Abstract
This study investigated the main target sites of chlorpyrifos (CPF), its effect on biochemical indices, and the pathological changes observed in rat liver and kidney function using gas chromatography/mass spectrometry. Adult female Wistar rats (n = 12) were randomly assigned into two groups (one control and one test group; n = 6 each). The test group received CPF via oral gavage for 21 days at 5 mg/kg daily. The distribution of CPF was determined in various organs (liver, brain, heart, lung, kidney, ovary, adipose tissue, and skeletal muscle), urine and stool samples using GCMS. Approximately 6.18% of CPF was distributed in the body tissues, and the highest CPF concentration (3.80%) was found in adipose tissue. CPF also accumulated in the liver (0.29%), brain (0.22%), kidney (0.10%), and ovary (0.03%). Approximately 83.60% of CPF was detected in the urine. CPF exposure resulted in a significant increase in plasma transaminases, alkaline phosphatase, and total bilirubin levels, a significant reduction in total protein levels and an altered lipid profile. Oxidative stress due to CPF administration was also evidenced by a significant increase in liver malondialdehyde levels. The detrimental effects of CPF on kidney function consisted of a significant increase in plasma urea and creatinine levels. Liver and kidney histology confirmed the observed biochemical changes. In conclusion, CPF bioaccumulates over time and exerts toxic effects on animals.
Introduction
Organophosphate (OP) pesticides have successfully replaced the persistent organochlorine (OC) pesticides used in the 1970s. OP pesticides are extensively utilized for agriculture and domestic purposes and account for more than half of all insecticides used in the world. 1 –3 Chlorpyrifos (CPF; O, O′-diethyl-O-(3,5,6-trichloro-2-pyridyl) phosphorothionate) is a widely used OP insecticide that is persistently found at alarming levels in a number of domestic vegetables and surface and groundwater sources from Bangladesh. 4,5 In developing countries, multiple OP insecticide poisonings, health disorders, and deaths are the consequence of deliberate self-ingestion among younger populations because these highly toxic pesticides are readily available. 2
CPF is a broad spectrum chlorinated insecticide that is readily absorbed from the intestine and lung in humans and experimental animals. 6 CPF is thought to be primarily metabolized in the liver by multiple specific cytochrome P450 mixed-function oxidases through several reaction pathways, and CPF oxon is the major toxic metabolite. 6,7 CPF oxon inhibits acetylcholinesterase activity in target tissues, and accumulation of endogenous acetylcholine causes overstimulation and disruption of nervous system activity. 8,9 CPF elicits a number of additional toxic effects, including reproductive, DNA damage, and genotoxicity. 6 One of the molecular mechanisms of CPF-induced toxicity involves the generation of reactive oxygen species (ROS) and subsequent oxidative stress in target tissues. 10,11 Generally, lipid peroxidation (LPO) due to OP poisoning is evidenced by enhanced levels of thiobarbituric acid reactive substances (TBARS) in the plasma, brain, lung, testes, and erythrocytes. 12 –15
The extensive and indiscriminate use of OP pesticides in agriculture has been a major concern because of its potentially known or suspected harmful effects to humans. 4 Moreover, some of these pesticides remain in the soil, ground water, drinking water, marine organisms, and various food products. 4,5,16 Therefore, CPF toxicity may occur from the direct or indirect exposure due to the consumption of food and water contaminated with CPF residues. 17
Several analytical methods have been used for the identification and quantification of pesticides in animal matrices, including high-performance liquid chromatography (HPLC), gas chromatography (GC), and other chromatographic techniques. The majority of these methods utilize urine and blood samples as the only target animal matrices. 6,18 Very few methods that can determine OP insecticide levels in animal tissues are available; however, fat or other body tissues are possible important CPF target sites. Therefore, the availability of a rapid, sensitive, reliable, and inexpensive high-throughput method for the detection of pesticide residues in a wide range of animal matrices is necessary.
The objective of this research was to determine the distribution of CPF in rats following a short-term subchronic oral intoxication using gas chromatography–mass spectrometry (GCMS).
