher dose, indicating immunomodulatory activity. 35 Interestingly, Compound 8 showed better activity than the extract, showing that the activity of the extract may be due to the presence of this new compound.
Immunomodulatory Activity of the Ethanolic Extract and the New Compound (Compound 8) Indicated by the Proliferation of Immune Cells.
Discussion
Our results show that one of the main antioxidants, glutathione, was significantly increased in the tested rat blood. In addition, the high phenolic content (GAE and QE) detected is highly associated with the ability of the extract to reestablish glutathione levels. Moreover, some of the compounds isolated in this study, such as gallic acid, ellagic acid, afzelin, kaempferol and quercetin, are likely to be responsible for the extract’s antioxidant properties.36-38 This observation indicates the ability of the fruit extract to prevent cells, especially diabetic ones (which are known for their high levels of oxidation), from fighting against reactive species, thus preventing their degenerative effect on cell components. An antioxidant effect was also reported in which the oral administration of S. terebinthifolia extract successfully reinstated glutathione levels when compared to a control group (with hepatotoxicity induced by carbon tetrachloride). 39
In line with its reputation as an anti-inflammatory plant in folk medicine, S. terebinthifolia here showed the ability to reduce edema as well as the number of writhes induced by acetic acid. Some compounds of this plant, such as the flavonoids, including afzelin, quercetin and kaempferol, which were all isolated in this research, appear to have anti-inflammatory activity. Indications of the anti-inflammatory potential of S. terebinthifolia extracts are in line with a previous study by Rosas’s group. Furthermore, another earlier study showed that S. terebinthifolia extract inhibited the synovial IL-6, IL-1β and TNF-α production. 40
It is well described that oxidative stress is linked to the development of different pathological conditions, ranging from aging to inflammation and carcinogenesis, and the link between free radicals and inflammatory response is well known, since those elaborated from phagocyte cells are essential to the inflammatory response due to their role in the initiation of nuclear factor-κB (NF-κB) activity, resulting in the induction of inflammatory cytokines and cyclo-oxygenase 2 transcription, resulting in the activation of inflammatory response. 41 This could explain the link between both antioxidant and anti-inflammatory activities and this could be due in part to the elevation of glutathione. Glutathione is already known for its anti-inflammatory role, as it inhibits the transcription factor NF-κB that initiates and controls the transcription of a number of inflammatory genes. Glutathione not only inhibits the production of most inflammatory cytokines, but it also plays an essential part in certain immune system activities, both innate and adaptive, such as the proliferation of T-lymphocytes and the phagocytic activity of polymorphonuclear neutrophils. 42
The cytotoxic activity of the extract was detected and may be attributed to the polyphenolic content, since phenolic compounds are reported to protect against cancer development and to suppress cancer cell activity via their ability to inhibit the metabolic enzymes that cause activation of potential carcinogens or arrest cell cycle progression. 43 In addition, Bendaoud’s group reported the cytotoxicity of the essential oil of Schinus species with higher IC50 (47 ± 9 mg/L). 44 Many of the isolated compounds here, such as quercetin and ellagic acid, are known to have cytotoxic properties that are shown to suppress the growth of cancer cells through different mechanisms such as antioxidant function.45, 46
The fruit extract revealed an inhibitory effect on E. coli and S. aureus growth in different ratios in relation to the standards of 58.6% and 43.8%, respectively, as well as an effect against C. albicans growth at a rate of 63.2% in comparison with the standard drug. This could be attributed to the high phenolic compound of especially isolated compounds, such as gallic and ellagic acids, which have previously been reported to have antimicrobial activities. 47
The newly isolated compound shows a significant increase in the proliferation of RAW 264.7 macrophage cells (p < .05), as does the extract, reaching 1.56- and 1.41-fold, respectively, which indicates the immunomodulatory activity of the extract. In addition, Compound 8 showed better activity than the extract, indicating that the activity of the extract can be attributed to this new compound. As mentioned above, glutathione has an essential role in immune response, and in this study, its level was elevated by the extract, suggesting that glutathione may be responsible for the immunomodulatory effect of the extract. This property can be utilized to develop alternative antimicrobial strategies to fight broad-spectrum microorganisms via the augmentation of host responses during infectious disease. Furthermore, treatment plans relying on manipulation of the immune response have gained research attention due to advantages such as providing a broad spectrum of activity against various microbial infections as well as bacterial, with nonspecific emergency-treatment options that are essential for events where a novel pathogen emerges, such as severe acute respiratory syndrome coronaviruses 1 and 2 (SARS-CoV-1 and 2). Furthermore, these immunomodulatory compounds may increase treatment variation for immunocompromised patients for whom poor outcomes are reported from traditional antimicrobials in many cases. 48 Since Compound 8 is new and this is the first time, it has been evaluated for its immunomodulatory effect, it merits further evaluation. In the current study, only low quantities of this new compound were available and we therefore suggest that more biological activities be tested in further research.
Conclusion
In this study, the antioxidant, anti-inflammatory, analgesic, cytotoxic and antimicrobial properties of S. terebinthifolia fruit have been reported, with bioguided isolation leading to the isolation of a new immunomodulatory polyphenolic compound. Compound 8 showed significant immunomodulatory activity, whereby treatment with the extract increased the proliferation of immune cells, indicating its potential to boost immunity. This study concludes that this plant compound is a significant immune modulator as well as a potential antimicrobial agent, and thus may be a good candidate for infectious disease treatment. More in vitro and in vivo pharmacological studies are needed to further evaluate this interesting species.
Abbreviations
GAE, Gallic acid equivalent; QE, Quercetin equivalent; 1D, One-dimensional; 2D, two-dimensional; ppm, Parts per million; LD50, Median lethal doses; PBS, Phosphate-buffered saline; NCCLS, National Committee for Clinical Laboratory Standards; MTT, 3-(4,5-dimethyl-2-thiazolyl)-2,5-diphenyl-2H-tetrazolium bromide; IC50, Half maximal inhibitory concentration; NF-κB, Nuclear factor-κB; SARS-CoV-1 and 2, Severe acute respiratory syndrome coronaviruses 1 and 2.
Authors’ Contributions
Aljawharah AlQathama and Mohamed F. Barghash shared the performance of biological activity tests (antimicrobial, antioxidant, cytotoxic, immunomodulatory, analgesic and antinflammatory); Mohamed I. S. Abdelhady, Mohsen S. Al-Omar and Aliaa Shallan shared the collection and identification of plant material, chromatographic separation of polyphenolic compounds, and interpretation of spectrums of the isolated compounds. All authors shared in writing the manuscript.
Footnotes
Declaration of Conflicting Interests
The authors declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Ethical Approval
Funding
The author received no financial support for the research, authorship, and/or publication of this article.
Informed Consent
Not applicable