Materials and methods
Chemicals
The CPF insecticide (purity 99.0%) was obtained from Dr Ehrenstorfer GmbH (Augsburg, Germany). 1,1,3,3-Tetraethoxy propane was purchased from Nacalai Tesque Inc. (Kyoto, Japan). Florisil (magnesium silicate, mesh size 60–100) was purchased from Sigma (St Louis, Missouri, USA). The solvents, including n-hexane (Merck, Germany), ethyl acetate, acetone, and diethyl ether (BDH, England) were of analytical grade. The acetonitrile (Scharlau, Europe) was of HPLC grade. Prior to use, all glasswares were washed with detergent, sonicated, rinsed with distilled water, followed by acetone and then heated at 180°C for 2 h to remove any residual contamination.
Animals
The experiments were conducted according to the ethical guidelines approved by the Bangladesh Association for Laboratory Animal Science. The experimental protocol was approved by the Biosafety, Biosecurity and Ethical Committee of Jahangirnagar University (approval no: BBEC, JU M (2013.2c). Female Wistar rats (n = 12) aged between 16 and 24 weeks (170–190 g) were used in this study. The animals were bred and housed in the animal facility of the Department of Biochemistry and Molecular Biology at Jahangirnagar University at a constant temperature (23 ± 2°C) with humidity ranging between 40% and 70%. The rats were housed in metabolic cages and received a natural 12-h light/12-h dark cycle. The rats were provided a standard laboratory pellet diet and water ad libitum. The rats were acclimatized to the laboratory environment at least 1 week prior to the start of the experiment.
Animal treatment
The rats were divided into two equal control (n = 6) and test (n = 6) groups. The test group received CPF via oral gavage at a dose of 5 mg/kg (approximately 1/27 of the 50% lethal dose (LD50)) daily for 21 days. CPF was dissolved in corn oil (0.4 mL/rat) prior to use. The control rats underwent the same treatment paradigm with corn oil only (0.4 mL/rat). Each rat’s body weight was recorded regularly, and the doses were adjusted accordingly. Urine and stool samples from both groups were collected in the morning for 21 consecutive days and were immediately stored at −80°C. After the 21-day treatment period, rats from both groups were deeply anaesthetized with a ketamine hydrochloride injection (100 mg/kg) before euthanasia by dissection. Blood (5 mL) was immediately collected from the inferior vena cava in 2.5 mL heparinized tubes.
To the best of our knowledge, this CPF dose was first to investigate the subchronic CPF distribution and its effects on rat liver and kidney for 21 days period, while most of the previous studies reported other treatments in rats. 6,19,20
Plasma preparation
Approximately 2.5 mL of blood was centrifuged at 2000 r/min for 10 min at ambient temperature. The plasma was aspirated and stored at −20°C for further biochemical analysis. The second 2.5 mL blood sample was stored at −80°C until use in the distribution study.
Tissue storage and preparation of liver tissue homogenates
Multiple organs, including the brain, heart, liver, lung, kidney, ovary, adipose tissue, and skeletal muscle, were properly perfused with ice-cold phosphate-buffered saline (PBS) via heart and then immediately excised. The liver sample was further perfused to remove any residual blood from the vasculature before being blotted, minced, and homogenized in PBS (25 mM, pH 7.4) to produce a 10% (w/v) homogenate. The homogenate was centrifuged at 1700 r/min for 10 min, and the supernatant was stored at −20°C for subsequent biochemical analysis. A portion of the liver samples was stored in 10% formalin for histopathological examination. Other tissues, including the liver samples, were stored at −80°C for the determination of CPF distribution using high-resolution GCMS.
Biochemical analysis
Biochemical parameters in plasma
The collected plasma was used to estimate the hepatic marker enzymes alkaline phosphatase (ALP), alanine transaminase (ALT), aspartate transaminase (AST), and lactate dehydrogenase (LDH) using commercially available standard assay kits (Standbio Laboratory, Boerne, Texas, USA). The plasma was also used for the estimation of total bilirubin (TB), total cholesterol (TC), triglycerides (TG), and high-density lipoprotein–cholesterol (HDL-C) using commercially available standard assay kits (Standbio Laboratory). Low-density lipoprotein–cholesterol (LDL-C) and very low-density lipoprotein–cholesterol (VLDL-C) levels in the plasma were calculated using the Friedewald formulae
21
:
The total protein (TP) content of the plasma was measured according to the method established by Lowry et al. 22 Kidney markers (creatinine and urea) were estimated using commercially available standard assay kits (Standbio Laboratory).
Biochemical parameters of liver tissue
LPO was assessed by measure malondialdehyde (MDA) levels in the liver. MDA, also referred to as TBARS, was measured using the method described by Ohkawa et al. 23 Briefly, 0.1 mL of tissue homogenates were mixed with 0.1 mL of 8.1% (w/v) sodium dodecyl sulfate, followed by the addition of 1.5 mL of 0.4% thiobarbituric acid in 20% acetic acid (pH 3.5) and 0.1 mL of distilled water. The mixture was heated at 95°C for 1 h and then cooled. Next, 1 mL of n-butanol:pyridine (15:1, v/v) was added, and the sample was vigorously shook for 10 min. The solution was centrifuged at 1000 r/min for 10 min at room temperature and was allowed to stand for 15 min. The absorbance of the supernatant was measured at 532 nm. TBARS was expressed as nanomoles of MDA per milligram of protein. The TP in liver tissue homogenates was estimated based on Lowry method. 22
Histopathological analysis
For light microscopy, the liver samples were fixed in 10% neutral-buffered formalin. The fixed tissues were dehydrated in ascending grades of ethyl alcohol and cleared in xylene until they became translucent. Following paraffin wax embedding, serial sections with a thickness of 5 µm were cut on glass slides before routinely staining with hematoxylin and eosin for histopathological examination. All of the slides were examined with a light microscope (MZ3000 Micros, Austria) and compared with the control tissue sections.
Sample extraction for CPF distribution analysis
Tissue extraction
Solvent extraction of various organs (liver, brain, heart, lung, kidney, ovary, and skeletal muscle), adipose tissue, and stool samples was performed according to the method developed from previously described methods by Carr et al. 1 and Panseri et al. 24 with some modifications. The extraction efficiency was monitored by adding known concentrations of CPF in tissue samples or blank extracts. The sample aliquots (1 g) were homogenized in 3 mL of 0.05 M Tris-hydrochloric acid (Tris-HCl) buffer (pH 7.4) containing magnesium chloride (0.005 M). Next, 1 g of sodium chloride and 2.5 mL of n-hexane:acetone (4:1) or ethyl acetate (only for adipose tissue) as a solvent was added. The mixture was vortexed for 5 min before the addition of anhydrous sodium sulfate (Na2SO4; 6 g). The mixture was centrifuged at 3000 r/min for 10 min, and the fraction containing the organic solvent was collected. The samples were reextracted with an additional 2.5 mL of solvent (twice) as described above. The solvent fractions were pooled and evaporated to dryness under a gentle stream of nitrogen.
Lipids in the solvent extract from the adipose tissue were completely removed as follows: Briefly, the dried solvent extract was redissolved in 25 mL of acetonitrile and stored in the freezer at −20°C for 20 min before immediate filtration with filter paper. The precipitated lipids were further redissolved in 25 mL of fresh acetonitrile before being filtered again using a similar procedure. The filtered extracts were pooled and evaporated to dryness under a gentle stream of nitrogen. The dry extract was dissolved in 1 mL of hexane before cleanup with florisil.
Blood extraction
A CPF extraction from blood samples was performed according to the method described by Agarwal et al. 25 with some slight modifications. Briefly, blood samples (2 mL) were diluted with 10 mL of distilled water, followed by the addition of 2 mL of saturated brine solution. Extraction of the analytes was accomplished by the addition of 25 mL of n-hexane:acetone (4:1) (thrice) followed by vortexing for 5 min. Anhydrous Na2SO4 was added before centrifugation at 3000 r/min for 10 min. The solvent fractions were collected and evaporated to dryness under a gentle stream of nitrogen. The dry extract was dissolved in 1 mL of hexane before cleanup with florisil.
Urine extraction
Urine was extracted based on the method described by Tewari and Sharma 26 with some slight modifications. Briefly, the urine was first treated with HCl (6 N) in a dropwise manner until the pH reached approximately 2.0. The acidified urine sample (10 mL) was then transferred to a separating funnel. Distilled water was added to give a final volume of 100 mL. Next, n-hexane:acetone (4:1) (40 mL) was added as another solvent before the funnel was shaken for 5 min to separate the solution into two layers. Following the separation of the layers, the upper solvent layer was collected into a tube. The bottom sample layer was transferred to another separating funnel for further extraction. The funnel was shaken with an additional 10 mL of n-hexane for 5 min, and the hexane layer was recollected. The hexane extracts were combined and then filtered using anhydrous Na2SO4 (10 g). Next, the extract was further concentrated to a final volume of 1 mL under a gentle stream of nitrogen before cleanup with florisil.
Sample cleanup
To eliminate any residual co-extracts, florisil column chromatography was performed as described by Chowdhury et al. 27 The top 1.5 cm of a 0.6-cm diameter column was packed with anhydrous Na2SO4. Briefly, the florisil (60–100 mesh) was activated at 200°C for 6 h and subsequently deactivated with 2% distilled water. Elution was performed with a solvent mixture of 2% diethyl ether in double-distilled hexane at 5 mL/min. The eluent was concentrated to a small volume of 2 mL using a rotary vacuum evaporator (Rotavapor-R215, Buchi, Switzerland) at 3.66 psi in a water bath at 45°C. Any residual solvent was completely removed under a gentle stream of nitrogen. The evaporated extract was reconstituted with acetonitrile to a total volume of 1 mL prior to the GCMS injection. A similar procedure was performed for all rat tissue, blood, urine, and stool samples.
GCMS instrumental conditions
GCMS analysis of the blood, tissue, urine, and stool samples was performed using a mass spectrometer detector (FinniganTRACE DSQ-Mass Spectrometer, MS 200710, Ramsey, Minnesota, USA) configured with a GC (FinniganTRACE GC ultra, GCK04244B24500070) system equipped with split/splitless electronic pressure control. A fused silica ZB-5 MS capillary column (30 m × 0.25 mm × 0.25 µm film thickness) was used. The GCMS method was based on our previous published method. 4 Selected ion monitoring was utilized for increased sensitivity with selected ions at an m/z of 97 and 197.
Preparation of standards and calibration curve
The standard stock solution containing CPF at 1000 mg/L was prepared as previously described by Chowdhury et al. 4 The amount of pesticide in each sample was calculated based on the slope of the standard curve. 28
Quality control and quality assurance
The method was validated for accuracy and precision in accordance with the European Commission guidelines.
29
Accuracy was measured by analyzing and comparing samples with measured values of known spiked samples. Precision was expressed as the relative standard deviation. For the recovery experiment, the homogenized animal matrices from the control rats were spiked with appropriate volumes of standard CPF at concentrations of 0.05 µg/g in triplicate (n = 3). Percentage recovery was calculated using the following formula:
The mixture was left standing for 1 h to allow for equilibration before undergoing the extraction and sample cleanup processes described above and subsequent GCMS analysis. The mean percentage recovery of the method ranged from 78.0% to 96.0%, and the mean precision ranged from 4.45% to 14.54%.
The mean recovery percentage of CPF in the spiked positive control samples using two different solvent systems followed by florisil cleanup is shown in Table 1. The solvent showing the maximum recovery was undertaken for final tissue extraction, which was considered acceptable. 4,30
Recovery (%) of CPF in different animal matrices using GCMS technique.

CPF: chlorpyrifos; GCMS: gas chromatography/mass spectrometry
aMean value of three replicates
LOD and LOQ
Limit of detection (LOD) was defined as the lowest concentration of analyte in a sample that could be detected but not necessarily quantified. The limit of quantification (LOQ) was defined as the lowest concentration of the analyte that could be quantified with acceptable accuracy and precision. The LOQ and LOD were evaluated as signal-to-noise ratios (S/N) of 10:1 and 3:1, respectively, by analyzing unspiked samples (n = 10). 31 In the present study, the LOD and LOQ were determined as 0.001 and 0.003 mg/kg, respectively.
Statistical analysis
Data were analyzed using Statistical Packages for Social Science (version 20.0, IBM Corporation, Armonk, New York, USA) and Microsoft Excel 2007 (Redmond, Washington, USA). Statistical analysis of the biochemical data were completed using Tukey’s test. The results are expressed as the mean value ± standard deviation (SD). Differences with p < 0.05 were accepted as statistically significant.
Results
The absolute and relative body and liver weights are depicted in Table 2. At the end of the experimental procedure, a significant (p < 0.001) loss in body weight was observed in rats treated with CPF. CPF exposure also caused a slight but nonsignificant increase in the absolute and relative liver weight. In addition, the CPF treatment group showed several clinical signs such as weakness with reduced activity, huddling, slight diarrhea and hair loss, reduced food, and increased water intake. The observed signs were related to cholinergic crisis, consistent with the OP toxicity.
The effects of subacute CPF exposure on BW as well as absolute and relative liver weights.a

BW: body weight; CPF: chlorpyrifos.
aThe results are expressed as the mean ± SD, n = 6.
bp < 0.001; % BW gain = [(final BW–initial BW)/final BW] × 100.
The distribution of CPF into various organ, blood, and tissue samples following a 21-day exposure is summarized in Figure 1. Overall, approximately 6.18% of the CPF was distributed in body tissues, 83.6% of CPF was excreted in the urine and the remaining 10% was excreted through the feces.

Distribution of CPF in rat organs. CPF: chlorpyrifos.
Based on the GCMS results, the highest CPF concentration was detected in the adipose tissue (Figure 2), and the ovary showed the lowest concentration of CPF (Table 3). CPF was not detectable in the heart, lung, or skeletal muscle following subacute oral administration. In addition to adipose tissue, the liver, brain, and kidney were other important target sites for CPF accumulation in rats. Approximately 0.008 µg/mL of CPF was found in the blood, indicating that the compound was highly lipophilic.

(a) Chromatogram of a CPF standard injected at 5 ng/g (RT 13.19 min); (b) mass-spectrum of the CPF standard (m/z = 96.88 and 196.87); (c) representative chromatogram of adipose tissue showing the presence of CPF (RT 13.18 min); and (d) mass spectrum of CPF in adipose tissue (m/z = 96.83 and 196.78). CPF: chlorpyrifos; RT: retention time.
Concentration of CPF (in µg/g) in various organs.a

CPF: chlorpyrifos; BDL: below detection limit. LOD: limit of detection.
aResults are expressed as the mean ± SD of six rats. LOD = 0.001 ng/g and LOQ = 0.003 ng/g.
CPF exposure significantly affected multiple biochemical parameters in rats (Table 4). There was a significant increase in plasma ALP, ALT (p < 0.001), and AST activities (p < 0.05); however, LDH activity in CPF-treated rats was not significantly different when compared with the control group. Conversely, the TP level was significantly decreased in the plasma (p < 0.01) of CPF rats. In addition, TB was significantly elevated in the plasma of the test group when compared with the control animals (p < 0.001). CPF administration caused a significant (p < 0.001) increase in LPO as shown by an elevation in liver MDA levels when compared with the control (Figure 3).
The effects of CPF exposure on multiple liver biomarkers.a

CPF: chlorpyrifos; ALP: alkaline phosphatase; ALT: alanine transaminase; AST: aspartate transaminase; LDH: lactate dehydrogenase; TB: total bilirubin; TP: total protein; SD: standard deviation.
aResults are expressed as the mean ± SD, n = 6.
bp < 0.001.
cp < 0.05.
dp < 0.01.

The effects of CPF exposure on liver LPO. The results are expressed as the mean ± SD, n = 6, ***p < 0.001. CPF: chlorpyrifos; LPO: lipid peroxidation; SD: standard deviation.
Subchronic exposure to CPF significantly altered the lipid profile of rats (Table 5). Plasma TC (p < 0.01), TG, VLDL-C, and LDL-C levels were significantly elevated in CPF-treated rats when compared with the control group (p < 0.001). HDL-C levels decreased in the plasma of CPF rats when compared with the control group.
The effects of CPF exposure on lipid profiles.a

CPF: chlorpyrifos; TC: total cholesterol; TG: triglycerides; VLDL-C: very low-density lipoprotein–cholesterol; LDL-C: low-density lipoprotein–cholesterol; HDL-C: high-density lipoprotein–cholesterol; SD: standard deviation.
aResults are expressed as the mean ± SD, n = 6.
bp < 0.01.
cp < 0.001.
The detrimental effects of CPF on kidney function were confirmed by the significant increase of plasma urea levels when compared with the control group (p < 0.001); however, plasma creatinine did not significantly change (Table 6).
The effects of CPF exposure on plasma urea and creatinine levels.a
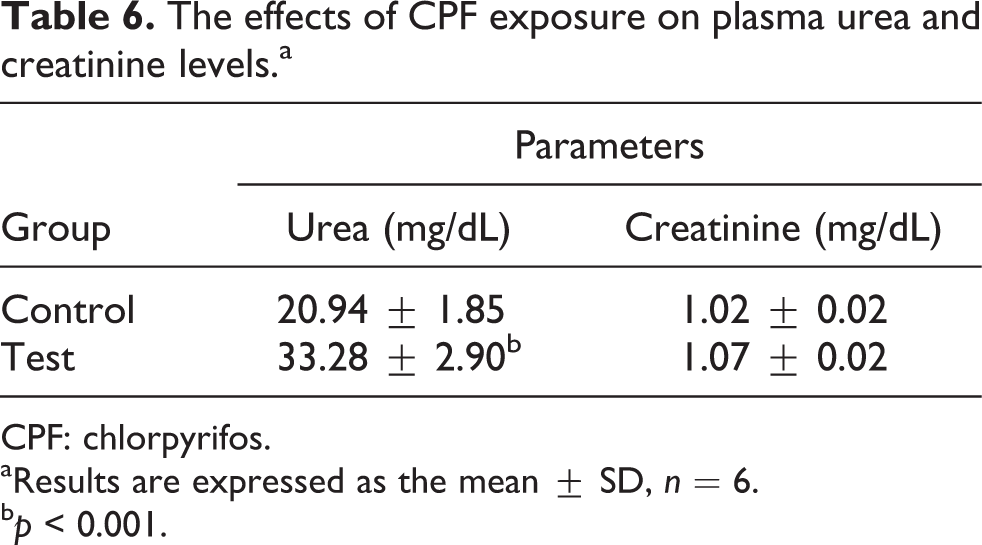
CPF: chlorpyrifos.
aResults are expressed as the mean ± SD, n = 6.
bp < 0.001.
Histopathology of control liver sections revealed normal histology, and the central vein (CV) was clearly observed in the center of the lobule. Hexagonal hepatocytes with clear and well-defined nucleus were regularly arranged in the lobule (Figure 4(a)). A microscopic examination of CPF-treated liver indicated the severe infiltration of inflammatory cells in the lobule with degeneration of hepatocytes at the peripheral CV areas (Figure 4(b)). In addition, congestion in the CV associated with inflammatory infiltrates was observed in CPF-exposed rats (Figure 4(c)).

Photomicrographs of H&E-stained sections of the liver of control rats showing (a) normal histological structure of the CV and surrounding hexagonal hepatocytes. CPF-treated rat livers showed (b) inflammatory cell infiltration in the CV and the lobule (red arrows), degeneration of hepatocytes at the peripheral area of the CV (black arrows) and (c) congestion in the CV associated with inflammatory infiltrates (yellow arrows). A kidney section from control rats (d) showing a normal morphology of renal parenchyma with well-defined glomeruli and tubules. CPF-treated rat kidneys (e and f) demonstrating a marked degeneration of tubular epithelium (black arrow) and intraluminal casts (green arrows). CPF: chlorpyrifos; CV: central vein; H&E: hematoxylin and eosin.
The control rats showed normal renal tubules and glomeruli (Figure 4(d)). However, rats treated with CPF showed severe tubular desquamation of epithelium, moderate cell necrosis, and vacuolization as well as presence of intraluminal casts (Figure 4(e) and (f)).
Discussion
To our knowledge, this is the first study to investigate the possible target sites of an OP insecticide using high-resolution GCMS techniques with simple solvent extraction procedures. This method was successfully applied to assess CPF distribution in various organs. The study also reports the significant changes in the lipid profile and liver and kidney function following subchronic intoxication with CPF in rats.
In toxicological studies, organ and relative organ weights are among the most important criteria for evaluation of toxicity. 20 The net body weight gain of CPF-intoxicated rats was markedly reduced when compared with the control group; however, a significant change in food consumption following CPF exposure was observed. A possible explanation for the reduction in body weight may be the overall increased degradation of lipids and proteins directly induced by CPF. 32 This is further supported by the decreased plasma TP observed in CPF-treated rats. Our findings support previous studies that showed a dose-dependent decrease in the body weight gain of animals intoxicated with OP, such as leptophos and isoprocarb. 33,34 Moreover, the slight increase in relative liver weight may be related to the reduction in body weight gain in experimental animals. 35,36
GCMS analysis for the detection and quantification of pesticides in animals provides a reliable screening method of contaminated samples due to its high accuracy and precision. 24 Nolan et al. 37 estimated that the bioavailability of orally administered CPF was at least 70% based on recovery levels in the urine following a single dose. Another study reported peak blood concentration at 109 ng/g following a single oral dose of CPF. 38 However, the authors also suggested that the actual bioavailability may be greater because a fraction of the administered dose could be eliminated through other pathways (e.g. bile/feces) or retained in the body tissue. 6 The findings from our study suggest that the approximate bioavailability of CPF is 83.60% based on the detection of CPF in blood and urine following subchronic oral administration. Timchalk et al. 9 reported that approximately 80% of CPF is absorbed. Our study suggests that approximately 6.18% of CPF is distributed in the body tissue, and adipose tissue is the main site of CPF accumulation. This result was expected because CPF has been reported to be highly lipophilic. 6
The solubility of CPF in nonpolar solvents and its accumulation in fatty tissues have been observed in a previous study. 39 Bakke et al. 40 reported the accumulation of CPF in adipose tissue only. In our study, we found that CPF also accumulated in the liver, brain, kidney, and ovary. CPF accumulation in the rat brain and kidney was also observed by Poulin and Krishnan 41 and Timchalk et al. 9 Our biochemical and histological findings strongly support the observed pattern of organ distribution in the liver and kidney.
The liver is the major site for detoxification or biotransformation of xenobiotics, 42 and it may be the final target site of CPF. Our study confirmed the significant accumulation of CPF in the liver. CPF may subsequently undergo biotransformation to induce severe oxidative stress in hepatocytes as evidenced by the significantly elevated liver MDA levels in experimental rats when compared with the control group.
One of the most important findings of this study was the detection of CPF in the rat ovary (0.03%), which is considered as an important risk factor for the development of ovarian cancer. The estrogenic property of CPF may contribute to the increased ovarian surface epithelium height which is thought to be the primary site for cancer development. 43,44 A disruption in the estrous cycle with prolonged metestrous and a significant increase in the uterine surface epithelium height and myometrium thickness was previously reported in rats exposed to CPF. Moreover, degeneration of oocyte and nucleus of the rat’s ovary has previously been reported. 44 CPF was also found to cause developmental defects in rats 45 further providing information about the biochemical, physiological, and toxicological interaction of CPF related to reproduction. Only 0.0082% of CPF was detected in the circulation; therefore, the binding tendency of CPF to various proteins, such as plasma albumin, might further lower the free level of CPF available for the distribution to other compartments. 6 In addition to multiple organs, we found that 10.224% of CPF was found in the stool. Similarly, Smith et al. 46 reported that 10% of CPF was excreted in the feces.
In our investigation, a considerable CPF concentration was detected in the urine of treated rats. According to Timchalk et al., 9 maximum excretion of CPF in the urine occurs after approximately 12 h; however, significant excretion can occur after 72 h. Our study found that approximately 83.6% of CPF was excreted through the urine. Similarly, Cochran et al. 47 reported the urinary excretion of approximately 70% of the administered oral dose. Smith et al. 46 reported a slightly higher (80–90%) urinary excretion percentage.
A number of studies have investigated the pathological changes induced by exposure to CPF for 7 or 28 days or for various extended periods of time at different experimental doses. 6,19,20 A summary of the previous findings is presented in Table 7. We investigated the pathological changes and biochemical parameters of the liver and kidney in rats orally exposed to CPF at 5 mg/kg for 21 days.
Summary of reports of the effects of CPF exposure to rats.

CPF: chlorpyrifos.
We observed a significant increase in ALT, AST, and ALP activities and TB levels in the blood, which is indicative of hepatic injury due to OP administration. CPF exposure has been reported to significantly increase diagnostic markers indicative of hepatocellular damage. 20,48 Therefore, the observed elevation could be attributed to the release of liver enzymes from the cytoplasm into the blood circulation after liver injury. This release of enzymes could lead to liver necrosis and inflammatory reactions. 20,49 Based on the accumulation of TB as a measure of binding, conjugation, and excretory capacity of hepatic cells, a rise in TB levels is an important clinical clue as to the severity of necrosis. 50 The present findings are similar with those reported from previous studies. 15,19,42 However, we found that LDH showed a nonsignificant increase.
OP insecticides have been reported to generate free radicals in biological systems. 51 As a lipophilic molecule, CPF can easily cross the cell membrane and induce high levels of cellular damage. 52 MDA is the major oxidation product of polyunsaturated fatty acids (PUFAs), and increased MDA levels are a crucial indicator of LPO. 53 As previously reported, we found that MDA levels were significantly elevated, indicating oxidative injury of the hepatocytes in CPF-treated rats. 32,54 Different mechanisms have been postulated to explain CPF-induced liver injury, including lipid peroxidation and interactions with membrane components. 55 LPO is the result of oxidative deterioration of the PUFAs of biological membranes, which results in impaired membrane function, structural integrity, 56 reduced membrane fluidity, and inactivation membrane-bound enzymes. 32 Our study explains the plausible peroxidation of PUFAs, the consequent degradation of phospholipids, and the cellular deterioration reported by Tappel. 57 In addition, significantly decreased TP levels were observed in the plasma of rats exposed to CPF. These data are consistent with previous reports. 2,20 OP-induced oxidative stress increased proteolytic enzymes and DNA fragments, 58 further explaining the observed reduction in TP observed in CPF-treated animals.
Alterations in the lipid profile due to CPF administration was observed in this study. Increased levels of TC and TG were found in CPF-treated rats. CPF-induced changes in liver cell membrane permeability 59 and/or increased cholesterol synthesis in the liver 60 are plausible mechanisms behind the increase in plasma TC levels. In addition, the blockage of the liver bile ducts and a consequent reduction in cholesterol secretion into the duodenum can result in elevated plasma cholesterol levels and indicates liver damage. 61,62 The hypertriglyceridemia observed in CPF-treated rats is good evidence of increased hepatic glyceride synthesis that is directly proportional to fatty acid and glycerophosphate concentrations. 63 This may be due to increased VLDL-C, which is involved in the transport of hepatic TG to extrahepatic tissues. 64
A marked increase in VLDL-C and LDL-C with a concomitant decrease in HDL-C was observed in CPF-treated rats. These findings are consistent with those reported in previous studies. 65,66 In addition, this pattern of change is closely associated with an increased risk for coronary artery disease 67 due to the suggestive and causative role of LDL-C oxidation in the pathogenesis of atherosclerosis. 68 Moreover, low HDL-C may be attributed to CPF-induced hepatic damage, resulting in the impairment of lipoprotein synthesis. 32,67 These observations suggest that prolonged exposure to CPF may be a major risk factor for the development of degenerative heart diseases. 67
We also measured the extent of the detrimental effects on kidney functioning following subchronic CPF exposure. Both urea and creatinine levels increased (even though the increase in creatinine level was not statistically significant). Urea and creatinine are waste products of protein metabolism that are excreted through the kidney. The marked elevation of plasma creatinine and urea levels in the CPF-treated group indicates functional damage of the kidney 69 and reduced glomerular filtration. Urea levels can also be increased by other factors, such as dehydration, antidiuretic drugs and diet; however, increased creatinine is more specific to kidney damage. 70 Therefore, a significant increase in urea with a nonsignificant increase in creatinine suggests a mild to moderate degree of kidney damage induced by CPF.
Histopathological examination is a widely accepted method to assess pesticide toxicity. 19 Microscopic examination of the liver of CPF-exposed rats revealed various pathological changes that included mild to severe infiltration of inflammatory cells in the CV and degenerative necrosis of the hepatocytes at the peripheral regions of the CV. Congestion of the CV with inflammatory infiltrates was observed in the CPF-treated liver. These changes are consistent with the biochemical parameters determined in our investigation. Similar observations were reported by previous studies of CPF and other OP insecticides. 42,62 The CPF toxin may reside in the liver, which is an organ responsible for detoxification; in addition, CPF may induce severe lesions of the hepatocytes due to ROS generation. 2,32 Moreover, degeneration of epithelium and necrosis was also found in the kidney tissue due to CPF intoxication. Our study confirmed the detrimental effects of CPF in the liver and its consequent oxidative degeneration on hepatocytes.
Our study assessed only the major tissues; however, the pancreas or testes of male rats may be important sites for CPF distribution that need to be investigated in the future. Further studies on the possible detrimental effects of pesticides on other organs are important to elucidate the complete toxicological profile of CPF.
Conclusion
This study confirms that CPF has multiple lipophilic targets, including adipose, liver, brain, and kidney tissues. We also described the pathological changes, potential biomarkers of liver and kidney functions, and lipid and lipoprotein status associated with CPF exposure.
Footnotes
Declaration of Conflicting Interests
The author(s) declared no potential conflicts of interest with respect to the research, authorship, and/or publication of this article.
Funding
The author(s) disclosed receipt of the following financial support for the research, authorship, and/or publication of this article: This research was supported by the National Science and Technology (NST) special allocation 2013-1014, No. 8 (BS 130).


